Abstract
Pemetrexed (Alimta, Eli Lilly) is a multi-targeted anti-folate originally approved for its use in malignant mesothelioma. Based on results from phase III clinical investigations, it is now approved for use as a single agent in the second-line setting and in combination with platinum therapy in the first-line setting for advanced non-small cell lung cancer. It is also under investigation in earlier stages of non small cell lung cancer including in the adjuvant setting and with radiation. It has shown to be particularly efficacious for non-squamous histology and is well tolerated. Toxicity includes, but is not limited to hematologic toxicity and gastrointestinal toxicity, which are minimized by vitamin B12 and folic acid supplementation. Recent analyses also suggest cost-effectiveness of this agent in patient with advanced, non-squamous cell non-small cell lung cancer.
Keywords
Introduction
In 2009, lung cancer represented the second leading cause of cancer in the United States with 219,440 new cases and was the leading cause of cancer death with 159,390 deaths in both men and women. 1 Non-small cell lung cancer (NSCLC) accounts for approximately 80% of lung cancer diagnoses, and unfortunately, the majority of patients present at diagnosis with unresectable, advanced disease. 2 In the recent past, advances in the understanding of the biology of lung cancer and importance of histology in NSCLC have contributed to an expansion of therapeutic options and improved clinical outcomes for patients with NSCLC.
Pemetrexed (Alimta, Eli Lilly) is a newer agent for use in NSCLC and is under further investigation for monotherapy and in combination therapy. Originally approved in 2004 for use in malignant mesothelioma, pemetrexed in combination with cisplatin was found to improve response rates (41.3% vs. 16.7%, P < 0.0001), time to progression (5.7 mos. vs. 3.9. mos., P = 0.001), and median overall survival (12.1 mos. vs. 9.3 mos., P = 0.02) in patients with unresectable malignant pleural mesothelioma over cisplatin monotherapy. 3 Pemetrexed has since demonstrated efficacy and tolerability in NSCLC and is now approved as monotherapy for use in the second-line setting and maintenance settings as well as first-line therapy in combination with cisplatin for advanced NSCLC.
Mechanism of Action, Activity
Pemetrexed (LY231514) is a newer generation multi-targeted anti-folate. It has very high affinity for folate receptors and enters the cell through the reduced folate carrier, similarly to methotrexate. It is then polyglutamated intracellularly to its active form, pentaglu-tamide, by folylpolyglutamate synthase (FPGS). The polyglutamated form of pemetrexed is a potent inhibitor of thymidylate synthase (TS), an enzyme needed for the synthesis of thymidylate nucleotides. The inhibitory process forms the primary mechanism of action of the drug in which transformation of deoxyuridine monophosphate (dUMP) to deoxythymidine monophosphate (dTMP) by TS can no longer take place, resulting in decreased thymidine necessary for DNA synthesis and cell replication, thus inhibiting cell growth. In addition to TS, other enzymes such as dihydrofolate reductase (DHFR) glycinamide rebonucleotide formyltransferase (GARFT), are inhibited by pemetrexed. The latter enzyme is involved in the de novo synthesis of purine. The ability of pemetrexed to target multiple enzymes involved in both thymidine and hypoxanthine synthesis pathways accounts for its cytotoxicity and inhibition of cell proliferation.2,4,5
In vitro and in vivo chemo-sensitivity to pemetrexed has been evaluated in numerous studies of various tumor types. TS, GARFT, and DHFR expression appear to inversely correlate with pemetrexed sensitivity. In a study evaluating 61 samples of freshly explanted tumor cells, a concentration-dependent sensitivity to pemetrexed was observed with 12%, 30%, and 46% of samples at 1 μg/ml, 10 μg/ml, and 100 μg/ml of pemetrexed, respectively. Additionally, a statistically significant inverse correlation between TS, GARFT, and DHFR mRNA expression in cell colonies and pemetrexed sensitivity was observed at concentrations of 10 μg/ml and 100 μg/ml, respectively. 6 These data suggest that the clinical efficacy of pemetrexed is potentially related to the concentration of its target enzymes in tumors.
As the primary enzymatic target of pemetrexed, thymidylate synthase expression in various cells has been studied extensively and may have prognostic and predictive significance in NSCLC. In a study of 47 stage I or II lung adenocarcinoma tissue samples, a positive correlation was observed between TS mRNA levels measured by real-time reverse transcriptase-polymerase chain reaction (RT-PCR) and tumor stage (P = 0.004), nodal metastases (P = 0.005), and lymphatic (P = 0.037) or vascular invasion (P = 0.018). In the same study, higher levels of TS correlated with a decreased postoperative cumulative survival (P = 0.029). However, in a multivariate analysis of these variables on survival, TS was not an independent prognostic indicator. 7 In another study of 104 patients with resected pathological stage I lung adenocarcinoma, TS levels were retrospectively evaluated by immunohistochemical (IHC) staining. By multivariate analysis high TS expression and performance status were found to be independent prognostic variables for 5 year overall survival. 8
Thymidylate synthase expression in NSCLC appears to vary by histological subtype in NSCLC. Tumors were evaluated for TS mRNA by RT-PCR and IHC in a study of fifty-six patients with resected stage IA-IIIA NSCLC. The median expression of TS was significantly higher for squamous cell carcinoma than adenocarcinoma by both RT-PCR and IHC. 9
The variability in TS expression by histologic subtype may also contribute to the variation in clinical efficacy of pemetrexed between squamous and non-squamous cell histology. A recent review of two phase III trials in NSCLC analyzed the efficacy of pemetrexed according to histology. The first trial compared pemetrexed to docetaxel in 571 previously-treated patients with NSCLC and the second study compared cisplatin plus pemetrexed versus cisplatin plus gemcitabine in 1,725 untreated patients with advanced NSCLC. Both studies, there demonstrated a statistically significantly improved overall and progression-free survival in patients with non-squamous histology who received pemetrexed. Specifically, patients with adenocarcinoma or large cell carcinoma histology had an improved overall survival when receiving pemetrexed compared to patients with squamous cell histology. 10 This data further supports the prognostic and predictive value of tumor histology in NSCLC as well as the significance of TS expression. Whether measurement of TS by IHC or RT-PCR is superior is still under investigation, but these results further support the predictive and prognostic significance of TS expression in NSCLC. The significance of TS expression may be increasingly important in an era when the prognostic and predictive role of potential biomarkers in malignancies is becoming more established and therapies are more targeted.
Safety and Toxicity
Antifolates have been associated with myelosuppression and gastrointestinal toxicity. These toxicities have halted clinical development of several antifolates. 11 Early clinical trials with pemetrexed demonstrated significant rates of myelosuppression, febrile neutropenia, diarrhea, and mucositis and therefore subsequent attempts have been made to identify associated risk factors in order to minimize these toxicities. Folic acid intake had been identified in a previously studied antifolate agent, LY249543 and based upon this identified risk factor for toxicity the role of folic acid in pemetrexed toxicity was investigated. Subjects who received pemetrexed at 500–600 mg/m2 in phase I and II trials between September, 1995 and November 1995 had toxicity data and had makers of vitamin deficiency collected including total homocysteine (tHcy), methylmalonic acid (MMA), and cystathionine. Increased total homocysteine indicates folate and/or vitamin B12 deficiency. Increased pre-treatment tHcy levels correlated significantly for grade 4 neutropenia (P = 0.0185), grade 4 thrombocytopenia (P = 0.0002), and grade 4 neutropenia with grade 3 or 4 infection (P = 0.0064). There was also a significant correlation between increased pre-treatment MMA levels and grade 3 or 4 diarrhea (P < 0.0001) or grade 3 or 4 mucositis (P < 0.0001). As a result of these findings, clinical trials with pemetrexed require patients to receive supplementation with folic acid (350–1000 μg daily) and vitamin B12 (1000 μg intramuscularly every nine weeks). 11
Dosing and Safety of Pemetrexed
Almost 80% of pemetrexed is bound to plasma proteins, thus resulting in rapid plasma distribution and elimination phases. It is rapidly eliminated from the plasma by urinary excretion with a half-life of 3.5 hours. Due to its predominant renal excretion, its plasma clearance decreases as renal function is reduced with a resultant increase in systemic exposure. Pharmokinetics of pemetrexed is not affected by elevated transaminases or hyperbilirubinemia due to its limited hepatic metabolism. 4
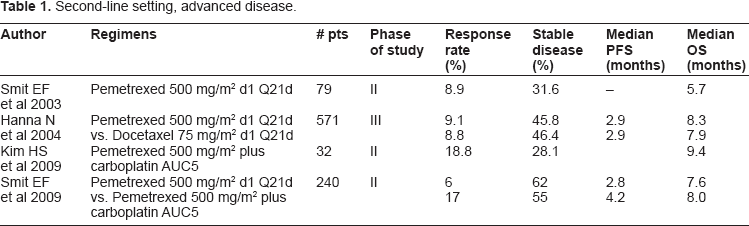
Second-line setting, advanced disease.
First line setting, advanced disease.
In early clinical trials with pemetrexed, prior to the routine use of B12 and folic acid supplementation, patients experienced significant toxicity. Hematologic toxicity included grade 3 and 4 neutropenia, anemia, and occasionally thrombocytopenia. Grade 3 or 4 non-hematologic toxicities that were frequently reported were skin rash, nausea, lethargy, anorexia, arthraligias, stomatitis, and diarrhea. 12 After these toxicities were recognized and after the report of a concurrent Canadian study of pemetrexed in colorectal cancer, the protocol was amended and the dose of pemetrexed was reduced to 500 mg/m2.
Various dosing regimens have been evaluated for pemetrexed. The maximum tolerated doses of pemetrexed and cisplatin were determined in a phase I study and found to be pemetrexed 600 mg/m2 and cisplatin 75 mg/m2 on day one of a 21 day cycle. 13 After studies showed that pemetrexed toxicity could be reduced with vitamin B12 and folic acid supplementation, pemetrexed at 500 mg/m2 was compared to 1000 mg/m2 in a randomized phase II trial as a second-line agent for advanced NSCLC, and has also been compared to 900 mg/m2 in a randomized phase III trial for second-line therapy in advanced NSCLC.14,15 In these studies patients received vitamin B12 and folic acid supplementation prior to and throughout treatment. Both studies demonstrated similar efficacy between the standard dose and the higher dose in response rates, median survival times, and one year survival rates. However, the 900 mg/m2 dose arm of the phase III trial was discontinued at interim analysis because there was a numerically higher frequency of adverse events (although no statistically significant difference).14,15 Accordingly, pemetrexed at 500 mg/m2 has remained as the standard dose for further trials.
Use of Pemetrexed in Lung Cancer
Second line advanced NCSCL Pemetrexed alone in second line NCSCL treatment
There are few chemotherapeutic agents that are considered efficacious as second line therapy for advanced NSCLC. Docetaxel, pemetrexed, and erlotinib are approved by the US Food and Drug Administration (FDA) for second line therapy for patients with advanced NSCLC based on improved quality of life and one-year survival in NSCLC patients with a performance status of 2 or less.16,17
Pemetrexed was approved though as a second-line agent for patients with advanced NSCLC in 2004. A phase II trial used pemetrexed in all patients who had progressed within three months of first-line chemotherapy for advanced disease. Single agent pemetrexed demonstrated clinical efficacy with a response rate of 8.9% in 79 evaluable patients. 18 As a single agent, pemetrexed was compared to docetaxel in a phase III trial for patients with stage IIIB or IV NSCLC who were not eligible for curative therapy who had progressed after only one prior regimen for advanced disease. 541 patients had an ECOG PS of 0–2 with measureable disease and were assigned to receive either pemetrexed 500 mg/m2 or docetaxel 75 mg/m2 on day 1 of a 21-day cycle. All patients who received pemetrexed were given vitamin supplementation. This study demonstrated similar efficacy for pemetrexed as docetaxel for overall response rate (9.1% and 8.8%, P = 0.105), median survival of 8.3 months versus 7.9 months (P not significant), and one year survival rate (29.7% in each arm). However, patients who received pemetrexed experienced less toxicity compared to those receiving docetaxel with less grade 3 or 4 neutropenia (5.3% vs. 40.2%, P < 0.001), febrile neutropenia (1.9% vs. 12.7%, P < 0.001), infections with neutropenia (0.0% vs. 3.3%, P = 0.004), hospitalizations for neutropenic fever (1.5% vs. 13.4%, P < 0.001), use of granulocyte colony-stimulating factor support (2.6% vs. 19.2%, P < 0.001) and alopecia (6.4% vs. 37.7%, P < 0.001). 19 Pemetrexed is a good option for patients with advanced disease who have progressed after first-line therapy.
Front-Line Therapy for Advanced NSCLC–-Platinum-Based Regimens
Platinum-based doublet therapy has historically formed the cornerstone of first-line therapy for patients with advanced NSCLC in patients with a good performance status. Platinum-based doublet therapy has been shown to improve the risk of death and one-year survival over best supportive care.20,21 A meta-analysis recently performed on 11 randomized phase III trials of platinum-based versus non-platinum-based combinations for the treatment of previously untreated advanced NSCLC demonstrated that patients who received a platinum-based regimen had a significant reduction in risk of death at one year with a survival rate at one year of 40% in the platinum-based arms versus 37% in the non-platinum based arms, respectively (OR: 0.88, 95% CI [.78–.99]; P = 0.044). There was no statistically significant difference in the risk of death due to toxicity between platinum and non-platinum containing regimens. 22
Efficacy of platinum-based versus non-platinum-based combination therapy was further evaluated in another meta-analysis including all randomized trials comparing platinum-based with non-platinum-based doublet regimens as first line treatment in advanced NSCLC. A total of 17 trials, with 4920 patients were included for analysis. This study concluded that the use of platinum-based doublet therapy, particularly with cisplatin, improved survival at one year (relative risk (RR) = 1.16, 95% CI 1.06–1.27; P = 0.001), complete response (RR = 2.29, 95% CI 1.08–4.88; P = 0.03), and partial response (RR = 1.19, 95% CI 1.07–1.32; P = 0.002), but increased anemia, neutropenia, neurotoxicity, and nausea. Interestingly, carboplatin was not associated with an increased one-year survival rate compared to non-platinum doublets and there was a small, but significant difference in the effect of cisplatin compared to carboplatin (P = 0.05) in favor of cisplatin. 23
Pemetrexed and Cisplatin in Combination
Pemetrexed in combination with cisplatin was originally approved as a first line therapy for malignant pleural mesothelioma. As previously discussed, pemetrexed in combination with cisplatin was found to improve response rates, time to progression, and median overall survival in patients with unresectable malignant pleural mesothelioma compared to cisplatin monotherapy. 3
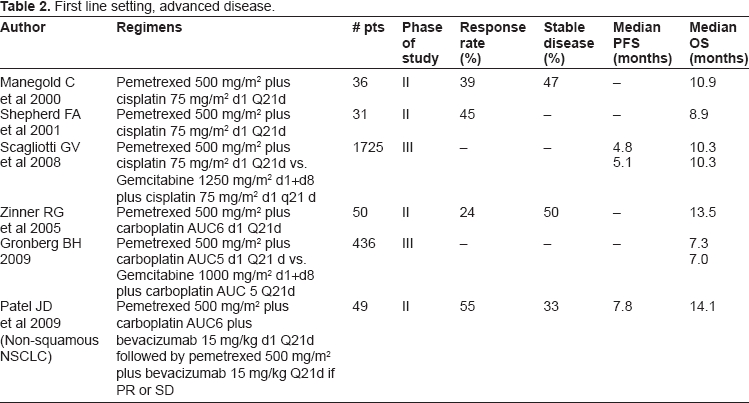
Two phase II trials of pemetrexed in combination with cisplatin showed activity in NSCLC and good tolerability.24,25 The first trial was a multicenter phase II study which assigned 36 chemo-naive patients with stage IIIB or IV NSCLC and an ECOG PS 0–2 to pemetrexed 500 mg/m2 plus cisplatin 75 mg/m2 on day one of a 21 day cycle with a median of 4 cycles given (range 1–11 cycles). This study demonstrated a partial response (PR) rate of 39% and stable disease in 47% of patients and a median survival of 10.9 months. Hematologic toxicity was the predominant toxicity with 59% of patients experiencing grade 3 or 4 granulocytopenia without fever or infection, 14% had grade 3 anemia, and 17% had grade 3 or 4 thrombocytopenia. 24 The second phase II trial also assigned pemetrexed 500 mg/m2 plus cisplatin 75 mg /m2 on day 1 of a 21 day cycle to 31 patients with untreated stage IIIB or IV NSCLC and an ECOG PS of 0–2. Twenty-nine patients were evaluable for response and received a median of 6 cycles. Results demonstrated an overall response rate of 45% and median overall survival of 8.9 months and 1-year survival rate of 49%. Hematologic toxicity was again the most common toxicity with 33% experiencing grade 3 or 4 granulocytopenia with only one episode of febrile neutropenia. 25
Based on efficacy observed in earlier phase trials, the combination of pemetrexed and cisplatin was evaluated in a multinational non-inferiority phase III trial for patients with stage IIIB or IV NSCLC and an ECOG PS of 0–1. 26 1,725 patients were randomly assigned to receive cisplatin 75 mg/m2 on day 1 plus gemcitabine 1250 mg/m2 on days 1 and 8 of a 21 day cycle (CG arm) or cisplatin 75 mg/m2 plus pemetrexed 500 mg/m2 (CP arm) on day one of a 21 day cycle for a maximum of 6 cycles. In this trial, the median overall survival was non-inferior for the CP arm compared to the CG arm and both had a median OS of 10.3 months. PFS between the CP arm and CG arm were 4.8 months and 5.1 months, respectively. Furthermore, objective response rates were also non-inferior between the CP arm and CG arm and were 30.6% and 28.2%, respectively.
Interestingly, in a pre-planned analysis the investigators did observe a statistically significant treatment-by-histology interaction (P = 0.0011). In patients with adenocarcinoma (n = 847) those who received the CP arm compared to the CG arm had an OS of 12.6 months vs. 10.9 months, respectively (P = 0.03). Patients with large cell carcinoma (n = 153) had an improved survival as well with CP vs. CG (10.4 months vs. 6.7 months; P = 0.03). Patients with squamous-cell histology appeared to have improved survival with the CG arm (n = 229) versus the CP arm (n = 244; 10.8 months vs. 9.4 months; P = 0.05). Other factors found to be significantly associated with prognosis and survival and appeared treatment-independent were sex, race, performance status, and disease stage (P < 0.05). For example, the median overall survival for females to males was 13.3 months versus 9.6 months in the CP arm and 11.4 months versus 9.9 months in the CG arm. The median OS for East or Southeast-Asians compared to Caucasians was 13.8 months versus 10.0 months, respectively in the CP arm and 11.9 months versus 10.1 months, respectively, in the CG arm.
The safety of CP and CG was also compared and the cisplatin plus pemetrexed arm had a more favorable toxicity profile compared to the cisplatin plus gemcitabine arm. The CP arm versus the CG arm experienced less neutropenia (15% vs. 27%, respectively; P ≤ 0.001), anemia (6% vs. 10%, respectively), thrombocytopenia (4% vs. 13%, respectively), grade 3 or 4 febrile neutropenia (1% vs. 4%, respectively; P = 0.002), transfusions (16.4% vs. 28.9%, respectively, P < 0.001), use of erythropoietic factors (10.4% vs. 18.1%, respectively; P < 0.001), granulocyte colony stimulating factors (3.1% vs. 6.1%, respectively; P = 0.004), and alopecia (12% vs. 21%, respectively; P < 0.001). 26
This data was recently re-analyzed retrospectively to evaluate the subset of patients who were East-Asian (EA) from either Korea or Taiwan (n = 126, 7.3%). East-Asian patients had a significantly longer OS compared to the non-EA study population with a median survival that was approximately 6 months longer than the median overall survival for the entire study population irrespective of which treatment was received (HR of EA patients compared to non EA patients was 0.5, 95% CI = 0.39–0.64; P < 0.001). This further supports ethnicity as an independent prognostic factor in NSCLC. 27 There were also similar trends in the EA subset towards improved PFS, OS, and ORR for patients who received CP compared to CG but due to the small number of patients, these results were not statistically significant. 27
Pemetrexed and Carboplatin Combination
Pemetrexed combined with carboplatin as a first-line regimen in advanced NSCLC is efficacious. In a phase II study, 56 patients with stage IIIB or IV NSCLC and an ECOG PS of 0–1 received pemetrexed 500 mg/m2 plus carboplatin AUC 6 on day one of a 3-week cycle. The partial response rate was 24%, one-year survival was 56%, and median survival was 13.5 months. These results were comparable other platinum containing doublets for advanced NSCLC and this regimen was well tolerated. 28
Pemetrexed combined with carboplatin has been compared to gemcitabine plus carboplatin in a randomized phase III trial as a first-line regimen in advanced NSCLC. Patients with stage IIIB or IV NSCLC and an ECOG PS 0–2 received pemetrexed 500 mg/m2 and carboplatin AUC5 (PC arm) on day one of a 21 day cycle or gemcitabine 1000 mg/m2 on days 1 and 8 plus carboplatin AUC5 (GC arm) on day 1 of a 21 day cycle for a maximum of four cycles. Patients who were 75 years or older received an automatic dose reduction in treatment of 25%. The primary outcomes of this study were global quality of life measured by the European Organization for Research and Treatment of Cancer (EORTC) quality of life questionnaire (QLQ), nausea or vomiting, fatigue, and dyspnea. Secondary endpoints were overall survival and toxicity. Significantly more patients in the PC arm compared to the GC arm completed four cycles (72% vs. 62%, respectively; P = 0.03), four cycles without delays (58% vs. 44%, respectively; P = 0.004), and four cycles without dose reductions (50% vs. 20%, respectively; P < 0.001). There were no significant differences in the primary health related quality of life (HRQoL) endpoints of global QoL, nausea or vomiting, fatigue, or dyspnea. Furthermore, there were no differences between treatment arms for median OS (n = 436; PC: 7.3 months, GC: 7.0 months; P = 0.63) or one year survival rate (PC: 34%, GC: 31%). The investigators did not observe a significant difference in OS or 1 year survival rate by histology. However, there was significantly more toxicity in the GC arm patients compared to the PC arm for grade 3 or 4 leukopenia (46% vs. 23%, respectively; P < 0.001), neutropenia (51% vs. 40%, respectively; P = 0.024), thrombocytopenia (56% vs. 24%, respectively; P < 0.001), need for red blood cell transfusions (43% vs. 29%, respectively; P = 0.003) and platelet transfusion (9% vs. 3%, respectively; P = 0.007). 29 Based on these results, pemetrexed and carboplatin are considered a reasonable front-line combination therapy for advanced non-squamous NSCLC and offers a more favorable toxicity profile compared to gemcitabine plus carboplatin.
Early Trials in the Second-Line Setting: Pemetrexed plus Carboplatin
The role of platinum therapy in the second-line setting combined with pemetrexed has been investigated. In a randomized phase II study, single agent pemetrexed 500 mg/m2 (arm A) was compared to pemetrexed 500 mg/m2 with carboplatin area under the curve (AUC) 5(arm B). 30 240 patients were enrolled with an ECOG PS 0–2 and stage IIIB or IV disease who had progressed at least three months after platinum containing regimens. The median time off of platinum therapy for arm A was 7.2 months and 8 months in arm B. There was a significantly increased TTP, the primary endpoint, in arm B compared to arm A (4.2 months vs. 2.8 months, P = 0.005). However, there were no significant differences between arm A and arm B in the secondary endpoints of objective response rate (17% vs. 6%), overall survival (7.6 months vs. 8.0 months), or toxicity between the two treatment arms. However, performance status and time since previous chemotherapy were significantly related to OS (P < 0.001 and P = 0.002). 30
A more recently published open-labeled phase II trial assigned 32 patients with inoperable stage IIIB or IV NSCLC to pemetrexed 500 mg/m2 plus carboplatin area under the curve 5 every three weeks. This study demonstrated similar results to that of the randomized phase II study with an overall response rate of 18.8% and median overall survival of 9.4 months. 31 Although median OS was comparable to the study by Smit et al differences in time to progression (TTP) may be secondary to differences in the patient populations between trials. This study enrolled very few women, had a higher than normal percentage of squamous-cell histology and shorter interval between previous therapy and progression of disease. Further studies are needed to evaluate the role of platinum combinations in the second-line setting for advanced NSCLC.
Pemetrexed in Combination with Targeted Agents
Pemetrexed plus bevacizumab
Bevacizumab is a monoclonal antibody to vascular endothelial growth factor (VEGF) that has been shown to improve survival in one randomized phase III study for patients with stage IIIB or IV non-squamous NSCLC who received carboplatin and paclitaxel with or without bevacizumab. 32 This regimen is approved in the United States for the frontline therapy of advanced non-squamous cell NSCLC. Given bevacizumab's activity in NSCLC, pemetrexed 500 mg/m2 and carboplatin AUC 6 were combined with bevacizumab 15 mg/kg on day one of a 3 week cycle for up to six cycles in a phase II trial of patients with previously treated stage IIIB or IV non-squamous NSCLC. Patients who experienced a response or had stable disease after 6 cycles continued maintenance pemetrexed and bevacizumab every 3 weeks until disease progression or unacceptable toxicity. The objective response rate was 55%, median PFS 7.8 months, and median OS was 14.1 months. The toxicity profile was acceptable. 33 Further studies addressing the role of maintenance therapy will be discussed.
These results served as the basis for a randomized phase III trial comparing pemetrexed, carboplatin, and bevacizumab to a standard of care arm. 900 patients with stage IIIB or IV NSCLC will be enrolled in a multi-center study to receive pemetrexed 500 mg/m2 plus carboplatin AUC 6 plus bevacizumab 15 mg/kg on day one of a 21 day cycle for up to four cycles, followed by maintenance pemetrexed plus bevacizumab or paclitaxel 200 mg/m2 plus carboplatin AUC 6 plus bevacizumab 15 mg/kg on day one of a 21 day cycle up to four cycles followed by maintenance bevacizumab. The primary endpoint is to compare OS between the two arms and secondary endpoints include efficacy, safety, quality of life, pharmacokinetics, and to evaluate correlative biomarkers. If the primary endpoint is met, this study will provide a well-tolerated alternative for advanced non-squamous NSCLC. It will also help to determine if maintenance pemetrexed improves outcomes when added to maintenance bevacizumab. 34
Future Directions for the Second Line Setting
Pemetrexed is being evaluated in combination with a number of novel therapies for the second line treatment of advanced NSCLC. Vandetanib is an oral inhibitor to vascular endothelial growth factor receptor (VEGFR), endothelial growth factor receptor (EGFR) and RET (rearranged during transfection) signaling that is orally administered. It has been evaluated in a randomized double blind phase III trial for patients with stage IIIB or IV NSCLC as a second line therapy. The primary endpoint was PFS and secondary endpoints were OS, ORR, and time to deterioration of symptoms (TDS). Patients received vandetanib 100 mg daily plus pemetrexed 500 mg/m2 every three weeks or placebo plus pemetrexed 500 mg/m2 every three weeks for a maximum of 6 cycles. There were significant differences in ORR (19.1% vs. 7.9%; P < 0.001) and TDS (HR 0.61; P = 0.004). There were trends towards improved PFS (HR = 0.86; 97.58% CI 0.69–1.06; P = 0.108) and OS (HR = 0.86; 97.54% CI 0.65–1.13; P = 0.219). Furthermore, there appeared to be reduced toxicity in the combination arm compared to the pemetrexed arm. While this trial did not meet its primary endpoint, the investigators concluded that there does appear to be efficacy with this combination and has a tolerable side effect profile. 35
Lapatinib is an oral small molecule inhibitor of EGFR and human epidermal growth factor receptor 2 (HER-2). In a phase I trial of patients with stage IIIB or IV NSCLC who had received one previous chemotherapy regimen, daily lapatinib was combined with pemetrexed in a dose escalation trial. 18 patients were treated and 3 patients experienced a partial response. Grade 3 or 4 toxicity included lymphocytopenia, neutropenia, diarrhea, nausea, decreased ejection fraction, and increased alkaline phosphatase and the optimal dose was found to be lapatinib 1250 mg daily and pemetrexed 500 mg/m2 IV every twenty one days. 36
Another combination that has been studied in the second line setting is pemetrexed and enzastaurin, an oral inhibitor to serine and threonine kinase, which targets the protein kinase C (PKC) and AKT pathways. Inhibition of these pathways can lead to decreased oncogene expression and increased apoptosis. 160 patients with stage IIIA, IIIB, or IV NSCLC who had progressive disease after one prior systemic chemotherapy for advanced disease at least 2 weeks prior to enrollment received either pemetrexed alone, or enzastaurin 375 mg three times a day on day 1 and 250 mg twice daily after day one for a 28 day cycle plus pemetrexed 500 mg/m2 on day 8 of the first cycle, and on day one of 21 day cycles after cycle one. However, at interim analysis, this study did not reach any of its endpoints and did not appear to have improved efficacy over pemetrexed alone. 37
Maintenance Therapy with Pemetrexed
The role of pemetrexed in a maintenance setting is not fully defined. In addition to the multi-center phase II trial that treated patients with maintenance pemetrexed and bevacizumab after response or stable disease on pemetrexed, carboplatin, and bevacizumab, maintenance pemetrexed has also been compared to best supportive care in patients with stage IIIB or IV NSCLC who had not progressed on four cycles of first-line platinum based chemotherapy. This randomized double blind phase III trial assigned 663 patients in a 2:1 ratio to pemetrexed 500 mg/m2 every three weeks plus best supportive care or placebo plus best supportive care. Patients who received pemetrexed maintenance had a statistically significant improved progression free survival (4.3 months vs. 2.6 months; HR 0.50 CI 0.42–0.61; P < 0.0001) and overall survival (13.4 months vs. 10.6 months; HR 0.79, P = 0.012) compared to patients who received placebo and best supportive care. There was also a histology-dependent interaction observed with improvements in PFS and OS limited to non-squamous histology. 38 However, this study does not address the role of pemetrexed maintenance after receiving pemetrexed in the first-line setting. A randomized trial that will compare maintenance pemetrexed to placebo in patients with non-squamous NSCLC who have not progressed after initial therapy with pemetrexed and cisplatin is planned.39,40
Future Directions for Locally Advanced Disease
Pemetrexed is under investigation for use in patients with locally advanced disease. One phase I trial administered cisplatin 80 mg/m2 and pemetrexed 500 mg/m2 concomitantly with radiotherapy 66 gray (Gy) administered over 45 days (2 Gy per day, 5 days per week) to patients with stage III NSCLC that was not surgically resectable. Unfortunately, this study was closed prematurely due to poor accrual after four patients were enrolled and treated. However, none of these patients experienced dose-limiting toxicity and all received 3 cycles of chemotherapy without dose reduction and no interruptions in radiotherapy. One of the four patients died in a car accident, two achieved partial response and one experienced a complete response. One patient died of progressive disease at 21 months and two are alive at three years. Although closed prematurely, the investigators of this trial conclude that pemetrexed and cisplatin at full doses is a tolerable regimen with radiotherapy and shows activity in locally advanced NSCLC. 41 Another phase I trial presented in abstract form also supports that pemetrexed 500 mg/m2 plus either carboplatin AUC5 or cisplatin 75 mg/m2 every three weeks, with radiotherapy is well tolerated. 42
A phase II Cancer and Leukemia Group B (CALGB) study has also been reported in abstract form. Previously untreated patients with stage III NSCLC received thoracic radiation at 70 Gy with carboplatin AUC 5 and pemetrexed 500 mg/m2 on day 1 of a 21 day cycle with (arm B) or without (arm A) weekly cetuximab for 6 weeks. All patients received an additional four cycles of pemetrexed 500 mg/m2 for consolidation. For arm A and arm B, response rates were 73% and 71%, respectively; median survival 22.3 months and 18.7 months, respectively. 43
Future Directions in the Early Stage, Adjuvant Setting
Pemetrexed is also being introduced in early stage disease as adjuvant therapy. The TREAT protocol (Trial on Refinement of Early stage non-small cell lung cancer. Adjuvant chemotherapy with pemetrexed and cisplatin versus vinorelbine and cisplatin) is an ongoing prospective, multi-center, open label randomized phase II study in patients with stage IB, IIA, IIB, or T3N1 disease. After surgical resection, patients will be randomized to either 4 cycles of vinorelbine 25 mg/m2 on days 1, 8, 15, and 22 and cisplatin 50 mg/m2 on day 1 and 8 for four cycles (VC arm) or pemetrexed 500 mg/m2 and cisplatin 75 mg/m2 (PC arm) on day1 of a 21 day cycle for four cycles. The primary objective of this study is to compare the feasibility of these two regimens and secondary objectives will be efficacy. 44
Survival without Toxicity
It is often difficult to weigh the risks and benefits between efficacy and morbidity from toxicity when comparing various regimens. Investigators have attempted to quantify a risk benefit analysis to better compare chemotherapeutic regimens with similar efficacy but different side effect profiles. A retrospective risk-benefit analysis was performed on the data from the randomized phase III trial of pemetrexed versus docetaxel for second line therapy for advanced NSCLC. The outcome evaluated was the survival without grade 3 or 4 toxicity, which was defined as the time from the date of randomization to the first date of any grade 3 or 4 toxicity or death for 541 patients who received treatment. Patients who received pemetrexed had a statistically significantly longer survival time without grade 3/4 toxicity compared to the patients who received docetaxel (HR = 0.06; 95% CI 0.50–0.72; P < 0.0001). The median survival time without toxicity was 1.2 versus 0.4 months, respectively. When specific grade 3/4 toxicities were analyzed (neutropenia >5 days, febrile neutropenia, anemia, thrombocytopenia, fatigue, nausea, vomiting, diarrhea, stomatitis, and neurosensory events) the median survival time without selected grade 3/4 toxicity was 5.8 months and 1.2 months for pemetrexed and docetaxel, respectively. 45
Scagliotti et al performed a similar risk-benefit analysis on the data from a phase III trial of pemetrexed plus cisplatin versus gemcitabine plus cisplatin for first line treatment in advanced NSCLC and retrospectively analyzed the survival without grade 3/4 toxicity. They determined that the survival without grade 3/4 toxicity was longer in patients who received pemetrexed plus cisplatin versus gemcitabine plus cisplatin. Furthermore, when the non-squamous cell histology was analyzed separately, there was also an improved survival without toxicity observed for the pemetrexed plus cisplatin arm versus the gemcitabine plus cisplatin arm (HR = 0.64; P < 0.001) but this effect was not seen within the squamous-cell population. 46
Cost-Effectiveness
Cost-effectiveness is an important consideration when choosing a chemotherapeutic regimen and therefore, investigators have started to evaluate the cost-effectiveness of pemetrexed. A cost-effectiveness analysis was performed by Klein et al that compared cisplatin plus pemetrexed to three other first-line regimens for advanced NSCLC. They evaluated the cost per life-year gained (LYG) during two years by a person who was starting first-line therapy for advanced NSCLC and also calculated the incremental cost-effectiveness ratios (ICERs) and incremental cost-utility ratios of cisplatin plus pemetrexed compared to three other first line regimens. In this analysis, the incremental LYG for cisplatin plus pemetrexed to cisplatin plus gemcitabine was $104,577 and $231,291 for cisplatin plus pemetrexed to carboplatin plus paclitaxel for all patients with advanced NSCLC, regardless of histology. In the US $100,000 is often cited as the “cost-effective” threshold. However, in a subset of patients with non-squamous histology, another analysis was performed and calculated the incremental LYG was $83,537 for cisplatin plus pemetrexed to cisplatin plus gemcitabine and $178,613 for cisplatin plus pemetrexed to carboplatin paclitaxel. So, based on this model, it appears that the combination of cisplatin plus pemetrexed is cost-effective as a first line therapy, particularly in patients with non-squamous histology. 47
Another cost-effectiveness analysis in Spain was performed that compared pemetrexed to docetaxel in advanced NSCLC with non-squamous histology and further supports the use of pemetrexed as a cost-effective regimen. The investigators calculated that comparing pemetrexed to docetaxel resulted in an ICER of €23967 ($32,308) per quality adjusted life year (QALY) and €17225 ($23,219). In Spain, the accepted threshold for QALY is €30,000 ($40,440). 48 Both of these cost-effectiveness analyses suggest that pemetrexed based regimens, particularly in patients with non-squamous cell histology, are cost-effective compared to similarly clinically efficacious regimens.
Pharmacogenetics
Proteins that are thought to be responsible for pemetrexed transport, activation, and inactivation, are the reduced folate carrier SLC19A1, folylpolyglutamate synthase (FPGS), and gamma-glutamyl hydrolase (GGH), respectively. Studies of these proteins with methotrexate, a related agent, suggest these mutations in these proteins may confer resistance to methotrexate. In the phase II trial of pemetrexed and bevacizumab in the second line therapy for patients with advanced non-squamous NSCLC the exon 6 (2522) C→T polymorphism in SLC19A1 correlated with 3 month progression free status and PFS (P = 0.01 and P = 0.05, respectively) and the IVS1 (1307) C→T polymorphism in GGH correlated with OS (P = 0.04). 49
In another phase II trial of advanced NSCLC patients comparing pemetrexed to pemetrexed plus carboplatin in platinum pre-treated patients, polymorphisms of TS, the reduced folate carrier y-glutamyl hydrolase, and MTHF by peripheral blood were investigated. Patients with a homozygous mutation for MTHFR 1298C had a significantly increased PFS compared to wild type or heterozygous mutations. 30 Future directions studying pharmacogenomics may help in predicting who may benefit from pemetrexed.
Conclusion
The treatment options for non-small cell lung cancer are improving with new therapeutics under investigation to improve efficacy and minimize toxicity. Pemetrexed is one such agent that has proven efficacious and well tolerated in second-line and-first-line therapy for advanced NSCLC. It is also promising as a chemotherapy that is cost-effective for certain subsets of the population who develop NSCLC. Future directions for pemetrexed include evaluation of this agent in earlier stages of NSCLC and in combination with other, more targeted agents in enriched patient populations so that therapy can become more individualized.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
