Abstract
Background
In the event of an MI, the presence of LV systolic dysfunction with or without heart failure is associated with a poorer prognosis. Elevation of aldosterone levels also results in a poor prognosis after an MI.
Objective
This review discusses the cardiovascular benefits of blocking the aldosterone receptor using eplerenone therapy in post MI patients.
Methods
A literature search of Pubmed was conducted for relevant English language articles published between 1990–2010 using the search terms eplerenone, aldosterone, myocardial infarction, heart failure and endothelial dysfunction. Additional articles were obtained from reference lists of identified publications.
Results
Eplerenone is a selective aldosterone receptor blocker that has been shown by a landmark study, EPHESUS to reduce all cause mortality, cardiovascular mortality and heart failure admissions in post MI patients with LV systolic dysfunction and heart failure or diabetes. It blocks the damaging effects of aldosterone including LV remodeling, endothelial dysfunction and arrhythmogenicity. Due to its selective action, there are less sexual side effects compared with spironolactone therapy. However, a small risk of hyperkalemia and renal dysfunction is associated with eplerenone therapy.
Conclusions
Eplerenone therapy should be commenced for post MI patients with LV systolic dysfunction and heart failure or diabetes provided there are no contraindications.
Introduction
Coronary artery disease is a significant public health problem in the developing and developed world. There have been advances in medical therapy and in coronary intervention, which have resulted in better mortality rates. However if an MI is associated with left ventricular (LV) dysfunction with or without heart failure, the prognosis is considerably worsened.1–3 These patients have longer hospital stays, higher rates of rehospitalization, higher in-house mortality and higher 6 months mortality rates. Therefore, it is important to introduce therapy that could improve their outcomes.
After an MI, there is acute neurohormonal activation which returns back to baseline rapidly unless associated with LV dysfunction when it tends to persist. 4 Aldosterone levels (plasma levels and local production by the heart) are elevated5–8 after an MI and such elevated levels have been found to be deleterious. This makes aldosterone a prime therapeutic target as in landmark studies such as the Randomized Aldactone Evaluation Study (RALES) 9 and Eplerenone Post-Acute Myocardial Infarction (EPHESUS). 10
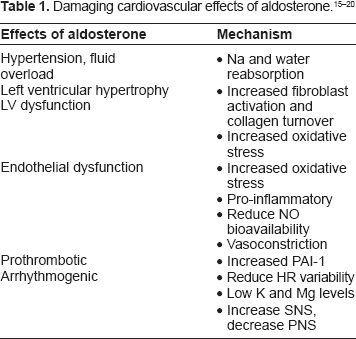
Aldosterone is mainly produced by the zona glomerulosa of the adrenal cortex although extra-adrenal sources include the heart, brain and vasculature. In a failing heart, the presence of increased expression of the aldosterone synthase gene leads to local aldosterone production by the heart. 11 After an MI, activation of the renin-angiotensin-aldosterone system (RAAS) also leads to an elevation of angiotensin II levels, thereby stimulating aldosterone production further. Other stimuli for aldosterone production include increased serum potassium levels, ACTH, catecholamines, AVP and endothelin. Aldosterone has detrimental cardiovascular effects resulting in adverse cardiovascular outcomes (see below).
Begui et al showed that high plasma aldosterone levels in ST elevation MI (STEMI) patients was associated with an increased rate of in-hospital mortality, cardiovascular mortality, heart failure, ventricular fibrillation and resuscitated cardiac arrest. 12 Additionally, Palmer et al showed that in a 5 year follow up of patients with STEMI and NSTEMI, the highest tertile aldosterone level (>141 pmol/l) was a significant independent predictor of mortality (HR 2.19, P = 0.018) and heart failure hospitalizations (tertile 3: 10.3%, tertile 1: 5.43%, P = 0.033). 13 In a neurohormonal substudy of SAVE, the aldosterone level at 3 months in post MI patients with asymptomatic LV dysfunction was associated with having severe heart failure at 2 years. 14 We would like to highlight that these studies only indicate an association of high aldosterone levels in post MI patients and not causation. Therefore, we cannot conclude from these studies that reducing aldosterone levels post MI will reduce cardiovascular morbidity or mortality.
Aldosterone independently produces cardiovascular target organ damage. Aldosterone acts via both slow genomic and fast non-genomic effects. Aldosterone retains sodium and water in the kidneys resulting in increased blood pressure, blood volume and peripheral oedema. Aldosterone is also proinflammatory and prothrombotic, reduces nitric oxide (NO) availability, 15 promotes vasoconstriction, causes endothelial dysfunction 16 and increases oxidative stress via NADPH oxidase superoxide production. Additionally, it is pro-arrhythmogenic by producing increased sympathetic activation, 17 reduced parasympathetic activity, 18 reduced heart rate variability 19 and increased loss of potassium and magnesium. Aldosterone stimulates collagen synthesis and myocardial fibrosis resulting in LV remodelling and LVH. 20 Aldosterone also feeds back on the RAAS and upregulates ACE and angiotensin II type 1 receptors. Hence, it is important to block the actions of aldosterone post MI and in heart failure.
Aldosterone levels are incompletely suppressed by ACE inhibitors and angiotensin receptor blockers. The RALES 9 and EPHESUS 10 study have respectively shown the benefits of prescribing aldosterone receptor blockers to chronic heart failure patients or post MI heart failure patients on top of optimal medical therapy including beta-blockers and ACE inhibitors.
The two currently available aldosterone antagonists are spironolactone and eplerenone. The latter is licensed in the UK and Europe to treat heart failure whilst in the US it is licensed to treat both heart failure and hypertension. Spironolactone is an effective mineralocorticoid receptor blocker that was synthesized in 1959. The RALES study showed that the addition of spironolactone to chronic heart failure patients NYHA class III and IV already on ACE inhibitors, diuretics and in most cases digoxin resulted in a 30% reduction in mortality rate and a 35% reduced rate of heart failure admissions. 9 However, due to its non-selectivity it also acts as an antagonist against androgen, glucocorticoid and progesterone receptors thereby resulting in undesirable sexual side effects such as gynaecomastia and impotence in men and menstrual irregularities in women. Therefore, eplerenone, a selective aldosterone receptor antagonist was derived from spironolactone in 1984 in order to avoid the sexual side effects whilst retaining its aldosterone blocking actions. This review discusses the cardiovascular benefits of eplerenone therapy in post MI patients.
Methods
A literature search of Pubmed was conducted for English language articles published between 1990–2010 using the search terms eplerenone, aldosterone, myocardial infarction, heart failure and endothelial dysfunction. Relevant articles were identified and additional articles were obtained from reference lists of identified publications.
Mechanisms of Action
Eplerenone is a selective, competitive mineralocorticoid receptor blocker which was developed from spironolactone by adding the 9α, 11α epoxy group and substituting the 17α-thioacetyl group with a car-bomethoxy group. 21
Eplerenone is more selective for the mineralocorticoid receptor than for the other steroid receptors mentioned above. In rats, the IC50 of eplerenone for the aldosterone receptor was 360 nmol/L compared to >10,000 nmol/L for the androgen, progesterone and estrogen receptors. 21 Compared to spironolactone, eplerenone itself is less potent i.e. it has 10- to 20- fold reduced affinity in vitro for the aldosterone receptor and is 50 to 75% as potent. 21 The mineralocorticoid receptors are located in epithelial cells (such as in the kidneys) and non-epithelial tissues such as brain, heart and vasculature.
Metabolism
Eplerenone undergoes hepatic metabolism by cytochrome P4503A4 into inactive metabolites, which have a relatively short elimination half-life of 4–6 hours. Its half-life is much shorter than spironolactone because the latter has active metabolites. Steady state is reached within 2 days and mean peak plasma level is achieved about 1.5 hours post oral intake. Food doesn't affect absorption. About 50% of eplerenone is bound to plasma proteins especially alpha 1-acid glycoproteins. Eplerenone has an apparent volume of distribution of 43 to 90 L whilst the apparent plasma clearance is 10 L/hr. 21
Pharmacokinetic Profile
The pharmacokinetics of eplerenone does not differ according to gender. Maximum concentration and the area under the curve concentration (AUC) of eplerenone are increased in ≥65 years old, renal and hepatic impairment. Haemodialysis doesn't remove eplerenone. Caution should be given to concomitant use of CYP3A4 inhibitors such as ketoconazole, erythromycin, verapamil and saquinavir as they result in increased plasma levels of eplerenone. 21 On the other hand, inducers of CYP3A4 such as St John's wort result in 30% reduced plasma level.
Cardiovascular Effects of Eplerenone
LV remodelling
LV remodelling occurs post MI partly because aldosterone stimulates cardiac collagen synthesis and fibroblast activation. 20 LV dilatation, hypertrophy and reactive fibrosis remote from the infarct site results in further LV dysfunction and heart failure.
Experimental studies have shown that eplerenone post MI reduces adverse LV remodelling.22,23 It reduces the increase in LVEDP and LVEDV whilst improving LVEF and diastolic dysfunction.23–25 Eplerenone reduces LV fibrosis in the remote noninfarcted myocardium, which plays a key role in LV remodelling. Originally there was concern that the antifibrotic effect of aldosterone blockade might weaken the scar tissue which replaces the infarcted myocardium but in fact it does not seem to have any detrimental effects on the essential infarct tissue healing. 22 In non-infarct areas of myocardium, eplerenone attenuates activation of key transcriptional factors, activator protein 1 (AP-1) and nuclear factor (NF-KB), collagen types I and III mRNA levels, and downregulation of SERCA2 ATPase. Additionally, it attenuates LV expression of AT-1 receptor, ACE and endothelin-1 gene.24,25 In human studies such as EPHESUS, eplerenone is shown to suppress circulating biomarkers of collagen turnover (PINP and PIIINP). 26
Weir and colleagues assessed the effects of eplerenone on LV remodelling using contrast enhanced cardiac MRI in 100 patients post MI with LVSD and optimal medical therapy but without heart failure or diabetes. 27 In this double blind, placebo controlled study, patients were recruited 1–14 days post MI and followed up for 24 weeks. At a mean dose of 43.7 mg, eplerenone only had a very modest antiremodelling effect, after adjusting for 15 covariates. Eplerenone, compared to placebo, reduced the primary end point (change in LV end-systolic volume index) by 6.1 ± 2.7 ml/m2 (P = 0.027) and LV end diastolic volume index by 7.5 ± 3.4 ml/m2 (P = 0.031) but only after adjustment of covariates. There was also no significant adjusted between-treatment difference in the change of LVEF (P = 0.18).
Giving eplerenone on top of an ACE inhibitor/angiotensin receptor blockade has been shown to have additional beneficial effects compared to monotherapy alone in terms of significantly attenuating LVEDP and LVEDV, improving LV function, suppressing NT-proANP and norepinephrine levels and markers of myocardial fibrosis.23,25
Reduce sudden cardiac death and arrhythmogenicity
A striking effect of aldosterone blockade is its ability to reduce sudden death (in both RALES and EPHESUS). The reduced risk of sudden cardiac death and arrhythmias post MI with eplerenone therapy is probably due to a combination of factors including reduced cardiac fibrosis, (thereby reducing cardiac remodelling, LV dysfunction and LVH) and increased potassium and magnesium levels. It also reduces early electrical remodelling that occurs post MI–-action potential prolongation, increase in myocardial calcium current (Ica) and reduction in potassium outward current (Ito). 28 Finally, aldosterone blockade has been shown to improve myocardial norepinephrine uptake, baroreceptor function 17 and heart rate variability. 19
Endothelial dysfunction
Aldosterone is a key neurohormone post MI that stimulates reactive oxygen species (ROS) production such as superoxide anion (O–2) by NADPH oxidase in vascular cells resulting in endothelial dysfunction. The superoxide anion O–2 combines with NO resulting in NO inactivation.
Early therapy with eplerenone post MI has been shown to reduce oxidative stress and endothelial dysfunction by a number of methods resulting in increased NO bioavailability. It significantly reduces superoxide anion production and NADPH oxidase activity, increases vascular antioxidant manganese superoxide dismutase (MnSOD) levels, reduces NF-κB-mediated induction of iNOS and inhibits vascular ACE level upregulation. Additionally, eplerenone stimulates endothelial NO synthase (eNOS) phosphorylation through Akt.29,30 Combination therapy with eplerenone and an ACE inhibitor has been shown to result in additive protective effects on the endothelial function. 31
Antithrombotic
Aldosterone receptor blockers have been shown to reduce expression of PAI-1, which inhibits fibrinolysis.32,33 This raises the possibility that aldosterone blockade may produce some of its benefits by being antithrombotic.
Prevent restenosis
An animal study has shown eplerenone to reduce constrictive remodelling and prevent restenosis post coronary angioplasty. 34
Clinical Study
Ephesus study (eplerenone post-acute MI heart failure efficacy and survival study) 10 In this multicentre, randomized, placebo controlled study, 6,632 post MI pts with LV systolic dysfunction (LVEF ≤ 40%) and heart failure or diabetes were randomized at 3–14 days post MI to receive 25 mg eplerenone or placebo which was subsequently increased after 4 weeks of therapy to 50 mg if tolerated. The mean follow-up period was 16 months. These patients had a mean age of 64 and were already on optimal medical therapy (aspirin 88%, beta-blocker 75%, statin 44%, ACE inhibitor/ARB 87%). Exclusion criteria for this study was use of potassium-sparing diuretics, serum creatinine concentration >2.5 mg/dl μ220 (mol/l) and serum K > 5.0 mmol/l.
At a mean follow up period of 16 months, eplerenone resulted in a 17% reduction (P = 0.005) in cardiovascular mortality, which was predominantly due to a 21% reduction (P = 0.03) in sudden cardiac death. Eplerenone also reduced cardiovascular hospitalizations (which were mainly due to heart failure), 10 significantly reduced the mean length of heart failure hospitalization by 1.6 days (P = 0.019) and reduced the total days of heart failure hospitalization by 3.6 days (P = 0.0006). 35 Table 2 shows the beneficial effects of eplerenone therapy post MI at 30 days and a mean follow up period of 16 months.
Relative risks of eplerenone therapy 30 days and 16 months post MI.
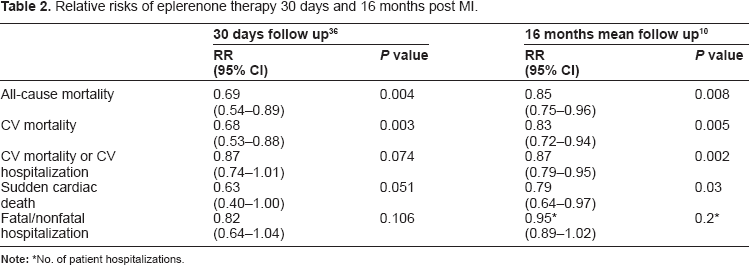
No. of patient hospitalizations.
Patients who were unable to tolerate the target dose of 50 mg/day still achieved a significant benefit in end points with eplerenone therapy. There was a reduction of 34% all cause mortality (P < 0.001), 36% cardiovascular mortality (P < 0.001), 35% sudden cardiac death (P = 0.03) and 22% cardiovascular mortality/hospitalization (P = 0.009). 37
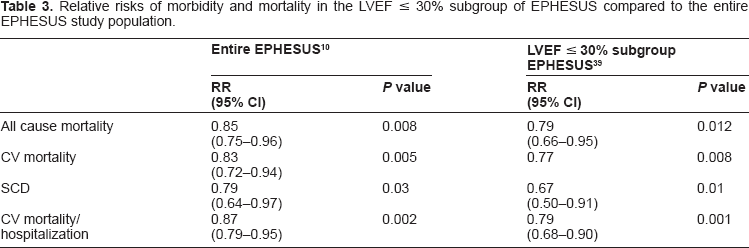
In a subgroup analysis of diabetics post MI with LVSD and CHF, eplerenone therapy resulted in a 17% reduction in a composite of CV mortality or hospitalization (P = 0.031). In fact, the absolute risk reduction was greater in the diabetics (5.1%) compared to non-diabetics (3%). 38 Eplerenone was also found to be even more beneficial amongst the high risk patients with an LVEF ≤ 30% compared to the entire EPHESUS population (see Table 3). 39
Relative risks of morbidity and mortality in the LVEF ≤ 30% subgroup of EPHESUS compared to the entire EPHESUS study population.
In EPHESUS, in the subgroup of patients with a history of hypertension (n = 4007), eplerenone had a significant effect in reducing all cause mortality (HR 0.71; P < 0.0001), composite endpoint of CV hospitalization/CV mortality (HR 0.82; P = 0.003) and sudden cardiac death (HR 0.71; P = 0.028). This was regardless of the baseline systolic blood pressure. However, this was not the case with the subgroup without a history of hypertension (n = 2625) where the only significant reduction was in the number of heart failure hospitalizations (HR 0.73; P = 0.028). There was a trend toward a reduction in mortality in this subgroup but this subgroup may have been underpowered to detect significant differences. 40 Hence, it maybe worthwhile for this to be assessed formally in a further study. However, a major limitation of the EPHESUS study was that eplerenone was compared to placebo instead of spironolactone.
Reversal of cardiac remodeling with eplerenone (REMODEL) study
This randomized, double-blinded, placebo controlled study did not show any benefit of eplerenone therapy on LV remodeling, assessed by radionuclide ventriculography, in 226 mild heart failure patients (NYHA class II and III) with an LVEF ≤ 35% who were on optimal medical therapy. 41
When is the Optimal Time to Prescribe Aldosterone Receptor Blockade?
Patients with LV systolic dysfunction with or without HF should be commenced on eplerenone early post MI as it has been shown to have significant early benefits, even at 30 days post randomization. It significantly reduces the risk of SCD, which occurs at its highest rate in the first month post MI. 36
An experimental study carried out by Hayashi and colleagues, found that immediate aldosterone receptor blockade with spironolactone resulted in an improved LV remodelling in patients with first anterior MI. 42 Additionally, Fraccarollo and colleagues found that immediate commencement of eplerenone in rats post MI improved healing of infarcts, promoted infarct neovascularization and reduced maladaptive LV remodelling by increasing macrophage infiltration and transiently increasing the chemokines that aid infarct healing. 43
In the EPHESUS study, eplerenone was commenced on day 3–14 post MI. However, early commencement of eplerenone (<7 days) was associated with a reduced risk of all cause mortality (31%), cardiovascular hospitalization/ mortality (24%) and sudden cardiac death (34%) compared to placebo therapy. This beneficial effect of eplerenone was not seen when it was commenced ≥7 days. 44 Additionally and importantly, early commencement of eplerenone was not associated with an increased incidence of hypotension.
Safety
Eplerenone therapy can be associated with renal dysfunction and hyperkalemia. In the EPHESUS, eplerenone therapy for one year was associated with a significant increase in serum creatinine of 5.3 μmol/l compared to 1.8 μmol/l in the placebo group. Serious hyperkalemia (serum K ≥ 6 mmol/l) also occurred more commonly with eplerenone therapy (5.5%) compared to placebo (3.9%) although it did not result in any deaths (P = 0.002). 10 The risk of severe hyperkalemia was significantly associated with a reduced baseline creatinine clearance of 50 ml/min. As shown by studies after the publication of RALES, the incident of hyperkalemia was much higher in real life (10%) 45 than in the RALES study (2%) 9 due to the inappropriate patient selection, prescription of higher dosages and inadequate monitoring and dose adjustment. 46 Hence, it is important to avoid such mistakes by only giving eplerenone to the correct patients, at the correct dose and to monitor them carefully thereafter.
The risk of developing hyperkalemia is increased if the subject is elderly, has diabetes, microalbuminuria, baseline renal dysfunction or high serum potassium levels. Prescribing high doses of an aldosterone antagonist can lead to hyperkalemia as eplerenone leads to a dose-dependent increase in serum potassium concentration (0.08 mmol/l at 100 mg/day to 0.36 mmol/l at 400 mg/day). 47 Concomitant medication such as NSAIDS, potassium sparing diuretics, potassium supplements, beta-blockers, ACE inhibitors or ARB should also be avoided. To avoid these complications, it is worth again emphasizing correct selection of patients, carefully monitoring them and adjustment of their dosages appropriately.
Eplerenone should not be commenced if baseline serum K > 5.0 mmol/l or GFR ≤ 30 ml/min. Renal function and serum K level should be checked 1 week after drug commencement, then at 1 month and then 3–6 monthly thereafter. If the patient has a GFR 30–60 ml/min, then more regular blood test monitoring should be performed. If serum K increases to μ5.5 mmol/l, the eplerenone dosage should be reduced by 50% whilst the drug should be discontinued if serum K increases to μ6 mmol/l.
Dose-dependent mild laboratory changes have been detected with eplerenone therapy such as increases in cholesterol, triglycerides, serum creatinine, liver transaminase and TSH. Eplerenone has been given a pregnancy B category by the FDA. Animal studies have not shown any evidence of teratogenicity and there is inadequate data from human studies.
Patient Preference
Eplerenone, due to its selective properties, reduces the sexual side effects of spironolactone and hence is better tolerated. In the RALES study, spironolactone was discontinued in 8% of patients due to adverse effects compared to 5% in placebo group. 9 The medication dosage and length of therapy correlated with developing side effects. 9% of men developed gynaecomastia compared to 1% in placebo group (P < 0.001). The action of spironolactone at androgen receptors and the peripheral conversion of testosterone to estradiol results in gynaecomastia in men. Additionally, breast pain in men occurred more commonly in the spironolactone group. In the EPHESUS study, eplerenone therapy was not associated with an increased risk of developing gynaecomastia (0.5% vs. 0.6% in placebo), breast pain (0.1% vs. 0.3% in placebo) or impotence (0.9% vs. 0.9% in placebo). 10
Place in Therapy
The ACC/AHA 48 and ESC post STEMI guidelines 49 have respectively given a class 1 A and 1B recommendation for the use of an aldosterone receptor blocker for post STEMI patients with LVEF ≤ 40% without significant renal dysfunction (<2.5 mg/dl in men and <2.0 mg/dl in women) and having serum K < 5 mmol/l. However, it should be noted that the ACC/AHA guidelines does specify that these patients should already be on therapeutic doses of an ACE inhibitor prior to commencing the aldosterone receptor blocker whilst the ESC guidelines does not. Additionally, both these guidelines do not specify which aldosterone receptor blocker should be used although the evidence backs using eplerenone in this situation.
Cost
Eplerenone is more expensive compared to spironolactone. 25 mg of eplerenone costs $2.93/tablet compared to $0.26/tablet for 25 mg spironolactone. 50 Despite this, it is still found to be a cost effective drug to be prescribed in post MI heart failure patients. Eplerenone has an incremental cost of $10,400 to $22,000 per life year gained which is much less than the threshold of $50,000 per life-year gained. 51
Possible Future Indications
Mild heart failure
The REMODEL study has shown negative results in this patient group. Results are eagerly awaited on the Effect of Eplerenone in Chronic Systolic Heart Failure (EMPHASIS-HF, ClinicalTrials.gov Identifier NCT00232180) study which is currently ongoing, assessing the use of eplerenone in patients with mild heart failure NYHA class II already on optimal medical therapy. They are aiming to recruit larger numbers of approximately 2,500 patients and follow them up for up to 4 years.
Acute coronary syndrome
Experimental studies have already shown beneficial effects of eplerenone in reducing endothelial dysfunction and oxidative stress, preventing atherosclerosis progression and reducing PAI-1. Randomized studies should be considered to assess for any beneficial effects of aldosterone receptor blockade in acute coronary syndrome patients. 52
Use in conjunction with ICD
ICDs have been found to be very effective in reducing SCD in chronic heart failure patients with severe LV dysfunction but not early post MI. On the other hand, eplerenone has been found to be very effective at reducing SCD early after an MI. Hence, studies should be considered assessing the combined use of aldosterone blockade and ICDs in patients with severe LVSD post MI. 53 Since the former is effective early after an MI and the latter effective later after an MI, their combination could be ideal.
Conclusion
In the event of an MI and heart failure, there is activation of neurohormones especially aldosterone which has detrimental cardiovascular effects. Eplerenone is a selective aldosterone receptor blocker that is very effective in reducing mortality and heart failure hospitalizations in patients with LVSD post MI and heart failure or diabetes as shown in the EPHESUS study. Its main effect on mortality is by reducing SCD. It blocks the damaging effects of aldosterone such as oxidative stress, endothelial dysfunction, thrombosis and electrolyte abnormalities. Experimental studies have shown beneficial effects of eplerenone therapy on LV remodeling although the evidence is more modest in human studies. It is a well tolerated drug with a very small risk of serious hyperkalemia and renal dysfunction. Hence, a lot of extra monitoring of renal function and serum electrolytes is required for eplerenone to be used safely. It does not have the unpleasant sexual side effects that have been associated with spironolactone. However, it is much more expensive compared to spironolactone. There have not been any head-to head comparison trials of eplerenone with spironolactone. Studies are currently ongoing to assess the use of eplerenone in the milder heart failure population.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The peer reviewers, Dr Rekhraj and Dr Szwejkowski have no conflicts of interest to declare. Professor Allan Struthers would like to declare that he has received speaking and consultancy fees from Pfizer who market eplerenone. The authors confirm that they have permission to reproduce any copyrighted material.