Abstract
Azelastine HCl (Astelin™) is an intranasal antihistamine indicated for use in patients with seasonal allergic rhinitis (SAR) and non-allergic vasomotor rhinitis (VMR). A side effect of this medication is bitter taste which many patients find aversive. To address the issue of bitter taste, azelastine has been reformulated to include sucralose and sorbitol. The new formulation (Astepro™ 0.1%) is approved for seasonal and perennial allergic rhinitis and has been demonstrated to have comparable pharmacokinetics, pharmacodynamics and therapeutic efficacy compared to Astelin. More recently, Astepro at a concentration 0.15% has been approved for once a day use. This review will provide an overview of the old and new formulations of Azelastine.
Introduction
Azelastine HCl is an intranasal antihistamine indicated for use in patients with seasonal allergic rhinitis (SAR) and non-allergic vasomotor rhinitis (VMR). Up to 20% of the US population or approximately 60 million people are thought to suffer with some form of chronic rhinitis.1,2 It has been estimated that approximately 2–5 billion dollars are spent each year to treat this disorder resulting in a significant economic burden on our health care system.2,3 This expenditure consists not only of direct costs which comprise office visits and treatment, but also includes indirect medical costs such as missed work and school resulting in lost productivity. 3 Not included in this economic picture are conditions that are either associated with or complications of chronic rhinitis such as asthma, sinusitis, otitis media and conjunctivitis. These conditions if not managed properly can often lead to surgical procedures that might have been prevented or inappropriate use of medications further increasing health care costs.1,3,4 The proper treatment of rhinitis requires accurately diagnosing patients with either allergic, non-allergic or mixed rhinitis (defined as having an allergic and non-allergic component) as response to treatment differs significantly between these conditions. 1 For example, patients with non-allergic rhinitis will have little to no effect from medications such as second generation non-sedating antihistamines or leukotriene modifying agents as the mechanism of this condition is not histamine or leukotriene mediator mediated. 1 The approval of Azelastine in 1996 for allergic rhinitis and subsequently in 1999 for non-allergic rhinitis has resulted in expanding the therapeutic options available for treating both allergic and non-allergic rhinitis. 5 The importance of this medication for treatment of chronic rhinitis is supported by the positioning of azelastine HCl as a first-line treatment for allergic rhinitis (AR) and non-allergic rhinitis (NAR) by the World Health Organization and Joint Task Force of the American Academy of Allergy, Asthma and Immunology.6–8 Its multi-mechanistic actions and effectiveness across the range of chronic rhinitis subtypes has significantly impacted the health of a vast number of patients suffering from chronic rhinitis. 5
One limitation of azelastine (Astelin™) was its very bitter taste that many patients found objectionable leading to non-compliance. Survey studies inquiring about patient satisfaction with their rhinitis medications have found that approximately two-thirds were disappointed with some aspect of their medications. Furthermore, these surveys found that two-thirds (67%) of allergists and primary care physicians were dissatisfied with treatment options due to patients stopping medications or having to switch frequently because patients did not feel the medications were effective.5,9 These results may reflect the inappropriate use of certain medications for different chronic rhinitis subtypes or a specific characteristic of the medication itself such as taste or other side effects.
These findings provided impetus for the makers of azelastine to reformulate their product to reduce its bitter taste in the hope of improving patient acceptance of this medication. In November 2008, a new formulation patent for azelastine HCl was granted. This new form of the intranasal spray maintains the same generic name, azelastine HCl, but bears a new trade name, Astepro™. The reformulated product replaces saline in the original azelastine HCl with sucralose and sorbitol in order to address the issue of bitter taste. 9
Another important attribute of the reformulated product is price. The Medical Letter recently published prices of $101.35 and $96.66, for 30 days of treatment of both forms of original azelastine HCl and the new form, respectively. 10 This reduction in price, coupled with better compliance with the new formulation of azelastine HCl (Astepro™), could potentially reduce health care costs associated with treatment of chronic rhinitis significantly each year. More recently, a higher concentration of Astepro 0.15% was approved by the FDA for once a day use which may prove to be even more cost-effective.
This review will provide the reader with an overview of chronic rhinitis subtypes followed by a comprehensive overview of azelastine HCl with some focus on the new reformulated product.
Background
The term rhinitis is defined as: inflammation of the nasal mucosa. Clinically, rhinitis is characterized by a spectrum of symptoms including nasal congestion, post-nasal drainage, rhinorrhea, itching of the nose with sneezing. However, symptoms alone have been previously demonstrated not to be useful for differentiating chronic rhinitis subtypes. 11 Allergic and non-allergic rhinitis, are differentiated by the presence or absence of specific IgE antibodies to seasonal and/or perennial allergens.6,12 The presence of specific IgE to aeroallergens that correlate with exposure resulting in rhinitis symptoms is consistent with a diagnosis of allergic rhinitis (AR) while the absence of IgE in the presence of symptoms is usually given a diagnosis of non-allergic rhinitis (NAR) by default provided there are no underlying structural, medication or other underlying clinical conditions (i.e. thyroid disease) that are responsible for these symptoms. Often times, patients have features of both allergic and non-allergic rhinitis and for lack of better terminology these individuals are referred to as having mixed rhinitis.6,12
Allergic Rhinitis
Allergic rhinitis can be subdivided into SAR or perennial allergic rhinitis (PAR). The classification of AR as intermittent and persistent in lieu of seasonal and perennial, respectively, has been proposed and adopted in Europe but consensus on proper terminology for chronic rhinitis subtypes in the US is still evolving. The seasonal subclass is triggered by exposure to outdoor aeroallergens such as tree, grass and weed pollens which vary geographically but typically are present for finite times during the year. Perennial triggers refer to animal allergens, dust mites, cockroaches, rodent allergens and molds. The most bothersome rhinitis symptoms reported by AR patients are nasal congestion and postnasal drip which can manifest seasonally (SAR) or perennially (PAR).4,6 Approximately 10%–30% of adults and 40% of children in the US (about 20–40 million people) are annually affected by AR.6,12 The current treatment guidelines from the World Health Organization and Joint Task Force recommend as first line treatment for AR oral antihistamines, intranasal antihistamines or intranasal corticosteroids.6,13
Non-allergic Rhinitis
Because AR and NAR have very similar symptomatology, differentiating between AR and NAR can be challenging especially when one considers that up to 50% of chronic rhinitis sufferers may have mixed rhinitis.1,14,15 NAR patients may have intermittent or persistent symptoms with or without identifiable triggers. An infectious etiology is usually able to be excluded because of its chronic persistent symptom pattern. 15 A difficulty in diagnosing NAR is our current lack of understanding of the underlying mechanism(s) for this condition. Unique features of this condition is that it is usually recognized later in life, there is no family history of allergies, no seasonality of symptoms or symptoms around cats or dogs and frequently patients are symptomatic in response to strong odorants or chemical exposures as well as temperature and/or barometric pressure changes. 11 Recently, an irritant index scale has been developed to identify objective quantitative irritant trigger cut points that could differentiate patients with AR and NAR. The purpose of this scale is to more objectively define chronic rhinitis subtypes. One limitation of the irritant index scale is that a significant number of NAR patients do not complain of symptoms in response to non-allergic triggers. 1 Whether this subset of NAR patient responds differently to treatments approved for NAR such as azelastine HCl requires further investigation.
Non-allergic rhinitis is subcategorized as vasomotor rhinitis (VMR) and non-allergic rhinitis with eosinophilic syndrome (NARES) based on the presence or absence of eosinophils in the nasal passages. Differentiating between VMR and NARES may have treatment ramifications as patients with NARES may respond better to anti-inflammatory agents such as intranasal corticosteroids. 1 However, a difficulty in using nasal histology as a way to differentiate rhinitis subtypes has been the methods used to collect nasal secretions and/or scrapings which are difficult to consistently reproduce.
Mixed Rhinitis (MR)
Patients who manifest allergic (AR) and non-allergic (NAR) triggers are referred to as mixed rhinitis (MR). Mixed rhinitis accounts for approximately 50% of chronic rhinitis cases. 16 The irritant index scale recently developed to differentiate NAR from AR has provided a more objective way to differentiate MR patients from AR patients. 1 These individuals tend not to respond as well to the classic medications approved for the treatment of AR.
Azelastic HCl: Chemistry, pharmacokinetics, and pharmacology
Chemistry
Azelastine HCl is a bitter tasting, white powder that is nearly odorless. The chemical name is, (±)-1-(2H)-phthalazinone,4-[(4-chlorophenyl) methyl]-2-(hexahydro-1-methyl-1H-azepin-4-yl)-, mono-hydrogen chloride (Fig. 1). Its molecular formula is C22H24ClN3O·HCl and its molecular weight is 418.37. The original formulation contains 0.1% azelastine HCl in an aqueous solution at pH 6.8. Icipients include benzalkonium chloride (125 ug/ml), edetate disodium, hydroxypropyl methyl cellulose, citric acid, dibasic sodium phosphate, sodium chloride, and purified water. 17
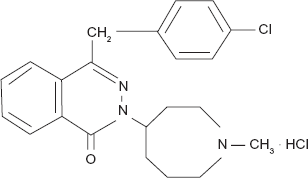
Chemical structure of azelastine HCl.
The new formulations contain either 0.1% or 0.15% azelastine HCl in an isotonic aqueous solution with sorbitol and sucralose added to alter the natural bitter taste of azelastine. The new formulation also exchanges sodium citrate for citric acid and hydrocellulose for hydroxypropyl methyl cellulose. In addition, dibasic sodium phosphate and sodium chloride have been removed from the new product but otherwise all other constituents in the reformulated products are similar to the original drug formulation. 17
Pharmacokinetics
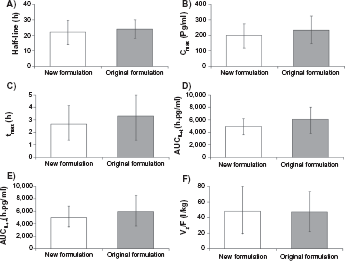
Figure 2 summarizes the pharmacokinetics of azelastine HCl. 17 The pharmacokinetics of the original and new formulations have not been altered. All values including absorption and elimination half-life remain constant between the two formulations. Therefore the change to a sucralose/sorbitol vehicle did not significantly alter the pharmacokinetic profile of azelastine HCl. 17
For the once a day Astepro 0.15% concentration administered 2 sprays per nostril (822 mcg total dose), the mean azelastine peak plasma concentration (Cmax) is 409 pg/mL, the mean extent of systemic exposure (AUC) is 9312 pg · hr/mL and the median time to reach Cmax (tmax) is 4 hours. 18
Absorption
Using a twice-daily (b.i.d.) administration of azelastine nasal spray delivers a 548 mcg dose. The onset of action measured in an environmental exposure chamber study was found to be as early as 15 minutes. 17 The peak plasma concentration (Cmax) is reached at 3 hours and yields a systemic bioavailability of 40%. Comparisons of the Cmax between the new and original compounds were 200–267 pg/ml and 235–288 pg/ml, respectively.5,9,17 Therefore the Cmax were very similar for both formulations, demonstrating that altering the original mixture did not significantly impact its administration or bioavailability. Although the systemic bioavailability for azelastine appears large, the systemic side effects are disproportionately low for this medication. Figure 3 illustrates the mean plasma concentrations of the old and new azelastine formulation over time. 9
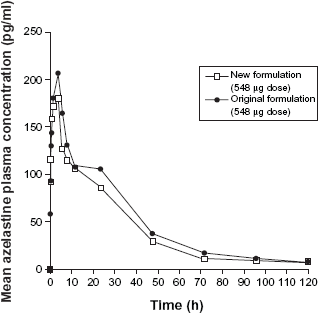
Mean plasma concentration versus time profile in patients treated with the new or original formulation of azelastine. 9
Distribution
The steady-state of the new formulation is maintained similar to the old formulation at 14.5 L/kg. The binding to carrier proteins is similar for both formulations. Plasma protein binding in serum has not been directly reported but is expected to be similar to the original formulation. For desmethylazelastine, the major metabolite of azelastine HCl, plasma protein binding was reported to be approximately 97% compared to the parent compound, azelastine HCl, which is 88% protein bound. 9
Metabolism
The cytochrome P450 enzyme system is the main metabolic pathway of the azelastine HCl. The specific isoform is still unknown. The mean desmethylazelastine Cmax is 23 pg/mL and the median Tmax is 24 hours after a single spray dosage. At steady-state, plasma concentrations of desmethylazelastine range from 20%–50% of azelastine HCl concentrations. 9 After a single dose, intranasal administration of Astepro Nasal Spray 0.15%, the mean desmethylazelastine Cmax is 38 pg/mL, the AUC is 3824 pg · hr/mL and the median tmax is 24 hours. 18
Elimination
The elimination half-life (t1/2) for the new azelastine HCl formulation is 22 hours compared to its metabolite, desmethylazelastine, which is 52 hours. The elimination t1/2 of Astepro 0.15% is 25 hours compared to its metabolite which is 57 hours. 18 Approximately 75% of an oral dose of radiolabeled azelastine HCl was excreted in the feces, with less than 10% as unchanged azelastine HCl. The t1/2 of the new and old formulations are 22–7.5 hours and 24–6.0 hours, respectively (Fig. 2). 9 Another pharmacokinetic property that is often measured is the AUC0→∞ (area under the curve) of the plasma azelastine HCl concentration over time. The AUC0→∞ was calculated for both the old and new azelastine formulations and were found to be 5122–1546 and 6122–2373 hours pg/ml, respectively which are considered to be very similar. 9 A renal dosing study using single-doses of azelastine, performed in patients with a creatinine clearance <50 mL/min, found that there was a 70%–75% higher Cmax and AUC compared to healthy subjects. 9
Pharmacology
Originally derived from phthalazinone, azelastine HCl has a high-affinity for H-1 and H-2 receptor sites.5,9,17 Its major metabolite, desmethylazelastine, has also been shown to have a high H-1 receptor antagonist activity. Studies have demonstrated that azelastine HCl was approximately ten-times more potent than chlorpheniramine. 19 Studies using the original azelastine formulation in animal and human models showed that azelastine HCl blocked the release of histamine from multiple cell types. This value was significant (P < 0.05) when the drug was administered using the recommended dose.
Azelastine HCl also has broad spectrum effects on a variety of inflammatory mediators including leukotrienes, cytokines, adhesion molecules and kinins.20–24 One study found that a twice daily 2 mg dosing of azelastine HCl significantly reduced histamine release (P < 0.05). 25 Other studies have shown that treatment with azelastine HCl also decreased LTB4, LTC4 and LTD4 levels.26,27 Treatment with azelastine HCl has been demonstrated to cause a significant decrease (P < 0.05) in Substance P and kinin levels.26,27 Studies investigating the effect of azelastine HCl on mast cells have demonstrated decreased release of inflammatory cytokines and inhibition of tumor necrosis factor alpha.27–29 In a double-blind study in AR patients, a significant decrease in neutrophils and eosinophils along with decreased expression of intercellular adhesion molecules was observed. 22
In order to assess the effect of azelastine HCl on airway inflammation, exhaled nitric oxide (eNO) levels were measured before and after treatment. 22 It was observed that nasal eNO and IL-8 levels were significantly decreased (P < 0.05) after a 2 week open-label study in patients taking azelastine HCl. 30
Although pharmacology studies of the new formulation have not yet been reported, since the parent chemical compound has not changed and there are very similar pharmacokinetics between the two formulations, it is not expected that the pharmacology of the new formulation would differ significantly from that of the old.
Drug interactions
Similar studies conducted on the original formulation were performed on the new formulations and no difference in drug-drug interactions was observed. These studies tested the interaction between azelastine HCl and oral erythromycin at a dose of 500 mg t.i.d for seven days. There was no change in ECGs and specifically no effect on the corrected QT interval (QTc). The same series of tests were performed using ketoconazole 200 mg b.i.d. for seven days. Similarly, no change in the QTc was observed for either preparation. Finally, testing to determine an interaction between theophylline (300 or 400 mg b.i.d.) and azelastine 4 mg bid demonstrated no significant interactions.9,17,31
Clinical efficacy
Astelin™ has been demonstrated in clinical trials to be effective in the treatment of SAR and NAR. The clinical efficacy of the new formulation of azelastine has been evaluated using the same standardized patient questionnaires used for the old formulation. These assessment instruments included the rhinitis quality of life questionnaire (RQLQ), total symptom complex (TSC), total nasal symptom score (TNSS) and the major symptom complex (MSC). 32 The RQLQ measures functional problems as well as the physical, emotional, social and occupational impact of rhinitis on daily life in adults between the ages of 17–70.32,33 The TSC records the number of nose blows, sneezes, runny nose/sniffles, itching of the nose, and watery eyes plus itchy eyes/ears/throat/palate, cough, and postnasal drip. The TNSS measures nasal congestion, rhinorrhea, sneezing, and nasal itching whereas the MSC measures rhinorrhea, sneezing, itchy nose, number of nose blows, and watery eyes.17,31
Original formulation studies
Seasonal allergic rhinitis efficacy and onset of action studies
Two park studies conducted by Weiler et al and Meltzer et al in 1994 evaluated 294 and 264 patients with seasonal allergic rhinitis, respectively, to assess dose ranging, safety and efficacy of azelastine.34,35 Both studies demonstrated that azelastine improved rhinitis symptoms within two hours after administration and lasted up to 24 hours suggesting that it could be administered once or twice a day34,35
Patel et al conducted a controlled environmental chamber study on 450 patients ages 18–65 with a known history of seasonal allergic rhinitis. 36 This study compared the effect of azelastine HCl (55 ug) to mometasone (200 ug) and placebo. Serial total nasal symptom scores (TNSS) were completed at 15, 30, 45, 60, 90, 120, 150, and 180 minutes, and up to 8 hours post exposure. A significant reduction in TNSS was observed from base line in the azelastine HCl group (P < 0.05) compared to the placebo group as early as 15 minutes. No difference in TNSS was observed in the mometasone verses the placebo group. 36
Ratner et al performed a two-week study evaluating 251 SAR patients ≥ages 12 years on azelastine two sprays b.i.d. versus placebo. Subjects receiving azelastine demonstrated significantly greater improvement in total symptom complex (TSC 26% versus 4%; P < 0.01) and major symptom complex scores (MSC 27% versus 6%; P < 0.01) compared to placebo. 37
Berger et al investigated 354 SAR patients ≥12 years. The study group was divided into two groups; one group received azelastine HCl plus a placebo pill and the other group received oral cetirizine 10 mg and a placebo nasal spray. A 24% increase in TNSS scores was observed for subjects taking azelastine compared to a 20% increase in those subjects taking cetirizine compared to baseline. The TNSS score between the two combination therapies was not statistically significant. However, two subcategories (nasal congestion and sneezing) were improved (P < 0.05) in favor of azelastine HCl compared to cetirizine. Furthermore, the rhinitis quality of life questionnaire scores were improved for subjects on azelastine (P < 0.05) compared to ceterizine.33,38,39
Non-allergic rhinitis studies
Leiberman et al conducted a large open label study consisting of 4,364 patients with SAR and VMR enrolled over two allergy seasons. Patients were treated with azelastine two sprays b.i.d. for two weeks; 78% of the patients with VMR and 90% of the seasonal-AR reported at least some symptom control using azelastine HCl. 3
Banov et al conducted a 21 day multicenter study of 426 non-allergic vasomotor rhinitis (VMR) patients randomized to receive azelastine HCl or placebo. A total vasomotor rhinitis symptom score (TVRSS) questionnaire was used to assess improvement in symptoms on drug versus placebo. There was a significant increase (P < 0.01) in the TVRSS for azelastine HCl from baseline. Specifically, rhinorrhea, sneezing, nasal congestion, and post nasal drip all improved from base line in response to azelastine compared to placebo. 40
Dose-ranging studies
Lumry et al conducted a two-week study to determine if reducing the dose of azelastine HCl could maintain its clinical efficacy while reducing its bitter taste. 41 This double-blind, placebo-controlled study enrolled 554 patients, 12 years and older with moderate-to-severe AR. Subjects were randomized to receive either one spray of azelastine HCl in each nostril b.i.d or one spray of placebo in each nostril b.i.d. 41 At this dose, a significant difference in efficacy between the treatment and placebo groups was observed. 41 Reduction in TNSS scores was statistically significant for the drug (3.18 or a 19% reduction) compared to the placebo (1.89 or an 11% reduction) treated groups (P < 0.01). In addition, the one-spray b.i.d. dosing regimen significantly reduced patient dissatisfaction with the bitter taste from 19% to 8% compared to two-sprays bid dosing. 41
The side effects of azelastine HCl nasal spray, using the two-spray b.i.d. dosing regimen resulted in 19.7% of patients reporting bitter taste compared to 8.3% with the 1-spray b.i.d. dosing. 41 Other studies have confirmed that one spray in each nostril b.i.d. regimen is therapeutically effective.41,42 In addition, Lumry et al described somnolence in only 0.4% of the patients using the 1-spray each nostril b.i.d. regimen compared with the 11.5% using the 2-spray b.i.d. regimen. 2
Based on these dosing studies, it was recommended that patients, above the age of 12, use one to two sprays in each nostril b.i.d dosing when treating SAR and two sprays in each nostril b.i.d. when treating NAR. The original azelastine formulation was approved for use by the FDA using either one to two sprays in each nostril b.i.d for children ages 5–11, but its use in children in this age range has not yet been approved for the new formulations.
New formulation studies
Efficacy and safety studies of astepro 0.1%
Bernstein et al conducted a two-week study involving 835 patients ≥12 years to compare the efficacy of the new and old formulations and to evaluate the efficacy of each formulation at both 1-spray and 2-spray b.i.d. dosing. 43 Patients were required to have a TNSS score of at least 8 out of 12 over a 12 hour period in order to participate. 43 Subjects were separated into 6 groups (approximately 128 per group); 97.6% of the patients completed the study with only 1% dropping out due to adverse effects of the medication. The results of the study verified the results of previous studies which showed that the original formulation improved TNSS scores at both one and two spray b.i.d. dosing. Furthermore, the newer formulation demonstrated greater efficacy than the original formulation at both dosing regimens. 43 Specifically, the new azelastine HCl formulation demonstrated that one spray each nostril dosing, provided significant improvement in the TNSS compared to the old formulation (P = 0.023). The new formulation demonstrated a 27.9% (P < 0.001) TNSS improvement from baseline, compared to 23.5% improvement (P < 0.01) for the original formula and 15.4% improvement for the placebo. 43
This study also investigated the onset of action between the two formulations at both one spray and two spray dosing regimens. The TNSS scores were measured at different time intervals to assess onset of action. It was found that both formulations, at low and high doses, provided relatively rapid symptomatic improvement when compared to placebo. The onset of action of the new formulation was more rapid than the old formulation when both were administered as two spray b.i.d. 43 Onset of action with two spray b.i.d. dosing for the new azelastine HCl was 30 minutes, while the older formulation was 45 minutes post administration. Both formulations maintained improvement in TNSS compared to placebo over the four hour observation period. With one spray b.i.d. dosing, the new formulation had a significant improvement over the placebo group at 15 and 30 minutes post administration, however, this difference was not sustained after 30 minutes. 43
Rhinitis quality of life was also measured in this study and was significantly improved for subjects receiving both formulations compared to placebo (P = 0.042 for the original formulation and P < 0.001 for the new formulation) when dosed 2-sprays per nostril b.i.d. 43 No significant improvement in RQLQ was observed for either formulation when dosed 1-spray b.i.d. 43
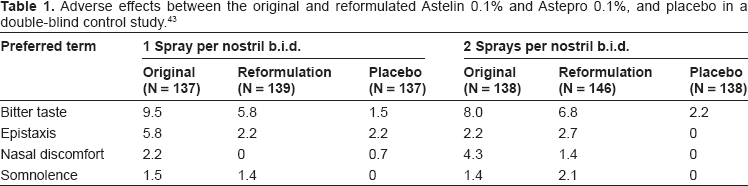
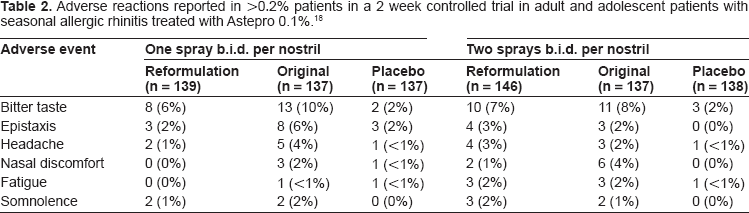
Table 1 summarizes the side effects reported for the two formulations in this study. The original MP430 approval studies for the reformulated azelastine HCl focused on the side effect profile of bitter taste, somnolence, and ulceration of the nasal mucosa. Comparison between the original and newer azelastine HCl formulations showed little difference in bitter taste at the 2-spray b.i.d. dosing (Table 2). This study also failed to demonstrate the well established increase in bitter taste with two spray dosing as opposed to one spray dosing with the original formulation. At one spray b.i.d. dosing, bitter taste from the original and newer formulation was found to be reported at 10% versus 6%, respectively. At two-spray dosing, 7% of patients reported bitterness with the new formulation and 8% with the original. 9 An explanation for, the lack of a large difference in bitterness between the original and new formulations is thought to be due to the terminology used to assess this side effect leading to high reports of “dysgeusia”. 43
Adverse effects between the original and reformulated Astelin 0.1% and Astepro 0.1%, and placebo in a double-blind control study. 43
Adverse reactions reported in >0.2% patients in a 2 week controlled trial in adult and adolescent patients with seasonal allergic rhinitis treated with Astepro 0.1%. 18
Somnolence has been another side effect commonly noted with the original formulation reported in the package insert. However, it has been reported that whereas the old formulation of azelastine HCl was associated with a 11.5% incidence of somnolence, only 2% of subjects receiving the newer formulation reported this side effect.9,43 This discrepancy has been suggested to be due to improper administration of the medication leading to increased absorption in the posterior pharynx causing this systemic side effect.9,43
Ulceration of the nasal mucosa was seen in 10% if patients treated with azelastine HCl. Comparison studies showed no significant difference in the incidence of nasal mucosal ulceration between the two formulations.9,10
Efficacy and safety studies for astepro 0.15%
The efficacy and safety of Astepro nasal spray 0.15% for seasonal allergic rhinitis was evaluated in five randomized, multicenter, double-blind, placebo-controlled clinical trials in 2499 patients ≥12 years with symptoms of seasonal allergic rhinitis and in two trials consisting of 578 patients ≥12 with perennial allergic rhinitis. 18 Efficacy was based on the reflective total nasal symptom socres (rTNSS), instantaneous TNSS (iTNSS) and other supportive secondary efficacy variables. The primary efficacy endpoint was the mean change from baseline in rTNSS over 2 weeks. Astepro 0.15% demonstrated greater improvement in rTNSS compared to placebo which was statistically significant for both SAR and PAR (Fig. 4). 18 The onset of action was shown to be within 30 minutes for Astepro 0.15%.
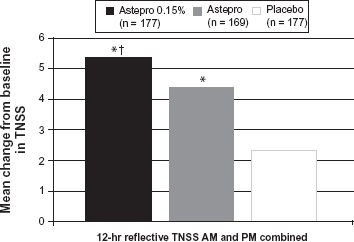
Comparison of mean change from baseline in TNSS of astepro 0.15% 2 sprays each nostril qd, Astepro 0.1% 2 sprays each nostril twice a day and placebo. 18
Safety and tolerability
Both the new and original forms of azelastine HCl are classified as pregnancy category C drugs. Studies performed with mice, rats and rabbits found various teratogenic effects ranging from cleft palate, short or absent tail, fused, absent or branched ribs, to death. 31 It is recommended that the new formulation be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. It is unknown if azelastine HCl is excreted in human milk, and as a result clinicians should be cautious when prescribing this medication to nursing mothers. 31
The new azelastine formulation has been approved for patients 12 years and above. Children below age 12 have not been studied and at this time it is not recommended for use in this age group. Azelastine HCl has been used in over 1400 studies involving asthmatic patients and is considered safe to use in this population. Howard et al reported no significant differences in side effects for azelastine used in elderly patients (>65) compared to younger patients (<65). 44 However, in patients with moderate renal insufficiency there was 65% increased Cmax of azelastine HCl. 44
Berger investigated the long term effect of the new formulation in a 1-year study. In this study, 862 patients were randomized into two groups, one receiving the original formulation and the other receiving the new formulation for treatment of allergic rhinitis. The results of the study showed that the new formulation is safe and well tolerated after one year use. 9 In this same study, it was found that participants using the original formulation had a higher incidence of non-compliance than the new formulation, (2.1% versus 1.4%) but this was not significant (P = 0.6041). 9 There were 48.4% and 49.1% overall adverse effects and 17.1% and 16.7% treatment related adverse effects in the new and original formulations, respectively. 9 Thus no significant difference in adverse effects or treatment related adverse effects were reported between the two formulations. Similarly, there was no difference in tolerability between Astepro 0.15% and Astepro 0.1% (Table 3). 18
Adverse reactions with ≥2% incidence in placebo-controlled trials of 2 to 4 weeks duration with astepro nasal spray 0.15% in adult and adolescent patients with seasonal or perennial allergic rhinitis. 18

Concomitant use of oral antihistamines and azelastine HCl
Studies comparing oral antihistamines have been previously conducted comparing loratiadine, cetirizine and fexofenadine to the original azelastine HCl formulation. These studies demonstrated that azelastine HCl was statistically superior to oral antihistamines (P < 0.05) using TNSS and RQLQ. Presently, there have not been any studies comparing the new formulation of azelastine to oral antihistamines. 39
Conclusions
The original azelastine formulation, though effective in the treatment of AR, NAR and MR, had mild side effects that some patients found objectionable resulting in poor adherence. The results of studies involving the new formulation of azelastine HCl demonstrate that its side effect profile is significantly improved. The most common side effects previously found to interfere with patient compliance, bitter taste and somnolence, are minimized with the new formulation. In that azelastine has multiple mechanisms of action and has previously been found to work for both SAR and PAR rhinitis subtypes, reformulation of this compound to make it more tolerable for patients to use long term should prove to be very beneficial in the management of chronic SAR and PAR. Unfortunately, the new formulation does not have the indication for NAR as the FDA has been unwilling to recognize therapeutic indications for this disorder unless a specific mechanistic action has been demonstrated. This will no doubt impede pharmaceutical development for NAR in the future.
Dosing flexibility is also another advantage azelastine provides as the new formulation (0.1%) has been demonstrated to be effective at reducing nasal symptom scores when administered 1 spray in each nostril twice daily while further reducing any potential adverse effects. The higher concentration of azelastine 0.15% now permits effective once daily dosing which may have a further favorable impact on compliance with this agent. 18
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
