Abstract
Diabetes Mellitus (DM) is a chronic disease, with a rapidly increasing worldwide incidence and prevalence. Diabetes accounts for 8% of the US population, according to the United States Centers for Disease Control and Prevention. In terms of individuals, this number comes to a staggering 24 million. 1 In 2007, the total direct medical cost of treating diabetes and its associated complications was $116 billion. More than half of this is spent in treating its complications, both micro and macrovasular. Indirect costs in terms of disability, loss of work or premature mortality amounted to an additional $58 billion. 2 Several trials have shown the benefits of improved glycemic control on microvasular complications3–7 and a propensity to reduce macrovasular disease. Furthermore tight glycemic control early in the disease, so called the legacy effect, has shown to reduce mortality. 5 However, these and several other trials have shown the progressive and unrelenting nature of the disease. Reduced efficacies of existing medications over prolonged periods, and continued beta cell dysfunction have lead to unmet glycemic targets. In addition, current antidiabetic medications have significant side effects most of which include hypoglycemia and weight gain. All the above points are rasion d'être that new additional therapies are needed. Recently, new classes of agents targeting the incretin system have become available. These can be divided into two broad categories; glucagon like peptide-1 (GLP-1) agonists/analogs (exenatide, liraglutide), and dipeptidyl peptidase-4 (DPP-4) inhibitors (sitagliptin, vildagliptin, and Saxagliptin (undergoing phase 3 trials)). Exenatide, a 39-amino acid peptide produced in the salivary gland of the Gila monster lizard, is a GLP-1 agonist. It is the first of its class approved for use as adjunctive therapy, in patients with Type 2 diabetes mellitus (T2DM). Current data suggests that exenatide, in combination with metformin, glyburide, or a glitazone, results in significant reductions in fasting and postprandial plasma glucose and hemoglobin A1c (HbA1c). Apart form gastrointestinal side effects, exenatide is relatively well tolerated and does not cause hypoglycemia when used alone. Additionally, the drug serves to promote moderate weight loss. The authors aim to provide a comprehensive overview of exenatide, detail its mechanism of action, and discuss its role in the present day treatment of patients with T2DM.
Introduction
Type 2 Diabetes Mellitus (T2DM) is a chronic complex and progressive disease, characterized by a relative lack of insulin and varying degrees of insulin resistance. Non pharmacological strategies such as diet and exercise are critical and an essential part of good diabetes management. Combining these measures with pharmacological options early in the disease process is recommended, as adherence to lifestyle changes are short lived. However, as physicians, we must continue to stress strict adherence to lifestyle modification, as these changes have pleiotropic effects apart from glycemic control. 8 Current approaches to improving glycemic control in diabetic populations include using medications which reduce hepatic glucose output (Metformin), increase insulin secretion (Sulfonylureaes, Meglitinides), augment insulin sensitivity (Glitazones), and reduce glucose absorption (Acarbose). Lastly, insulin and its analogues also come into play. Given the progressive nature of the disease, all except insulin fail to maintain glycemic control on their own. To date, less than 45% of patients with diabetes have met the recommended glycemic targets of having a glycalted hemoglobin (HbA1c) of less than 7%. Hence, a combination therapy is often required.9,10
Incretins
Incretins are hormones that are released from the gut into the bloodstream in response to oral nutrition, following which they then modulate the insulin secretory response. The insulin secretory response of incretins, called the incretin effect, accounts for at least 50% of the total insulin secreted after oral glucose load. 11 Despite the concept of a Gut hormone which increases insulin secretion being almost a century old, 12 the theory only came into force when reliable assays for insulin were available. In 1964, Elrick et al 13 were the first of three groups to show that glucose, when given orally, induced a greater insulin response (by radioimmunoassay) than intravenous (i.v.) glucose injection. This was despite higher blood glucose levels attained because of the i.v. vs. oral glucose.14,15 This lead to the discovery of local gut hormones called incretins. Subsequently in 1973, Brown and colleagues found that an enzyme that inhibited gastric acid secretion in dogs also had insulinotropic properties. They coined the term GIP (including both the synonyms gastric inhibitory peptide and glucose-dependent insulinotropic peptide). 16 However GIP, did not fully account for the increase in insulin. In 1985, another peptide produced in gut, glucagon-like peptide-1 (GLP-1), was found to be a potent insulinotropic factor. 17
GLP-1 agonist
GLP-1 is secreted by the enteroendocrine L cells that are scattered among the enterocytes throughout the small bowel and ascending colon, where they are secreted into the bloodstream when food containing fat, protein or glucose enters the duodenum. 18 Incretins account for approximately 60% of the insulin response to glucose intake. 19 Currently there is poor evidence that the deficiency of GLP-1 plays any role in the pathogenesis of T2DM. Recent data shows that GLP-1 may be increased in early phase of the disease. 20 However, as the disease progresses the level of GLP-1 is reduced when measured in patients with poor glycemic control, and in those with longer duration of DM.21,22 Thus, it seems reasonable to presume that reduced GLP-1 in addition to disease progression may be one of the reasons for increasing difficulty in maintaining adequate glycemic control. GLP-1 infusion in T2DM can normalize blood glucose, improve β-cell function, and restore first-phase insulin secretion and “glucose competence” to β cells.23,24 Hence, GLP-1/GLP-1 analogues are novel therapeutic targets for treating T2DM. Continuous GLP-1 administration is required for maintenance of glucose homeostasis because of its short half-life (1.5–2 min). GLP-1 is degraded by dipeptidyl peptidase-4 (DPP-4). Hence, therapeutic options include analogues which provide supra physiological levels of GLP-1 and are resistant to cleavage by DPP-4, or agents which inhibit the activity of DPP-4. DDP-4 has effects beyond its proteolytic action, including T-cell proliferation, 25 and is also widely expressed in human tissues including the brain, lungs, kidneys, adrenals, pancreas, and intestines. 26 Thus, there is some concern that DDP-4 inhibitors may have pleiotropic effects unrelated to diabetes control and may contribute to side effects which are not initially seen in early clinical trials.27,28
Exenatide is a 39-amino acid peptide produced in the salivary gland of the Gila monster lizard (Heloderma suspectum). 29 The drug is resistant to degradation by the DDP-4 enzyme and has an affinity for GLP-1 receptor, that is greater than endogenous GLP-1 itself. 30 Exenatide is the first GLP-1R agonist proved as an adjunct therapy in the general population suffering from T2DM.
Pharmacology
Mechanism of action
Patient's with type 2 diabetes mellitus have decreased post prandial secretion of GLP-1 and hence, an impaired incretin response. The amino acid sequence of exenatide shares a 50% sequence identity with human GLP-1, enabling exenatide to bind with and activate the human GLP-1 receptor in vitro. Binding triggers cAMP mediated and/or other intracellular signaling pathways and results in the glucoregulatory actions of exenatide. Exenatide is impervious to DPP-IV inactivation, consequently giving it a longer duration of action. It promotes the first phase of insulin secretion and the suppression of glucagon, without hindering the counter regulatory hormonal response to hypoglycemia. In patients with type 2 diabetes, this first-phase insulin response is absent. 29 As depicted by Figure 1, therapeutic plasma concentrations of exenatide significantly increase first-phase and second-phase insulin secretion in response to an IV bolus of glucose, when compared with saline (P < 0.001 for both). 30
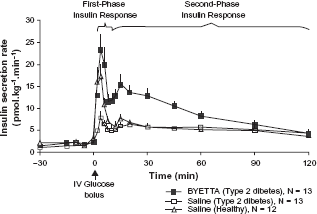
Mean (+SEM) insulin secretion rate during infusion of BYETTA or saline in patients with type 2 diabetes and during infusion of saline in healthy subjects. 30
The above effects are glucose dependant. Exenatide also slows gastric emptying, causes early satiety and increases beta cell responsiveness to glucose. Lastly, in vitro and in vivo animal models show promoted islet cell neogenesis/pancreatic beta cell proliferation. 29
Pharmacokinetics
The drug is meant for subcutaneous injection with each mL containing 250 micrograms (mcg) synthetic exenatide, 2.2 mg metacresol as an antimicrobial preservative, mannitol as a tonicity-adjusting agent, and glacial acetic acid and sodium acetate trihydrate in water for injection as a buffering solution at pH 4.5. 30 Aside from the subcutaneous route, animal data shows the nasal and intra-pulmonary routes of administration to be the next most viable. 31
Exenatide reaches median peak plasma concentrations in 2.1 h in patients with T2DM, has a half life of 2.4 hours and demonstrates pharmacological activity for approximately 6–8 hours.29,30 Exenatide exposure (AUC) is shown to increase proportionally across the therapeutic dose range of 5 mcg to 10 mcg. The mean volume of distribution following a single dose is 28.3 L. The drug is largely eliminated by glomerular filtration and following this, proteolytic degradation, with a mean clearance of 9.1 L/hrs.
Age, gender, race and body mass index do not seem to affect the pharmokinetics of exenatide. In patients with end stage kidney disease on dialysis, clearance of the drug is reduced to 0.9 L/hr. In those with mild to moderate renal impairment (creatnine clearance of 30 to 80 ml/min), clearance is shown to be only mildly reduced thus requiring no dose reduction. However, exenatide is not recommended in patients with creatinine clearance of less than 30 mls/min. At present, no data is available on exenatide use in patients with hepatic insufficiency as this is not expected to affect drug concentrations given that it is primarily renally cleared.
The co-interaction of exenatide with certain drugs like Lovastatin, Digoxin, Acetaminophen, Warfarin and Lisinopril has also been studied. These have been tabulated in Table 1. 30
Drug interactions resulting from the co-administration of exenatide (10 mcg BID). 30

Pharmacodynamics
Exenatide in patients with type 2 diabetes mellitus is shown to decrease postprandial plasma glucose concentrations, as shown in Figure 2. 30
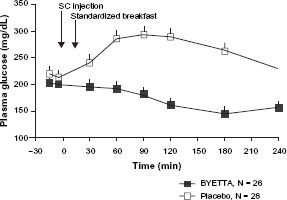
Mean (+SEM) postprandial plasma glucose concentrations on day 1 of BYETTAa treatment in patients with type 2 diabetes treated with metformin, a sulfonylurea, or both (N = 54). 30
In terms of fasting hyperglycemia, data shows immediate insulin release post exenatide injection. This is depicted in Figure 3. 30
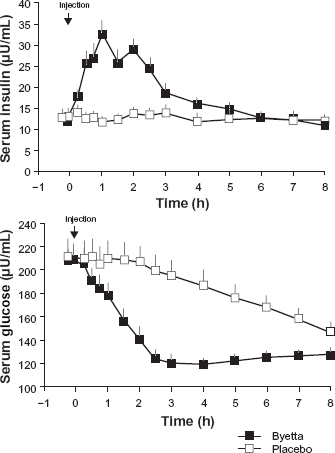
Mean (+SEM) serum insulin and plasma glucose concentrations following a one-time injection of BYETTAa or placebo in fasting patients with type 2 diabetes (N = 12).
Clinical Studies
So far, all clinical studies have measured the efficacy of anti diabetic medications using HbA1c as a marker of good diabetes care. HbA1c alone as a guide is an oversimplification of the metabolic consequences associated with diabetes, as this addresses only part of the clinical conundrum that we face when dealing with T2DM. Data showing improvements in definite macrovascular end points such as cardiovascular events and all cause mortality will take several years to show any difference given the chronicity of the disease. In T2DM, a reduction in HbA1c is shown to reduce microvascular complications, but its role in acting as a surrogate marker for reduction in macrovascular events is a contentious issue.32–34
Glycemic control
Data from randomized, triple blinded, multicentered trials involving a total of 1600 patients on metformin, or sulfonurea or both, showed an improvement in HbA1c. The study cohort were 16–75 years old, had elevated HbA1c (7.5%–11%), and were on metformin, or a sulfonylurea, or a combination of both for 30 weeks. All the aforementioned trials had an improvement in HbA1c as their primary end points, which was a dose dependent improvement with higher percentage of cohorts achieving a target of less than 7%.35–37 Similar effects were seen in patients who were on thiazolidinediones (mean difference HbA1c was -0.98%). 38 However the short duration of the study and lack of maximizing oral hypoglycemic agents (OHAs) were the main criticism for this trial. 39
Meta analysis of all published trials has shown that exenatide reduced HbA1c compared with placebo (weighted mean difference, -0.97% [95% confidence interval {CI},-1.13% to -0.81%] for GLP-1 analogues) and were non-inferior to other hypoglycemic agents. Exenatide decreased both fasting and postprandial glycemia; however, improvements in postprandial glycemic excursions were larger. 40 Patients receiving exenatide were more likely to achieve glycemic goals (A1C ≤ 7 percent) compared with patients receiving placebo (45 versus 10 percent, respectively). The modest effectiveness may, at least in part, be also attributed to participants’ relatively low baseline HbA1c (~8%) compared with older trials with other currently available therapies, wherein baseline HbA1c was often in the 9% to 10% range.
All available randomized controlled trials (RCTs) either published or unpublished showed similar results (HbA1c in comparison with placebo (-1.0 [-1.1: -0.8]) 41
These meta analyses included studies ranging from 12 weeks to 30 weeks duration in the case of the first meta analysis and up to a year in the second meta analysis. However open-label extension studies have shown maintain of reduction of HbA1c up to 82 weeks.42,43 Of note, 92 patients of the 150 cohorts completed the full duration of treatment in the second study group.
There has been some controversy regarding the effect of GLP-1 agonist on long term beta cell dysfunction. Animal studies have shown GIP has antiapoptotic properties 44 and may indeed increase beta cell mass, the holy grail of diabetic treatment. This theory has been tested by a small RCT comparing exenatide to insulin as an add on to metformin for a year. They found treatment induced change in combined glucose and arginine stimulated C-peptide secretion was 2.46 fold [95% CI,2.09 to 2.90,P < 0.0001] greater following 52-week exenatide treatment as compared to insulin glargine. Both exenatide and insulin glargine reduced hemoglobin HbA1c similarly: -0.8 ± 0.1% and -0.7 ± 0.2% respectively. Exenatide reduced body weight compared to insulin glargine (difference -4.6 kg, P < 0.0001). 45 Also in the three and a half year follow up Reductions in A1C from baseline to week 12 (-1.1 ± 0.1% [mean ± SEM]) were sustained to 3 years (-1.0 ± 0.1%; P < 0.0001), with 46% achieving A1C < or = 7%. 46 The short comings of this analysis is discussed below.
Weight loss and BMI
GLP-1 analogues resulted in weight loss in all clinical trials. 40 With respect to placebo, the difference in endpoint BMI was -0.35 [-1.14; -0.10] kg/m2. With comparison to insulin, the difference was even larger (-1.57 [-1.98; -1.15] kg/m2; P < 0.001). 41 More importantly, this weight loss was sustained in the follow up studies46,47 and retrospective analysis. 48 There was no strong co relationship between side effects of nausea and weight loss. Participants who did not report nausea also lost weight. 40
Metabolic parameters
Diabetes is a complex metabolic process and patients frequently have other cardiovascular risks such as hypertension, dyslipidemia, high BMI and fatty liver, all of which could be influenced by the medications in question. It is interesting to review data regarding Exenatide and its potential not only to reduce microvasular outcomes by reducing HbA1c, but also to influence macrovascular outcomes by modifying other risk factors.
Two opened labeled trials have shown that exenatide positively impacts other risks factors. In the first study in which analyses were conducted on data from all subjects who had the opportunity to achieve 2 years of exenatide exposure, irrespective of their treatment arm in the 30-week placebo-controlled trials. Patients with elevated ALT at baseline (151/283 [53%]) had a mean (SEM) reduction of ALT (-11 IU/L from baseline 38 [1] IU/L; P < 0.05) and 39% achieved normal ALT by week 104. Furthermore, beta-cell function as measured by the homeostasis model assessment (HOMA-B), blood pressure, and aspartate aminotransferase (AST) all have improved. 47
However, of a total of 974 patients enrolled in this 2-year extension phase, four hundred fifty-three patients were excluded due to late enrollment date and site closure. Of the remaining 521 patients that were considered eligible, only 283 completed the study. Also the percentage of patients on ACE inhibitors, statins and Aspirin, which are now the standard of care, was very low (37%–39%). The same group reported lipid profiles in a sub group of patients (151) who completed the three and a half year study period and found a 12% reduction in triglycerides, a 5% reduction in total cholesterol, a 6% decrease in low density lipoprotien (LDL), and an increase in high density lipoprotien (HDL-C) by 24%. 46 Similar effects were seen in a retrospective data analysis of approximately 300 patients. 48
Cardiovascular effects
Several animal studies have shown that GLP-1 agonist improve hemodyanamic parameters and left ventricular contractility, especially in an ischemic myocardium.49–54 At least part of this effect is thought to be insulin independent. 51 Although no studies with subcutaneous exenatide exists, I.V. infusion of GLP-1 agonists in post MI patients with reduced EF, 55 CABG, 56 and heart failure 57 showed improvement in cardiovasular outcomes. These include improvement in EF,55,57 quality of life and 6 minute walk test, 57 and the reduced use of ionotropes. 56 The benefits seen were similar in patients with and without diabetes, however the number of subjects in these studies was small (all less than 20). The studies themselves were of short duration. Nevertheless, they should pave the way for further trials.
The above data should not be confused with the effect of tight glycemic control on the rates of major cardiovascular events. As noted above, the relationship between a reduction in HbA1c and cardiovascular events has always been a contentious issue.
Till date only two trials have shown macrovascular benefit with improved glycemic control. Studies like the United Kingdom Prospective Diabetes Study (UKPDS) have shown a decrease in cardiovascular events in a 10 year follow up study with improved glycemic control. The Epidemiology of Diabetes Interventions and Complications (EDIC) trial, a 10 year follow up of the Diabetes Control and Complications Trial (DCCT), showed a delayed reduction in cardiovascular events.
Recently, the Action to Control Cardiovascular Risk in Diabetes Study Group (ACCORD) trial concluded that intensive therapy (aiming for a HbA1c <6%) did not significantly reduce major cardiovascular events, when compared to standard therapy (aiming for an HbA1c of 7.0 to 7.9%) [hazard ratio,0.90; 95% confidence interval [CI], 0.78 to 1.04; P = 0.16]. The primary outcome was the first occurrence of nonfatal myocardial infarction or nonfatal stroke or death from cardiovascular causes (myocardial infarction, heart failure, arrhythmia, stroke etc). ACCORD also showed an increased mortality in the intensive therapy group and raised the possibility of harm when intensively lowering HbA1c in high risk patients i.e. patients with a median glycated hemoglobin level of 8.1% and either previous cardiovascular events or multiple cardiovascular risk factors [hazard ratio,1.22; 95% CI,1.01 to 1.46; P = 0.04]. 58
The Glucose Control and Vascular Complications in Veterans with Type 2 Diabetes (VADT) trial looked at patients with poorly controlled T2DM and targeted a HbA1c of 6.9% in the intensive therapy group. The primary outcome in the study was the time from randomization to the first occurrence of a major cardiovascular event, a composite of myocardial infarction, stroke, death from cardiovascular causes, congestive heart failure, surgery for vascular disease, inoperable coronary disease, and amputation for ischemic gangrene. Again, there was no difference in terms of the primary outcome between the intensive therapy group and the standard therapy group (mean HbA1c 8.4%) [hazard ratio in the intensive-therapy group,0.88; 95% confidence interval [CI], 0.74 to 1.05; P = 0.14]. 59
Interestingly, this study also did not show much difference in the rates of microvascular complications. However, again the study took place in elder individuals (mean age 60.4 years) who had long standing poorly controlled T2DM (mean duration 11.5 years). 40% of the study cohort had had a cardiovascular event and likely already had microvascular disease. The mean follow-up periods in ACCORD and VADT were 3.5 years and 5.6 years, respectively. A 10 year follow up of the VADT cohort may possibly reveal a delayed benefit of intensive control. 59
Thus, we again reach an impasse. The addition of exenatide to an oral hypoglycemic regime could certainly aid in the reduction of microvascular events via a reduction in glycated hemoglobin. However, the benefit of the drug on/cardiovascular events is unclear given our current knowledge on tight glycemic control and the reduction of major cardiovascular events. Further long term data is needed to elucidate this.
Efficacy vs. other diabetic medications
Exenatide vs. DDP-4 inhibitors
No long term data are available directly comparing the two medications which act on the same pathway in a different manner. However, compared to placebo, DPP-4 inhibitors are weight neutral whereas exenatide causes a modest and sustainable weight loss. Also the other main difference is the route of administration. Exenatide is given twice a day subcutaneously and DPP-4 inhibitors are given orally. A two week double-blind, randomized cross-over, multi-center study conducted in metformin-treated T2DM patients compared exenatide to stiagliptin. This study showed exenatide had a greater effect than sitagliptin to lower postprandial glucose, and a more potent effect to increase insulin secretion and reduce postprandial glucagon secretion. In contrast to sitagliptin, exenatide slowed gastric emptying and reduced caloric intake by increasing satiety. 60 This study, though of short duration, confirmed the finding of previous trials; exenatide increases GLP-1 levels more so than DPP-4 inhibitors.
Exenatide vs. insulin
The comparison of exenatide vs. Insulin and its analogues is confounding. A meta analysis of 4 trials showed no difference between glycemic control between the two groups. 41 However, in this meta analysis, all the four trials were open labeled and the mean HbA1c was 8.4%. Higher HbA1c could suggest more beta cell dysfunction and hence resistance to exenatide. Also in the glargine study, the target level was reached in only 21.6 percent of the subjects randomly assigned to glargine, and the average dose used (25 units) was substantially lower than in most studies. This relatively non aggressive insulin therapy may have tilted the balance of overall glycemic control in favor of exenatide. 61
This theory has been vindicated in a RCT of patients with type 2 diabetes for a mean duration of 9 years and mean HbA1c of 9% showed improved glycemic control in the Insulin group when compared to exenatide (HbA1c < 7%: 37% vs. 20%, P = 0.006). 62 This finding was substantiated by another study which examined replacement of insulin with exenatide in patients with type 2 diabetes; glycemic control deteriorated in 38 percent (11 of 29 patients) when exenatide was substituted, compared with 19 percent (3 of 16 patients) who continued insulin. Patients who lost glycemic control were more likely to have a longer duration of disease, lower C-peptide concentrations (suggesting less endogenous beta cell function), and larger insulin requirements at baseline. 63 Combining the results of these two studies correlates with our current knowledge that diabetes is a progressive disease with ongoing beta cell dysfunction, hence the relative lack of improvement in glycemic control with exenatide when compared to insulin in patients with long standing disease.
There has been no head to head comparison of exenatide vs. other oral hypoglycemic agents, however given the average HbA1c reduction of approximately 1%, one can safely assume that it is as efficient as other drugs in reduction of HbA1c.
Safety
Hypoglycemia
Hypoglycemia is a common side effect of most anti diabetic medications. However, incretins exhibit their actions in a glucose dependent manner and therefore the rates of hypoglycemia are less. When all the evidence from the trials reporting hypoglycemia was combined in a meta analysis, it was found that hypoglycemia with exenatide was mainly observed in combination with sulfonylureas (MH-OR 4.35 [1.89;11.21], P = 0.001 and 1.37 [0.72;2.36], P = 0.34, in trials with or without sulphonylureas, respectively). When compared with insulin, exenatide twice daily was not associated with increased risk for hypoglycemia (MH-OR 0.61 [0.33;1.14], P = 0.125). Severe hypoglycemic episodes (7 in total) were also only exclusively in combination with sulphonylureas. 41 In trials comparing exenatide with insulin, 12 patients experienced severe hypoglycemia (5 with exenatide, 7 with insulin P = 0.61). 41
Acute pancreatitis
Initial reports showing an increased incidence of acute pancreatitis (especially necrotizing hemorrhagic pancreatitis), caused concern. However, important confounders may have been missed in these initial reports. Obesity (a body-mass index of more than 30), for example, is associated with an increase in the risk of a severe clinical course of pancreatitis by a factor of 2 to 3. 64 In addition, many patients with type 2 diabetes mellitus have hypertriglyceridemia, which could be playing a role in causing acute pancreatitis. An observational study was published looking at a healthcare database of approximately 28,000 cohorts of exenatide initiators and matched to an equal number of metformin or glyburide initiators. During follow-up of up to 1 year, acute pancreatitis occurred among 0.13% of patients treated with exenatide. The risk of acute pancreatitis was comparable for initiators of exenatide (RR 1.0; 95% confidence interval (CI) 0.6–1.7) relative to the comparison cohorts. 65
Deaths and mortality
The number of deaths was too low in the reported trials to derive statistical significance difference in the drug and placebo groups. This is to be expected in studies of short duration.
Tolerability
Nausea (44 vs. 18%) and vomiting (13 vs. 4%) were the main side effects, when compared to placebo. Nausea tended to occur most on initiation of therapy and commonly subsided over the first few weeks.66,67 It was reduced by 34% (P = 0.005), when a target dose of exenatide was achieved in patients with gradual dose escalation compared with patients receiving the equivalent target dose without titration. 43 A post hoc analysis of the three AMIGO trials showed that the reduction in HbA1c and weight was not likely to be a direct effect of nausea. Patients with mild nausea had similar changes in Hba1c and weight compared with the other sub groups. 68
Place in Therapy
Over the years, a number of different agents have been developed to treat type 2 diabetes mellitus, ranging from oral hypoglycemic agents like metformin to various insulin formulations like glargine. In 2008, Nathan et al developed a consensus algorithm for the initiation and adjustment of therapy in patients with T2DM. Bearing in mind the goal to keep HbA1C levels less than 7%, this algorithm takes into consideration the characteristics of individual interventions, their costs and their synergies (Table 2).
Algorithm for the initiation and adjustment of therapy in patients with T2DM. 69
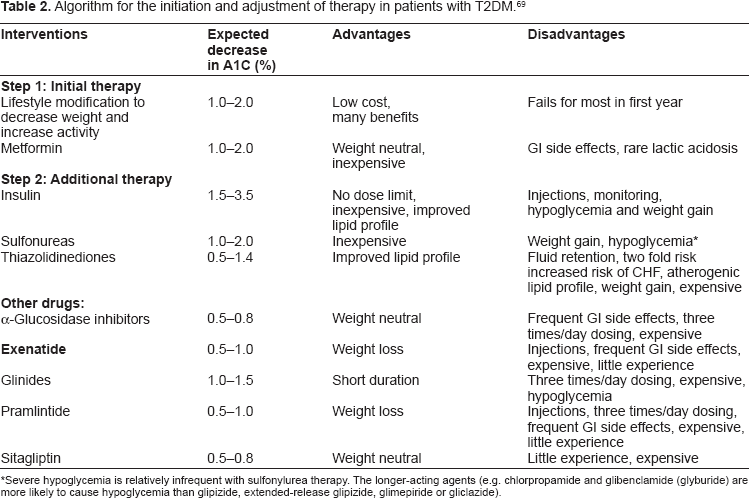
Severe hypoglycemia is relatively infrequent with sulfonylurea therapy. The longer-acting agents (e.g. chlorpropamide and glibenclamide (glyburide) are more likely to cause hypoglycemia than glipizide, extended-release glipizide, glimepiride or gliclazide).
Tier 1 encompasses well-validated core therapies and Tier 2, less well-validated therapies. Under the umbrella of Tier 1, this treatment algorithm recommends the initiation of lifestyle modification and starting patients on metformin. Following this, if there is a failure to achieve glycemic goals after titration of metformin to its maximal effective dose, either insulin or a sulfonurea should be added. For HbA1C's greater than 8.5 or symptomatic hyperglycemia, insulin is the better choice. The application of Tier 2 therapies comes into play in selected clinical settings, especially where one is trying to avoid hypoglycemia. Here, one may add pioglitazone or exenatide to metformin. If HbA1C is less than 8% and weight loss is a consideration, then exenatide is recommended. Off note, DPP-4 inhibitors, metaglinides and amylin agonists are not included in these two tiers due their equal or lower effectiveness in lowering blood glucose and/or their relative expense or limited clinical data. 67
Conclusion
GLP-1 analogues like exenatide serve the function to restore GLP-1 levels that are found to be decreased in T2DM, and thus hold great potential in treating patients with T2DM. The vast majorities of patients with T2DM are obese and have other associated cardiovascular risk factors. GLP-1 analogues appear to be ideal drugs due to their effects on weight loss. In animal studies, exenatide has shown to improve pancreatic function by decreasing apoptosis and increasing beta cell proliferation. Therefore, it is an excellent adjunctive therapy for many patients who are on a sulfonurea, metformin or a combination of both, but still have not achieved desirable glycemic control. Microvascular benefits of tighter glycemic control are well documented. The effect on macrovascular/cardiovascular outcomes is yet to be determined. Most common side effects of exenatide include nausea, vomiting, but these are short lived as patients generally overcome them with time. Caution should be exercised in patients who have very high triglycerides. Lastly, exenatide is not recommended in patients with end stage renal disease.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
