Abstract
Background
Rituximab (RTX) is a biological agent used for the treatment of refractory rheumatoid arthritis (RA). The present systematic review and meta-analysis update the safety and efficacy of RTX for the treatment of RA in data published since 2006.
Methods
We searched PubMed from January 2000 to March 2010, and recent ACR Annual meeting abstracts for randomized (RCT) and non-randomized, controlled clinical trials (CCT) investigating the effects of RTX in RA. Included studies were at least six months, included participants who were ≥16 years of age meeting ACR revised criteria for RA, and compared RTX in combination with any DMARD or RTX alone versus placebo or other DMARDs or any biologic. Studies were excluded if they included patients previously exposed to RTX. Two reviewers independently extracted the data, and disagreement was resolved by discussion and consensus. The primary outcome was the Disease Activity Score (DAS28). The secondary outcome was American College of Rheumatology (ACR50) response. The efficacy results were combined in a meta-analysis. The primary endpoint was analyzed as a continuous variable using a random-effects analysis model to account for the fact that intervention effects were not uniform across all the studies. The secondary endpoint was analyzed as an odds ratio using the Mantel-Haenszel estimator under a random effects model to account for heterogenity in intervention effects across the studies. Descriptive statistics were used to compare adverse event (AE) rates and included both randomized and observational trials.
Results
For the meta-analysis of efficacy, we initially examined 45 studies, devolving to 6 studies after applying inclusion/exclusion criteria. 5 were RCTs and 1 was a controlled clinical trial. Improvements in DAS28 and ACR50 statistically favored RTX (Figure 1). For the safety evaluation, 22 studies (5 RCTs and 17 observational studies) were examined. Infusion-related reactions were higher in the RTX group (mean: 28%) vs. placebo (18%). Overall infection incidence was similar for both groups (RTX: 39 vs. placebo: 40%).
Conclusions
Our updated review supports RTX as a safe and efficacious therapy for treatment-naïve and methotrexate and/or TNF-alpha refractory patients with RA. AE's revealed no more frequent infections than control. Future trials need to assess the longer-term efficacy and safety of RTX for RA, as well as the optimal time of re-administration of this agent.
Introduction
Rheumatoid arthritis (RA) is a chronic, systemic autoimmune disorder affecting approximately 1%–3% of the population 1 and is primarily characterized by inflammation and swelling of synovial joints. Pain, fatigue, disability, and impaired social functioning are the most common symptoms. It can cause deformity and destruction of joints leading to substantial loss of function and quality of life,2,3 and is associated with reduced life expectancy. 4
Treatment of RA requires a multidisciplinary approach with both pharmacological and non-pharmacological interventions, such as physical and occupational therapy. With regard to pharmacotherapy, non-biological disease modifying anti-rheumatic drugs (DMARDs) and/or biological agents as single or combination therapy are the mainstay of treatment. 5 Both non-biological DMARDs and biological agents reduce joint damage 6 and improve quality of life, 7 and are target-oriented molecules that suppress inflammation and decrease progression of joint damage.8,9 In patients who fail to respond to non-biological DMARDs, studies have demonstrated that biological agents serve as effective alternatives.10,11
Rituximab is one such biological agent that was introduced for the treatment of refractory RA. 12 It is a mouse/human chimeric IgG1–κ monoclonal antibody, which targets the CD20 antigen found on the surface of B lymphocytes. Rituximab is depletes CD20+ B lymphocytes implicated in the immunopathogenesis of RA. The American College of Rheumatology 2008 treatment guidelines for RA recommend the use of rituximab in patients with severe rheumatoid arthritis who experience an inadequate response to methotrexate or in combination with DMARDs or sequential administration of other nonbiologic DMARDs. 5 The standard dose consists of two 1000 mg intravenous infusions separated by a period of two weeks.
A Cochrane review found that rituximab showed similar efficacy when compared with abatacept, adalimumab, etancercept, infliximab, and was more efficacious than anakinra. 13 That review analyzed the results of three rituximab trials, the most recent of which was published in 2006. 11 – 12 – 14 A number of rituximab clinical trials have been conducted since that time. The purpose of the present meta-analysis is to include the results of recent clinical trials of rituximab to evaluate the efficacy of rituximab for the treatment of RA. In addition, we undertake a systematic review, particularly to examine additional data regarding adverse effects of this medication.
Methods
Study selection
To identify potentially relevant citations, two reviewers (HA, ERV) independently searched PubMed from 2000 to March 2010 using the following Medical Subject Headings (MeSH) and text words: rituximab and rheumatoid arthritis. The search was limited to human studies, clinical trials, and English language studies. The same reviewers manually searched American College of Rheumatology (ACR) Annual Conference abstracts for potentially relevant studies. The reference lists of retrieved articles were also reviewed. Studies were included if they (1) utilized 1000 mg rituximab in 2 doses per course; (2) were randomized controlled trials (RCTs) or controlled clinical trials (CCTs); (3) compared rituximab in combination with any DMARD or rituximab alone versus placebo or other DMARDs or any biologic; (4) were a minimum trial duration of 6 months; (5) included participants who were at least 16 years of age meeting the ACR 1987 revised criteria 15 for rheumatoid arthritis with active disease as described by authors in relation to the outcome measures; (6) were in the English language. Studies were excluded if they included patients who had previously been exposed to rituximab.
Data abstraction
Two reviewers (HA, ERV) independently extracted data, and disagreement was resolved by discussion and consensus. The extraction of data included study design, participant characteristics and outcome measures. For each study, the following participant characteristics were retrieved when available: number of participants; geographical location of study; type of cohort; follow-up period; proportion of females; mean age; proportion of participants with positive RF (anti-CCP was not usually included in the articles and so was not extracted); mean RA disease duration; proportion of patients with prior non-MTX DMARD, MTX, or other biologic use; proportion of participants currently on MTX; mean current MTX dosage; proportion of participants currently on glucocorticoids; mean current glucocorticoid dosage; proportion of participants currently on NSAIDs. Study authors were solicited via email to supply any missing information.
Efficacy outcomes
The primary efficacy outcome measure evaluated in this meta-analysis was the Disease Activity Score (DAS-28), which measures joint swelling and tenderness, ESR(or CRP) and a measure of patient derived global disease activity. 16 The secondary outcome measure was a modification of the American College of Rheumatology (ACR 50) response, 17 which represents ≥50% improvement in tender and swollen joints counts plus ≥50% improvement in 3 of the 5 other measures (e.g. patient and physical global assessments of disease activity, pain, functional status and an acute phase reactant).
Safety outcomes
Safety outcomes included proportion of deaths and withdrawals for toxicity or lack of efficacy. Safety outcomes also included selected adverse events, including infusion reactions (i.e. anaphylactic events, urticaria, hypotension, angioedema, hypoxia, bronchospasm, pulmonary infiltrates, acute respiratory distress syndrome, etc.), additional cardiovascular events (i.e. hypertension, myocardial infarction, cerebral vascular accidents, ventricular fibrillation, cardiogenic shock), malignancy (i.e. melanoma, non-melanoma skin carcinoma, etc.), lymphoma, or breast carcinoma, etc., infections, serious infections (i.e. infections requiring intravenous antibiotics and/or hospitalization, cellulitis, pneumonia, abscess, sepsis, tuberculosis), gastrointestinal effects (i.e. nausea, vomiting, diarrhea, dyspepsia, epigastric pain, oral ulcers, peptic ulcers, abdominal pain, or intestinal obstruction), central nervous system effects (i.e. headache, dizziness, convulsions, PML, or epilepsy), pulmonary effects (i.e. dyspnea or cough), rheumatologic effects (i.e. exacerbation of RA, arthralgias, fatigue), hematological effects (i.e. thrombocytosis, metorrhagia, myelodysplastic syndrome), dermatologic effects (i.e. rash, pruritis). Safety outcomes were abstracted from all of the studies included in the meta-analysis. Given the paucity of safety data presented in the included studies, safety data were also obtained from observational studies (both controlled and non-controlled studies) retrieved in the initial literature search.
Data analysis
Both primary and secondary efficacy endpoints were meta-analyzed using the Cochrane Reviews software, RevMan. The primary endpoint, comparison of change in DAS28 from study baseline to follow-up between Rituximab and control treatment arms, was analyzed as a continuous variable using a random-effects analysis model to account for the fact that intervention effects were not uniform across all the studies, i.e. treatment and control regimens varied to some degree among studies. In addition, the standardized mean difference (rather than the absolute mean difference) was chosen as the summary statistic in order to account for the possible use of different versions of the DAS28. The secondary endpoint, comparison of ACR 50 response from baseline to follow-up between Rituximab and control, was analyzed as an odds ratio using the Mantel-Haenszel estimator under a random effects model, again to account for the fact that intervention effects were not uniform across the studies. Due to the dissimilarity one study from the others (Finckh et al 18 ), in terms of intervention effect and study design (not randomized, lack of blinding/concealment of allocation), we carried out a second version of the meta-analysis of the primary endpoint, omitting this particular study and the two meta-analyses were compared. Funnel plots were created to detect publication bias. For the safety analysis, descriptive statistics were used to compare adverse event rates. Given the heterogeneity of study groups evaluated in the safety portion of this review, no further comparative statistics were performed.
Results
Study characteristics
A total of 42 articles were identified for review (Fig. 1). Contact with a content expert revealed three other potentially eligible studies which were reviewed following completion of the manuscript.19–21 Of these three additional studies, two were determined to meet all inclusion criteria and therefore were included for analysis.19,21 After screening the other titles and abstracts based on the predefined inclusion criteria, 20 studies were excluded. The remaining 21 studies were obtained for more thorough review. Fifteen were excluded for the reasons outline in Figure 1. Manual review of reference lists of reviewed studies revealed no additional studies for consideration. In total, 6 studies were included in this meta-analysis consisting of five RCTs11,12,14,19,21 and one CCT. 18
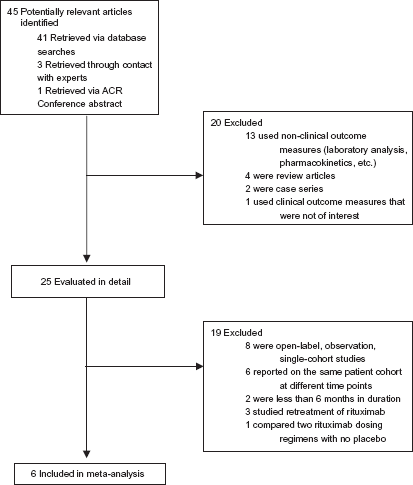
Selection of studies for meta-analysis.
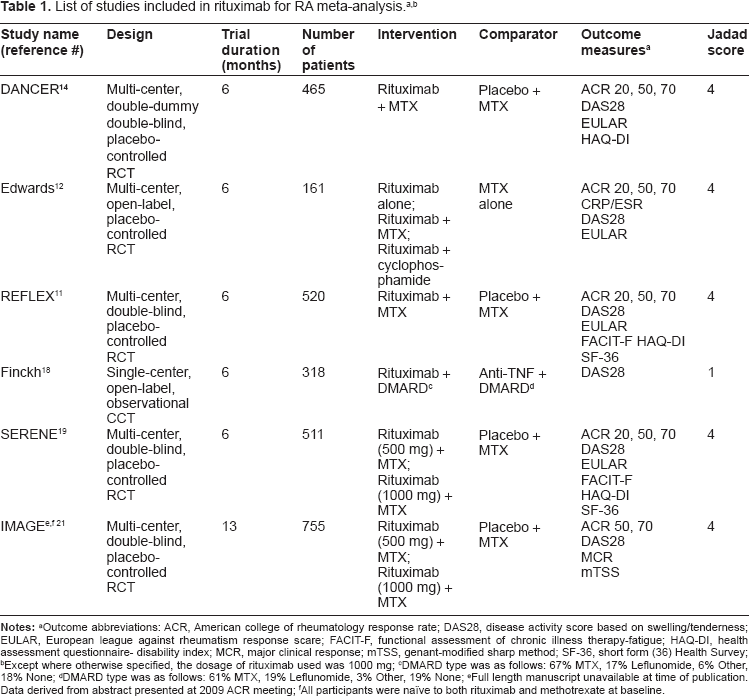
Table 1 outlines the design and quality assessment of each study. The majority of studies compared rituximab to a placebo and methotrexate.11,12,14,19,21 One study compared rituximab to an anti-TNF agent. 18 In terms of follow up, all of the studies assessed outcome variables at 6 months from initiation of therapy, with the exception of the IMAGE trial, 21 which assessed clinical outcomes at 13 months. All of the studies evaluated the primary outcome measure for this meta-analysis (DAS-28). Five studies (Finckh et al 18 excepted) evaluated the secondary outcome measure of ACR 50 response rate. Quality assessment was based on the Jadad score for RCTs. 22
Notes:
Outcome abbreviations: ACR, American college of rheumatology response rate; DAS28, disease activity score based on swelling/tenderness; EULAR, European league against rheumatism response scare; FACIT-F, functional assessment of chronic illness therapy-fatigue; HAQ-DI, health assessment questionnaire- disability index; MCR, major clinical response; mTSS, genant-modified sharp method; SF-36, short form (36) Health Survey;
Except where otherwise specified, the dosage of rituximab used was 1000 mg;
DMARD type was as follows: 67% MTX, 17% Leflunomide, 6% Other, 18% None;
DMARD type was as follows: 61% MTX, 19% Leflunomide, 3% Other, 19% None;
Full length manuscript unavailable at time of publication. Data derived from abstract presented at 2009 ACR meeting;
All participants were naïve to both rituximab and methotrexate at baseline.
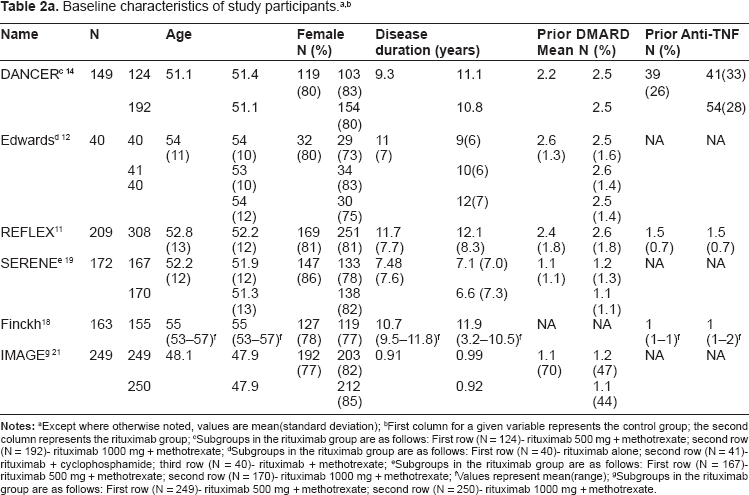
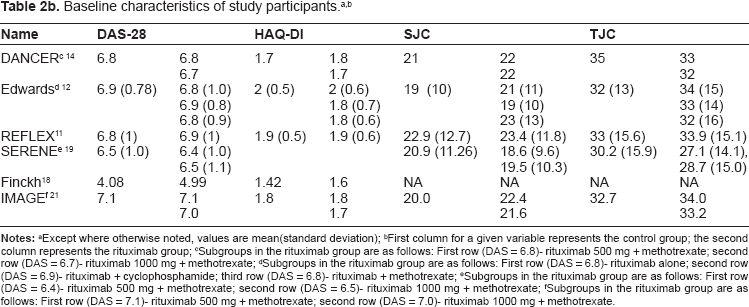
Study recruitment and enrollment occurred internationally at over 200 centers. Sample size varied among the included studies, ranging from 161 to 748 participants (Table 2a). A total of 2728 patients were enrolled. Of these patients, 73% to 85% was female. Seventy-four to 87% of patients were RF positive (among studies where RF was available). Mean age ranged from 48 to 55 years, and mean disease duration ranged from 0.91 to 11.9 years. Baseline DAS28 scores were similar among the patients in both study groups (Table 2b).
Notes:
Except where otherwise noted, values are mean(standard deviation);
First column for a given variable represents the control group; the second column represents the rituximab group;
Subgroups in the rituximab group are as follows: First row (N = 124)- rituximab 500 mg + methotrexate; second row (N = 192)- rituximab 1000 mg + methotrexate;
Subgroups in the rituximab group are as follows: First row (N = 40)- rituximab alone; second row (N = 41)-rituximab + cyclophosphamide; third row (N = 40)- rituximab + methotrexate;
Subgroups in the rituximab group are as follows: First row (N = 167)-rituximab 500 mg + methotrexate; second row (N = 170)- rituximab 1000 mg + methotrexate;
Values represent mean(range);
Subgroups in the rituximab group are as follows: First row (N = 249)- rituximab 500 mg + methotrexate; second row (N = 250)- rituximab 1000 mg + methotrexate.
Efficacy
The results of the meta-analysis for the primary endpoint are reported in Figure 2. Improvements in DAS28 score were significantly larger in the rituximab group compared with the placebo group, with a standard mean difference of -0.73 (95% CI: -1.05 to -0.41). Excluding the Finckh et al trial, 18 which was the only non-randomized trial in the meta-analysis, improvements in DAS28 score were still significantly larger in the rituximab group compared with the placebo group, with a standard mean difference of -0.90 (95% CI: -1.04 to -0.75) (Fig. 3).
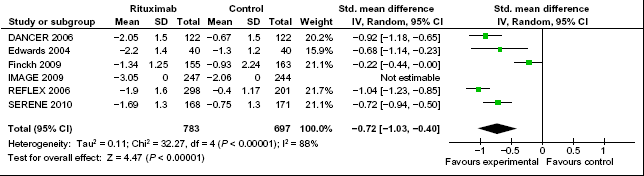
Meta-analysis of DAS28 improvement among patients receiving rituximab versus patients receiving placebo and/or methotrexate. Green squares represent the point estimate and their sizes represent their weight in the pooled analysis. Horizontal lines signify the 95% confidence intervals. The diamond shows the 95% confidence intervals for the pooled mean differences. Values less than one indicate mean differences that favor rituximab.
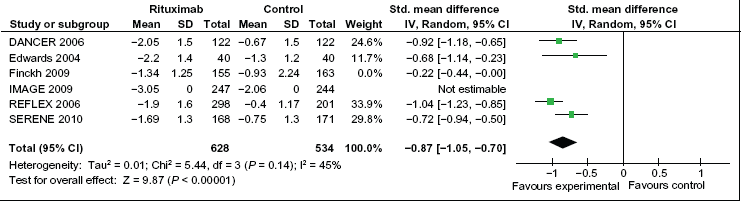
Meta-analysis of DAS28 improvement among patients receiving rituximab versus patients receiving placebo and/or methotrexate. This analysis only includes randomized clinical trials (Finckh et al excluded). Green squares represent the point estimate and their sizes represent their weight in the pooled analysis. Horizontal lines signify the 95% confidence intervals. The diamond shows the 95% confidence intervals for the pooled mean differences. values less than one indicate mean differences that favor rituximab.
As with the DAS28, improvements in the ACR 50 criteria were significantly greater in the rituximab group compared to the placebo group, with OR = 4.19 (95% CI: 2.92–6.03) (Fig. 4). The ACR 50 improvement analysis did not include the Finckh et al trial, 18 as this study did not measure ACR 50 improvement.
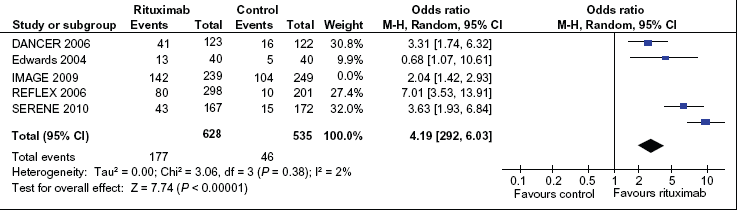
Pooled Odds Ratio (OR) of ACR 50 response among patients receiving rituximab versus patients receiving placebo and/or methotrexate. Blue squares represent the point estimate and their sizes represent their weight in the pooled analysis. Horizontal lines signify the 95% confidence intervals. The black diamond shows the 95% confidence intervals for the pooled OR's. Values greater than one indicate OR's that favor rituximab.
Visual inspection of the funnel plots revealed no significant asymmetry (Figs. 5, 6).
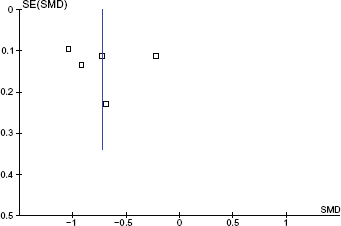
Funnel plot for DAS28 data. Y-axis is the Standard error (SE) of the DAS28 change Standardized Mean Difference (SMD).
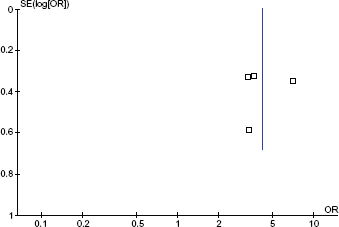
Funnel plot for ACR 50 data. Y-axis is the Standard error (SE) of the ACR 50 Odds Ratio (OR).
Safety
Tolerability and safety of rituximab were examined in 22 studies and presented in a detailed and extensive listing, which can be used descriptively (Table 3). Given that few studies examined rituximab alone (60 patients overall), compared with rituximab plus DMARD (3947 patients overall), we did not include rituximab alone in our presentation of adverse events. The incidence of any adverse effect ranged from 22% to 95% in the rituximab group and 70% to 88% in the control group, with a mean incidence of 73% and 79%, respectively. The most commonly reported adverse events were infusion related reactions with a higher incidence reported in the rituximab group (mean 29%) compared with the placebo group (mean 18%). Overall infection incidence was similar for both groups (39% for rituximab group versus 40% for placebo). The most commonly occurring infections for both groups were nasopharyngitis, upper respiratory tract infections, urinary tract infections and bronchitis. Serious infections, including cellulitis, pneumonia, gastroenteritis, and pyelonephritis, occurred slightly more frequently in the rituximab group (mean 3.5%) compared with the control group (mean 2.6%). No opportunistic infections or tuberculosis occurred; however, reactivation of herpes virus was observed in the rituximab population (1.1%), but not in the control population.
Notes:
Except where otherwise noted, values are mean(standard deviation);
First column for a given variable represents the control group; the second column represents the rituximab group;
Subgroups in the rituximab group are as follows: First row (DAS = 6.8)- rituximab 500 mg + methotrexate; second row (DAS = 6.7)- rituximab 1000 mg + methotrexate;
Subgroups in the rituximab group are as follows: First row (DAS = 6.8)- rituximab alone; second row (DAS = 6.9)- rituximab + cyclophosphamide; third row (DAS = 6.8)- rituximab + methotrexate;
Subgroups in the rituximab group are as follows: First row (DAS = 6.4)- rituximab 500 mg + methotrexate; second row (DAS = 6.5)- rituximab 1000 mg + methotrexate;
Subgroups in the rituximab group are as follows: First row (DAS = 7.1)- rituximab 500 mg + methotrexate; second row (DAS = 7.0)- rituximab 1000 mg + methotrexate.
Notes:
Except where indicated otherwise, values are total number of patients, mean percentage, range of percent of patients (na%–-nb%) and N’ for total number of patients;
Dosage of Rituximab administered ranged from 500 mg to 1000 mg I.V infusion on days 1 and 14. Some patients were taking background non-steroidal anti-inflammatory drugs including COX2 inhibitors and corticosteroids;
DMARDs, Disease modifying anti-rheumatic drugs, including methotrexate and sulfasalazine, azathioprine, penicillamine, gold, hydroxychloroquine, chloroquine, leflunomide, anakinra, salazosulfapyridine, bucillamine, mizoribine;
Control groups included the following: placebo and DMARDs, and/or DMARD alone and/or NSAID's and/or background corticosteroids and/or Biologics;
All related and unrelated to the drug treatment, including those by concomitant interventions adverse effects seen in the study;
Includes serious and severe adverse effects;
Infusion reaction–-1 or 2nd infusion were headache, hypertension, nausea, pruritus, urticaria, diarrhea, flushing, pyrexia, dizziness, hot flush, throat irritation, tachycardia, ear puritus, oropharyngeal swelling, hypotension, asthma, vomiting, rash, anaphylactic reactions, laryngeal edema, cough, hoarseness, including severe infusion reactions;
Infections were nasopharyngitis, URI, pyrexia, bronchitis, sinusitis, UTI, flu like symptoms, cystitis, erysipelas, burn wound infections, FUO, cutaneous mycosis, oral and vaginal candidiasis, pulmonary infections, bronchiectasis, due to presence and excavation of pulmonary nodule;
Serious infections were infections requiring intravenous antibiotics, hospitalizations, prolongation of hospital stay, causing death.
Sepsis- incluing neutropenic sepsis, septic shock, urosepsis, septic arthritis, staph aureus septecemia;
Abscess–-any part including cornea, foot, abdominal wall, perirectal abscess;
Other serious infections included, gasteroenteritis, bronchitis, cat bit infection, influenza, FUO, de-novo hepatitis, bronchitis, epiglottitis, bursitis, ARDS, Not otherwise specified (NOS).
Other malignancies included Pancreatic neoplasm, Intestinal adenocarcinoma, Renal cell carcinoma, ovarian carcinoma, transitional cell carcinoma, lung adenocarcinoma, squamous cell carcinoma of cervix, oesophageal adenocarcinoma, NOS;
Cerebral vascular accident, Hemorrhagic stroke, convulsions, epilepsy, essential tremor, NOS;
Pulmonary including Interstitial Lung Disease, Status asthamaticus, respiratory insufficiency (NOS);
CVS events include cardiac tamponade, Mmyocardial Iinfarction, Ccoronary artery disease, labile BP, Supraventricular Ttachycardia, Chest pain, hypotension, Thromboangitis obliterans, Pulmonary emboli, arterial emboli, cardio-resp arrest, ventricular asystole, cardiac faliure, NOS;
Musculosketal Disorders include: Arthralgias, fatigue, fractures, process requiring joint surgeries, faliure of implants, lower limb fracture;
Skin manifestations include Rash, Pruritus;
Hematological- thrombocytosis, metorrhagia, myelodysplastic syndrome;
Ob/gyn- spontaneous abortion, ovarian cyst.
Non-melanotic skin carcinoma occurred more commonly than other carcinomas in the rituximab treatment groups (0.8% in rituximab group versus none reported in the control group). Moreover, overall incidence of malignancies was higher in the rituximab group (2.1%) compared with the control group (0.6%). There was only one case of lymphoma (Hodgkin's) reported in the rituximab population.
Mortality rates were relatively similar (1.2% in the rituximab group versus 0.9% in the control group). Withdrawals and discontinuation secondary to adverse events were also the same in both groups (3%). Among patients receiving rituximab, human anti-chimeric antibodies against rituximab were reported in 3% to 23%; however, no increase in drug resistance or opportunistic infections was reported. No studies included pregnant women.
Discussion
Rituximab is currently indicated for the treatment of patients with moderate to severe RA who show no response, experience a loss of response with time, or have adverse effects to anti-TNF alpha agents. 5 The results of the present meta-analysis add to the support for the efficacy of rituximab in RA, as measured by DAS28 and ACR 50. 13
The results of the safety analysis should be interpreted with caution. The percentages reported may be misleading as for some sub-categories the number of patients is small and a single adverse event may substantially change the percentages. The most frequent adverse effects when using rituximab are infusion-related reactions, such as hypotension, hypertension, chills and rash. The reported incidence of infusion-related side effects varied considerably among studies (mean percentage in rituximab group was 28%), although they were surprisingly frequent in the placebo-treated patients as well (mean percentage in placebo group was 18%).
Consistent with a prior systematic review of rituximab, 23 the present meta-analysis did not find an increase in the risk of serious infections compared to placebo during rituximab therapy, although the use of glucocorticoids or the RA itself may increase the risk of serious infections. 24 The fact that no tuberculosis was found likely reflects the fact that all patients are screened for tuberculosis before study entry. While the FDA has received reports of patients who developed fatal progressive multifocal leukoencephalopathy (PML), following rituximab treatment for SLE, 25 there were no cases of PML from JC virus activation reported in any of the studies. In terms of malignancy, non-melanotic skin carcinoma occurred more commonly than other carcinomas in the rituximab treatment groups, as previously described.
The quality of a meta-analysis is always a reflection of the quality of the included studies. All of the trials included in this meta-analysis were moderate to large, blinded, randomized controlled-clinical trials (Finckh et al 18 excepted). Masking of allocation was inconsistently reported and the method used to generate the sequence of randomization was rarely described. However, the absence of these details likely had minimal impact on the clinical endpoints and the mean Jadad score was 3.5 (range: 1 to 4).
Important strengths of this meta-analysis include homogeneity of design and methodology among the included studies. For example, all of the studies assessed the same dosage of rituximab administered at the same timing intervals. Although there was a range of disease durations, there were no significant differences in disease severity, patient age or gender, prior DMARD or anti-TNF agents use between experimental and control groups.
The included studies also used strategies to mitigate bias. For instance, selection bias was minimized in all studies through either consecutive enrollment or a randomization scheme. All studies used prespecified definitions for outcomes measures as a means to limit the effect of interobserver variability.
In addition, the primary and secondary endpoints are well-validated measures of disease outcomes in RA. Finally, the analysis used a random-effects model which, although it gives a higher weight to large studies, has a lower large to small study weighting gradient than fixed-effects models and this helps, in our view, to enhance the credibility of the overall analysis.
Limitations of this meta-analysis include publication bias. It is possible that negative trials of rituximab may not have been published and therefore were not included in this review. However, publication bias risk was minimized by including presentations from ACR Annual meetings and creating funnel plots. Second, the present analyses were limited to the data presented and/or shared by authors of the source studies. In certain cases, there was incomplete information. However, we consulted authors directly in these studies and they kindly supplied additional data, thus mitigating these two potential problems. Third, this meta-analysis was based on aggregate data, not on individual patient data. Thus, it was not possible to explore whether patient factors contributed to the statistical heterogeneity we observed in treatment effects. Fourth, the generalizability of the present findings is limited by the populations enrolled in the analyzed studies. All of the studies (Finckh et al 18 excepted) were international, multi-centered studies and some of the studies included DMARD naïve patients while others included anti-TNF-alpha incomplete responder; however, there were not large numbers of patients in each of these sub-groups, so the results should be applied to general RA patients with caution. Fifth, outcomes beyond 6 months were only assessed in one study, 21 so these results may not reflect long term outcomes.
Despite these limitations, the present meta-analysis and systematic review demonstrates that rituximab is efficacious both in treatment naïve and methotrexate and/or anti-TNF-alpha refractory patients with RA. In addition, the safety review is encouraging in that there are no new signals with respect to infusion reactions, serious infections or malignancy. In fact these data are somewhat re-assuring in this regard. Long-term data regarding rituximab efficacy and safety in RA are awaited.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. ERV, HA + PM report no conflicts of interest. DEP has received research grants or been on advisory boards for Roche, Genentech, Brogen-Ldec, Abbott, Amgen, Centocare, UCB and BMS. The authors confirm that they have permission to reproduce any copyrighted material.