Abstract
Menstrual migraine is a common neurological condition reported to affect up to 60% of women with migraine. Most women manage migraine adequately with symptomatic treatment alone. However, in women with menstrual migraine, menstrual attacks are recognised to be more severe, last longer, and are less responsive to treatment compared with attacks at other times of the menstrual cycle. In these situations, prophylactic treatment may be necessary. Short-term perimenstrual and continuous prophylactic treatments have shown efficacy in clinical trials but none are licensed for menstrual migraine. This article reviews the evidence for acute and prophylactic drugs in the management of this condition and considers future therapeutic options.
Introduction
Migraine is prevalent neurological disorder, affecting four of every 10 women and two of every 10 men, mostly before age 35 years. 1 By age 30 years migraine is 3-fold more prevalent in women than in men with the peak periods for migraine risk in women being at age 25 ± 8.6 years and 50 ± 15.8 years. 2 Diagnostic features accompanying headache include photophobia, nausea and limitation of usual daily activities. 3 Recent research by the World Health Organization has established migraine as a leading cause of years of life lived with a disabling condition–-12th for women, compared to 19th for men. 4
The two most frequently encountered types of migraine differ only in their presence or absence of ‘aura’ 5 Table 1. Menstruation is a significant risk factor for migraine without aura, even in women who have attacks with aura at other times of the cycle.6–10 In population and clinic-based studies, between 20% and 60% of women with migraine report an association with menstruation.9,11–16 Attacks are most likely to occur on or between two days before menstruation and the first three days of bleeding.5,7–9,17–22 Although long-recognised as a clinical entity, the research definitions for menstrual.
Diagnostic criteria for migraine without aura and migraine with aura (adapted from 5 ).

Fewer than 10% of women report migraine exclusively with menstruation and at no other time of the month (“pure” menstrual migraine) Table 2.5,9,11,14–16 The majority of women with regular menstrual attacks also experience migraine at other times of the month (“menstrually-related” migraine).5,9 The term “menstrual migraine” is often used to encompass both conditions. Although some women report a link between their migraine attacks and ovulation, this has not been confirmed in epidemiologic studies.9,20,23 A prospective study confirmed that the observed number of attacks associated with ovulation was not significantly different from the expected number of attacks. 21
Diagnostic criteria for pure menstrual migraine and menstrually-related migraine (adapted from 5 ).

For the purposes of this classification, menstruation is considered to be endometrial bleeding resulting from either the normal menstrual cycle or from the withdrawal of exogenous progestogens, as in the case of combined oral contraceptives and cyclical hormone replacement therapy.
Menstrual attacks are more severe and disabling, last longer, and are less responsive to symptomatic medication compared to attacks at other times of the cycle.13,16,20,24–29 Menstrual migraine is also associated with increased menstrual distress and disability.30–33
The timing of menstrual attacks is consistent with the natural fall in estrogen during the late luteal phase of the menstrual cycle Figuer 1. This ‘estrogen-withdrawal’ trigger is consistent with increased risk of migraine during the hormone-free interval of combined hormonal contraceptives and during puberty and the perimenopause, times of fluctuating hormone levels.2,21,34 Stable low, high, or rising hormone levels, such as during pregnancy and postmenopause are associated with reduced risk of migraine.
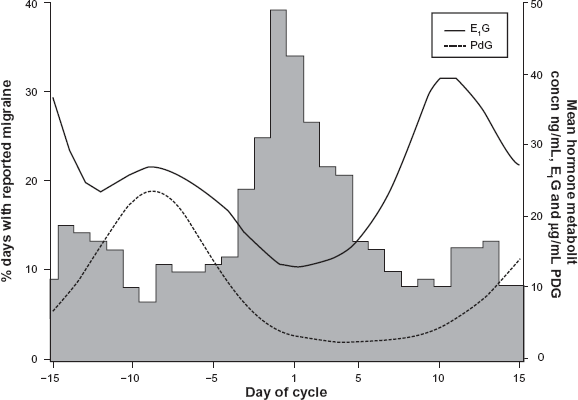
Incidence of migraine, urinary estrone-3-glucuronide (E1G) and pregnanediol-3-glucuronide (PdG) levels on each day of the menstrual cycle in 120 cycles from 38 women. Reproduced with permission from. 21
Treatment Options
All treatments licensed for migraine can be used to treat menstrual migraine. Summaries of Product Characteristics provide information on the licensed indications. However, if the posology and method of administration of a drug used to treat or prevent menstrual migraine is different from the license, the drug must be prescribed ‘off-license’. This is particularly the case for prophylaxis of menstrual migraine.
Lifestyle Recommendations
Assuming the concept of multiple factors acting in combination to trigger migraine, hormonal factors combine with non-hormonal triggers to increase the overall susceptibility to attacks at the time of menstruation. 35 Therefore, every effort should be made to identify and cope with non-hormonal triggers. 36 A case-control study of 85 women with migraine compared with 85 controls identified a significant relationship between diet, eating habits, resting and sleeping patterns. 37 Of the migraine group 37.6%, did not have a regular diet program, compared with 17.6% of the control group (P = 0.004); 37.6% of the migraine group, compared with 23.5% of the control group, did not eat meals on a regular schedule (P = 0.046), and 29.4% of the migraine and 9.4% of the control groups had less than 3 meals per day (P = 0.001). Study findings related to the sleep and rest patterns showed that 50.6% of migraineurs and 29.4% of the control group did not have regular sleep hours (P = 0.005). Further, 23.5% of the migraine and 4.7% of the control subjects slept less than 6 hours per night, while 28.2% of the migraine and 25.9% of the control group subjects slept more than 8 hours per night (P = 0.001). In some cases, attention to modifiable lifestyle factors may reduce the frequency and severity of all attacks. In others, non-hormonal attacks are eliminated while menstrual attacks persist and may require specific management.
Acute Treatment
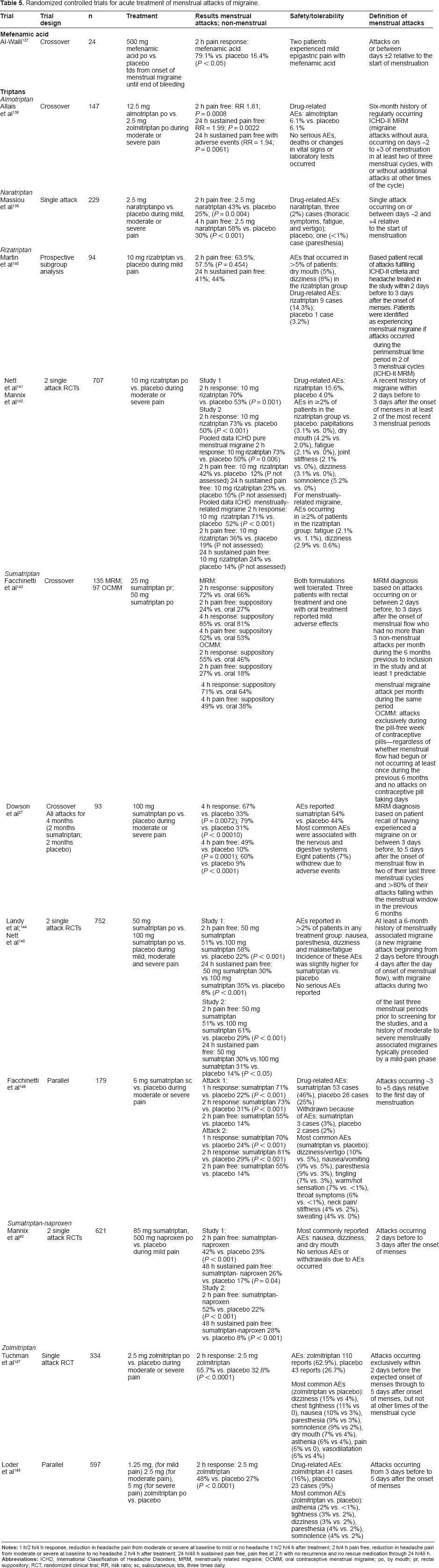
A variety of non-prescription and prescription drugs are available for the acute treatment of migraine. 38 A number of open-label studies Table 3, post-hoc analyses Table 4 and prospective randomized controlled trials Table 5 have been undertaken to assess comparative efficacy and tolerability of menstrual versus non-menstrual attacks. There is considerable variation in the definition of menstrual attacks as well as in the diagnosis of menstrual migraine. An evidence-based systematic review and meta-analysis concluded that, based on trial quality, evidence supported grade B recommendations for use of sumatriptan 50 and 100 mg, mefenamic acid 500 mg, and rizatriptan 10 mg for acute treatment of menstrual migraine. 39 Trial quality also supports grade B recommendations (good evidence of efficacy; benefits outweigh harms; improves important health outcomes) for the combination sumatriptan 85 mg-naproxen 500 mg. 40
Open-label trials for acute treatment of menstrual migraine.
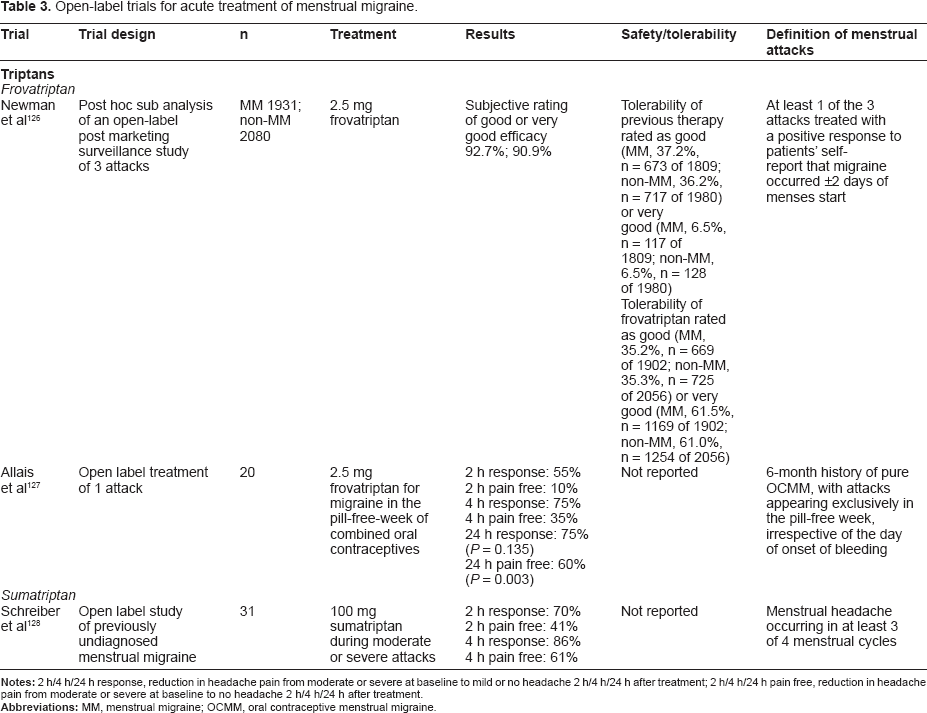
Post hoc analyses of menstrual attacks in clinical trials for acute treatment of migraine.
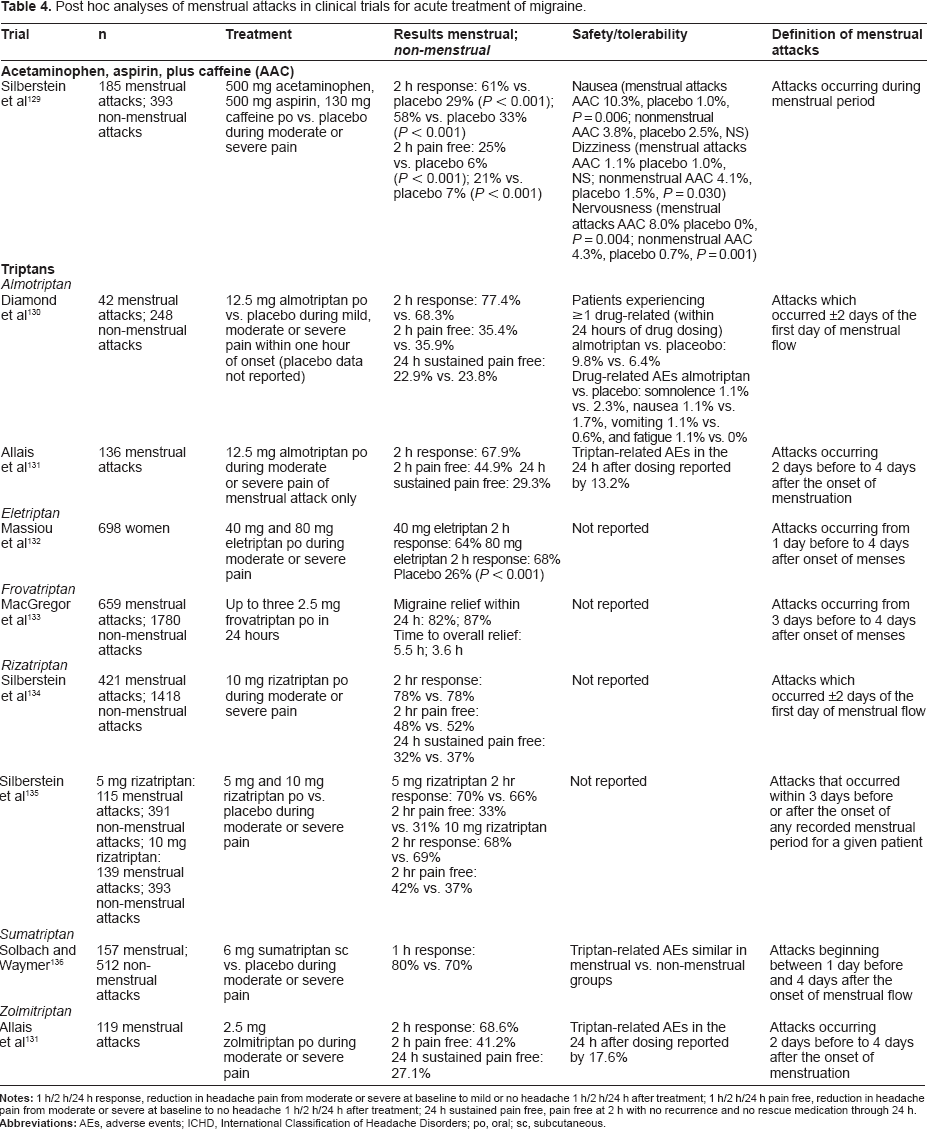
Randomized controlled trials for acute treatment of menstrual attacks of migraine.
In all studies, treatment was well tolerated with adverse events similar to those reported throughout the clinical trial programmes for each drug. The most frequent triptan-related adverse events include tingling, paresthesias, and warm sensations in the head, neck, chest, and limbs; less frequent are dizziness, flushing, and neck pain or stiffness. 41
While these studies confirm efficacy of acute treatments for menstrual and non-menstrual attacks of migraine, there are differences between results of analyses of attacks diagnosed as menstrual and results from clinical trials in women diagnosed with menstrual migraine at baseline. While the former suggest no difference in efficacy, the latter confirm the clinical impression that in women with menstrually related migraine, menstrual attacks do not respond as well to acute treatment as nonmenstrual attacks.24,27
Prophylaxis
Most women with pure menstrual migraine manage their condition with acute treatment. Prophylaxis should be considered for women who have inadequate relief from the usual forms of acute therapy or who are troubled by headache recurrence and require multiple doses of acute migraine medications. Diary evidence to confirm the diagnosis should be a prerequisite before prescribing prophylaxis for menstrual migraine as the patient history of a menstrual association can be unreliable.12,27
Prophylaxis of menstrual migraine using non-drug approaches such as relaxation biofeedback, person-centered insight therapy or acupuncture appears to be ineffective.42–44 For women with frequent non-menstrual attacks in addition to menstrual migraine, standard prophylactic agents are recommended. These include tricyclic anti depressives, beta-blockers and anti-epileptics.38,45 A post-hoc analysis suggests that to piramate reduces the frequency, but not severity or duration, of perimenstrual migraines in women with menstrually-related migraine. 46 No prophylaxis prevents all attacks but they can reduce the frequency and severity of attacks and improve response to acute treatment.
Specific management strategies have been studied but none are licensed specifically for management of menstrual migraine. Given that there are no investigations to identify the most effective prophylactic, an empirical approach is necessary, considering the individual needs and wishes of each woman. In order adquately to assess efficacy, each method should be tried for at least three cycles before considering alternative prophylaxis.
Perimenstrual (Short-term/Intermittent Prevention)
Short-term prevention strategies have the advantage that treatment is only used at the time of need, thus avoiding continuous exposure to active drug and the potential for adverse events associated with daily prophylaxis. 47 However, many studies are open label Table 6 and results from randomized controlled trials are limited Table 7. None of the drugs and hormones recommended below is licensed for management of menstrual migraine.
Open label trials of perimenstrual migraine prophylaxis.
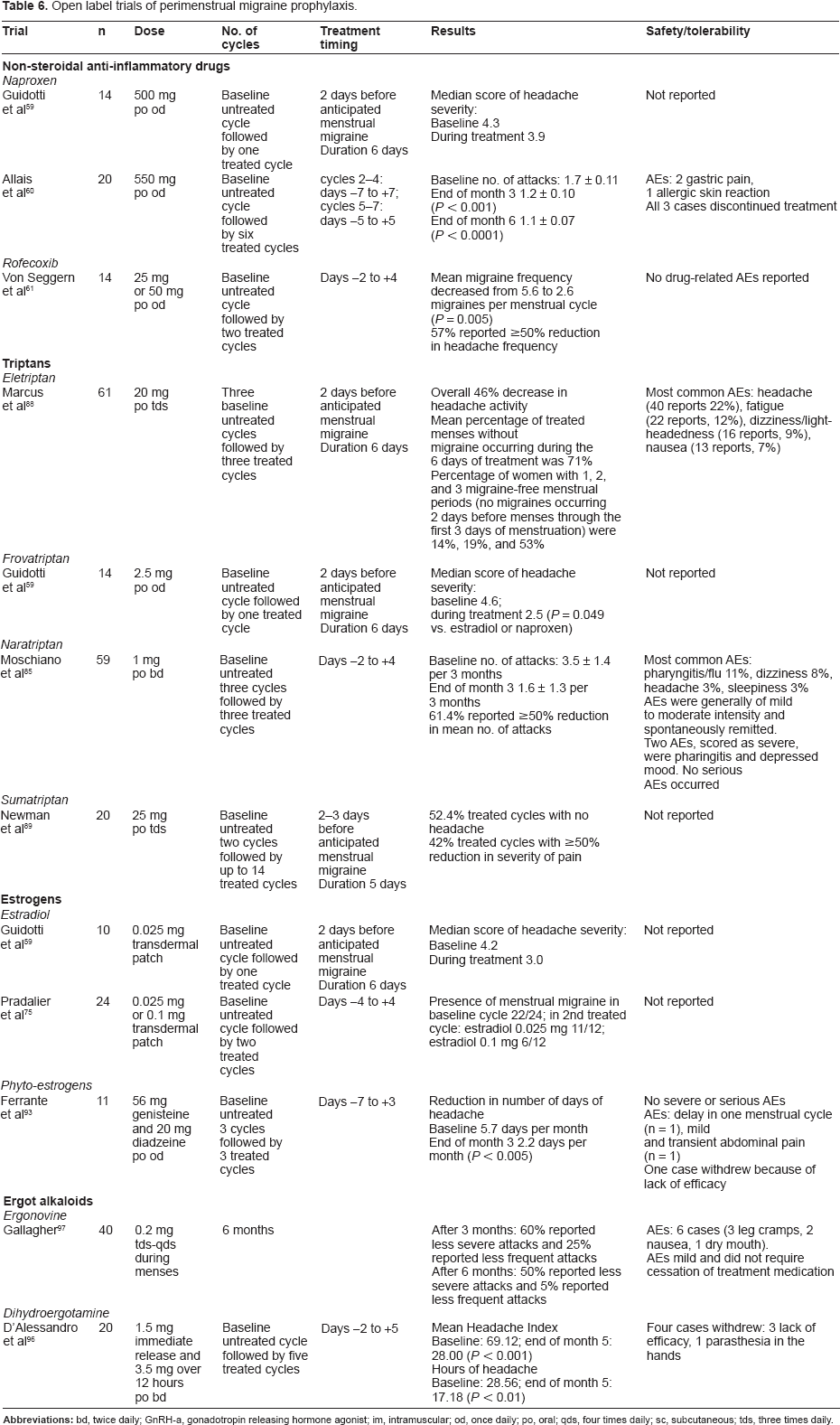
Randomized placebo-controlled trials of perimenstrual migraine prophylaxis.

Women using perimenstrual prophylaxis must have regular periods and a predictable relationship between migraine and menstruation as treatment is typically started a few days before expected onset of menstruation or the anticipated menstrual attack. A home-use fertility monitor can be used to predict menstruation accurately. 48
Non-steroidal anti-inflammatory drugs
Prostaglandins have been implicated in the pathophysiology of menstrual migraine. 49 In particular, entry of prostaglandins into the systemic circulation can trigger throbbing headache, nausea and vomiting. 50 In the uterus prostaglandins are synthesised primarily by the endometrium. There is a three-fold increase in prostaglandin levels in the uterine endometrium from the follicular to the luteal phase, with a further increase during menstruation. 51 As a result of the “withdrawal” of estrogen and progesterone the endometrium breaks down and prostaglandins are released. This causes vasoconstriction within the endometrium and disruption of endometrial cells, stimulating further prostaglandin synthesis. When an excessive amount of prostaglandins gain entrance to the circulation, other systemic symptoms occur that are characteristically associated with menorrhagia and/or dysmenorrhoea such as headache and nausea.52,53 Plasma taken during the premenstrual phase from women with dysmenorrhoea and reinfused postmenstruation into the same women resulted in premenstrual symptoms, including headache. 54 Thus prostaglandins may have a specific role in migraine associated with dysmenorrhoea and/or menorrhagia. In support of this, prostaglandin inhibitors are effective for the prevention of menstrual attacks of migraine. 55
Non-steroidal anti-inflammatory drugs (NSAIDs) are effective prostaglandin inhibitors. They should be tried as first-line agents for migraine attacks that start on the first to third day of bleeding, particularly in the presence of dysmenorrhoea and/or menorrhagia.52,56,57 Side effects of NSAIDs include gastro-intestinal disturbance. Misoprostol 800 μg or omeprazole 20 to 40 mg daily may give some gastroduodenal protection. 58 NSAIDs may cause fluid retention, leading to edema and elevated blood pressure. Kidney failure can also occur. Contraindications include peptic ulcer, aspirin-induced allergy, severe hepatic, renal and cardiac failure. Interactions include anticoagulants, diuretics and antihypertensive agents.
Open-label studies using perimenstrual naproxen 500–550 mg once daily suggest efficacy of this approach for menstrual migraine.59,60 Rofecoxib was not associated with gastrointestinal symptoms in an open-label study. 61 However, the drug has since been withdrawn because of cardiovascular safety concerns with high-dosage long-term use.
Randomized studies using naproxen 550 mg twice daily perimenstrually have confirmed efficacy with good tolerability.62,63 Sances et al, noted that 25% of women taking naproxen reported mild or moderate nausea and epigastric distress but all continued treatment. 62 Limited data from a single randomized placebo-controlled trial suggest nimesulide may be an effective alternative. 64
Estradiol
Research has focused on ‘withdrawal’ of estrogen and progesterone/progestogens as occurs during the luteal phase of the menstrual cycle and during the hormone-free interval of combined hormonal contraception.65–68
The rationale for perimenstrualestrogen supplementation is based on evidence that the natural decline in estrogen in the late luteal phase of the menstrual cycle, just prior to menstruation, is associated with increased risk of migraine. 69 Somerville noted that a period of estrogen ‘priming’ with several days of exposure to high estrogen levels is necessary for migraine to result from estrogen ‘withdrawal’, as occurs in the late luteal phase of the menstrual cycle.70–72 This would explain why migraine is not associated with the transient estrogen surge at ovulation. Since women using combined hormonal contraceptives still experience migraine occurring during the hormone-free interval, it would seem that ovulation is not a prerequisite for menstrual attacks.
Somerville showed that migraine could be postponed by maintaining high plasma estradiol levels with an intramuscular injection of long-acting estradiolvalerate in oil; migraine subsequently occurred when the plasma estradiol fell. 70 Somerville further attempted to control estrogen fluctuations with oral estrogens and estrogen implants. Both of these routes of delivery failed to provide stable plasma levels of estradiol and so, not surprisingly, were of no benefit to migraine. 72 This supports the hypothesis that prolonged estrogen exposure is necessary for ‘withdrawal’ to trigger migraine.
Several trials have confirmed the efficacy of transcutaneous estradiol for menstrual migraine prophylaxis.73–78 Transdermal administration is preferred since it provides more stable serum levels than oral estrogen. The recommended strategy is estradiol gel 1.5 mg applied daily from two to three days before expected menstruation for seven days. 79 Alternatively transdermal estrogen 0.1 mg can be used from two to three days before expected menstruation or menstrual migraine up to the fourth or fifth day of menstruation, ie, two twice-weekly patches or one seven-day patch, although an additional patch may be necessary if menstruation is late. 79 Lower doses of 0.025 mg and 0.05 mg estradiolare not effective.59,74–76
Estradiol is well tolerated although post-treatment migraine can occur. Somerville noted that migraine estradiol treatment delayed migraine between three and nine days in all 6 women studied. 70 De Lignières reported that 1 of 20 women had migraine three days after stopping estradiol treatment. 78 MacGregor found an increase in migraine occurrence in the 5 days immediately following estradiol use compared to placebo, relative risk [RR] 1.40 (95% CI 1.03 to 1.92, P = 0.03). 73 Possible reasons for this posttreatment migraine may be that the dose of estradiol was inadequate; the duration of treatment was too short; or perhaps that exogenous estrogen prevents the normal secretion of endogenous estrogen. Although there are no trial data, clinical practice suggests that for these women the duration of supplement use can be extended until day 7 of the cycle, tapering the dose over the last two days.
Menstrual irregularity can occur, probably due to suppression of endogenous estrogen during treatment. 73 There is no evidence that estradiol supplements increase the risks of cancer or thrombosis in premenopausal women. 80
Triptans
Randomized placebo-controlled trials and open-label studies of frovatriptan, naratriptan, and zolmitriptan for perimenstrual prophylaxis have suggested efficacy although direct comparison of the results is limited by the different endpoints used.59,81–87 Only results from single open-label studies are available for eletriptan and sumatriptan.88,89 Although the definitions of menstrual migraine appear similar, only one trial required review of documented diary data as confirmation for inclusion. 82 Only two studies required confirmation of menstrual migraine as a specific diagnosis.82,85
Perimenstrualtriptan prophylaxis is well tolerated and the incidence and type of adverse events reported is consistent with those reported in trials of acute treatment. The high completion rates in the clinical trials are notable. However, there is potential concern that treatment may defer attacks or result in ‘rebound’ migraine following treatment. This has been noted with naratriptan, with the percentage of patients reporting migraine during the immediate post-treatment period being higher in those treated with naratriptan compared to patients receiving placebo. 84 Increased migraine post-treatment has been reported not to occur following perimenstrual frovatriptan. 82
Specific analyses of safety and tolerability have been undertaken on long-term trials of frovatriptan and naratriptan. During treatment of up to 12 perimenstrual periods over a 12- to 15-month period, adverse events with frovatriptan were generally mild or moderate in severity and were similar to that observed with acute use of triptans. 90 Results of subgroup analyses of women whose medical histories included comorbidities that might suggest increased cardiovascular risk but were not themselves contraindications to frovatriptan, provide preliminary evidence of safety of frovatriptan in this population. 91
Patients completing 6- to 12-months perimenstrual prophylaxis with naratriptan noted that no specific adverse event considered at least possibly to be related to study medication occurred in more than 2% of patients. No serious drug-related adverse events were reported and no patient experienced clinically relevant drug-related changes in 12-lead ECGs, vital signs, or clinical laboratory tests. 92
Other treatments used for perimenstrual prophylaxis
In an open-label of 10 women with pure menstrual migraine taking phyto-estrogensgenistein 56 mg and daidzeine 20 mg daily, 10 days per month (starting 7 days before the predicted onset of menses) for three cycles, the average number of days with migraine during the baseline period decreased significantly after 3 months of therapy (P < 0.005). 93
Magnesium prolidone carboxylic acid 360 mg decreased the duration and intensity of premenstrually occurring migraine in a placebo-controlled, double-blind study of 24 women with premenstrual syndrome and migraine. 94 This study was principally aimed at identifying the effect of magnesium on a number of premenstrual problems, not just headache. The generalizability of the results to women whose menstrual headaches do not occur in association with other premenstrual symptoms is unclear. Diarrhea was reported by one woman during treatment with magnesium.
A trial of vitamin E 400IU, an anti prostaglandin agent, given perimenstrually in a cross-over study of two menstrual cycles showed limited effect as a prophylactic, although headache pain and associated symptoms were reduced compared to placebo. 95 Tolerability was not reported.
The ergot alkaloids ergonovine and dihydroergotamine have shown efficacy in open-label studies of menstrual migraine prophylaxis but there are no data from randomized controlled trials to support their use.96,97
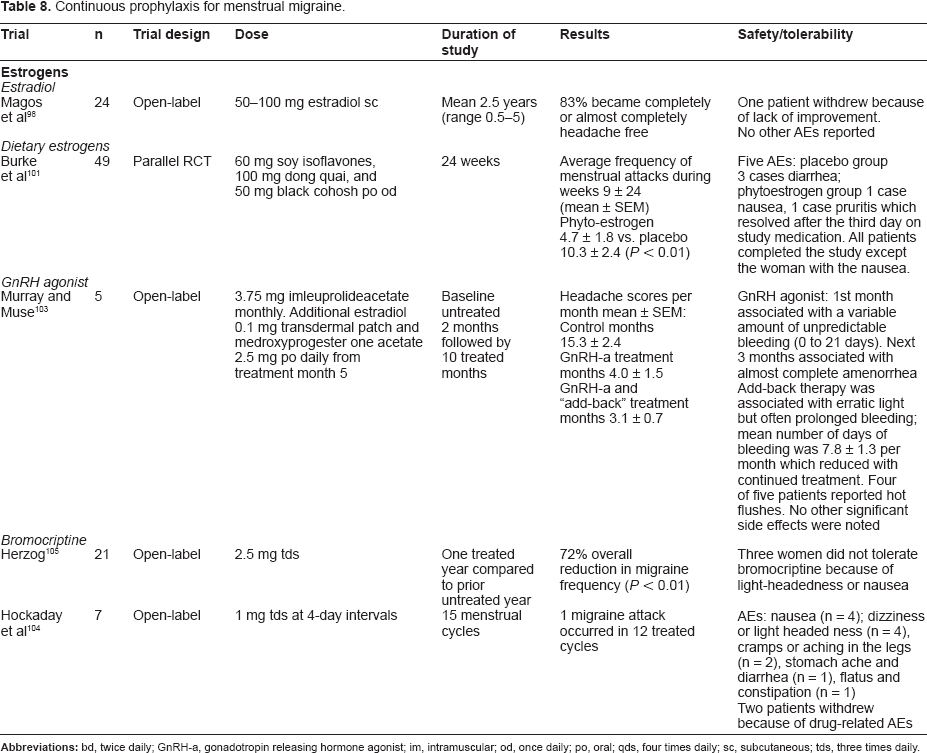
Continuous Prophylaxis
For women who are unable to predict menstruation, several continuous prophylactic strategies specifically for women with menstrual migraine have been studied Table 8.
Continuous prophylaxis for menstrual migraine.
Estradiol
Estradiol implants are the most effective method of obtaining high stable estrogen levels. In an open-label study estradiol implant given in doses large enough to suppress ovulation and produce constant plasma estrogen levels achieved an 96% response rate in 24 women with menstrual migraine treated for up to five years, with 46% of women becoming completely headache free. 98 Treatment was well tolerated.
In unhysterectomised women, progestogen opposition is necessary to protect the endometrium, as unopposed oestrogens increase the risk of endometrial cancer. However, cyclical progestogen can mimic premenstrual symptoms, including headache. 99 Although there are no clinical trials for migraine, suppression of ovulation with 100 μg patches used continuously together with continuous progestogen are likely to be effective with fewer progestogenic side-effects. 100
Dietary estrogens
Phyto-estrogens are estrogen-like molecules derived from soy, which have gained interest as a “natural” alternative to prescription estrogens. They have estrogenic effects in some tissues, without stimulation of the endometrium. Theoretically, this confers greater long-term safety than with estradiol treatment, although this has yet to be established. Black cohosh (Cimicifugaracemosa) contains several compounds with estrogen receptor activity similar to soy isoflavones. Dong quai (Angelica polymorpha), known as Chinese Angelica, also has estrogen-like effects. In a randomized placebo-controlled trial, a combination of 60 mg soy isoflavones, 100 mg dong quai, and 50 mg black cohosh the average frequency of menstrual attacks in women treated with the phytoestrogen preparation was 4.7 ± 1.8 (mean ± SEM) during weeks 9–24 compared with 10.3 ± 2.4 in placebo treated patients (P < 0.01). 101
Gonadotropin-releasing hormone analogues
These drugs inhibit ovulation, reducing levels of estrogen. Although effective, adverse effects of estrogen deficiency, eg, hot flushes, restrict their use. 102 The hormones are also associated with a marked reduction in bone density and should not usually be used for longer than six months without regular monitoring and bone densitometry. ‘Add-back’ continuous combined estrogen and progestogen can be given to counter these unwanted effects. 103
Bromocriptine
This dopamine agonist inhibits gonadotrophin releasing hormone and luteinizing hormone, reducing estrogen levels. Two studies have suggested efficacy of bromocriptine in migraine although larger double-blind placebo controlled studies are necessary before it can be recommended.104,105 Although generally well-tolerated, adverse events related to treatment included light-headedness or nausea.
Anti-estrogens
There is some limited evidence of efficacy for danazoland tamoxifen but symptoms of estrogen deficiency such as hot flushes, menstrual irregularity, fatigue and joint pains, restrict their use.106–110
Contraceptive Strategies
Combined hormonal contraceptives minimize the hormonal fluctuations associated with migraine and are particularly useful when a woman also requires contraception. 111 They have additional non-contraceptive benefits on premenstrual syndrome, menorrhagia and dysmenorrhoea. International guidelines for safe prescribing are available. In particular, contraceptive doses of synthetic estrogens should not be used by women who also have with migraine with aura because of the synergistic increased risk of ischemic stroke. 112
Most licensed combined hormonal contraceptives include a hormone-free interval during which a withdrawal bleed occurs. However, this withdrawal of hormones is associated with increased risk of migraine.113–115
Supplementing estrogen during the hormone-free interval can minimize this risk of migraine. In a small open-label study, 11 women with menstrual migraine were treated with a 28-day cycle of 0.02 mg ethinylestradiol oral contraceptive for 21 days followed by 0.9 mg conjugated equine estrogen daily for 7 days. All women achieved at least a 50% reduction in number of headache days per cycle (mean 77.9% reduction). 116 Transdermal estrogen in an alternative option. A study using 0.05 mg estradiolpatches during this time suggested that this dose is suboptimal for prophylaxis although post-trial treatment with 0.1 mg doses was effective. 114
Continuous use of combined hormonal contraceptives is a more simple strategy to prevent ‘estrogen-withdrawal’ migraine. This regime is well tolerated, although unscheduled bleeding is a common reason for withdrawal from clinical trials in the first 6-months of treatment. Continued use induces amenorrhea in 80% to 100% of women by 10 to 12 months of treatment. 117 For women who experience migraine during the seven-day hormone-free interval of standard regimes, data from an open-label study suggest that continuous use of combined hormonal contraceptives is effective. 118
To date, larger trials have only assessed headaches in the hormone-free interval and not specifically migraine. A trial of 102 women taking 3 mg of drospirenone and 0.03 mg ethinylestradiol continuously showed that, compared to the usual 21/7-day regimen of combined hormonal contraceptives, a 168-day extended placebo-free regimen led to a decrease in headache severity along with improvement in work productivity and involvement in activities. 119 Similarly, an extended 84-day regimen of a transdermal contraceptive reduced the total incidence of mean headache days compared to a 21/7-day regimen. 120
Progestogen-only contraceptives
In general, standard contraceptive oral progestogens have little place in the management of menstrual migraine since most are associated with a disrupted menstrual cycle. 121 In contrast, unlicensed higher doses of oral progestogen, sufficient to suppress oestrogen fluctuations and achieve amenorrhoea, have shown benefit.122,123 Depot medroxyprogesterone acetate should, in theory, provide stable low levels of oestrogen to prevent menstrual migraine but there are no studies to support or refute this.
Future Therapeutic Approaches
While trial data can support the efficacy, safety and tolerability of treatments, data on patient satisfaction and compliance in clinical practice are much needed.
A new therapeutic approach being studied is with the calcitonin-gene related peptide antagonist telcagepant. Results are awaited from a six-month phase 2/3, randomized, double-blind, placebo-controlled clinical trial to evaluate the safety, tolerability, and efficacy of telcagepant for prevention of menstrually-related migraine in 4500 female patients with episodic migraine (NCT01125774). In this study, women were randomized to 140 mg telcagepant or placebo once daily at bedtime for 7 days, beginning at onset of menses, for 6 menstrual cycles.
Genetic research is also underway, with the hope of identifying genes that will enable an objective method of diagnosing menstrual migraine. Studies investigating the role of the estrogen receptor 1 (ESR1) gene in migraine show a significant association of the A allele of the G594A SNP and the progesterone receptor (PGR) PROGINS insert with migraine.124,125 Women who carry a copy of both PR and ESR1 risk alleles were 3.2 times more likely to suffer from migraine, an effect that is greater than the independent effects of these genetic variants on disease susceptibility. It is anticipated that this association will be stronger in women with menstrual migraine, who have a strong hormonal trigger for attacks.
Accurate diagnosis of menstrual migraine can aid the selection of currently available treatments and could ultimately lead to the development of more specific treatments, targeted to the affected genes.
Conclusions
The majority of women with menstrual migraine need only to optimize symptomatic treatment. Trial data to support grade B recommendations (good evidence of efficacy; benefits outweigh harms; improves important health outcomes) are available for sumatriptan 50 and 100 mg, mefenamic acid 500 mg, rizatriptan 10 mg and the combination sumatriptan 85 mg-naproxen 500 mg. 39
If acute treatment is insufficient for effective control, perimenstrual prophylaxis is usually considered in addition to symptomatic medication. A number of different treatment strategies have been studied for the prevention of menstrual migraine in randomized placebo-controlled trials and open-label studies. Choice of prophylaxis depends on the regularity of the menstrual cycle, timing of attack in relation to bleeding, presence of dysmenorrhoea and/or menorrhagia, and the need for contraception. Each prophylactic should be tried for a minimum of three menstrual cycles before considering an alternative option, using diary cards to record outcomes. Trial data to support grade B recommendations exist for use of transcutaneous estradiol 1.5 mg, frovatriptan 2.5 mg twice daily, and naratriptan 1 mg twice daily for perimenstrual prophylaxis. 39
Contraceptive approaches should be considered for women with menstrual migraine who also wish to prevent pregnancy. Continuous combined hormonal contraception has limited evidence of efficacy but should not be used by women with migraine with aura.
Results from a prophylactic trial of telcagepant, a calcitonin-gene-related peptide antagonis are awaited. Research to identify relevant genes has the potential to improve diagnosis and enable targeted management.
Disclosures
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The peer reviewers of this paper report no conflicts of interest. The author has acted as a paid consultant to, and/or her department has received research funding from Addex, Astra Zeneca, BTG, Endo Pharmaceuticals, Glaxo Smith Kline, Menarini, Merck, Pozen, and Unipath. She received no financial support for the preparation of this manuscript. The author confirms that she has permission to reproduce any copyrighted material.
