Abstract
Diabetic peripheral neuropathy (DPN) is one of the most common complications of diabetes which primarily affects the sensory nervous system. Pain is the most common complaint that prompts patients to seek medical advice. With various presentations and intricate pathological mechanisms, diabetic peripheral neuropathic pain is currently the most crucial and challenging aspect of managing diabetic complications. As a heterogeneous disorder, there is no medication or treatment modality that is effective for all types of DPN and its associated neuropathic pain. Peripheral nerve decompression provides a new option for treating patients with diabetic peripheral neuropathic pain in the lower extremities. However, the clinical applicability of nerve decompression has been debated since it was first proposed. This review discusses the theoretical basis of nerve decompression, the clinical indications, and the progress of basic research based on the pathological mechanisms and nerve impairment patterns of diabetic peripheral neuropathic pain. The heterogeneity of DPN patients is summarized in terms of three aspects: complex pathophysiological mechanisms, multilevel nervous system involvement, and various nerve impairment properties. Identifying the presence of nerve entrapment among complex pathophysiological mechanisms is the key to successful outcomes. Tinel signs, focal pain, mechanical allodynia, and two-point discrimination were reported to be prognostic factors for good surgical outcomes, and their predictive ability might stem from their association with the early stage of entrapment neuropathy.
Keywords
Diabetic peripheral neuropathy and neuropathic pain
According to the latest reports of the International Diabetes Federation Diabetes Atlas 10th edition, 537 million adults are living with diabetes globally, and the number is continually increasing. 1 Diabetic peripheral neuropathy (DPN) is one of the most common complications of diabetes. It has been reported that more than 50% of individuals with diabetes will develop diabetic neuropathy over time. However, due to its heterogeneity, the prevalence of DPN is highly dependent on multiple factors, including the type of diabetes, age, disease duration, disease severity, country, and access to modern treatment. 2 A recent worldwide meta-analysis consisting of 29 studies with a total of 50,112 participants reported that patients with type 2 diabetes had a greater incidence of DPN (31.5% [95% CI 24.4–38.6]) than did those with type 1 diabetes (17.5% [13.1–36.5]). 3 With respect to age and diabetes duration, the prevalence of DPN is reportedly lower in adolescents than in adults.4,5 According to an international study, including 2733 subjects with type 2 diabetes from 14 countries, the DPN incidence varies greatly between different countries, ranging from 0.58% to 79.55%. 6 With such a large patient population, DPN has a negative physical and psychological impact on people and their families as well as posing financial strains on health care and society. The variation in prevalence may also be associated with different assessment methods and definitions of DPN. 7 While separate definitions for different varieties of DPN exist, an internationally acknowledged simple definition of DPN for clinical practice was proposed as ‘the presence of symptoms and/or signs of peripheral nerve dysfunction in people with diabetes after the exclusion of other causes’.8,9
The clinical presentations of DPN are diverse, with sensory nervous system involvement being the most common, mainly manifesting as numbness and pain. As the most distressing symptom of DPN, neuropathic pain seriously affects quality of life and is the main cause of medical treatment. The estimated prevalence of DPN pain (DPNP) in patients with diabetes ranges between 6% and 34%. 10 In addition to common spontaneous pain, there are forms of evoked pain, including hyperalgesia and allodynia. The diverse manifestations imply the existence of complex underlying pathophysiological mechanisms, which are associated with the involvement of the multilevel nervous system and various properties of nerve impairments (Figure 1).
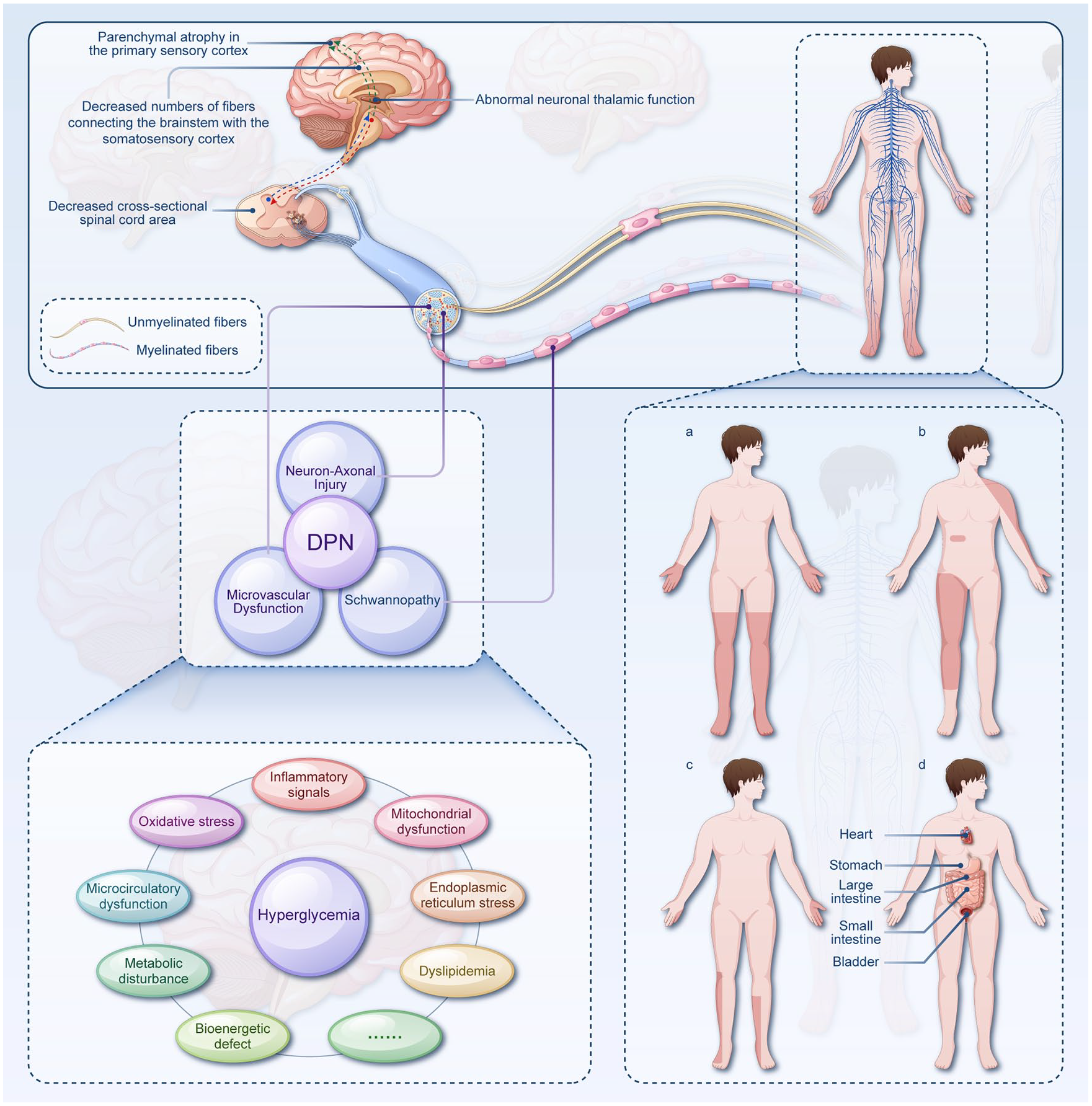
Schematic illustration showing the involvement of the multilevel nervous system (upper panel), complex underlying pathophysiological mechanisms (lower, left panel), and different patterns of nerve impairment (lower, right panel) in patients with DPN and DPNP. In the upper panel, several pathological changes in the CNS have been illustrated at the level of both the spinal dorsal horn and the brain. Myelinated afferents (sensory axons that are myelinated by associated Schwann cells) and unmyelinated afferents (sensory axons that are grouped into Remak bundles) are shown to be distributed throughout the whole human body. The lower, left panel shows that the interactions between Schwann cells, axons, and microvessels have been proposed to be intimately associated with DPN. As hyperglycemia-mediated cellular injury is likely the central factor in DPN, multiple pathophysiological mechanisms are included in the development of DPN and DPNP. Different patterns of nerve impairment in patients with diabetes are displayed in the lower. (a) distal symmetric polyneuropathy affecting both sides of the upper and/or lower extremities; a similar pattern is also shown in small-fiber-predominant neuropathy and treatment-induced neuropathy; (b) radiculoplexopathy or radiculopathy; (c) mononeuropathy or multiplex mononeuritis affecting single or multiple peripheral nerves; (d) autonomic neuropathy affecting multiple organs of the cardiovascular system, digestive system, and urinary system.
Complex pathophysiological mechanisms
Although the mechanisms of DPN are not fully understood, there are some proposed pathophysiological alterations underlying peripheral nerve injury in diabetes patients.
According to current experimental and clinical studies, hyperglycemia-mediated cellular injury is likely the central factor in DPN.11–13 Increased glucose metabolism results in overactivation of the hexosamine, 14 polyol, 15 and protein kinase C pathways, 16 as well as excessive generation of advanced glycation end products (AGEs) and/or receptor-associated AGE activation (RAGE). 17 The interaction between AGEs and their receptor (RAGE) further activates intracellular signaling pathways. 18 Oxidative stress, activation of inflammation, dysfunction of Na+/K+ ATPase activity, and nuclear DNA degradation are involved in the above pathways, leading to neurovascular dysfunction and nerve conduction deficits.14–20,
Disturbance of lipid metabolism is also involved in the development of DPN.21,22 Like hyperglycemia, dyslipidemia is also associated with the release of proinflammatory cytokines and chemokines, causing inflammation-mediated and immune-mediated neurotoxicity.23–25 Other pathophysiological changes also include microvascular dysfunction, 26 endoplasmic reticulum stress, 27 and mitochondrial dysfunction. 28
The pathways involved in metabolic abnormalities are also injurious to Schwann cells, which are essential for the structural and functional integrity of the peripheral nervous system (PNS). 29 As such, Schwannopathy has become an integral factor in the pathogenesis of DPN, and interactions between Schwann cells, axons, and microvessels have been proposed to be intimately associated with DPN29,30 (Figure 1).
Multilevel nervous system involvement
Recent reports have revealed that the pathological changes in DPN patients are not limited to, as per the traditional view, the PNS. A number of MRI studies have implicated the central nervous system (CNS) in the pathogenesis of DPN, including decreased cross-sectional spinal cord area,31,32 decreased numbers of fibers connecting the brainstem with the somatosensory cortex, 33 abnormal thalamic function, and parenchymal atrophy in the primary sensory cortex.34,35 Likewise, the evolution of DPNP has been shown to involve multilevel sensory nervous system lesions. These range from peripheral nerves to the dorsal horn to higher regions of the CNS 36 (Figure 1). Understanding the role of the CNS in the development of DPN or DPNP is highly important, especially given that it is not certain that CNS involvement results from the progression of PNS impairment, from the direct insult of diabetes, or from a combination of both. This question determines, to a large degree, the main level of therapeutic targets.
Different patterns of nerve impairment
DPN has different patterns of nerve impairment, including distal symmetric polyneuropathy, small-fiber-predominant neuropathy, treatment-induced neuropathy [Figure 1(a)], radiculoplexopathy or radiculopathy [Figure 1(b)], mononeuropathy [Figure 1(c)] and autonomic neuropathy [Figure 1(d)]. 37 These patterns of nerve involvement are closely associated with various nerve impairments, including metabolic disturbances, mechanical compression, genetic susceptibility, and microcirculatory dysfunction. 38 Usually, these impairments coexist and interact with each other. For example, peripheral nerves subject to metabolic injury are more vulnerable to local mechanical compression. 38 In addition, interactions between vascular and metabolic factors are synchronized with the pathogenesis of DPN. 39 Thus, in addition to the symptoms, course, risk covariates, and pathological alterations, the various properties of nerve impairment involvement in DPN are another important reason for its heterogeneity.
Thus, as a heterogeneous disease, there is no medication or treatment modality that is effective for all types of DPN and its associated neuropathic pain. The therapeutic regime, which commonly includes lifestyle modification, glycemic monitoring and management, and control of cardiovascular risk factors, has not changed significantly in recent decades. The current treatments for painful DPN focus on symptom relief rather than disease modification due to its elusive mechanisms. As a consequence, the efficacy of these treatments is limited, and the incidence of side effects is high. Further research on the underlying pathological mechanisms and the development of mechanism-based treatment modalities is needed. 36 In this review, the focus was on nerve decompression for treating DPNP with nerve entrapment. In addition, the associated theoretical basis, clinical indications, and progress of basic research were discussed.
Search strategy
We searched PubMed for articles published up to and including March 2024 using the search terms ‘diabetic neuropathy’ or ‘diabetic peripheral neuropathy’ in combination with ‘neuropathic pain’, ‘mechanisms’, ‘diagnosis,’ ‘nerve entrapment’, ‘surgical decompression’, and ‘nerve decompression’. We further searched the reference lists of publications identified in this research strategy and selected those that were judged relevant. We focused mainly on publications in English and preferentially selected those published in recent years but did not exclude widely referenced older articles.
Proposal and application of nerve decompression
According to the classification of DPNs proposed by the Neurodiabetes Consensus (Toronto) Group, DPN is separated into generalized polyneuropathies and focal (e.g. CN III neuropathy and median neuropathy at the wrist from carpal tunnel syndrome), as well as its multifocal varieties (e.g. multiple mononeuropathy, lumbosacral, thoracic, and cervical radiculoplexus neuropathies). 40 Entrapment neuropathies (ENs) are the latter type, and the incidence of EN in the diabetic population is more than one-third. 9 EN should be distinguished from mononeuropathies, which are other focal neuropathies that result from vasculitis and subsequent ischemia or infarction of nerves and usually have an acute onset and self-limiting course. 41 Moreover, EN results from chronic mechanical compression of peripheral nerves, and treatment may include surgery due to the chronic onset and progressive course of the disease.41,42
Although it is generally accepted that surgical decompression of the median nerve at the wrist, for carpal tunnel syndrome, or of the ulnar nerve at the elbow, for cubital tunnel syndrome, is appropriate in the diabetic population, the application of this concept to the lower extremity is not widespread. The surgical procedure of triple-nerve decompression for treating nerve entrapment in lower-extremity DPN was first proposed by Dellon in 1992 based on the following evidence: (1) The susceptibility of diabetic nerves to chronic compression was revealed in animal studies,43,44 (2) sites of nerve compression in lower extremities have been identified in anatomically,45–48 and (3) axons are more susceptible to being compressed distally when proximal mechanical compression is present on the same axon, and vice versa (‘double-crush’ theory).49,50 The first two serve as experimental bases for surgical decompression, and the ‘double-crush’ theory provides a theoretical basis for multiple-site nerve decompression. The Dellon approach to neurolysis for nerves in the lower extremity specifically includes the common peroneal nerve passing through the fibular neck, the superficial peroneal nerve in the leg, the deep peroneal nerve over the dorsum of the foot, and the tibial nerve at the medial ankle. 51 From 2000 through the end of 2009, more than 360 surgeons were trained to perform this procedure in more than 40 states and over 17 countries. 38
In prior clinical studies, peripheral nerve decompression for treating DPN has been reported to be effective at relieving pain and restoring sensation, preventing ulcers and amputations, improving quality of life, and changing the natural history of DPN.52–58 A multicenter clinical study led by Dellon reported that decompression of the tibial nerve in DPN patients can prevent ulceration and amputation and decrease hospitalization after foot infection. 59 Nevertheless, earlier clinical studies on nerve decompression for DPN were mostly retrospective. 60 Due to certain flaws in the research design, such as the nature of the research, selection of the preoperative evaluation indicators, and endpoints, evidence of the effectiveness of nerve decompression is still lacking. Therefore, in 2006, the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology concluded that the evidence of the efficacy of nerve decompression for DPN remained unproven (Level U) due to a lack of randomized controlled trials (RCTs). 61 Hopefully, high-quality and well-designed prospective clinical trials will be conducted by scholars in this province. 62
Over the last decade, three RCTs assessing the efficacy of nerve decompression of lower-extremity nerves for the treatment of painful DPN have been conducted in different countries. The efficacy of nerve decompression for treating painful DPN has been advocated in all of these trials. The earliest study was reported by Macare
In these three RCTs, electrophysiological studies were only performed in two prior trials,64,66 but no marked improvement or change was observed in either of these trials. The incidence of surgical complications was low in these trials. Macare
The historical hesitance to advocate nerve decompression for DPN has been critiqued in view of recent studies using objective, measured outcome protocols. 67 As the number of researchers participating in clinical practice and research on surgical decompression for DPN has increased, more well-designed RCTs will be conducted and published in the near future.68–70
Theoretical evidence, indications, and prognostic factors for nerve decompression
The effectiveness of surgery depends largely on the correct choice of indications. From the perspective of the basis for nerve decompression treatment of DPN and DPNP, nerve entrapment is the direct indication for surgical decompression. In addition to the nerve conduction velocity across the entrapment site, which is considered the gold standard for diagnosis, several potential prognostic predictors of a good outcome following nerve decompression in patients with DPN and DPNP have been reported.
Tinel sign
The Tinel sign was first described by Hoffman from Germany and Tinel from France in 1915 and was defined as a tingling feeling elicited when an injured nerve trunk is percussed at or distal to the lesion site. 71 This sign indicates the level of regeneration or localization of the site of nerve injury. After being proposed and applied in clinical practice, this sign has become associated with the diagnosis of carpal tunnel syndrome and other compression neuropathies. In 2004, Dellon first proposed that a positive Tinel sign is a reliable prognostic indicator for a successful outcome from decompression of the tibial nerve in patients with diabetes with symptomatic neuropathy, as well as in patients with symptomatic idiopathic neuropathy. 72 This conclusion was further confirmed in a multicenter prospective study in which 628 subjects were enrolled, and all the subjects had a positive Tinel sign over the tibial nerve in the medial malleolus. Of these patients, 465 (74%) had a VAS score >5. After decompression of the tibial nerve and its branches, the mean VAS score decreased significantly from 8.5 to 2.0 at 6 months and remained at this level for 3.5 years. During this same time period, plantar sensibility improvement from a loss of protective sensation to recovery of some two-point discrimination was also observed. 73
Nevertheless, as the value of the Tinel sign in assisting in the diagnosis of entrapment has been heavily debated, Datema

A hypothesis diagram illustrating the role of the Tinel sign as a prognostic predictor in nerve decompression for DPN patients. A transformation from positive to negative of the Tinel sign might occur in the course of DPN. At the early stage of diabetic EN, the Tinel sign will be positive as the irritated peripheral nerve still maintains its conduction function. As the compression continues, the conduction of peripheral nerves will be blocked without regeneration. The Tinel sign would be negative at this advanced stage. There might be an intermediate state of transition in which the boundary between positive and negative Tinel signs becomes blurred. Tai Chi Diagram was used here to symbolize the transition between positive and negative Tinel signs. It seems difficult to predict the surgical outcome according to the presence or absence of the Tinel sign at this intermediate state of transition. Nevertheless, a better surgical outcome will undoubtedly be achieved at the early stage.
Distribution and characteristics of pain
In a prior study, the distribution of pain was also reported to play a predictive role in the prognosis of surgical decompression for DPN patients. Patients were divided into two groups according to their description of pain distribution. It was found that while patients in both the focal and diffuse pain groups could benefit from surgical decompression, pain relief, and morphological restoration could be better achieved in the focal pain group during the 2 years of follow-up. 82 As mentioned above, EN, as a subtype of focal DPN, often involves a single nerve in the early stages, with focal clinical manifestations. As the disease progresses, both the proximal and distal segments of the nerve or multiple peripheral nerves become involved. This is known as the ‘double crush’. 83 Furthermore, persistent impairment in the PNS is followed by structural remodeling and reorganization in the CNS, making pain diffuse and chronic.84–86 At this stage, it is difficult for surgical decompression of peripheral nerves to relieve or reverse chronic pain and other sensory disturbances resulting from structural and functional plasticity in the CNS.
Alternatively, the nerve fibers responsible for transmitting focal pain predominantly consist of myelinated fibers, which exhibit heightened sensitivity to mechanical stimuli compared to unmyelinated fibers.87,88 Therefore, one can speculate that the presence of focal pain suggests the potential for mechanical compression. In the case of a central (spinal cord level) disorder, myelinated fibers mediate mechanical allodynia, which has also been suggested to be a valuable prognostic predictor of nerve decompression in DPNP patients. 89 In the EN, the association of myelinated fibers with mechanical allodynia indicates, to some extent, the association of compression injury with mechanical allodynia.
Two-point discrimination
In addition to the Tinel sign and the distribution of pain, two-point discrimination could also be a potential predictive factor for the prognosis of painful DPN after nerve decompression surgery in a retrospective study. 90 Assessment of two-point discrimination has been commonly employed as a parameter of high-order perceptual function since its first proposal by Weber in 1834. 91 As the two-point discrimination threshold is reported to be proportional to the size of the area in the brain representing this region, it can be used as a quantitative measure to detect the loss of nerve function. 92 In addition to central somatosensory function, the threshold of two-point discrimination also depends on peripheral innervation density. 93 Loss or increased sensory phenomena, which may present as altered thresholds of two-point discrimination, are early and prominent manifestations of DPN, especially at compression and entrapment sites.84,90 The complete disappearance of two-point discrimination occurs in the advanced stage of compression since there is no further regeneration.73,81 Thus, better two-point discrimination is thought to be associated with a relatively early stage of nerve compression, and patients in this stage are prone to benefit from surgical decompression.
Basic research on the underlying mechanisms
Establishment of an animal model and the verification of double crushes
Investigations into the mechanistic aspects related to surgical decompression for DPN and DPNP patients began with the establishment of a rat model simulating nerve compression in DPN patients. 49 In this study, the researchers investigated the impact of single- or double-band placement on neural electrophysiological function in a model of sciatic nerve compression. Their findings confirmed that the presence of two simultaneous compression sites, or the addition of a second compression site either proximal or distal to the initial site, resulted in diminished neural function compared to a single compression site. Based on these findings, multiple nerve decompressions, including the common peroneal nerve, deep peroneal nerve, superficial peroneal nerve, and posterior tibial nerve, as well as its plantar branches, have been proposed for treating nerve entrapment in DPN patients. 51
Nerve swelling and new connotation of ‘double crush’
Peripheral nerve entrapment in DPN patients originates from mechanical compression as swollen nerves pass through anatomically constrained channels. In chronic hyperglycemia, neurons take up,

Schematic showing ‘double crush’ formed by nerve swelling inside and the thickened fibro-osseous tunnels outside, as well as nerve glucose metabolism in both euglycemic and hyperglycemic states. Under euglycemic conditions, the size-matching nerve passes through the fibro-osseous tunnels (upper panel). Under hyperglycemic conditions, the hexokinase enzyme becomes fully saturated, and the aldose reductase pathway is activated to convert excess glucose to sorbitol. The accumulation of sorbitol will produce an osmotic pressure gradient across the intracellular and extracellular membranes, causing axonal and nerve trunk swelling by transporting extracellular fluid into the neuron. Focal entrapment neuropathy will be induced when enlarged nerve trunks pass through size-constrained fibro-osseous tunnels (lower panel).
Peripheral sensitization and central reorganization
As the hallmark of neuropathic pain, mechanical allodynia, resulting from the paradoxical conversion from innoxious tactile stimulation to pain sensation under some pathological conditions, commonly occurs in patients with DPN. 99 There are certain patterns of nerve fiber impairment and neuronal activation (at the level of both the dorsal root ganglion and spinal dorsal horn) in diabetic rats with mechanical allodynia. 100 More precisely, impairment of primary myelinated fibers and activation of associated dorsal root ganglion neurons are particular features of diabetic rats with MA. At the level of the spinal dorsal horn, neuronal activation in both the superficial and deep laminae was observed. From the perspective of gate control theory,101,102 neuronal activation in deep laminae was suggested to result from enhanced input of impaired myelinated fibers (peripheral sensitization). However, neuronal activation in superficial laminae was attributed to the reconnection (disinhibition) between low-threshold mechanoreceptor inputs and pain transmission pathways (central reorganization), which is disconnected by inhibitory interneurons under physiological conditions.
It has been acknowledged in prior reports that injury or lesion in the PNS could be followed by multiple pathophysiological changes in the CNS, and both peripheral sensitization and central reorganization were suggested to collaborate for the development of neuropathic pain in DPN patients.99,103 If there is sufficient evidence of peripheral nerve involvement as the initiation factor, it would be rational to establish a treatment modality targeting peripheral nerves for DPN and DPNP. Taking mechanical allodynia in DPN as an example, impairment of myelinated fibers and activation of the associated neurons in the dorsal root ganglion were attributed to initiation factors based on the following findings in a diabetic rat model: (1) myelinated fibers are vulnerable to mechanical compression 104 ; (2) the threshold of mechanical pain is decreased by peripheral nerve compression47,105; and (3) mechanical allodynia is relieved by removing compression. 106 These findings also provide experimental evidence suggesting that mechanical allodynia is a prognostic predictor of DPN and DPNP after nerve decompression. 89
Controversy, dilemma, and future direction
Given the traditional concept that medication is the mainstream treatment for DPN and that the latter is a common complication of diabetes that is prone to infection and gangrene, surgical treatment has been subject to numerous doubts and disputes since it was proposed. Preliminary clinical observations, studies on relevant mechanisms, and outcomes of high-level clinical trials will provide solid evidence to determine whether surgical decompression can effectively treat nerve entrapment in DPN and DPNP patients.
The heterogeneity of DPNP makes the identification of underlying pathological mechanisms particularly important. When a pain symptom becomes a disease per se, symptom-based treatment appears insufficient for a disorder that should be stratified by mechanism. 99 Therefore, in addition to the straightforward and intuitive notion that ‘where there is compression, there should be decompression’, 107 actions need to be taken to further understand the heterogeneity of DPNPs and stratify them based on the various mechanisms, explore the indications for treatment, and maximize therapeutic effectiveness. The application of neuronal electrophysiological biomarkers was recently reported to provide effective support for the exploration of mechanisms and phenotype confirmation. 108 The disappearance of rate-dependent depression, which is the measure of the change in amplitude of the H-wave component of the Hoffmann reflex over consecutive stimulations, can be used as a biomarker to distinguish neuropathic pain caused by spinal disinhibition from that of peripheral origin. 109 This discrimination is of great importance in clinical practice, as prescribing drugs that target the CNS when the pain is being generated in the PNS is likely to fail.
The combined nerve impairment of diffuse lesions and focal compression is almost inevitable in DPN. In most cases multiple mechanisms coexist, and identifying the dominant mechanisms of nerve injury or primary nerve fiber involvement would provide a theoretical basis and clinical evidence for justifying the rationale for emerging therapies as well as improving their effectiveness.
The limitation of this review is that it mainly focuses on the role of nerve decompression in treating diabetic sensory disorders, especially DPNP, without elaborating on the role of nerve decompression in other types of peripheral nerve dysfunction, such as motor and autonomic nerve dysfunction, as well as microvascular dysfunction. However, the role of peripheral nerve decompression in improving autonomic nerve function and relieving microcirculation has been extensively described in a recent review. 110
Conclusion
Based on experimental investigations and clinical evaluations, peripheral nerve decompression provides an effective option for treating DPN with nerve entrapment. Identifying the presence of nerve entrapment among complex pathophysiological mechanisms is the key to successful outcomes. Tinel signs, focal pain, mechanical allodynia, and two-point discrimination were reported to be prognostic factors for good surgical outcomes, and their predictive ability might stem from their association with the early stage of EN.
