Abstract
Diarrhoeal diseases continue to play a major role in the lives of HIV-positive people, impacting negatively on quality of life and causing significant morbidity and mortality. Within the global HIV pandemic, there are distinct geographical variations in the aetiology of chronic diarrhoea and the strategies towards its diagnosis and management. This article aims to highlight the contemporary approach to chronic diarrhoea in HIV-positive people, expand upon the myriad agents responsible for this presentation, and explore present and future therapeutic and management options.
Introduction
Despite improvements in nutrition, sanitation, and hygiene over the past decades, diarrhoeal diseases remain common, representing one of the five leading causes of death worldwide.1,2 Even with improved rates of breastfeeding and access to oral rehydration solution, it was estimated that, in the year 2000, diarrhoeal diseases killed up to 2.5 million people globally.3,4 Chronic diarrhoea is also a major health burden in the HIV-positive population.
The advent of Highly Active Anti-Retroviral Therapy (HAART) has led to substantial improvement in the health and life expectancy of people living with HIV.5,6 This improvement has meant an overall decrease in opportunistic infections in areas of the world where HAART is readily available and, more specifically, less people with advanced HIV immunocompromise (CD4 counts < 200 cells/mm3) developing infective diarrhoea. 7 However, diarrhoeal illness continues to represent a significant co-morbidity within the HIV-positive population. The inequality found within the geographical and socio-economical distribution of HIV infection itself—two-thirds of all deaths attributable to HIV/AIDS occur in sub-Saharan Africa 8 —can also be found within the burden of diarrhoeal disease in HIV-positive people worldwide. In high-resource settings, 40% of people with HIV reported one episode of diarrhoea in the previous month. 9 Approximately 90% of HIV-positive people in low-resource settings experience diarrhoea lasting over one week at some point during their illness.10–13 AIDS-associated diarrhoea still represents a leading cause of death in HIV-positive adults and this is most notable in low-resource settings. 14 Chronic diarrhoea is in itself an AIDS-defining illness and has long been recognised in sub-Saharan Africa—where AIDS was first termed “slim disease”—to be associated with HIV infection. 15
The aetiology of HIV-related chronic diarrhoea is wide-ranging. In terms of infectious agents, a plethora of causative organisms must be considered from viral to bacterial to fungal to protozoal—or even as a direct effect of the virus itself, ‘HIV enteropathy’. 16 Non-infectious causes are also expansive: an adverse effect of prophylactic antibiotics, antivirals or antifungals; AIDS-associated malignancy; vitamin deficiencies; adverse effects of HAART; and many other causes that go beyond the scope of this article.
Aside from the obvious physical health burden that chronic diarrhoea poses, one must also not forget the psychosocial and emotional impact upon patients; chronic diarrhoea has been found to be an independent predictor of decreased quality of life in HIV-positive individuals17,18 especially amongst middle-aged and older HIV-infected adults. 19 Moreover, the additional economic cost in loss of days of work and productivity may be high. The negative effects of chronic diarrhoea in the HIV-positive population are notably multi-layered and far-reaching.
This review is not exhaustive and will not cover acute diarrhoeal illnesses, the paediatric population or the HIV-negative population. Nor will the article focus in great depth on clinical presentation and diagnostics. It will, however, illustrate the geographical variation in chronic diarrhoea in HIV-positive people worldwide, expand on the infectious and non-infectious aetiology of HIV-related chronic diarrhoea, examine the treatment options available, explore the relationship of chronic diarrhoea with certain HAART medications, and look toward future treatment options. Furthermore, as there is a distinct lack of evidence as to use of antimicrobial and antimotility medication in the context of chronic diarrhoea in HIV, the article will also highlight two recent and relevant Cochrane reviews in this area.10,20
The article is intended for a wide audience including general and specialist physicians, HIV and non-HIV pharmacists, and allied healthcare professionals.
Definition and Presentation
The definition of chronic diarrhoea, in itself, has been a subject of debate, with many varying definitions to be found in the literature. For the purposes of this article, the definition will be in line with that suggested by the American Gastroenterological Association—a decrease in faecal consistency lasting more than four weeks. 21 It must be noted, however, that there is no standard definition of AIDS-associated diarrhoea, with HIV-positive patients often citing greater frequency of loose stools (compared to HIV-negative individuals) as their daily norm. 22 The chronology of 4-weeks to establish “chronic diarrhoea” may be excessive in the HIV-setting (in the same way as definition of pyrexia of unknown origin in the HIV-patient may be reached earlier than in the immunocompetent patient) and other authors have suggested 2 weeks as a more suitable duration for definition. 20 This polemic has created issues in interpretation of studies concerning HIV- or AIDS-related diarrhoea as parameters used to define the diarrhoea were not validated in this population; clearly, for future investigations, an accepted definition and validated tool to measure diarrhoea in the HIV-positive population would be beneficial. 23
Whilst it will not be covered in great depth within the confines of this article, it is clear that, for the clinician, the clinical history gleaned from a patient is essential in streamlining investigations to establish a causative mechanism for their presentation. For example, in many cases, a few focussed questions are likely to elicit whether the diarrhoea is small bowel (dysregulation of secretory and nutrient-absorbing properties that leads to watery, high volume loss with dehydration and malabsorption) or large bowel (frequent, small volume stools, often painful). This distinction will likely aid further investigation: in stool examination of patients with documented history of small bowel involvement, small bowel organisms are more likely to be isolated. 24 In addition, symptoms of tenesmus or dyschezia may point towards anorectal involvement and possible sexually transmitted infection or malignancy. However, it is not only the diarrhoeal history that the patient gives that may focus the clinician's investigations. Thorough HIV history is essential, covering date of diagnosis, co-infections, and medications (including previous HAART). Sexual history is of relevance especially in those who have anal intercourse. Travel and occupational history that may point to previously overlooked exposures (ie, Cryptococcus in the pigeon-fancier) should be included and, of course, family history—which may reveal an established history of inflammatory bowel disease (IBD). Although clearly a patient's HIV serostatus will influence the likelihood of certain causative aetiologies, non-HIV related causes must always be excluded. Despite good history-taking and clinical examination, it has been noted that clinical findings do not adequately predict positive organism identification during subsequent microbiological investigation. 25
HIV Immunodeficiency and Chronic Diarrhoea
While the incidence of infectious diarrhoea has declined in high-resource settings since the advent of HAART, the incidence of diarrhoea itself has remained relatively constant. 26 The clinician must be alert to non-infectious causes of chronic diarrhoea: such as Kaposi's sarcoma (KS) or bowel carcinoma in the ageing HIV population. The infectious aetiologies encountered are closely correlated with the individual's degree of immunodeficiency as expressed by CD4 cell count (Fig. 1). The HIV virus depletes CD4+ T cells, leading to varying degrees of immunosuppression and leaving the individual susceptible to myriad infective organisms, but especially those in which the immune response is T-cell mediated (ie, fungal, mycobacterial, protozoal and viral infections). Higher CD4 counts and other factors, such as use of cotrimoxazole prophylaxis—used to prevent Pneumocystis jirovecii pneumonia but also protective against certain bacterial and protozoal diarrhoeal agents—have been shown to be protective against both acute and chronic diarrhoea. 27 Diarrhoea in HIV-positive individuals with preserved CD4 counts is more likely to be pathogen-free on culture than in those encountering chronic diarrhoea at low CD4 counts. People with advanced immunosuppression are more likely to be infected with organisms such as Cryptosporidium species and other parasites. 25 In addition to infection with organisms specific to degree of immunosuppression, it is imperative to be aware that other organisms, while not being specific to CD4 count, may present more severely, persistently, 28 and chronically in the immunodeficient HIV-positive individual: entero-aggregative E. coli being one example. 29 Indeed, the disease-to-infection ratio (ie, those with symptomatic infection versus asymptomatic carriers) appears to be raised in all stages of HIV infection. 30
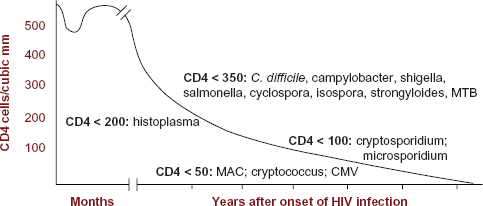
Natural history of diarrhoeal infections in HIV disease with regards to CD4 count.
Diarrhoeal infections do not only exert direct effects on the individual. As opposed to acute diarrhoeal illness, the chronic form may contribute to malnutrition which is, in itself, a strong predictor of morbidity and mortality especially in those with small bowel mucosal damage 31 and in the paediatric population. 32
Uncontrolled HIV infection can be viewed as a chronic inflammatory state that has negative effects throughout multiple body systems. It has been shown to contribute directly to increased risk of ischaemic heart disease and worsening lung functions over time independently of opportunistic infections or adverse effects of HAART. 33 The gastrointestinal system is no exception, with HIV thought to be implicated through direct infection of enterocytes and gut-associated lymphoid tissue (GALT)—an early site of infection and a reservoir of HIV replication—with subsequent dysregulated cytokine production.31,34–36
Geographic and Socioeconomic Variations
There still exists a wide gulf between the health of populations living in low- and high-resource settings. Of course, geographical variations between the causative diarrhoeal organisms are marked, with levels of parasitosis high in regions such as Sub-Saharan Africa, 37 South and East Asia 38 or, with a potential increase in cases of HIV and visceral leishmaniasis, 39 South America. Within certain geographical areas, there are variations in diarrhoeal incidence depending on the season ie, the effect of monsoon seasons on water quality and sanitation in affected countries.40,41 Nevertheless, it is not merely the spectrum of causative aetiology of diarrhoea found in a region that predicts the levels of chronic diarrhoea: socioeconomic and population factors are intrinsically linked with the prevalence of chronic diarrhoea.
Access to medications and, more importantly, adequate healthcare facilities may be scarce for people in a low-resource setting, especially those living rurally. HAART may not be scaled-up to meet population needs and therefore diarrhoeal disease associated with the subsequent immunodeficiency in HIV-positive people may be more prevalent. 42 Even where medications such as HAART are available, their price may be prohibitive. These issues encountered in low-resource settings also extend to the investigation and management of chronic diarrhoeal disease. Clearly, the greater the availability of investigative tools, the better the diagnostic work-up, and the more likely an identifiable pathogen will be established in a patient with chronic diarrhoea. 43 Indeed, in one study, nearly half the patients previously termed to have “idiopathic diarrhoea” had causative organisms identified via colonoscopy and terminal ileal biopsies. 44 Extensive work-up and focussed treatment strategies may be neither achievable nor appropriate in resource-limited settings. In these areas, empirical treatment involving vigorous fluid-resuscitation, nutritional support, and rational antibiotic use may be preferable.
Infectious Aetiologies
The infectious causes of chronic diarrhoea in the HIV-positive population are wide and varied: bacterial infection such as shigellosis and campylobacter occur more frequently than in the negative population45,46; viral infections—which may be exclusive to HIV-positive or immunosuppressed people such as cytomegalovirus (CMV) and HIV-enteropathy, 31 - or not exclusive but presenting more persistently or severely in immunodeficient individuals such as rotavirus, caliciviruses and adenovirus 47 ; protozoal infections such as cryptosporidium and isospora; and fungal and mycobacterial infections. Overall, infections are the most commonly implicated aetiology in chronic diarrhoea associated with HIV/AIDS 25 and can be due to multiple pathogens. 48 Methodical investigation and logical management is essential. This section of the review will aim to describe some of the more important and frequently found infectious aetiologies and their treatment regimens.
Viruses
Previous studies into causative aetiology of infective diarrhoea appeared to reveal an association with acute or relapsing diarrhoea and presence of rotaviruses and adenoviruses in the stool. 49 Other related studies in both adults and children went on to show that these viruses were also found, albeit at lower frequency, in the stool of HIV patients without diarrhoea.47,50 It was therefore postulated that these organisms were not always directly associated with the chronic diarrhoea and may be incidental findings, opening up the possibility of other—as yet undiscovered—organisms causing the clinical picture. Subsequent work revealed the effect of the HIV virus itself on the GI tract which will be covered in a later section. For the main part, rotaviruses, astroviruses, adenoviruses, picobirnaviruses, and caliciviruses cause acute diarrhoeal illness in HIV-positive people and rates vary according to region, nutritional and socioeconomic status. 51 Apart from in those with genetic immunodeficiency (ie, severe combined immunodeficiency disease) or on immunosuppressive agents, chronic diarrhoea caused by caliciviruses was not previously reported. However, we recently reported the first known case of chronic norovirus (a member of the caliciviridae family) diarrhoea in a grossly immunodeficient HIV-positive patient which responded favourably to newer HAART regimens. 52 There may be an argument that there are certain organisms leading to chronic diarrhoea that are being underdiagnosed. What is clear is that, aside from improving a patient's immune status with appropriate HAART, encouraging good hygiene practice, and giving supportive fluids and nutritional adjuncts, there is no specific treatment against the viruses themselves. The sole exception to this being CMV infection.
The major viral pathogen causing chronic diarrhoea in the HIV-positive individual, mainly with advanced immunosuppression (below CD4 counts of 50 cells/ml), 53 is CMV. Prior to HAART it affected up to 5% of AIDS patients 54 but even in modern day medicine it remains a diagnostic and management challenge. Of all CMV's multi-system—most notably retinal—manifestations, GI infection accounts for up to 15%. 55 CMV can affect the gastrointestinal tract at any point from mouth to anus but CMV oesophagitis with or without ulceration and, more importantly, colonic involvement are notable sites. Presentations can include oesophagitis, gastritis, and enteritis. Involvement of the large bowel can lead to abdominal pain, tenesemus, and per-rectal bleeding or bloody stools, with CMV colitis being found, in some series, in up to 16% of HIV patients. 56 Ideally, diagnosis will be arrived at by excluding other possible infections and marrying appropriate clinical symptomatology with macroscopic tissue destruction (ie, CMV ulcers) and microscopic visualisation of viral inclusion bodies, but this is not always possible. 57 CMV detected in peripheral blood by polymerase chain reaction (PCR) is not uncommon in advanced HIV disease and does not always equate to end-organ damage.
First line treatment of CMV gastrointestinal disease is an initial induction course of intravenous ganciclovir for 2–4 weeks depending on patient response.53,57–60 In the absence of renal impairment, Ganciclovir IV is dosed at 5 mg/kg twice daily. Ganciclovir is very poory absorbed enterally. Although a valine ester of ganciclovir (valganciclovir) has been developed 61 to improve oral bioavailability of ganciclovir and licensed for CMV retinitis, caution must be exercised in the context of gastrointestinal disease as malabsorption may still occur, particularly if the ileum is affected. Furthermore, there is not enough evidence to recommend using valganciclovir as an initial therapy for gastrointestinal disease or the appropriate juncture to switch to it in the improving patient. However, practically employing valganciclovir allows the clinician to switch from IV to PO therapy, usually when symptoms are alleviated, enteral absorption can be felt reliable and/or the CMV PCR becomes undetectable. Oral therapy also facilitates the discharge of patients from inpatient care if besides requiring intravenous therapy, the patient is fit.
During initiation treatment with ganciclovir, patients’ full blood count and renal function should be monitored at least three times a week. Anaemia and leukopaenia are the most likely blood dyscrasias to occur, though thrombocytopaenia is also possible. Renal function should be monitored as nephrotoxicity has been reported and a dose reduction of ganciclovir for reduced glomerular filtration rate.
Second line treatment is foscarnet. Foscarnet is associated with higher levels of electrolyte disturbances and nephrotoxicity. Nephrotoxic side-effects can be reduced by pre-hydration. Due to these side effects, and the comparatively more complicated method of administration, patients and treatment centres may prefer to use ganciclovir.
Cidofovir is an alternative agent but is not routinely recommended in guidelines as it causes potential irreversible nephrotoxicity. Due to its long half life, induction therapy is a weekly infusion reducing to every two weeks for maintenance therapy. This characteristic is useful for patients in whom valganciclovir is inappropriate and for whom daily attendance for IV therapy is inconvenient. Furthermore, its nephrotoxicity can be ameliorated by the use of adequate prehydration, administration of probenecid and avoidance of concurrent nephrotoxic drugs. 62
Initiation of HAART leads to a substantial decline in mortality after CMV diagnosis (up to one-third) but the clinician must be vigilant to the possibility of immune reactivation syndrome (IRS) with a corresponding flare in symptomatology. 55
Sexually transmitted infections including HSV may also be found, with HSV-2 being more closely associated with anogenital lesions than HSV-1. In the absence of HIV therapy, primary genital herpes can be prolonged with the risk of severe, multifocal and coalescing mucocutaneous anogenital lesions. Treatment includes the use of high dose and extended course aciclovir, valaciclovir or famciclovir. Severe lesions may require the use of intravenous acyclovir. 63 Dependent upon the frequency of recurrent episodes of herpes, episodic or continuous suppressive therapy may be indicated.
Bacteria
It is not only the spectrum of bacterial organisms that differ between the HIV-immunodeficient population and the immunocompetent population but also their frequency, severity and persistence. Escherichia coli, Salmonella sp., Campylobacter and Shigella are amongst the most prevalent in both populations. 64
Enteroaggregative E. Coli (EAEC) has been implicated as a cause of persistent diarrhoea with higher severity and likelihood of isolation in the HIV-positive than HIV-negative population. 65 EAEC has also been found to be a significant pathogen in children from low-resource settings in Sub-saharan Africa and East Asia.66,67 Shigella and Salmonella species are also found at a much higher frequency than in the HIV-negative population and are more likely to cause symptoms of gastroenteritis.25,37 Of note, while mainly causing acute diarrhoea in immunocompetent individuals, non-typhoidal Salmonella infection (ie, Salmonella enteritidis, Salmonella typhimurium) may cause severe, relapsing, invasive infection with colitis, bacteraemia and high mortality rates in those with advanced HIV disease and impaired nutrition.68,69
One of the most common causes of “traveller's diarrhoea”, Campylobacter jejuni, whilst being self-limiting in the immunocompetent host can cause watery diarrhoea that persists for weeks in immunocompromised HIV patients. This is also true of non-jejuni species. 70 Prevalence of infection may be decreasing due to increase coverage of Pneumocystis jirovecii prophylaxis with co-trimoxazole that is also active against C. Jejuni. This may cause problems with resistance, however, which is explored in more detail below.
A further pathogen that has received much attention over the past decade is Clostridium difficile. As with the immunocompetent population, this pathogen is associated with previous antibiotic use in the HIV-positive patient. 71 Antibiotics such as clindamycin, cephalosporins, fluoroquinolones, cotrimoxazole and co-amoxiclav are particularly associated with subsequent C. difficile infections and their use must be weighed against this complication, especially in the elderly or immunocompromised patient. C. difficile is an enterotoxin producer that can lead to a variety of clinical manifestations ranging from asymptomatic to pseudomembranous colitis and, in some studies, has ranked as one of the most common bacterial gastrointestinal infections in diarrhoeal HIV-patients. 72 We favour early presumptive treatment of suspected C.difficile toxin-induced diarrhoea because this reduces length of stay in hospitalised patients. A high neutrophil count often points to the diagnosis in the immunocompetent patient, but this is usually absent in advanced HIV disease (McAllister & Blanchard, manuscript in preparation). Fortunately, antibiotic resistance to the two mainstay treatments for C. difficile diarrhoea—metronidazole and vancomycin—is, as yet, uncommon. However, in the case of other bacterial diarrhoeal pathogens, antibiotic resistance is uniformly high relating to widespread inadequate antibiotic control policies, poor prescribing practices, and easy availability of these antibiotics over-the-counter. High rates of resistance have been found in Campylobacter species to cotrimoxazole, nalidixic acid, and ciprofloxacin (ie, fluoroquinolones), especially in low-resource settings. 67 In fact, resistance of Salmonella, Shigella and E. coli to co-trimoxazole has now become widespread.25,29,67,73
E. coli diarrhoeal infections should not be treated with antibiotics, which can exacerbate the condition, but with supportive treatment and hydration. The same may be said for mild to moderate C. jejuni diarrhoea although treatment with a macrolide antibiotic can decrease symptom and organism-shedding duration. 68 Azithromycin has an advantage in patients treated with antiretrovirals as it does not inhibit CYP450 3A4 to the extent of erythromycin and clarithromycin, thereby providing a way to avoid undesirable drug interactions whilst providing effective treatment. Ciprofloxacin should be used with caution due to resistance rates. Salmonellosis, again, may not always require antibiotic treatment but if bacteraemic intravenous ciprofloxacin 400 mg twice daily followed by oral ciprofloxacin 500 mg twice daily or ceftriaxone 2 g daily followed by azithromycin 500 mg daily may be used. Moderate disease requiring antibiotics but not intravenous therapy may be treated for the duration with ciprofloxacin or azithromycin. Caution must be employed when using fluoroquinolones as strains with reduced susceptibility are prevalent in Asia. 74 Ciprofloxacin can be used in severe cases of shigellosis (often caused by S. dystenteriae species). 75
Mycobacteria
Both Mycobacterium tuberculosis and Mycobacterium avium intracellulare complex (MAC) can cause gastrointestinal disease but the former is a rare, often ileocaecum-affecting, presentation. This article will focus on MAC. MAC is an ubiquitous environmental organism and, in the HIV-positive population, mainly causes disease in those with advanced immunodeficiency with CD4 counts under 50 cells/mm3. Disseminated MAC infection can lead to chronic fever, weight loss, abdominal pain and non-bloody diarrhoea. Recognised guidelines now recommend prophylaxis of MAC in patients with CD4 counts below 50 cells/mm3 commonly with azithromycin or clarithromycin 57 although rifabutin has been shown to reduce incidence of disease (albeit to a lesser extent) when used prophylactically. 76 Clearly, another approach to avoidance of this illness is by initiation of HAART prior to such a depleted CD4 cell count if possible; HAART and chemoprophylaxis have led to a decreased incidence and prevalence of disseminated MAC disease. 72 Overall, despite a distinct risk of IRIS, starting HAART in patients with low CD4 counts and MAC disease is lifesaving.
Recommended first-line treatment of confirmed MAC disease is a macrolide (azithromycin preferred over clarithromycin due to greater experience with this drug) and ethambutol (which is associated with lower rates of relapse) with duration of treatment being sometimes lifelong and dependent on degree of immune-restoration 57 ; addition of rifabutin may be considered in patients with high mycobacterial burden. 77 Interactions between medications must be considered in the case of the macrolides which are enzyme-inhibitors and rifampicin (and to a lesser extent rifabutin) which has a degree of enzyme-induction (for further information see Liverpool University's useful website www.hiv-druginteractions.org).
Fungi
The most common fungi to cause chronic diarrhoeal symptoms are Candida species, Cryptococcus, Microsporidia (previously classified as protozoa but recently reclassified as fungi) 78 and, to a lesser extent, Histoplasma. Fungal infection is rare in patients with preserved CD4 counts.
Candidal infection can affect the GI tract throughout its entirety but is most commonly encountered on clinical examination of the oropharynx, sometimes causing dysphagia, in particular with oesophageal involvement. Candida species (including a higher proportion of non-albicans species than in immunocompetent individuals) may be isolated from the GI tract and, if no other organisms are encountered, may be the sole organism responsible for diarrhoeal disease. 79 The diarrhoeal disease is non-bloody and watery and candidal plaques and ulceration may be found on endoscopy. Risk factors—apart from HIV-related immunosuppression—include prolonged antibiotic use, other reasons for immunosuppression and malnutrition. 80 Severe candidal infection may precipitate a candidaemia. There is more experience in treatment of oesophageal disease but oral fluconazole, ketoconazole or itraconazole are often sufficient for treatment of lower GI disease. 81 Intravenous Amphotericin B may be employed if there is azole resistance (non-albicans species exhibit higher rates) but can lead to renal impairment and biochemical disturbance. Although these effects can be ameliorated by using lipid based formulations (eg lipid complex or liposomal), cost may prove a limiting factor to availability especially in low-resource settings. The clinician and pharmacist must also beware changes in dosage between these preparations.
Although there are over 1000 species of Microsporidia, infection in the immunocompromised HIV-positive patient is comprised in the main part of two: Enterocytozoon bieneusi and Encephalitozoon species. Unfortunately, rates seen in low-resource settings are comparable with that seen in the pre-HAART era in high-resource settings. 82 E. bieneusi causes chronic watery diarrhoea of varying severity with malnutrition and weight loss whereas Encephalitozoon species can, in addition, disseminate to involve other organs.
Treatment against E. bieneusi has been shown to be successful with the anti-angiogenic antibiotic fumagillin although bone marrow toxicity causing blood dyscrasias are a recognised complication limiting its license. Although used in veterinary medicine, fumagillin is for the most part unavailable though in some instances it can be sourced on special request from Novartis Pharmaceuticals, France. Albendazole is used to treat Encephalitozoon species. Other therapies include nitazoxanide, metronidazole and octreotide but robust evidence for these therapies is limited. 83 Again, HAART therapy is also essential to improve immune function and clearance of the organism but, in the case of E. intestinalis, protease inhibitors may have a direct effect on the organism. 84 With the lack of effective treatments, the mainstay of managing microsporidial disease is increasing the patient's CD4 count with ART.
Protozoa and Parasites
While a significant problem, clearly not all stool samples need to be sent for examination for ova and parasites. A careful history that would point the clinician to possible parasitic infection and warrant sending the relevant stool sample would include: persistent diarrhoea; relevant travel history; occupational exposure (ie, day-care centres); relevant local outbreak; and sexual history (higher risk in men who have sex with men).
Cryptosporidium infection remains a troublesome infection to the immunocompromised HIV-population in both high-resource and, to a greater extent, low-resource settings. 85 The differences in prevalence may, in part, be explained by drinking water quality regulations leading to eradication of cryptosporidial oocysts previously present in the water. 86 Cryptosporidia are protozoa that are found worldwide and manifest as chronic, voluminous, watery diarrhoea (related to villous atrophy and increased intestinal permeability) in 90% of patients affected. 87 The diarrhoea can be relapsing—thought to be related to autoinfection—and can lead to dehydration, abdominal cramping and weight loss. It is more common in those with CD4 counts below 100 cells/mm3 and can present as fulminant in those with CD4 < 50 cells/mm3. 88 There are a number of agents with variable activity against Cryptosporidium infection: paramomycin, an aminoglycoside, acts by binding the parasite at the apical membrane in the gut epithelium but has been shown to be only partially effective and can lead to relapses on discontinuation89,90; azithromycin is the preferred macrolide against cryptosporidium causing varied clinical improvement but incomplete eradication of the parasite 91 ; nitazoxanide, a thiazolide with a license in the US, has activity against both protozoa but also helminthic infection with good clinical effect and decreased duration and burden of cryptosporidial shedding.89,92 Albendazole may be used but is associated with unacceptably high relapse in half the patients treated. 93 Similar to microsporidial infections, controlling their HIV and allowing immune reconstitution is the most effective treatment. Other novel and future therapies for cryptosporidium will be covered in a further section.
Isospora belli is the other notable protozoan in the immunosuppressed HIV-patient, causing chronic diarrhoea with symptoms similar to those found in cryptosporidiosis, again most notably in low-resource settings. 94 Current first-line treatment is co-trimoxazole but ciprofloxacin, or pyrimethamine (with folinic acid), are suitable alternatives. After resolution of symptoms a period of secondary prophylaxis is recommended. 95
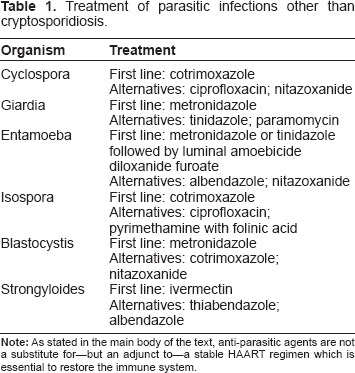
Treatment of other parasitic infections such as Cyclospora, Giardia, Entamoeba, Blastocystis, and Strongyloides can be found in Table 1.
Treatment of parasitic infections other than cryptosporidiosis.
HIV Enteropathy
Also known as “idiopathic, pathogen-negative diarrhoea”, HIV enteropathy represents its own diagnostic and management challenges and has been found in up to 60% of HIV patients with severe, refractory diarrhoea. 96 Since Kotler et al first identified decreases in intestinal IgA plasma cells of AIDS patients in the 1980s, it was thought that HIV itself may be directly infecting enterocytes. 16 Presence of other viruses in the GI tract of AIDS patients made establishing HIV as a sole causative diarrhoeal agent difficult. However, further studies went on to show that HIV directly infects enterocytes and lamina propria cells 97 ; impairs cellular transport mechanisms 98 ; and destroys gut-associated lymphoid tissue (GALT) in the early stages of infection and replication. 35 It has also been discovered that HIV's transactivating factor protein (Tat) interferes with the enterocytes own Tat protein leading to colonic mucosal ion secretion and, subsequently, diarrhoea. 99
Treatment for HIV enteropathy is HAART and supportive therapies. However, as the gastrointestinal tract and GALT have been recognised as a sanctuary site for HIV replication, this compartmentalisation of HIV infection means that, if a clinician wishes to treat HIV enteropathy, the HAART regimen chosen must achieve adequate levels in the GALT.
Conventionally CD4 count is monitored in peripheral blood because this is the most accessible compartment. In fact CD4 depletion occurs earlier, is more profound and reconstitutes more slowly following HAART in GALT. The main measures likely to alleviate this problem are earlier commencement of HAART and the use of antiretroviral drugs that are concentrated in gut tissues. Although nearly all antiretrovirals are administered orally and would be expected to achieve good gut penetration, in fact cellular levels of antiretrovirals in the gut may well be depleted by the many efflux transporters in gut epithelium. 100 To our knowledge the only antiretroviral concentrated in gut tissue is the CCR5 blocking drug maraviroc, but it is only licensed for use relatively late in HIV disease. This is most unfortunate because retroviral coreceptor usage is more likely to switch from CCR5 to CXCR4 as HIV disease progresses. Interestingly, maraviroc also boosts levels of circulating CD4 cells, although it is not clear if this represents an absolute increase in numbers due to a reduction in inflammation and immune activation, or just redistribution between compartments. 101
Diarrhoea as a Side-Effect of HAART or other Medications Related to HIV
Gastrointestinal symptoms—most commonly nausea and diarrhoea—have long been recognised as side effects of anti-retroviral therapy. These symptoms are almost universal at the start of HAART therapy but are often transient. When the clinician is presented with an HIV patient established on treatment, whose sole complaint is diarrhoea, HAART must be considered as a causative agent.
The decrease in opportunistic infections as a cause for chronic diarrhoea and the increase in HAART use have meant that drug-related toxicity is becoming increasingly recognised. 102 Older agents such as the nucleoside analogues (including didanosine, lamivudine, stavudine, zidovudine) whilst causing a relatively low frequency of diarrhoea in their own right were also uncommonly associated with nausea and abdominal pain related to mitochondrial toxicity with lactic acidosis. 103 Other agents, conversely, appear to improve diarrhoeal and other GI symptoms in patients with low CD4 counts, such as the fusion inhibitor enfuvirtide (T20) as shown in the TORO trial 104 and etravirine in the DUET trial. 105 Moreover, certain new drugs have not shown an association with increased GI side-effects: the integrase inhibitor raltegravir was well tolerated with respect to GI symptoms in naive patients 106 as was the CCR5-inhibitor, maraviroc, in both naive (MERIT trial) 107 and experienced patients (MOTIVATE trial 108 ).
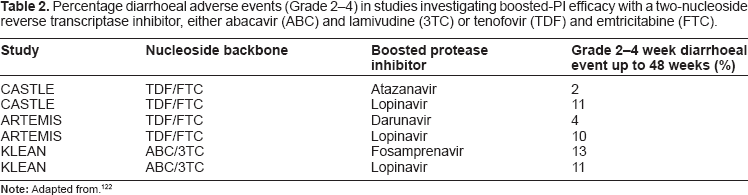
It is apparent that one group, the protease inhibitors (PIs), are associated with diarrhoea and GI upset to a greater degree than other HAART medication groups. This is clearly an issue in itself as it negatively affects a patient's quality of life and may reduce compliance with HAART. The mechanisms by which PIs are thought to exert their GI effects are due to inflammation of the gut. This is caused by the drug inducing stress on endoplasmic reticulum, which leads to disruption of the barrier integrity in intestinal epithelial cells, subsequent cellular leakage, and an overall secretory state with increased fluid loss. 109 In order to increase drug bioavailability and activity, most PIs are “boosted” by a drug in the same class called ritonavir. Ritonavir itself was shown to cause diarrhoea at high doses (600 mg twice daily) in the initial mono-therapy trials (it was also shown to have poor anti-HIV activity). 110 When given to “boost”, ritonavir is given at low dose (100 mg once or twice daily) with another PI and it is difficult to establish diarrhoeal rates at this dose as it is often inextricable from that caused by the other PI. There are variations between the severity of diarrhoeal reactions between the other PIs with boosted lopinavir and fosamprenavir showing higher rates of moderate to severe diarrhoea than boosted atazanavir or darunavir (see Table 2). The ARTEMIS trial showed boosted darunavir to be non-inferior to boosted lopinavir in action but also with preferable rates of gastrointestinal side effects including diarrhoea 111 ; similar improved rates of diarrhoea were found with the use of boosted darunavir instead of boosted lopinavir in the CASTLE study. 112
Percentage diarrhoeal adverse events (Grade 2–4) in studies investigating boosted-PI efficacy with a two-nucleoside reverse transcriptase inhibitor, either abacavir (ABC) and lamivudine (3TC) or tenofovir (TDF) and emtricitabine (FTC).
Aside from substitution of a PI with one known to have fewer GI side effects or switching to another drug class, the evidence for management of PI-associated diarrhoea is limited and anecdotal. Once other causes are ruled out, empirical antimotility agents may be used. This will be expanded on in a later section.
Other Non-Infectious Aetiology
The wide differential of chronic diarrhoea in the HIV-positive patient is summarised in Table 3. A full explanation of each goes beyond the realm of this article. It is important for the clinician or other healthcare professional to note, however, that as the HIV population lives longer, it too will be affected by diseases of ageing. Some of these diseases may cause chronic diarrhoea, such as bowel carcinoma. In a similar vein, the issue of polypharmacy will become more relevant in the ageing HIV population who are likely to develop hypercholesterolaemia (especially an issue with certain protease inhibitors) and type 2 diabetes; both the treatments (for example statins and metformin) of these conditions and symptoms related to the conditions themselves (autonomic neuropathy affecting the digestive tract or ischaemia of the bowel) can cause chronic diarrhoea.
Differential of infective chronic diarrhoea in HIV-positive people.

Cochrane Reviews of Antimicrobial Therapy and Empirical Antidiarrhoeal Therapy
The Cochrane collaboration currently has an intervention protocol out for review concerning antimicrobials for chronic AIDS-associated diarrhoea in adults. 20 It cites the importance of its review as being that “90% of people living with HIV-infected people living in low-resource settings experience AIDS-associated diarrhoea” and that “evidence for specific antimicrobial use remains inconclusive”. In particular, it will try to answer the “controversy” of the optimum treatment for Cryptosporidial infection and also Isospora. The results of this review may help to guide clinicians in the future. It is certain that the need for good evidence for empirical antimicrobial therapy is essential in low-resource settings where diagnostic facilities may be limited; patients with suspected parasite or protozoal infection may be helped by medications such as albendazole or metronidazole in the absence of positive culture. 87
A completed review by the Cochrane team focuses on the use of antimotility agents in people with HIV/AIDS. 10 In the review, the authors argue that there may be a need for empirical antidiarrhoeal therapy in half the non-pathogenic or idiopathic cases of diarrhoea or those resulting from antiretroviral therapy. It looked at studies into both antimotility drugs such as opioids (loperamide; diphenoxylate; and codeine) as well as adsorbents (bismuth; kaolin/pectin; attapulgite). It found little robust evidence to support their use in the HIV-positive population, citing that much of the available evidence and WHO guidance on the use of antimotility agents was based on expert opinion and studies in HIV-negative people. The only study to fully meet their criteria showed that attapulgite was no better than placebo in controlling diarrhoea in HIV/AIDS patients. 113 This review did not consider the importance of oral rehydration solution (ORS) which, with appropriate use in the home or peripheral health centres, could dramatically reduce both hospital admission and subsequent morbidity and mortality. 114 It is currently estimated that global ORS coverage rates are less than 50%.
New and Novel Therapeutic Agents
Zinc, Vitamin a and micronutrients
Zinc supplementation is recommended by WHO in HIV-positive children with acute diarrhoea. The mechanism by which this occurs is thought to relate to the ability of zinc to limit the action of Tat-induced secretion of fluid. 115 Zinc, vitamin A and micronutrient deficiency have been found in HIV-infected individuals with chronic diarrhoea, especially children in low-resource settings. 116 Despite the advice of organisations such as WHO and American Gastroenterological Association related to micronutrient supplementation in children, a randomised-controlled trial 117 and Cochrane review found no conclusive evidence for such supplementation in HIV-infected adults, and the issue remains polemic. 118
Cryptosporidium
There is ongoing work into new thiazolide compounds (which substitute the nitrogen containing part of the compound for bromine or chlorine) which look promising, having been found to have excellent Cryptosporidium inhibition in vitro; in vivo trials are ongoing. 91 Other compounds that show potential are: isoflavone derivatives which may also act on biliary cryptosporidiosis; bisphosphonates which inhibit the growth of Cryptosporidium; and sinefungin that blocks Cryptosporidium's own polyamine synthesis.
Other Miscellaneous Treatments
Curcumin
There has been recent interest into a biologically active compound of the spice “turmeric” called curcumin. It has an anti-inflammatory effect through inhibition of a number of inflammatory cytokines including TNF-alpha and other enzymes including Cox-2. It has been shown to be effective in other gastrointestinal conditions such as inflammatory bowel disease 119 and irritable bowel syndrome. 120 A small study in HIV-positive individuals given curcumin 1000–3000 mg per day in three divided doses showed improvement in stool frequency, weight gain and a favourable side-effect profile with regard to bloating and abdominal pain. 121 Further work is required into this compound.
Others
Other therapies such as green bananas, crofelemer, oat bran, psyllium, and probiotic agents have all been speculated as empirical therapies for chronic diarrhoea in both HIV-negative and positive patients but, as yet, there is insufficient evidence to recommend any of these specific treatments.
Vaccines
Although work has been done into vaccines for infectious diarrhoea in the HIV-negative population, there is to date no good evidence for use of vaccines to prevent any form of HIV-related diarrhoea. This—along with the ongoing quest for a safe, effective, and affordable vaccine for HIV itself—is an important area for future research.
Conclusion
Chronic diarrhoea in HIV-positive people is a complex disorder with myriad causative agents, both infectious and non-infectious. The evidence for empirical antimicrobial or antimotility is limited with gaps in the scientific evidence being most detrimental to those patients being managed in low-resource settings. It is apparent that reducing late diagnoses of HIV and increasing HAART coverage would have a beneficial effect on opportunistic infections causing chronic diarrhoea. However, the approach to this condition must be broad and multi-layered: governmental action to increase availability of clean and sanitary water and avoid malnutrition; health promotion to encourage better hand hygiene; and, where possible, management by a multi-disciplinary team.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
Footnotes
Conflict of Interests
The Authors declare no conflicts of interest.
Funding
The Authors received no funding in production of this manuscript.
Acknowledgements
TW would like to thank Dr. Peter Flegg of Blackpool Victoria Hospital and Dr. Andy Ustianowski of The Monsall Unit, North Manchester General Hospital for their advice, support and encouragement prior to and during the writing of this article. Thanks also go to Michael Reid, Susan Beames and their library team at Blackpool Victoria Hospital for their invaluable contribution to the initial literature search.
