Abstract
On the strength of two phase III clinical trials, palonosetron hydrochloride was granted FDA approval in March 2008 for the prevention of postoperative nausea and vomiting (PONV) in the period up to 24 hours after surgery. Palonosetron is superior to the established first-generation 5-hydroxytryptamin-3 receptor antagonists (5-HT3-RAs) in respect of pharmacokinetic data such as a high receptor binding affinity (pKi = 10.45) and a prolonged mean elimination half-life (40 hours). Clinically, palonosetron 0.075 mg was superior to placebo within the 0 to 24 h period usually investigated. A pooled data analysis of the complete response (CR) rates revealed efficacy in the 0 to 24 h period (when compared to placebo CR = 1.67 [1.38–2.02; P < 0.001, n = 370]) and between 24–72 h after surgery (CR = 1.29 [1.10–1.51; P = 0.002, n = 273]). Overall, the effect of palonosetron in reducing delayed vomiting was not as promising as expected. In the approval studies for PONV the rates of AEs including headache (3%), constipation (2%) and prolongation of the QTc interval (5%) were indistinguishable between palonosetron and placebo. In studies on chemotherapy-induced nausea and vomiting, palonosetron increased the QTc interval (between one and three ms) to a lesser extent than ondansetron or dolasetron (5 ms). The safety profile of palonosetron therefore seems to be favourable so far, making it a preferred perioperative antiemetic in the geriatric population or in multimorbid patients. However, further studies are needed to permit general recommendations, and we still lack comparative trials with older (and less expensive) 5-HT3-RAs, trials with combined PONV prophylaxis, and trials in the paediatric population.
Introduction
Besides postoperative pain, the occurrence of postoperative nausea and/or vomiting (PONV) is the most frequent and most unpleasant adverse outcome of surgery and general anaesthesia. Moreover, from the patient's point of view the anaesthetist is clearly responsible for this “big little problem” of the early postoperative period. Although a considerable amount of basic and clinical scientific research has been conducted in recent years, giving us a detailed insight in the complexity of PONV, a global panacea for its total prevention has not been found (so far). Therefore, further research into new antiemetics is justified and to be welcomed. A new approach to managing PONV is the clinical use of neurokinin-1 receptor antagonists, which represent a new antiemetic drug class. A further advance may be the development of favourable molecules of established drug classes with improved efficacy, prolonged action and reduced side effects. Palonosetron is a recent introduction in the prevention of PONV and seems to meet these expectations as a new, “second generation” 5-hydroxytryptamin-3 receptor antagonist (5HT3-RA). This review aims to briefly describe some aspects of PONV in the context of the latest consensus recommendations by societies and expert panels and will focus on the newly approved 5HT3-RA palonosetron for the prevention of PONV.
PONV as an important outcome
Not only do patients suffering from PONV feel unwell; they also feel exposed to an embarrassing situation. They therefore rate PONV as an important clinical outcome of anaesthesia that should be avoided.1,2 Furthermore, patients are willing to pay US$ 56–-US$ 100 out of their own pocket for a totally effective anti-emetic.3,4 Not surprisingly, patients who have already experienced PONV would invest much more than PONV-naive patients to keep free of these symptoms. Despite improvements in anaesthesia and surgical techniques, 25% of all patients still experience nausea and vomiting after surgery, and this figure may be tripled in high-risk patients.5,6 Assuming that approximately 10% of all inhabitants of the industrialized countries have to undergo an anaesthetic procedure annually, PONV really does matter. 7 Besides patient satisfaction there are also “objective” reasons for preventing and not merely treating PONV. Although vomiting is not life-threatening in most cases, it can cause haematomas and increased wound pain as a result of unfortunate movements; it may provoke ruptures of the surgical suture and therefore jeopardize the success of the surgery performed. 8 The latter also applies to all situations in which an increase in intracavital pressures (e.g. intraocular, intracranial) should be avoided. Although rare, hazardous situations and complications may result from severe PONV; they include oesophageal rupture,9,10 aspiration pneumonitis, 11 bilateral pneumothoraces, 12 subcutaneous emphysema,13,14 tracheal rupture, 15 prolapse of the eyeball 16 and painless loss of vision. 17 So PONV has a direct influence on “true” outcome parameters like morbidity and quality of life. Last but not least, PONV considerably increases healthcare costs due to delayed discharge from the recovery room, unanticipated hospital admission 18 and greater demands on the time resources of the postoperative care staff. These indirect costs usually far exceed the direct costs of antiemetics.
How to identify patients at risk
Risk factors for PONV
Considering that the majority of patients do not suffer from PONV and every drug may cause adverse reactions, a balanced antiemesis by individual risk stratification is currently recommended.19,20 But this individual risk stratification remains a problem in clinical practice because of many contributing factors with regard to the anaesthetic, the type of surgery, and the patient himself. Table 1 lists numerous risk factors for PONV; most of them are still in the column “conflicting results”. In the last decade many efforts have been made to identify risk factors with a strong clinical impact, or one that is weak but significant, and to disprove factors historically thought to influence PONV. Using logistic regression analysis and validation of the original evaluation data set in a second validation data set resulted in accepted, proven risk factors for PONV that were finally included in several PONV risk scores. Unfortunately, all of them only have an accuracy of approximately 70% in predicting the patient's individual risk.5,6,21–23 The simplified risk score developed by Apfel and Koivuranta, based on a cross-validation project between a Finnish and a German PONV study group, seems to be the most popular today. 6 This score simply counts four risk factors (female gender, history of PONV or motion sickness, non-smoking status, expected use of postoperative opioids) to determine a risk of approximately 10, 20, 40, 60 or 80% when 0, 1, 2, 3 or 4 factors are present (Fig. 1). For children the POVOC Score has recently been evaluated (Fig. 2).24,25 If 0, 1, 2, 3 or 4 of the factors “history of PONV (including first-degree relatives)”, “duration of surgery >30 min”, “age ≥3 years” and “strabismus surgery” are present, the risk that the child will vomit after the operation is 9, 10, 30, 55 or 70%. Although such simplified risk scores are far from perfect, their easy use may represent a practical approach to an individualised PONV prophylaxis. Patients at high risk for PONV should be provided with a combination prophylaxis and as many potential risk factors as possible should be eliminated, whereas patients with a low risk of PONV might be supported with one antiemetic or managed with a “wait-and-see” strategy. 26 This means that patients with a very low risk of PONV should not be exposed to the potential side effects of antiemetics and should only be treated with antiemetics if PONV really occurs.
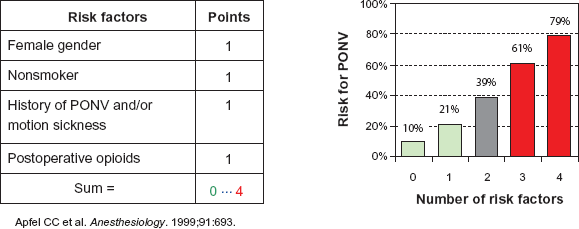
Apfel's simplified risk score for PONV.
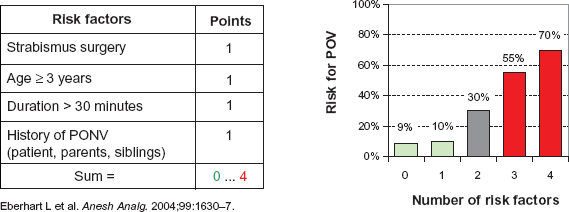
POVOC score to predict the probability of postoperative vomiting in children.
Risk factors for postoperative nausea and/or vomiting.
Genetic considerations
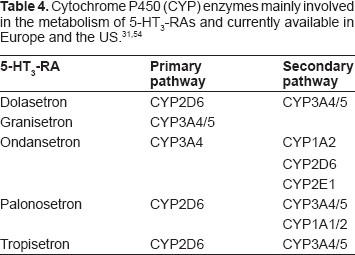
Another approach to detecting patients at high risk for PONV might be a preoperative lab screening of genetic polymorphisms known to be associated with the occurrence of PONV. We know that a considerable percentage of patients does not respond satisfactorily to antiemetics used prophylactically or therapeutically despite the use of combination prophylaxis and adherence to current organizational PONV management guidelines.27,28 Moreover, as long as 10 years ago a twin study by Reavley et al. revealed that genetic factors contribute to the risk of postoperative nausea and vomiting. 29 A genetic component may, therefore, play an important role in the development of PONV and three main pathways are currently under investigation. Firstly, genetic polymorphisms in the cytochrome P450 system result in “slow, extensive, rapid or ultrarapid” metabolizers. Whereas slow metabolizers have a higher risk of suffering side effects through individual overdosing, the status of an ultra-rapid metabolizer leads to a faster breakdown of the antiemetic and inefficacy of PONV prophylaxis due to an extremely short mean elimination half-life. So it is that the efficacy of 5HT3-RAs may be reduced considerably by a duplication of the CYP2D6 gene. 30 The regional distribution of such genetic polymorphisms varies enormously and might influence the choice of first-line antiemetics. Secondly, genetic polymorphisms can occur in the genes for several receptors involved in the development of PONV. The association between 5-HT3 receptor gene polymorphisms and the efficacy of antiemetic treatment with 5HT3-RAs has so far only been investigated in cancer patients receiving emetogenic chemotherapy.31,32 One pilot study investigated both 5HT3-A and 5HT3-B receptor genes in patients with postoperative vomiting after general anaesthesia. 33 Of the 35 polymorphisms detected, only five genetic variants were independently able to influence the incidence of PONV significantly. Thirdly, genetic polymorphisms can occur in transporter proteins like the adenosine triphosphate-binding cassette subfamily B member 1 transporter. This may result in increased physiological transmembrane transportation followed by higher CNS levels of 5HT3-RAs. 34 At present, the great variability of genetic polymorphisms and the considerable effort and cost involved preclude general preoperative screening of genetically determined PONV susceptibility.
The current armamentarium against PONV
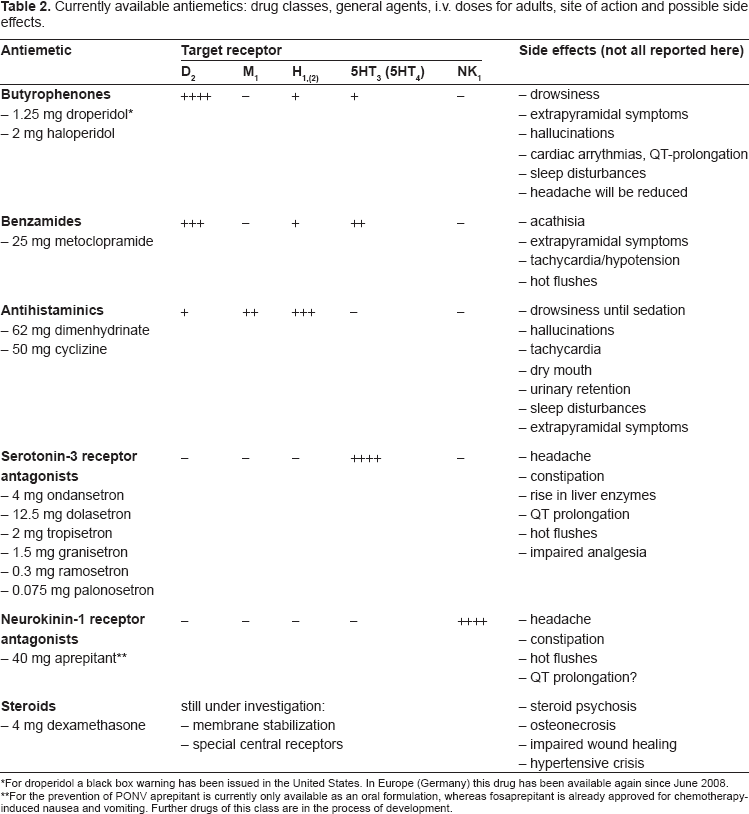
At present, a 25%–30% relative reduction of the risk of PONV can be achieved with all antiemetic drugs considered to be effective.27,35,36 In a Cochrane Review including 737 studies and 103,237 patients, more than 60 drugs with supposed antiemetic properties were investigated by using a random-effects model. Only nine of them were effective against all four endpoints (nausea, vomiting, nausea and vomiting, use of rescue medication). 35 Cyclizine, dexamethasone, dolasetron, droperidol, granisetron, metoclopramide, ondansetron, ramosetron and tropisetron demonstrated efficacy in preventing PONV with relative risks between 0.60 and 0.80 compared to placebo. Table 2 gives an overview of the currently available drug classes and receptors involved in antiemetic action. Most of the antiemetics exert their effect independently of each other.27,35,37 So the higher a patient's baseline risk, the more single interventions have to be combined. There seems to be no major interaction between any of the antiemetics currently available, and the overall effect of combination therapy can be predicted by simply adding up the relative effect of each single component. 38 Multimodal prevention of PONV includes the use of many pharmacological interventions with different sites of action (e.g. a combination of dexamethasone, metoclopramide and ondansetron) and reduction of the emetogenic potential of the anaesthetic itself (use of propofol instead of volatile anaesthetics; supplementation with regional blocks or peripheral analgesics to save on opioids; appropriate fluid management with balanced crystalloids and colloids; reduction of preoperative anxiety and/or agitation; avoidance of profound and prolonged hypotension). Although every clinic should have its own treatment policies or standard operating procedures adapted to specific settings and patient populations, several guidelines for the management of PONV have been developed and published as a consensus of experts based on current knowledge.19,20,39–43 These evidence-based consensus guidelines provide valuable information to clinicians for different clinical settings and patient populations. Adherence to these guidelines may reduce (but still not eliminate) the occurrence of PONV and improve the efficacy of PONV prophylaxis. 44
Currently available antiemetics: drug classes, general agents, i.v. doses for adults, site of action and possible side effects.
*For droperidol a black box warning has been issued in the United States. In Europe (Germany) this drug has been available again since June 2008.
For the prevention of PONV aprepitant is currently only available as an oral formulation, whereas fosaprepitant is already approved for chemotherapy-induced nausea and vomiting. Further drugs of this class are in the process of development.
Palonosetron
Palonosetron (see Fig. 3) is a “second generation” 5HT3-RA newly approved for the prevention of PONV since March 2008; of the 5HT3-RAs it has the highest binding affinity to the 5-HT3 receptor and at approximately 40 hours the longest elimination half-life. Unlike the representatives of the first generation with competitive inhibition of the 5-HT3 receptor, palonosetron seems to exhibit allosteric binding and positive cooperativity leading to effects persisting beyond the mere receptor binding time. 45 From this a superior effect in cases of CINV (chemotherapy-induced nausea and vomiting) and also PONV has been deduced. Advanced age, postoperative use of opioid analgesics and early PONV are some major factors contributing to late PONV. 36 The first is a fact contributed by the patient himself, and the second is an essential part of adequate postsurgical analgesia and cannot be avoided generally. Long-acting antiemetics like palonosetron may therefore offer advantages over commonly used antiemetics.
Palonosetron (2-[(S)-Chinuclidin-3-yl]-(3aS)-2,3,3a,4,5,6-hexahydro-1 H-benzo[de]isochinolin-1-on; C19H24N2O).
Mechanism of action, metabolism and pharmacokinetic profile
Serotonin (5-hydroxytryptamine, 5-HT) is a biogenic amine resulting from hydroxylation and decarboxylation of the essential amino acid tryptophane. It acts on 5-HT1–7 receptors, and of these the 5-HT3 receptor is mainly responsible for PONV. The 5-HT3 receptor is a pentamer with a centrally permeable cylindrical body. 46 Genes for five 5-HT3 subtypes (5-HT3A-E) have been identified to date. 46 Above all the 5-HT3A and 5-HT3B receptor subunits are expressed in the anatomical structures associated with triggering PONV–-centrally in the Area postrema and the Nucleus tractus solitarii and peripherally in the enterochromaffin cells of the gastrointestinal tract. Whereas all 5-HT receptors are G-protein coupled receptors, the 5-HT3 receptor is an ionotropic ligand-gated ion channel. Activation of presynaptic 5-HT3 receptors leads to a rapid rise in cytosolic Ca2+ concentration by inducing calcium influx and mobilization of intracellular calcium stores. 46 Furthermore, presynaptic activation of 5-HT3 receptors also modulates the release of various neurotransmitters such as dopamine, neurokinine, cholecystokinine and acetylcholine, which are all substances known to be involved in the pathogenesis of PONV. On the other hand, activation of postsynaptic 5-HT3 receptors leads to an influx of Na+ and K+. All first-generation 5-HT3-RAs act at an extracellular binding site as competitive antagonists. 47 Rojas et al. performed receptor site saturation, diagnostic equilibrium binding and kinetic dissociation experiments to examine competitive versus potential allosteric interactions between ondansetron, granisetron and palonosetron and the 5-HT3 receptor. As a result they confirmed the unique action of palonosetron through exhibiting allosteric binding and positive cooperativity when binding to the 5-HT3 receptor. 45 In addition, palonosetron triggered functional effects that persisted beyond its binding to the 5-HT3 receptor, e.g. a prolonged inhibition of Ca2+ influx.
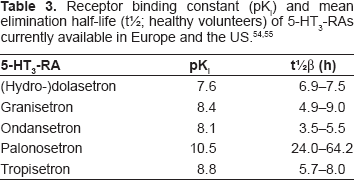
Palonosetron is characterized by high receptor binding affinity (pK = 10.45) and a prolonged mean elimination half-life (40 hours) after intravenous administration.48,49 There may be some ethnic differences, for the elimination half-life is 33.7–54.1 hours in U.S. subjects and 30.8–36.8 hours in Japanese subjects. 50 However, the elimination half-life of palonosetron greatly exceeds that reported for the first-generation 5-HT3-RAs, which is between 3.5 and 9 hours (Table 3).51,52 Approximately 62% of the palonosetron is bound to plasma proteins. Palonosetron is eliminated from the body chiefly by renal excretion (~85%) and in the faeces. In healthy human volunteers approximately 40% of the dose was recovered as the unchanged drug in the urine. 53 Another 50% was metabolized to form two primary metabolites: N-oxide-palonosetron and 6-S-hydroxypalonosetron. These metabolites both have less than 1% of the 5-HT3-RA activity of palonosetron. 48 As with other 5-HT3-RAs, the metabolic pathways were mediated by multiple CYP enzymes (Table 4). According to in vitro studies, palonosetron is neither an inhibitor nor an inducer of CYP2D6, CYP1A2 and CYP3A4.
Clinical pharmacokinetic parameters did not seem to differ significantly between poor and extensive metabolizers of CYP2D6 substrates. 48 Although the elimination half-life of 5-HT3-RAs increases and plasma clearance decreases with greater age, dosage adjustment is not necessary in geriatric patients. 30 The same applies to patients with hepatic or renal impairment. After intravenous dosing of palonosetron in patients undergoing surgery (abdominal surgery or vaginal hysterectomy), the pharmacokinetic characteristics of palonosetron were similar to those observed in cancer patients. 48
Clinical studies
Efficacy of palonosetron in preventing CINV
Palonosetron was granted initial US approval by the FDA in 2003 for the prevention of acute and delayed nausea and vomiting associated with initial and repeated courses of moderately emetogenic cancer chemotherapy. This approval was based on clinical studies performed by Eisenberg et al. and Gralla et al. published in 2003.54,55 Both studies, which included 592 and 570 patients respectively, investigated palonosetron in doses of 0.25 mg and 0.75 mg and were designed as non-inferiority studies with 100 mg dolasetron or 32 mg ondansetron. In the study by Eisenberg et al., complete response rates (CR, defined as no emetic episode and no use of rescue medication) during the first 24 hours were 63.0% for palonosetron 0.25 mg, 57.1% for palonosetron 0.75 mg and 52.9% for dolasetron 100 mg and thus indicated non-inferiority in the prevention of acute CINV. 54 In the delayed phase (24–120 hours) CR was 54.0%, 56.6% and 38.7% respectively, thus indicating superiority of palonosetron over dolasetron in the prevention of delayed CINV. In the study by Gralla et al., CR rates were significantly higher for palonosetron 0.25 mg than for ondansetron during the acute period (81.0% vs. 68.6%) and the delayed period (74.1% vs. 55.1%) (P < 0.01). 55 Surprisingly, CR rates achieved with palonosetron 0.75 mg were numerically higher but not statistically different from ondansetron during both periods. In a recent study, 1143 cancer patients who were receiving highly emetogenic chemotherapy were randomly assigned to either single-dose palonosetron (0.75 mg) or granisetron (40 microg/kg) 30 min before chemotherapy on day 1, both in conjunction with dexamethasone (16 mg intravenously on day 1 followed by additional doses on days 2 and 3). 56 According to this study, the efficacy of palonosetron in preventing CINV is not inferior to that of granisetron in the acute phase (75.3% vs. 73.3%) and is better than that of granisetron in the delayed phase (56.8% vs. 44.5%; P < 0.001) when administered in combination with dexamethasone. However, the recommended dosage of palonosetron indicated for CINV is a single 0.25 mg i.v. dose administered over 30 seconds approximately 30 minutes before the start of chemotherapy. In another recent trial this dose of palonosetron was investigated in comparison with granisetron 3 mg in 208 patients receiving highly emetogenic chemotherapy. 57 CR rates for acute CIV were 82.7% for palonosetron and 72.1% for granisetron, which demonstrated that palonosetron was not inferior to granisetron in preventing acute CIV (P = 0.07). Comparisons of CR rates for delayed CIV yielded no statistical difference between palonosetron and granisetron (61% vs. 55.5%). However, palonosetron is the only 5-HT3-RA approved for the control of delayed CINV in patients receiving moderately emetogenic chemotherapy and is repeatedly described as the preferred 5-HT3-RA in reviews of palonosetron and CINV.
Efficacy of palonosetron in preventing PONV
In March 2008 palonosetron was granted FDA approval for a new indication: prevention of PONV for up to 24 hours after surgery. This extended approval was based on two clinical studies performed by Kovac et al. and Candiotti et al., published in 2008.58,59
Interestingly, an initial randomized, placebo-controlled, dose-ranging study had already been published 10 years earlier. 60 In 1998 Tang et al. reported on the efficacy of RS-25259, a long-acting selective 5-HT3-RA for preventing PONV after hysterectomy procedures under general anaesthesia maintained with isoflurane and nitrous oxide in oxygen. Complete response rates (as the inverse value of the treatment failures reported in the paper) with palonosetron 0, 0.1, 0.3, 1.0, 3.0 and 30 μg/kg were 22%, 30%, 37%, 40%, 42% and 49% respectively. Because of the small number of patients per treatment arm (between 27 and 40 subjects per group), only the largest dose studied significantly decreased the incidence of vomiting and the need for rescue medication. Furthermore, the substance failed to exhibit anti-nausea activity, and larger doses were associated with an increased incidence of headache in the postoperative period. The dose of 30 μg/kg (=2.25 mg in a patient with 75 kg body weight) greatly exceeds the current recommended dosage of palonosetron 0.075 mg for the prevention of PONV 48 However, the results of this early dose-ranging study suggested that smaller doses of palonosetron are ineffective for preventing PONV. This contrasted with results from another trial, only published as an abstract, which indicated that oral RS-25259 1 μg/kg is effective for preventing PONV in outpatients undergoing laparoscopic surgery. 61 In 2005, results of a dose-ranging study with a similar setting and performed by the same main investigators under Dr. Tang, namely Dr. White and Dr. Scuderi, were presented at the ASA congress and published as an abstract in Anesthesiology. 62 The number of patients included in each treatment arm was now increased to between 47 and 67. Complete response rates achieved with palonosetron 0, 0.1, 0.3, 1.0, 3.0 and 30 μg/kg were now 19%, 34%, 34%, 44%, 30% and 45% respectively. So palonosetron 1 and 30 μg/kg significantly reduced PONV compared to placebo (P < 0.05). Of all dosages, palonosetron 1 μg/kg was described as the most effective antiemetic prophylaxis with the least side effects in the high-risk population undergoing hysterectomy procedures.
These initial dose-ranging studies were followed by the two phase III clinical trials performed by Kovac et al. at 28 centres in Europe (Germany, Poland and the Czech Republic) and by Candiotti et al. at 35 centres in the United States and 7 centres in Romania.58,59 Both trials investigated palonosetron 0.025 mg, 0.050 mg and 0.075 mg vs. placebo, the first in patients undergoing hysterectomy or breast surgery who were scheduled to be hospitalized for at least 72 h after surgery and the second in outpatients scheduled to undergo elective laparoscopic abdominal or gynaecological surgery. The primary efficacy hypotheses were that at least one dose of palonosetron was superior to placebo, for the CR rate, in the 0–24 h and 24–72 h periods.
In the study by Kovac et al., 684 patients were enrolled. 58 Because of a potential unblinding event with the clinical supplies label, the first 130 patients were included in the safety analysis (n = 673) but excluded from the efficacy analysis (n = 544). In the 0–24 h period the CR rates were placebo 36%, palonosetron 0.025 mg 46% (P = 0.073), palonosetron 0.050 mg 47% (P = 0.069) and palonosetron 0.075 mg 56% (P = 0.001). For delayed PONV the corresponding CR rates were 52%, 56% (P = 0.511), 61% (P = 0.151) and 70% (P = 0.002) respectively. So of the three doses of palonosetron investigated, only the 0.075 mg dose was consistently statistically superior to placebo. Palonosetron 0.075 mg was further associated with less intense nausea as compared to placebo during the 0–24 h time interval (P = 0.001) and significantly delayed the median time to emesis (P = 0.002) and treatment failure (P = 0.004). No statistical difference in the incidence of vomiting was detected between placebo (10%) and palonosetron 0.075 mg (4%; P = 0.061) in the 24–72 h time interval.
In the study by Candiotti et al., 639 patients were screened, 574 were randomized and the data of 546 patients were analyzed. 59 In the 0–24 h period the CR rates were placebo 26%, palonosetron 0.025 mg 33% (P = 0.187), palonosetron 0.050 mg 39% (P = 0.017) and palonosetron 0.075 mg 43% (P = 0.004). For delayed PONV the corresponding CR rates were 41%, 44% (P = 0.638), 47% (P = 0.249) and 49% (P = 0.188) respectively. In this outpatient population, therefore, none of the doses of palonosetron investigated was statistically superior to placebo in preventing delayed PONV. Compared to placebo, palonosetron 0.075 mg was associated with a significant downward shift towards less intense nausea (P = 0.042) and with a significant reduction in the impact of PONV on patient functioning (P = 0.004; appetite, social life and enjoyment of life, but not sleep and physical activity) during the 0 to 24 h interval. The times to treatment failure were significantly longer for any dose of palonosetron vs. placebo (P = 0.024). The incidence of emesis was generally low in the delayed phase and no statistical difference was detected between placebo (9%) and palonosetron 0.075 mg (9%; P = 1.00) in the 24–72 h time interval.
Overall, the effect of palonosetron in reducing delayed vomiting was not as promising as expected. This may be attributed primarily to the generally low incidence as shown in the placebo groups in both studies. According to a review by Kolodzie and Apfel, nausea and vomiting within the 24–72 h period occur with an incidence of approximately 30% and 5% respectively. 63 So in order to achieve statistical proof of efficacy in preventing delayed vomiting after general anaesthesia, a much larger population or patients at higher risk, especially for delayed vomiting, has to be investigated. Nevertheless, taking the endpoint “complete response” Kovac et al. were able to demonstrate long-lasting efficacy of palonosetron 0.075 mg in preventing PONV. A pooled data analysis of the CR rates in the 0 to 24 h period usually investigated and the delayed period of PONV is given in Figures 4 and 5. Figure 6 shows the efficacy of palonosetron with regard to the endpoint “vomiting within 0 to 24 h”.
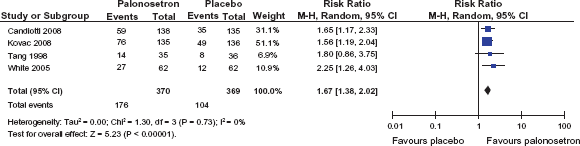
Pooled data analysis for the endpoint “complete response” for palonosetron 0.075 mg vs. placebo within the 0–24 h period (data extracted from 61, 62, 63 and 65).
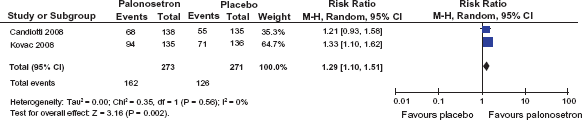
Pooled data analysis for the endpoint “complete response” for palonosetron 0.075 mg vs. placebo within the 24–72 h period (data extracted from 61 and 62).
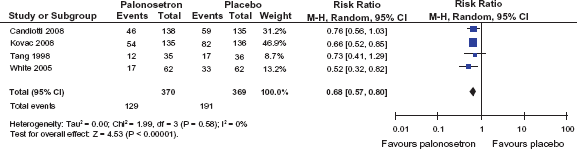
Pooled data analysis for the endpoint “postoperative vomiting” for palonosetron 0.075 mg vs. placebo within the 0–24 h period (data extracted from 61, 62, 63 and 65).
Unlike the earlier trials, the studies by Kovac et al. and Candiotti et al. also revealed anti-nausea efficacy of palonosetron 0.075 mg. A pooled data analysis of the endpoint “nausea within 0 to 24 h” is shown in Figure 7. In the study by Kovac et al. the incidence of nausea within 0–24 h was reduced by palonosetron 0.075 mg from 70.6% to 49.6% (P < 0.001). Although the CR rates for both the 0.025 mg and the 0.050 mg palonosetron doses were not statistically superior to placebo for the 0–24 h or 24–72 h periods, both lower doses reduced nausea severity during the 0–24 h period (P = 0.036 and P = 0.004). 58 For the delayed period a significant difference compared to placebo was reported for palonosetron 0.050 mg (P = 0.049) but not for 0.025 mg and 0.075 mg respectively. In the study by Candiotti et al. the incidence of nausea within 0–24 h was significantly reduced by palonosetron 0.075 mg (P = 0.033). The same applies to the distribution of nausea intensity in the 0–24 h period (P = 0.042) but not to the delayed period from 24–72 h postoperatively (P = 0.504).
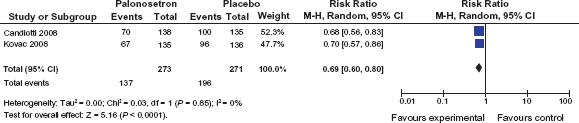
Pooled data analysis for the endpoint “postoperative nausea” for palonosetron 0.075 mg vs. placebo within the 0–24 h period (data extracted from 61 and 62).
Another clinical trial investigating the safety and efficacy of palonosetron in preventing PONV in paediatric patients was completed in January 2009. 64 Unfortunately, no results have yet been reported. So in the prescribing information for palonosetron the statement “safety and effectiveness in patients below the age of 18 years have not been established” is still to be found. 48
Safety
The safety profile of 5-HT3-RAs is well documented. Headache (OR = 1.2–1.7), constipation and transient elevation of liver enzymes occur a little more frequently than with placebo.35,65 Although the clinical use of 5-HT3-RAs rose rapidly after the FDA black box warning on droperidol, the drug class of 5-HT3-RAs is associated with a prolongation of the QTc interval too. 66
In two phase I studies, pharmacokinetic and safety data from 112 healthy, male subjects were evaluated after administration of different single i.v. doses of palonosetron. 50 The most frequently reported adverse events (AEs) were headache, transient elevation of liver enzymes and constipation, all known to be associated with the use of representatives of the drug class 5-HT3-RA. However, the incidence and severity of these AEs were similar for subjects receiving palonosetron and those receiving placebo. Moreover, no dose-related increase in the frequency of AEs was detected. Despite the known association of 5-HT3-RAs with QTc interval prolongation, no clinically significant changes from baseline in ECG and Holter monitoring were found. In a small cross-over study, eleven subjects receiving 2 doses of palonosetron 0.25 mg within 15 days, as either a 15-minute infusion or a 30-second infusion, were investigated. 67 No difference was found between the groups, and only headache and dyspepsia were considered possibly related to palonosetron.
In the approval studies for CINV conducted by Eisenberg et al. and Gralla et al., AEs were similar in frequency and severity with palonosetron, ondansetron and dolasetron.54,55 Headache (9%) and constipation (5%) were reported most often. Palonosetron increased the QTc interval (between one and three ms) to a lesser extent than ondansetron or dolasetron (5 ms). In the approval studies for PONV the rates of AEs including headache (3%), constipation (2%) and prolongation of the QTc interval (5%) were indistinguishable between palonosetron and placebo.58,59 Figure 8 shows a pooled data analysis for the endpoint “headache” for palonosetron 0.075 mg vs. placebo within the 0–24 h period. Post-marketing experience confirms the safety profile of palonosetron. Very rare cases (<1 in 10,000) of hypersensitivity reactions and injection site reactions like burning, discomfort and pain were reported. 48
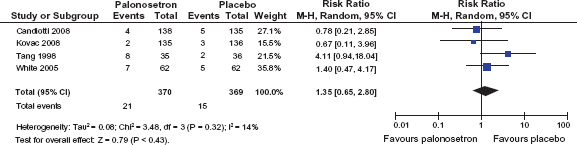
Pooled data analysis for the endpoint “headache” for palonosetron 0.075 mg vs. placebo within the 0–24 h period (data extracted from 61, 62, 63 and 65).
Interactions
Since palonosetron is neither an inhibitor nor an inducer of CYP2D6, CYP1A2 and CYP3A4, the risk of drug interactions appears to be low.48,68 No significant pharmacokinetic interactions were found after co-administration of palonosetron and dexamethasone, aprepitant or metoclopramide respectively. Palonosetron has been administered safely with corticosteroids, analgesics, antispasmodics and anticholinergic agents.
When using 5HT3-RAs, anaesthetists should be aware of negative interactions with analgesics. Ondansetron significantly lowers the analgesic effects of tramadol. 69 Tropisetron and granisetron are able to reverse an acetaminophen-mediated analgesia completely. 70 We do not have enough information on this phenomenon at present, especially in respect of palonosetron.
Place in Therapy
Six years after its initial approval, palonosetron has become the recommended first-line 5HT3-RA for the prevention of CINV because of its prolonged action and favourable safety profile. The same applies to its use in the prevention of PONV. 63 However, palonosetron is still a newcomer in the armamentarium against PONV and will be compared with the efficacy, safety and also the cost of the 5HT3-RAs already established. So far, no clinically relevant drug interactions and no severe prolongation of the QTc interval have been reported; this makes palonosetron a preferred perioperative antiemetic in the geriatric population and in multimorbid patients, including those with a high cardiac risk.
Conclusions
Palonosetron is superior to the established first-generation 5-HT3-RAs in respect of pharmacokinetic data such as a high receptor binding affinity (pKi = 10.45) and a prolonged mean elimination half-life (40 hours) after intravenous administration. In clinical trials palonosetron 0.075 mg is statistically superior to placebo in preventing PONV. Efficacy in the delayed period of 24–72 hours postoperatively is not as overwhelming as expected. This may be attributed mainly to a low general incidence of PONV in this period. We still lack comparative trials with older (and less expensive) 5-HT3-RAs, trials with combined PONV prophylaxis and trials in the paediatric population. The safety profile of palonosetron seems to be favourable to date, but further studies are needed to permit general recommendations.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.