Abstract
Liraglutide–-a once-daily human glucagon-like peptide-1 receptor agonist for treatment of Type 2 diabetes–-provides effective glycemic control with a lower incidence of hypoglycemia than therapies such as glimepiride and exenatide, and reduces body weight and systolic blood pressure. This article briefly discusses efficacy and safety results from the Liraglutide Effect and Action in Diabetes (LEAD) program, before considering practical issues of identifying and educating patients who may be suitable for liraglutide therapy.
Introduction
Due to the aging population and increasing rates of obesity, the prevalence of Type 2 diabetes (T2D) is rising in the UK. Indeed, it is predicted that by 2010 over 5% of people in England will have T2D, a 15% increase from 2001. 1 Thus, treatment of T2D will represent an increasingly significant burden on the workload of healthcare professionals, 2 particularly those in primary care and to health care providers.
In the UK, the Quality and Outcomes Framework (QOF) system for diabetes has been revised for 2009/2010 with new glycosylated hemoglobin (HbA1c) targets, 3 with financial rewards for primary care physicians if patients achieve these targets. Previous government reimbursement triggers are being replaced by three lower HbA1c target levels (7%, 8%, and 9%), and the potential benefits and drawbacks associated with these new targets are currently the subject of much debate.4,5
In 2005/2006, approximately 42% of all UK patients with T2D did not reach an HbA1c target of <7.5%, 6 despite the large number of available agents. Furthermore, problems exist with many standard treatments for T2D: sulfonylureas (SUs), insulin and thiazolidinediones (TZDs) cause weight gain, and hypoglycemia is a major limitation of SUs and insulin. Thus, novel agents are required that will improve glycemic control to help achieve lower HbA1c targets while producing less frequent or less severe hypoglycemia than SUs or insulin, and addressing other treatment challenges, such as weight gain, more effectively.
T2D occurs as a result of chronic insulin resistance (present in 92% of patients with T2D 7 ) and subsequent beta-cell dysfunction that appears to be reversible, particularly in the early stages of disease. 8 Patients with T2D are also deficient in glucagon-like peptide-1 (GLP-1), 9 an incretin hormone that is secreted in response to food intake, stimulating insulin secretion, and inhibiting glucagon secretion. Therefore, treatments that may preserve beta-cell function and address the GLP-1 deficiency in patients with T2D are required.
Liraglutide is a human GLP-1 receptor agonist that is administered as a subcutaneous injection and can be taken independently of mealtimes. Unlike the GLP-1 receptor agonist exenatide, which is administered twice daily, liraglutide's long half-life allows it to be taken once daily. 10 Liraglutide is indicated as an add-on to metformin and/or an SU or to metformin and a TZD for patients whose T2D is not adequately controlled with these regimens alone. 11 The recommended starting dose is likely to be 0.6 mg daily, increasing to a maintenance dose of 1.2 mg after at least 1 week. Based on clinical response, the dose may then be increased to a maximum of 1.8 mg after at least 1 more week. Recently, the European Medicines Agency (EMEA) approved the use of liraglutide in patients with T2D 11 largely based on results from a series of six Phase III clinical studies–-the Liraglutide Effect and Action in Diabetes (LEAD) program.
Main Outcomes of the LEAD Program
The LEAD program was designed to investigate the efficacy and safety of liraglutide in a range of different T2D treatment scenarios. More than 4000 patients with inadequately controlled blood glucose were enrolled as part of the six Phase III studies, which investigated liraglutide treatment as monotherapy, or in combination with other antidiabetic agents.12–18 The liraglutidecontaining and comparator regimens used in each of the LEAD studies are summarized in Figure 1.
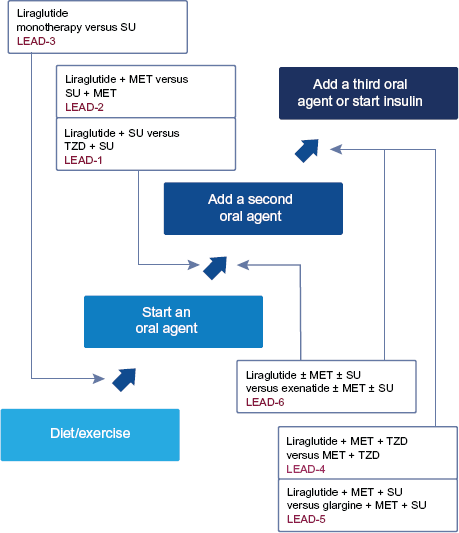
The Liraglutide effect and action in diabetes (LEAD) program.
Primary manuscripts have now been published for all 6 LEAD studies.12,14–16,18 Here, results from the LEAD studies are summarized with particular reference to glycemic control, effects on body weight and blood pressure, and tolerability.
Glucose control
In general, HbA1c reductions with liraglutide (1.2 or 1.8 mg, either alone or in combination with metformin and/or an SU, or metformin and a TZD) ranged between 0.8% and 1.5%.12,14–18 Furthermore, liraglutide improved beta-cell function, as indicated by analyses performed in three LEAD studies. 19 In general, rates of hypoglycemia were low with liraglutide,14,16 although the risk of hypoglycemia may be increased in those receiving an SU. 15 Importantly, significantly lower rates of minor hypoglycemia were recorded with liraglutide compared with exenatide in the LEAD-6 study (where each of these agents was combined with an SU and/or metformin). 12
Effects beyond glucose control
Mean body weight reductions ranged between 1.0 and 3.2 kg in patients treated with liraglutide (1.2 or 1.8 mg) either alone 14 or in combination with other agents (metformin, 16 metformin and rosiglitazone, 18 metformin and glimepiride, 17 or metformin and/or an SU 12 ). These results are particularly noteworthy given that even moderate weight loss has been shown to improve glycemic control and reduce cardiovascular risk. 20
Similarly, treatment with liraglutide (1.2 or 1.8 mg) produced reductions in systolic blood pressure of between 2.1 and 6.7 mmHg.12,14–16,18,21 These results contrast with those from Phase III studies of exenatide, which did not report any significant impact on blood pressure.22–24 Moreover, given that reductions in systolic blood pressure of only 5.6 mmHg have been associated with an 18% decrease in the relative risk of death from cardiovascular disease, 25 the modest reductions observed with liraglutide may have a considerable beneficial impact on long-term outcomes.
Safety/tolerability
Overall, liraglutide was well tolerated in the LEAD studies. Although gastrointestinal side effects were common, these were mostly transient. With some other antidiabetic agents, pancreatitis is a potential concern. In particular, postmarketing data have revealed cases of spontaneous pancreatitis among a small number of patients receiving exenatide, 26 leading to recommendations for permanent exenatide discontinuation if pancreatitis is diagnosed. However, whether pancreatitis proves to be a class effect of GLP-1 therapies remains to be seen. Given that patients with T2DM have approximately three times higher the risk of developing pancreatitis than the general population, 27 it may be difficult to establish a clear cause-and-effect relationship.
Implications of the LEAD Program for UK Primary Care
With the recent publication of the LEAD program studies and the arrival of liraglutide as an additional treatment option for T2D, it seems timely to look at the impact that liraglutide will have on primary care prescribing practices. The efficacy and safety results described above indicate that liraglutide could prove to be a valuable addition to the T2D treatment armamentarium. Given that patients with T2D are at high risk of developing cardiovascular disease, it is important that primary care physicians adopt a multifaceted approach to diabetes treatment which includes improving blood glucose levels, body weight and systolic blood pressure and treating dyslipidemia if present. The reductions in body weight and systolic blood pressure observed with liraglutide are particularly important in this context.
However, the introduction of any new agent such as liraglutide raises issues surrounding NHS constraints in the UK; for example, new agents must meet stringent cost-effectiveness criteria. One approach that primary care providers might adopt involves identification and selection of those patients likely to obtain the greatest benefit. In addition, primary care providers need to consider how best to educate patients about new agents such as liraglutide to ensure that patients have a clear understanding of why their physicians may recommend a change or addition to their current antidiabetic treatment. The sections below provide a practical guide to some of these issues.
Identifying patients suitable for treatment with liraglutide
Several groups of patients are likely to obtain marked benefits from liraglutide therapy (Table 1). In particular, liraglutide has been associated with a low risk of hypoglycemia relative to other T2D therapies (glimepiride and exenatide) and therefore may be suitable for groups of patients in whom hypoglycemia risk with these medications has an unacceptable impact or occupational safety risk.12,14,16 Patients who drive for a living are one such group; indeed, the UK Driver and Vehicle Licensing Agency receives police notifications of >45 accidents due to hypoglycemia per month. 28 Other patients include those with an erratic lifestyle (such as shift workers) and those who restrict their food intake. For example, there is a three- to five-fold increase in severe hypoglycemia among Muslim patients with T2D during the Ramadan fasting period. 29
Patients with Type 2 diabetes who are likely to derive maximum benefit from liraglutide.

Given the positive effects of liraglutide on body weight and systolic blood pressure, liraglutide treatment may also be appropriate for patients with a high body mass index (BMI), those in whom weight gain is a concern, and those with raised systolic blood pressure. National Institute for Health and Clinical Excellence guidelines currently consider the GLP-1 receptor agonist exenatide as an option for patients of European descent with a BMI ≥ 35 kg/m2 (with appropriate adjustments for other ethnic groups) or for patients with a BMI < 35 kg/m2 for whom weight loss would benefit other obesity-related comorbidities. 30 To date, data indicate that liraglutide may be offered as an effective treatment for these patients.
Some patients receiving exenatide develop neutralizing antibodies; 23 however, as the structure of liraglutide is more similar to native GLP-1 than that of exenatide (97% vs. ~50% homology, respectively 10 ), it is potentially less immunogenic. In addition, by the end of the LEAD-6 study, persistent nausea rates were lower in patients treated with liraglutide than with exenatide. 12 Therefore patients who are eligible for GLP-1 therapy but are intolerant of exenatide could benefit from liraglutide treatment.
The extension of liraglutide therapy beyond its current license may also be a realistic option in the future. For example, as an alternative to receiving an increased dose of insulin, some patients could receive liraglutide in addition to their current dose. Indeed, a clinical study investigating insulin detemir combined with liraglutide and metformin is currently recruiting volunteers. 31 Furthermore, some patients who have had negative experiences with insulin therapy (e.g. weight gain) and have sufficient remaining beta-cell function could potentially be treated with liraglutide rather than insulin; however, clinical studies of this approach would be required.
Educating patients and care givers about the use of liraglutide
Educating patients about their condition and medications poses a major challenge for primary healthcare professionals during the average 10-minute consultation. In particular, the time taken to educate patients about insulin and the required blood glucose monitoring has always been a problem for primary care physicians. The time required to teach a patient about liraglutide may prove to be shorter. Table 2 sets out a step-by-step guide to educating patients and their care givers about the mode of action and administration of liraglutide.
A patient or care giver education guide for liraglutide.

Conclusion
In summary, the LEAD studies showed that liraglutide improves glycemic control in patients with T2D at all stages of the treatment continuum. The low incidence of hypoglycemia relative to some other T2D therapies means that liraglutide treatment may be an attractive option for patients in whom hypoglycemia risk with these medications has an unacceptable impact. In addition, reductions in body weight and systolic blood pressure were achieved with liraglutide, indicating that patients with comorbidities such as high BMI and high blood pressure are likely to achieve marked benefits from liraglutide therapy. Evidence from the LEAD program resulted in the approval of liraglutide by the EMEA, meaning that it is now available for use in the UK. Consequently, primary care physicians need to be prepared to identify and educate those patients who are likely to obtain maximum benefit from liraglutide therapy.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
Footnotes
Acknowledgement
Editorial support was provided by Medi Cine International supported by Novo Nordisk, UK.
