Abstract
Aldosterone receptor antagonists have recently been added to the American College of Cardiology/American Heart Association/Heart Failure Society of America 2017 guideline update for serving a role in the reduction in morbidity in patients with heart failure with preserved ejection fraction and an ejection fraction greater than 45%. This recent addition to the heart failure with preserved ejection fraction recommendations is supported by the findings of the Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist study. All trials with spironolactone or eplerenone in the treatment of patients with heart failure with preserved ejection fraction are reviewed for both cardiovascular morbidity and mortality and symptom improvement. Limited positive data exist on the role aldosterone receptor antagonists has on mortality and symptom improvement in patients with heart failure with preserved ejection fraction. Ongoing trials with angiotensin receptor neprilysin inhibitor and sodium-glucose co-transporter 2 inhibitors in patients with heart failure with preserved ejection fraction are reviewed. This review provides the current clinical and scientific data pertaining to the safety and efficacy of aldosterone receptor antagonists in patients with heart failure with preserved ejection fraction.
Keywords
Introduction
Heart failure with preserved ejection fraction (HFpEF) has previously been referred to as diastolic dysfunction heart failure. Risk factors for HFpEF include advanced age, female sex, hypertension (HTN), and coronary artery disease (CAD). The diagnosis of HFpEF can be made on the basis of left ventricular hypertrophy (LVH), clinical evidence of heart failure (HF), a normal ejection fraction (EF) which is defined as a value ≥50% per the American College of Cardiology (ACC)/American Heart Association (AHA), and echocardiography findings. 1 The ideal treatment for HFpEF has not been extensively validated, as there have been a number of neutral outcome trials with commonly prescribed medications for heart failure with reduced ejection fraction (HFrEF). No drug selectively enhances myocardial relaxation without having associated effects on left ventricular (LV) contractility or on the peripheral vasculature.2–6
The most common cause of HFpEF is HTN that leads to LVH and decreased cardiac compliance. 7 The 2013 ACC/AHA guidelines recommend treating associated HTN in accordance with the national guidelines. 1 Drugs that cause regression of LVH (eg, angiotensin converting enzyme [ACE] inhibitors, angiotensin receptor blockers [ARBs], β-blockers) may also slow or reverse structural abnormalities associated with HFpEF.
In the perindopril in elderly patients with chronic heart failure (PEP-CHF) trial, the ACE inhibitor perindopril failed to reduce the incidence of the primary endpoint (all-cause mortality or HF hospitalizations) but did decrease HF symptoms and improved functional capacity. 8 The Candesartan in Heart failure: Assessment of Reduction in Mortality and morbidity (CHARM-Preserved) trial also failed to show any difference in cardiovascular (CV) mortality, but fewer hospitalizations were seen in the candesartan group. 9
The VALsartan in Diastolic Dysfunction (VALIDD) trial compared the effects of valsartan or placebo added to standard antihypertensive therapy (which included diuretics, β-blockers, calcium channel blockers [CCBs], or α-blockers) in patients with mild HTN and diastolic dysfunction. 10 The hypothesis of this trial was that renin angiotensin aldosterone system (RAAS) inhibition with an ARB would be associated with greater improvement in diastolic function due to more regression of LVH or myocardial fibrosis. Patients with a history of HTN were randomly assigned to receive either valsartan 160 mg (titrated up to 320 mg) or matching placebo. Patients who did not achieve a target blood pressure (BP) goal of <135/80 mm Hg received additional therapy starting with a diuretic followed by a CCB or a β-blocker, and then an α-blocker. Angiotensin receptor blockers, ACE inhibitors, and ARAs were excluded. The primary endpoint was the change in diastolic myocardial relaxation velocity from baseline to 9 months with a secondary endpoint of change in LV mass. A small, but significant, increase was seen in diastolic relaxation velocity in both groups from baseline to follow-up, but there was no significant difference between the treatment groups. The authors concluded that aggressive BP control—even in mild HTN—was associated with improvement in diastolic dysfunction, irrespective of whether BP reduction was achieved with an RAAS inhibitor or other antihypertensive agent.
The CHARM-Preserved study investigated the role of candesartan in patients with HFpEF. 9 The trial enrolled 3023 patients who met the overall CHARM trial inclusion criteria with an EF of more than 40% (mean 54%). Patients had symptomatic HF with normal (preserved) EF. They received either an ARB alone (n = 1514) or placebo; only 20% of subjects in both groups were taking an ACE inhibitor at randomization, 56% a β-blocker, and 11% spironolactone. After a median follow-up of 36.6 months, a trend was noted toward reduction in the primary outcome of CV death or hospital admission for HF in the candesartan group (22%) compared with placebo (24.3%; P = .118). Cardiovascular deaths and all-cause mortality were nearly identical in both groups, but the total number of hospitalizations for HF was significantly reduced in the candesartan group. Overall, in symptomatic patients with HFpEF, no significant improvement in mortality occurred with the use of candesartan compared with placebo, but there was a significant reduction in HF-related hospitalizations for those who received candesartan.
The Irbesartan in Heart Failure with Preserved Ejection Fraction (I-PRESERVE) study evaluated irbesartan titrated to 300 mg daily or placebo for the management of HFpEF. 11 The patient population was 60 years of age or older with the New York Heart Association (NYHA) class II through IV symptoms, EF of at least 45%, and hospitalized for HF within 6 months prior or have persistent class III or IV symptoms (n = 4128). There was no difference in the primary endpoint (death from any cause or hospitalization for a CV cause) between irbesartan (36%) and placebo (37%; P = .35). More patients in the irbesartan group experienced hyperkalemia than those in the placebo group. One possible reason for the neutral results of I-PRESERVE included the high rate of dual RAAS blockade at baseline (39% ACE inhibitor use in the irbesartan group and 40% in the placebo group; 28% spironolactone use in the irbesartan group and 29% in the placebo group). Overall, irbesartan showed no benefit in reducing morbidity or mortality in HFpEF patients.
Beta-blockers or nondihydropyridine CCBs are other classes of drugs of interest in HFpEF. Part of their value is to control HTN, a risk factor for all forms of HF. More specific to HFpEF, β-blockers and CCBs (especially verapamil) possess negative inotropic properties that may favorably influence the pathophysiology of diastolic dysfunction by slowing the heart rate to allow more time for complete ventricular filling particularly during exercise, reducing myocardial oxygen demand, and controlling BP. Both pharmacologic classes are beneficial in decreasing ischemia in patients with CAD.
Most HF trials that used β-blockers and demonstrated decreased morbidity and mortality have focused on HFrEF. The Study of the Effects of Nebivolol Intervention on Outcomes and Rehospitalization in Seniors with Heart Failure (SENIORS) evaluated β-blocker use in elderly HF patients irrespective of LV function. 12 The trial randomly assigned patients to nebivolol or placebo. Nebivolol is a selective β1-adrenergic receptor blocker with vasodilator properties that are mediated through nitric oxide (NO) release. This effect may be beneficial in elderly patients, who tend to have decreased endothelium-dependent vasodilation. 13 The primary endpoint of the study was the combination of all-cause mortality and CV hospital admissions. There was a 14% relative risk reduction in the primary endpoint in the nebivolol group versus placebo (P = .039). Prospective subgroup analyses of the primary outcome by left ventricular ejection fraction (LVEF; ≤35% or >35%), sex, or age (≤75 years or >75 years) showed benefits across all subgroups. Patients with EF >35%, however, appeared to benefit a little more than those with low EF%, and all-cause mortality was lower in patients older than 75 years treated with nebivolol compared with placebo. The study reinforces the current recommendations that all HF patients with reduced EF should receive β-blockers. Only 35% of the patients had preserved LV function and were mostly men. Further studies are required to define the role of β-blockers in HFpEF.
The prEserveD left ventricular ejection fraction chronic heart Failure with ivabradine studY (EDIFY) was performed to evaluate the benefit of ivabradine in the HFpEF population. 14 In this study, patients with NYHA class II/III HF with and EF >45% were randomized to ivabradine 7.5 mg twice daily or placebo and followed for 8 months. The study showed no significant changes in the co-primary endpoints of echo-Doppler E/e′ ratio, distance on the 6-minute walking test, and plasma-proBNP concentrations. Based on these findings, there is not a current role for ivabradine in the treatment of patients with HFpEF.
The role of ARAs in patients with HFpEF has been evaluated in a number of clinical trials over the last few years, which warrants attention. These trials have included evaluations relative to diastolic dysfunction clinical markers, symptom improvement, and hard CV outcomes. This review provides a detailed look at the ARA trials in the HFpEF population and highlights the current ACC/AHA recommendations on ARA use in this population.
ARA Trials in Patients With HFpEF
Spironolactone
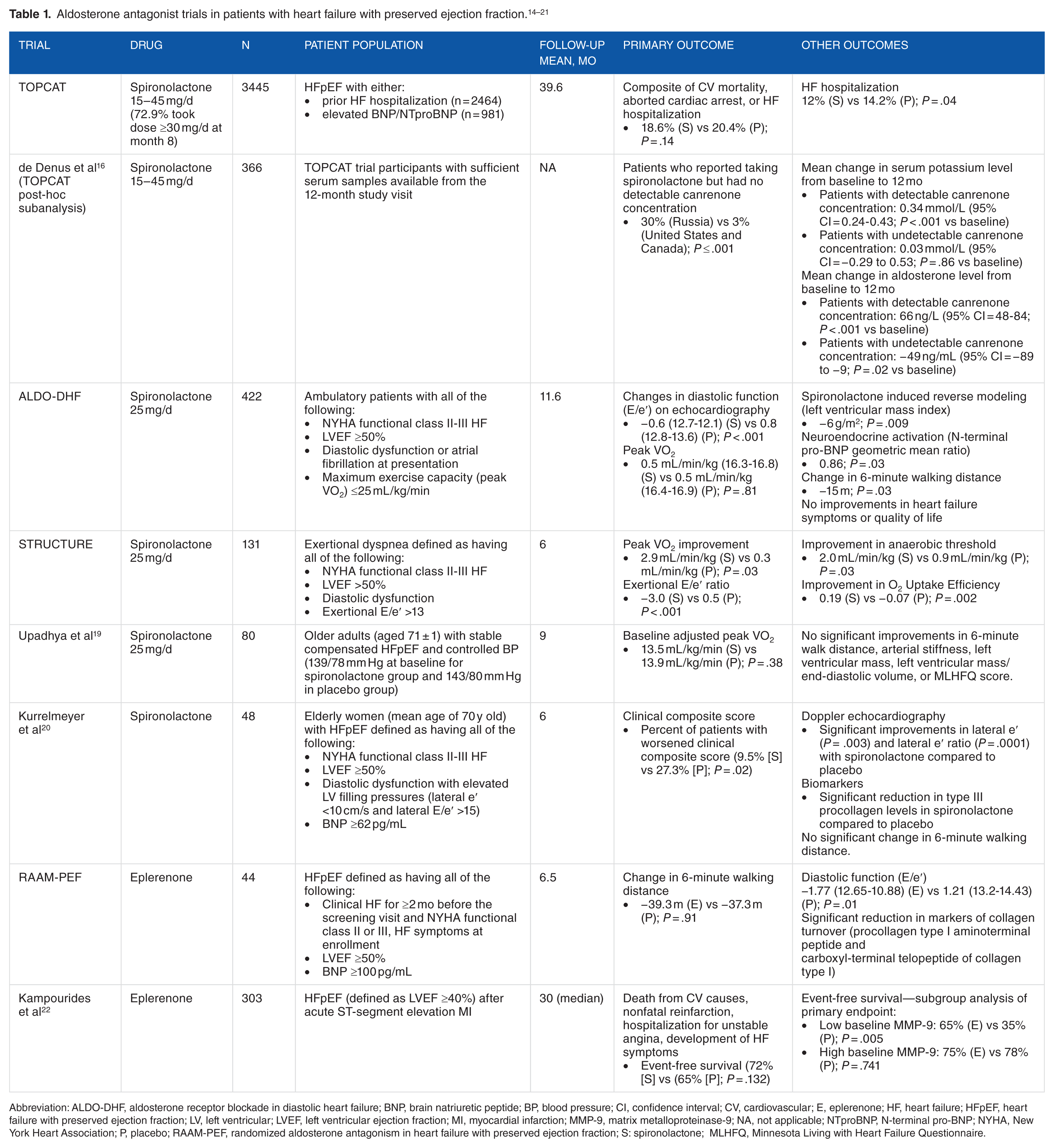
A complete review of all clinical trials evaluating ARA trials in patients with HFpEF can be found in Table 1.15-22 The Spironolactone for Heart Failure with Preserved Ejection Fraction (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist [TOPCAT]) trial evaluated the effects of spironolactone 15 to 45 mg daily versus placebo in 3445 patients from the Americas, Russia, and Georgia with symptomatic HF and an EF ≥45%. 15 Patients were eligible to enroll if they had either an HF hospitalization in the last 12 months or elevated brain natriuretic peptide (BNP; level ≥100 pg/mL or an N-terminal pro-BNP [NTproBNP] level ≥360 pg/mL) within 60 days before randomization. After a mean follow-up of 3.3 years, patients treated with spironolactone had no statistically significant difference in the primary composite outcome of CV mortality, aborted cardiac arrest, or HF hospitalization (18.6% with spironolactone versus 20.4% with placebo; P = .14). However, the incidence of HF hospitalizations was shown to be significantly reduced in spironolactone-treated patients (12% with spironolactone versus 14.2% with placebo; P = .04). This result was driven by the 28% of patients enrolled on the basis of elevated BNP and/or NTproBNP levels, where spironolactone showed a significant benefit on the primary outcome (hazard ratio [HR] = 0.65; 95% confidence interval [CI] = 0.49-0.87; P = .003). Spironolactone showed no benefit in those enrolled based on a history of HF hospitalization (HR = 1.01; 95% CI = 0.84-1.21; P = .92). Treatment with spironolactone was associated with increased serum creatinine levels, increased hyperkalemia, and reduced hypokalemia, with no significant differences in the incidence of serious adverse events between treatment and placebo.
Abbreviation: ALDO-DHF, aldosterone receptor blockade in diastolic heart failure; BNP, brain natriuretic peptide; BP, blood pressure; CI, confidence interval; CV, cardiovascular; E, eplerenone; HF, heart failure; HFpEF, heart failure with preserved ejection fraction; LV, left ventricular; LVEF, left ventricular ejection fraction; MI, myocardial infarction; MMP-9, matrix metalloproteinase-9; NA, not applicable; NTproBNP, N-terminal pro-BNP; NYHA, New York Heart Association; P, placebo; RAAM-PEF, randomized aldosterone antagonism in heart failure with preserved ejection fraction; S: spironolactone; MLHFQ, Minnesota Living with Heart Failure Questionnaire.
An exploratory post-hoc analyses by the investigators showed that patients enrolled from Russia or Georgia had much lower event rates (9.3% with spironolactone versus 8.4% with placebo; HR = 1.10; 95% CI = 0.79-1.51; P = .576) than those treated in North and South America (27.3% with spironolactone versus 31.8% with placebo; HR = 0.82; 95% CI = 0.69-0.98; P = .026). 16 In addition, the majority of patients from Russia and Georgia were enrolled in the hospitalization stratum (n = 1488 in hospitalization stratum and n = 190 in BNP/NTproBNP stratum), while enrollment in the Americas was more evenly balanced between the hospitalization and BNP stratum (n = 976 in hospitalization stratum and n = 791 in BNP/NTproBNP stratum). To further investigate the regional discrepancy in event rates, a post-hoc subanalysis was performed by de Denus et al 16 that measured concentrations of canrenone, an active metabolite of spironolactone, in 366 patients in the TOPCAT trial from the United States, Canada, and Russia. Their analysis of serum samples from the 12-month study visit showed that canrenone concentrations were undetectable in a significantly higher percentage of patients from Russia than from the United States and Canada (30% versus 3%; P = <.001). In addition, patients with detectable canrenone concentrations had significant increases in both potassium and aldosterone levels, which are expected effects of spironolactone. Conversely, there were no significant increases in potassium or aldosterone in patients with undetectable canrenone concentrations. Based on these results, authors of this analysis raised concerns regarding study conduct in some sites in Russia and Georgia, suggesting that results obtained in these regions may not reflect the true therapeutic response to spironolactone.
The Aldosterone Receptor Blockade in Diastolic Heart Failure (ALDO-DHF) trial assessed the safety and efficacy of long-term aldosterone receptor blockade by examining the effects of spironolactone on diastolic function and maximal exercise capacity. 17 There were 422 ambulatory patients with NYHA functional class II to III HF, EF >50%, and evidence of diastolic dysfunction enrolled to receive spironolactone 25 mg daily (n = 213) or matching placebo (n = 209) with 12 months of follow-up. After a mean follow-up of 11.6 months, diastolic function on echocardiography improved in spironolactone-treated patients compared to placebo, evidenced by E/e′ changes of −0.6 (12.7-12.1) versus 0.8 (12.8-13.6), respectively; P < .001. Spironolactone also showed statistically significant benefits in reverse modeling and neuroendocrine activation. However, there was no difference compared to placebo in respect to maximal exercise capacity (peak VO2), improvements in HF symptoms, or quality of life. The authors concluded that further investigation in larger populations is needed to determine the clinical significance of the improved LV function seen in the ALDO-DHF trial.
The Effect of Aldosterone Antagonism on Exercise Tolerance in Heart Failure with Preserved Ejection Fraction (STRUCTURE) trial aimed to identify improvement in exercise capacity with spironolactone in the subset of HFpEF patients with exercise-induced increase in E/e′. 18 The trial enrolled 131 patients with exertional dyspnea (defined as NYHA functional class II to III HF, EF >50%, diastolic dysfunction, and exertional E/e′ >13) and randomized patients to receive spironolactone 25 mg daily (n = 64) or placebo (n = 67). Significant benefits noted in patients treated with spironolactone after 6 months of follow-up included the primary outcomes of peak VO2 improvement (2.9 mL/min/kg with spironolactone vs 0.3 mL/min/kg with placebo; P = .03) and change in exertional E/e′ ratio (−3.0 with spironolactone vs 0.5 with placebo; P < .001). Significant improvements in anaerobic threshold and O2 uptake efficiency were also noted. Although STRUCTURE and ALDO-DHF demonstrated improvements in E/e′, the reliability of E/e′ in assessing the effects of pharmacologic interventions on diastolic function is not well elucidated. 18 Conversely, peak VO2 has been noted as a consistent marker of disease severity and prognosis in patient with HF. 23
Eplerenone
The Randomized Aldosterone Antagonism in Heart Failure with Preserved Ejection Fraction (RAAM-PEF) trial aimed to examine the effects of eplerenone on functional outcomes. 21 Patients with HFpEF (defined as NYHA functional class II to III HF, EF ≥50%, and BNP >100 pg/mL) were treated for 24 weeks after randomization with eplerenone (25 mg daily for 2 weeks followed by 50 mg daily for 22 weeks, if tolerated) or matching placebo. In patients treated with eplerenone, no improvement was noted in the primary outcome of change in 6-minute walking distance (−39.3 m with eplerenone versus −37.3 m with placebo; P = .91). However, compared to placebo, eplerenone was associated with significant improvements in diastolic function, evidenced by change in E/e′ of −1.77 (12.65-10.88) with eplerenone versus 1.21 (13.2-14.43) with placebo; P = .01. Eplerenone use was also associated with significant reduction in markers of collagen turnover (procollagen type I aminoterminal peptide and carboxyl-terminal telopeptide of collagen type I). The authors concluded that it remains to be determined if the observed benefits on collagen turnover have any impact on morbidity and mortality in HFpEF.
Kampourides et al 22 examined the usefulness of matrix metalloproteinase-9 (MMP-9) plasma levels (a marker of extracellular matrix turnover) to identify patients with preserved LV systolic function after an acute myocardial infarction (AMI) who could potentially benefit from eplerenone use. This single center, open-label, randomized, parallel-group, nonplacebo-controlled study examined 303 patients who were included if they had an AMI 1 to 7 days before enrollment, had no previous or current aldosterone antagonist treatment, and had an LVEF ≥40% on day 7 after the index event. On day 7 after their AMI, patients had plasma MMP-9 levels drawn and were randomized to either receive (n = 201) or not receive (n = 102) eplerenone 25 mg daily. After a median follow-up of 30 months, no difference was found in patients who did or did not receive eplerenone regarding the primary endpoint of death from CV causes, nonfatal reinfarction, hospitalization for unstable angina, or development of HF symptoms. However, a significant improvement in event-free survival for the primary endpoint was noted in patients treated with eplerenone that had low (≤12.7 ng/mL) baseline MMP-9 levels (65% with eplerenone vs 35% with placebo; P = .005). Eplerenone had no effect in the subgroup of patients with high baseline MMP-9 levels. The MMP-9 cut point of 12.7 ng/mL was derived from receiver operating characteristics curve analysis.
ACC/AHA/HFSA Guideline Recommendations
In 2016 and 2017, the ACC/AHA/Heart Failure Society of America (HFSA) published an updated to the 2013 guidelines for the management of HF.24,25 The 2017 update added new recommendation for the use of ARAs in the management of HFpEF based on the findings from the TOPCAT study. Specifically, the updated ACC/AHA/HFSA guidelines recommend a class IIb (Level of evidence B-R) recommendation to initiate ARAs in patients with HFpEF and an EF >45% with elevated BNP or an HF admission within 1 year to reduce morbidity 24 The same restrictions for use of ARAs has previously identified in the 2013 ACCF/AHA guidelines requiring patients to have an estimated glomerular filtration rate (eGFR) >30, creatinine <2.5 mg/dL and potassium <5.0 mEq/L for HFrEF also apply to HFpEF patients.1,24,25 In addition, the updated ACC/AHA/HFSA guidelines recommend a class IIb recommendation (level of evidence B) for the use of ARBs in patients with HFpEF to reduce morbidity. The 2017 ACC/AHA/HFSA updated guidelines align with the previous 2013 ACCF/AHA guidelines, as it relates to the management of edema symptoms. They recommend a class I (Level of evidence C) recommendation for the use of loop diuretics for HFpEF patients who have edema to help reduce edema symptoms.1,24,25
The 2017 ACC/AHA/HFSA added two additional key class III recommendations with their update calling out specific approaches not to use in the HFpEF patient as they could result in harm. The first of these class III (level of evidence B-R) recommendations is to not use nitrates or phosphodiesterase-5 inhibitors to increase activity or quality of life in the HFpEF patients, as these have not resulted in demonstrating benefit in clinical trials. 25 The second class III (level of evidence B-C) recommendation is to not recommend nutritional supplements in patients with HFpEF, as there has not been any demonstrated benefit seen with this approach. 25
Future Trials of ARA in HFpEF
The TOPCAT trial concluded that spironolactone is effective at reducing hospitalization for HF (HHF) in patients with HFpEF (HR = 0.83; 95% CI = 0.69-0.99; P = .04), but more data are needed to determine whether spironolactone improves mortality in this population. 15 Major limitations of the TOPCAT study included the heterogeneity of enrolled patients in the distinct geographical regions and questions regarding whether the patients enrolled in the Russia and Georgia cohorts indeed had HFpEF and received study drug.16,26 The Spironolactone Initiation Registry Randomized Interventional Trial in Heart Failure With Preserved Ejection Fraction (SPIRRIT-HFpEF) is an upcoming registry-based clinical trial designed to test the hypothesis that spironolactone reduces all-cause mortality in patients with HFpEF. 27 The design of this study aims to mitigate study design confounders that occurred in TOPCAT. This prospective randomized, multicenter, intention-to-treat, open-label treatment, phase IV, event-driven interventional registry-based trial is scheduled to start in December, 2017. The SPIRRIT-HFpEF trial will use the Swedish Heart Failure Registry (SwedeHF) to randomize patients 50 years of age and older with HFpEF (defined as EF ≥40%), receiving usual care to the addition of spironolactone 25 mg orally daily or placebo.27,28 The primary outcome measure is time to death from any cause. 27 Secondary outcomes include time to first HHF, adverse events, and drug therapy adherence. 29 Investigators have determined that 3500 patients are needed to demonstrate a 15% reduction in all-cause death. Trial enrollment will take 3 years with a study duration of 5 years. 29 Benefits to the registry-based approach for SPIRRIT-HFpEF include sponsorship and oversight by an independent, academic research organization, expedited enrollment, and automated data collection (including population, patient, and medication dispensing data). 29
Ongoing Non-ARA Pharmacotherapy Trials in HFpEF
In addition to spironolactone, several other non-ARA agents are undergoing CV outcomes trials in patients with HFpEF. The angiotensin receptor neprilysin inhibitor (ARNI) sacubitril/valsartan is currently being studied in two outcomes trials in patients with HFpEF. The Prospective Comparison of ARNI with ARB Global Outcomes in HF with Preserved Ejection Fraction (PARAGON-HF) trial is designed to determine the efficacy and safety of sacubitril/valsartan versus valsartan in patients with HFpEF. 29 Sacubitril/valsartan has demonstrated benefit in the HFrEF population, and there is rationale that it provides benefit in the HFpEF population as well. 30 In the phase II Prospective comparison of ARNI with ARB on Management Of heart failure with preserved ejectioN fracTion (PARAMOUNT) trial, sacubitril/valsartan demonstrated a significant reduction in NTproBNP in patients with HFpEF compared to valsartan alone (ratio sacubitril/valsartan to valsartan; HR = 0.77, 95% CI = 0.64-0.92, P = .005). 31 These positive findings set the stage for the PARAGON-HF outcomes trial, which began in July, 2014 and is expected to complete in March, 2019. 29 This trial has enrolled patients 50 years of age and older with HFpEF (defined as EF ≥45%) at 788 centers across 43 countries.29,32 To avoid issues of misdiagnosis seen in the TOPCAT trial, patients are required to have an elevated natriuretic peptide (NP) level and some degree of LVH. After a sequential run-in phase, the enrolled patients are randomized to either sacubitril/valsartan at a target dose of 97/103 mg twice daily or valsartan at a target dose of 160 mg twice daily. The primary objective is to compare sacubitril/valsartan and valsartan on the reduction in the rate of the composite endpoint of CV death and total HHF in patients with HFpEF. Secondary endpoints include comparing the two drug therapy interventions on changes in clinical summary score of HF symptoms and physical limitations, improving NYHA functional classification, a delaying in the time to first occurrence of a renal composite endpoint (renal death, reaching end-stage renal disease [ESRD], or ≥50% decline in estimated eGFR from baseline), and delaying the time to all-cause mortality. If these data are positive, sacubitril/valsartan may offer an evidence-based therapeutic option for the HFpEF population in addition to the HFrEF population.
The randomized, double-blind controlled study comparing LCZ696 to medical therapy for comorbidities in HFpEF patients (PARALLAX) is a study comparing sacubitril/valsartan to individualized medical therapy in reducing NTproBNP and improving HF symptoms and function capacity in patients with HFpEF. 33 This study began in September, 2017 and is expected to complete in December, 2019. Eligibility criteria include age 45 years and older and like PARAGON-HF, presence of HFpEF (defined as EF ≥45%) and an elevated NP level and some degree of LVH. Enrolled patients are randomized to sacubitril/valsartan, enalapril, valsartan, or matching placebo to one of the three active comparator arms. The primary endpoint is change in NTproBNP from baseline to week 12. Secondary endpoints include changes in clinical summary score of HF symptoms and physical limitations, NYHA functional classification, and 6-minute walk test from baseline to week 24. This study aims to demonstrate that sacubitril/valsartan improves symptoms and surrogate markers of cardiac function in patients with HFpEF.
Finally, the sodium-glucose co-transporter 2 (SGLT-2) inhibitor empagliflozin is being studied in a CV outcomes trial in the HFpEF population. The EMPagliflozin outcome tRial in Patients With chronic heart Failure With Preserved Ejection Fraction (EMPEROR-Preserved) will investigate once-daily empagliflozin versus placebo in patients with HFpEF with and without type 2 diabetes (T2D). 34 This clinical trial follows the Empagliflozin Cardiovascular Outcomes Event Trial in Type 2 Diabetes Mellitus Patients (EMPA-REG OUTCOME) that demonstrated patients with T2D who were at higher risk for CV events (including presence of cardiac failure) and received empagliflozin had a lower rate of the primary composite CV outcome and death from any cause (HR = 0.62; 95% CI = 0.49-0.77; P < .001). 35 In addition, these same patients had a significantly lower risk of HHF (HR = 0.65; 95% CI = 0.50-0.85; P = .002). Based on this information, the EMPEROR-Preserved study investigators’ aim to determine a composite endpoint of time to first event of adjudicated CV death or adjudicated HHF over 38 months in patients with HFpEF who receive empagliflozin 10 mg orally daily versus placebo in addition to usual care. Secondary endpoints include occurrence of adjudicated HHF, eGFR, time to first occurrence of sustained reduction in eGFR, time to all-cause mortality, time to onset of T2D, change in clinical summary score of HF symptoms and physical limitations, and occurrence of all-cause hospitalization. 35 If the data are positive, this will further define the high-risk CV population who may benefit from empagliflozin and whether this benefit extends beyond patients with T2D and HFpEF to patients with only HFpEF.
Discussion
Of all the trials that have investigated the potential benefit of ARAs in patients with HFpEF, the results of the TOPCAT trial are the most promising to date. The reduction in hospitalizations for HF seen in TOPCAT patients enrolled from the elevated BNP stratum is a clinically significant and practice-changing outcome that is reflected by the incorporation of ARAs for treatment of patients with HFpEF meeting the inclusion criteria of TOPCAT in the most recent ACC/AHA/HFSA treatment guidelines.24,25 Unfortunately, multiple confounders related to the international study with mixing of populations across various countries in the overall in study make it difficult to determine the true benefit that ARAs may have in patients with HFpEF. The low event rates of TOPCAT patients from Russia and Georgia raise questions about whether or not patients from these regions actually had HFpEF despite meeting the inclusion criteria at baseline. In addition, the non-adherence to treatment, evidenced by a lack of active metabolites in many patients from Russia and Georgia, likely blunted the true clinical benefit that may have been seen otherwise with the primary composite endpoint of death plus HF hospitalization. Considering that close to 50% of TOPCAT patients were from either Russia or Georgia, one could hypothesize that spironolactone’s true clinical benefit is potentially greater than what was seen in TOPCAT. Future trials like SPIRRIT-HFpEF hope to further elucidate the true benefit of ARAs in patients with HFpEF by addressing the study design confounders that occurred in TOPCAT.
Regarding the potential for ARAs to improve function outcomes, current evidence is conflicting. Maximal exercise capacity, which is a marker of disease severity and prognosis in patients with HF, has been assessed in a handful of ARA trials with mixed results. Although spironolactone-treated patients from the STRUCTURE trial showed improvements in peak VO2, no such benefit was seen in the ALDO-DHF trial. E/e′ is another assessment commonly used in HFpEF trials to assess left ventricular filling pressure (LVFP) and diastolic dysfunction. Spironolactone-treated patients in the ALDO-DHF and STRUCTURE trials both showed significant improvements in E/e′, as did eplerenone treated patients in the RAAM-PEF trial. However, E/e′ has not shown to be a reliable way to assess changes in LVFP. A 2017 systematic review showed that E/e′ was not useful for assessing changes in preserved LVEF in seven of nine studies. The authors of this review suggested that prospective multicenter studies are required for evaluation and validation before E/e′ can be recommended for clinical and research purposes. 23 Overall, further studies evaluating ARAs in randomized controlled trials and registry evaluations are needed to determine the role and ultimate potential benefit ARAs may have on functional outcomes.
The 2017 ACC/AHA/HFSA-focused update to the HF guidelines took into account the major landmark TOPCAT trial and the findings when advocating for the use of ARAs in the management of patients with HFpEF and an EF >45% to further reduce the risk of morbidity in this patient population. Based on the controversies with the findings across geographic locations and subpopulation analysis of the TOPCAT study, the guidelines made no recommendation for reductions in mortality with the use of ARAs in the HFpEF population. There remains a need for further trial analysis or registry evaluation to answer whether there is a mortality benefit in addition to a morbidity benefit with the use of ARAs in the patient with HFpEF. This new recommendation advocating for ARAs in addition to the recommendation for ARBs as potential options for reducing the risk of hospitalizations in patients with HFpEF is a major approach change as there have been numerous neutral or negative clinical trials with traditional HFrEF beneficial medications including β-blockers and ACE inhibitors. In addition, the guidelines advocate for a BP target of <130/80 mm Hg for patients with HFpEF, and with this, more aggressive BP target which aligns with the new ACC/AHA HTN guideline recommendations, the role of ARAs should increase as a means to reduce the risk of HF hospitalization and obtain optimal BP control.25,36
The ongoing SPIRRIT-HFpEF trial will further evaluate the role ARAs have in the management of patients with HFpEF to reduce both CV morbidity and mortality. Beyond ARAs, the ongoing studies of PARAGON-HF and PARALLAX have been designed to see if treatment with ARNIs in the HFpEF population will demonstrate the mortality and morbidity benefit seen with the HFrEF population. The completion of these trials is anticipated in the next 2 years in which the hope is we will have more data to support the role of ARAs in HFpEF at that point in addition to the findings of these ARNIs studies. The role of SGLT-2 inhibitors is currently being evaluated in the HFpEF patient population in the EMPEROR-Preserved trial. It is anticipated that the use of empagliflozin may have promise in the HFpEF population based on the findings of EMPA-REG-OUTCOME which demonstrated a statistically significant lower rate of HF hospitalization in patients with diabetes mellitus who were prescribed empagliflozin versus placebo. With the ongoing trials evaluating the role of ARNIs and SGLT-2 inhibitors in the HFpEF population, we will need to wait and see if CV benefit is demonstrated when they report out in the next 2 years.
Conclusions
Spironolactone and eplerenone are recommended by the 2017 ACC/AHA/HFSA guidelines and the TOPCAT trial to reduce the risk of morbidity in patients with HFpEF. Additional studies are needed to determine whether there is a role for ARAs to reduce mortality in patients with HFpEF. There remains mixed data on the role of ARAs to improve symptoms in patients with HFpEF. Further ongoing trials with ARAs, ARNIs, and SGLT-2 inhibitors will answer the question in the next few years if there is an expanded role for ARAs and if there is any role for ARNIs and SGLT-2 inhibitors in reducing CV morbidity and/or mortality for patients with HFpEF. A key take away in managing patients with HFpEF is that ARAs have a major role in reducing the risk of HF hospitalizations in patients with an EF >45% and an elevated BNP or recent hospitalization.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
JCM, SLA, and CG conceived and designed the experiments, analyzed the data, wrote the first draft of the manuscript, contributed to the writing of the manuscript, agreed with manuscript results and conclusions, jointly developed the structure and arguments for the paper, made critical revisions and approved and reviewed the final version of the final manuscript.