Abstract
Type 2 diabetes is a well known risk factor for cardiovascular disease (CVD). While glycemic control has consistently been shown to prevent microvascular complications, large randomized trials have not demonstrated the same consistent beneficial effects of intensive glycemic control in improving cardiovascular (CV) outcomes. Thus, optimal glucose control alone is not sufficient to reduce CV risk. Aggressive management of CV risk factors such as blood pressure, lipids, and body weight is also necessary. A growing body of evidence suggests that the recently available glucagon-like peptide 1 receptor (GLP-1R) agonists have beneficial CV effects beyond glucose control. Studies have demonstrated beneficial effects in the myocardium, endothelium, vasculature and various markers of cardiovascular risk such as body weight, blood pressure and dyslipidemia. Despite the growing evidence, large, randomized, blinded clinical trials with hard CV endpoints have not been performed. Most human studies have been small, and have focused on surrogate endpoints. The findings need to be confirmed by prospective, randomized cardiovascular outcomes trials. In this review we examine the GLP-1R agonist data on weight reduction, blood pressure lowering, beneficial changes in dyslipidemia, and improvements in myocardial and endothelial function. The safety as well as potential role of these agents in treatment regimens for type 2 diabetes is also addressed.
Introduction
Type 2 diabetes is a well established risk factor for cardiovascular disease (CVD) [Fox et al. 2007; Sarwar et al. 2010]. Data from the Framingham Heart Study demonstrated a 78% lifetime risk of developing CVD in men with diabetes compared with 54.8% in men without diabetes [Fox et al. 2008]. In women, the lifetime risk was 67.1% with diabetes and 38% without diabetes. A meta-analysis of 102 prospective studies that included approximately 700,000 patients showed that, independent of other conventional risk factors, diabetes was associated with a twofold increased risk of CVD [Sarwar et al. 2010]. Population-based studies have demonstrated that the incidence of myocardial infarction (MI) in patients with diabetes without a history of MI is similar to that in patients without diabetes with a history of MI [Haffner et al. 1998; Juutilainen et al. 2005]. Historically, epidemiologic studies have reported a linear and graded association between increasing hemoglobin A1c (HbA1c) levels and risk of cardiovascular (CV) events [Sarwar et al. 2010; Selvin et al. 2005]. However, large randomized, clinical trials including ACCORD (Action to Control Cardiovascular Risk in Diabetes) [Gerstein et al. 2008], ADVANCE (Action in Diabetes and Vascular Disease Preterax and Diamicron Modified Release Controlled Evaluation) [Patel et al. 2008], and VADT (Veterans Affairs Diabetes Trial) [Duckworth et al. 2009] have failed to show a consistent reduction in CVD events with tighter glucose control. The ACCORD trial [Gerstein et al. 2008] randomized 10,251 patients at high risk for CV events to intensive glucose control (target HbA1c < 6.0%) or standard therapy (target HbA1c 7.0–7.9%). The primary outcome was a composite of nonfatal MI, nonfatal stroke, or death from CV causes. After a mean follow up of 3.5 years, the primary outcome was not significantly different between groups (p = 0.16). Furthermore, compared with patients receiving standard care, intensive glucose control was associated with a higher number of total and CV deaths [hazard ratio (HR) 1.22, 95% confidence interval (CI) 1.01–1.46], a finding that led to early termination of the study after 3.5 years. The reasons for the increased risk of CV mortality among patients in the intensive glucose control arm remain unclear. The ADVANCE trial [Patel et al. 2008] examined the impact of intensive glycemic control on microvascular and major CV events (MI, stroke, and CVD death) in 11,140 patients with diabetes. While microvascular events were reduced with intensive glucose control, there were no significant reductions in CV events. In VADT [Duckworth et al. 2009] no significant reduction in CV events was noted in patients who received intensive glucose lowering for a median duration of 5.6 years despite an average 1.5% lower median HbA1c throughout the trial. In contrast to ACCORD, results from ADVANCE and VADT did not demonstrate excess mortality with intensive regimens that achieved HbA1c levels comparable with the 6.0% to 6.5% achieved in ACCORD. Although the individual trials failed to show consistent beneficial effects on CV events, an inadequate sample size may have underpowered these trials to detect a significant difference. Recently, two independent meta-analyses of five large prospective randomized controlled trials (including ACCORD, ADVANCE, and VADT) suggest that an association does exist between intensive glucose control and CV risk reduction [Mannucci et al. 2009; Ray et al. 2009]. The results of the meta-analyses showed that intensive therapy achieving an HbA1c level 0.9% lower than standard therapy resulted in significant reductions in coronary events without an increased risk of death [Mannucci et al. 2009; Ray et al. 2009].
Beyond optimal glucose control, aggressive management of CV risk is necessary for patients with type 2 diabetes. Standard glucose-lowering agents have been associated with undesirable side effects such as weight gain and hypoglycemia and have demonstrated either no effect or a very small effect on CV outcomes. Therefore, the need exists for new drugs that have a low risk for hypoglycemia and weight gain, and in addition, have beneficial CV effects. The recently introduced glucagon-like peptide-1 receptor (GLP-1R) agonists may address some of the challenges. There is evidence to show that, in addition to providing glycemic control, GLP-1R agonists have beneficial effects in improving multiple CVD risk factors. In this article we examine the emerging data on weight reduction, blood pressure lowering, beneficial changes on lipids, and improvements in cardiac and endothelial function. The safety and potential place of GLP-1R agonists in treatment regimens for type 2 diabetes is also addressed.
Pharmacology
GLP-1R agonists target an important pathophysiological defect in type 2 diabetes known as the ‘incretin effect’. It has been known for some time that glucose given orally elicits a much larger insulin response than when administered intravenously [Nauck et al. 1986]. The incretin effect may account for up to 50–70% of the total insulin secreted after glucose ingestion. In patients with type 2 diabetes the incretin effect is significantly reduced to less than 50% compared with individuals without diabetes. The incretin hormones, glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 are released from endocrine cells of the intestinal mucosa within minutes after food ingestion. GLP-1, the more physiologically relevant incretin, exerts multiple effects [Drucker and Nauck, 2006; Holst, 2007]. It enhances glucose-dependent insulin secretion and concurrently inhibits postprandial glucagon secretion [Gautier et al. 2008; Nauck et al. 1986]. In addition, GLP-1 slows gastric emptying and increases CNS-mediated satiety that leads to reduced food intake and subsequent weight loss [Gautier et al. 2008; Nauck et al. 1986; Mudaliar and Henry, 2009].
In patients with type 2 diabetes the postprandial secretion of GLP-1 is significantly decreased [Vilsbøll et al. 2001]. Exogenously administered GLP-1 has a very short half life of approximately 1–2 min because it is rapidly inactivated by the enzyme dipeptidyl peptidase 4 (DPP-4) [Deacon et al. 1995]. Therefore, a continuous infusion of GLP-1 would be required to achieve a clinical effect, limiting the practical use of this option. Efforts to overcome this barrier led to the development of GLP-1R agonists resistant to DPP-4 inactivation. Exenatide was the first GLP-1R agonist approved by the US Food and Drug Administration (FDA). With an amino acid sequence that is 53% identical to that of human GLP-1, exenatide activates GLP-1 receptors similarly to native GLP-1, but is resistant to DPP-4 inactivation and consequently has a longer half life [Amylin Pharmaceuticals, 2011]. Exenatide is administered subcutaneously twice daily at any time within a 60-min period before the morning and evening meals [Neumiller, 2009]. Because of its pharmacokinetics in renal impairment, exenatide should not be used for patients with a creatinine clearance of 30 ml/min or less.
Liraglutide, a human GLP-1 analog, has an amino acid sequence that is 97% identical to that of human GLP-1. Two modifications in the amino acid sequence of human GLP-1 plus a fatty acid side chain confer resistance to DPP-4 degradation and slow absorption of the drug into the circulation [Ross and Ekoe, 2010]. With a half life of 11–15 h, liraglutide is administered once daily, regardless of meal times. Dosage adjustment is not necessary in patients with renal impairment [Novo Nordisk, 2011].
Glucagon-like peptide-1 receptor agonist effects beyond glycemic control
Clinical studies have demonstrated that exenatide and liraglutide have beneficial effects beyond glucose lowering. Significant reductions in body weight, blood pressure, and beneficial changes in dyslipidemia have been reported. Several small trials have shown improvements in vascular inflammation, endothelial dysfunction, and beneficial effects on left ventricular function during ischemic conditions and heart failure.
Effects on body weight
In contrast to most therapies for type 2 diabetes that promote weight gain, GLP-1R agonists have been associated with weight loss. A meta-analysis performed by Vilsbøll and colleagues found that use of GLP-1R agonists in 6411 overweight [body mass index (BMI) ≥ 25] patients resulted in a mean weight loss of −2.9 kg (95% CI −3.6 to −2.2) [Vilsbøll et al. 2012]. Studies have shown that the weight loss achieved with continued GLP-1R agonist treatment is sustained over years [Astrup et al. 2011; Blonde et al. 2006; Garber et al. 2011; Klonoff et al. 2008]. The mechanism for weight loss is not clearly understood. It may be secondary to nausea, a common side effect of GLP-1R agonist therapy; however, patients who do not experience nausea also show weight loss [Blonde et al. 2006]. Further, when nausea occurs, it is during the initial weeks of therapy and decreases over time, whereas weight loss is reportedly progressive [Blonde et al. 2006]. Other postulated mechanisms for weight reduction include early satiety that may be mediated through delayed gastric emptying [Nauck et al. 1996; Drucker et al. 2008] as well as activation of central GLP-1 receptors [Baggio et al. 2004; Pannacciulli et al. 2007].
Clinical trials have consistently demonstrated significant weight loss with exenatide [Buse et al. 2004a, 2011; Kendall et al. 2005; Heine et al. 2005; Zinman et al. 2007; Drucker et al. 2008; Moretto et al. 2008; Liutkus et al. 2010]. Selected trials are summarized in Figure 1. In a randomized, double-blind, placebo-controlled trial, drug-naïve patients treated with exenatide 10 µg twice daily for 24 weeks experienced a weight loss of 3.1 kg from baseline (p < 0.001 versus placebo), while those receiving exenatide 5 µg twice daily experienced a 2.8 kg reduction in weight (p = 0.004 versus placebo) [Moretto et al. 2008]. Added to metformin, exenatide twice daily at the 5 µg and 10 µg doses produced weight reductions of 1.6 kg (p < 0.05) and 2.8 kg (p < 0.001), respectively [DeFronzo et al. 2005]. When added to a sulfonylurea, exenatide at 5 µg and 10 µg doses produced a weight loss of 0.9 kg [p = nonsignificant (NS)] and 1.6 kg (p < 0.05) respectively [Buse et al. 2004a]. In a 30-week double-blind, placebo-controlled randomized trial of 733 subjects with diabetes, exenatide at doses of 5 and 10 μg added to metformin and sulfonylurea elicited a weight loss of 1.6 kg in both groups compared with 0.9 kg in the placebo group (p < 0.01) [Kendall et al. 2005]. In an open-label trial comparing exenatide with insulin glargine, patients on metformin plus sulfonylurea randomized to add-on therapy with exenatide 10 µg lost an average of 2.3 kg compared with a weight gain of 1.8 kg in the insulin glargine group (p < 0.0001) [Heine et al. 2005].
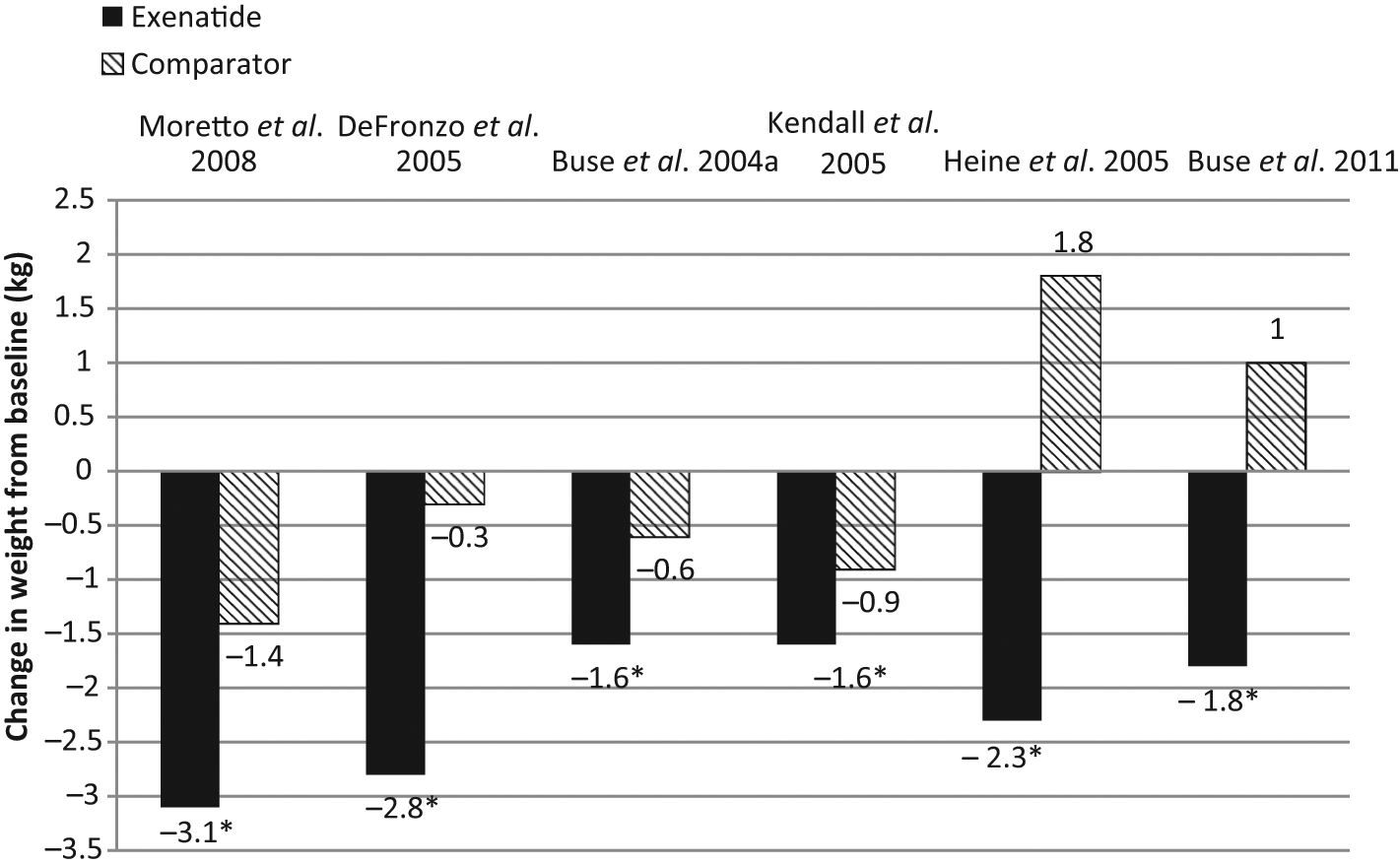
Weight reductions with exenatide from selected clinical trials.
Once-weekly exenatide, a compound recently approved in the USA, has also demonstrated the potential for weight loss. In a randomized 30-week open-label study, similar weight loss occurred in groups receiving once-weekly exenatide (3.7 kg) and exenatide twice daily (3.6 kg), p = 0.89 [Drucker et al. 2008]. Compared with sitagliptin or pioglitazone, once-weekly exenatide demonstrated significantly more weight loss [Bergenstal et al. 2010]. Once-weekly exenatide added to metformin ± sulfonylurea, produced an average weight loss of 2.6 kg versus a mean weight gain of 1.4 kg among patients receiving insulin glargine (p < 0.0001) [Diamant et al. 2010].
Similar to exenatide, beneficial reductions in body weight have been demonstrated with liraglutide [Buse et al. 2009; Marre et al. 2009; Nauck et al. 2009; Garber et al. 2009; Zinman et al. 2009; Russell-Jones et al. 2009]. Selected trials showing liraglutide effects on weight loss are summarized in Figure 2. In a double-blind, randomized controlled trial comparing liraglutide with glimepiride, patients assigned to liraglutide monotherapy lost an average of 3.1 kg compared with a mean gain of 1.1 kg in the glimepiride group (p = 0.0001) [Garber et al. 2009]. As add-on therapy to metformin, liraglutide was associated with a mean 2.8 kg weight loss, while glimepiride added to metformin produced an average 1 kg weight gain (p < 0.01) [Nauck et al. 2009]. Marre and colleagues found that the addition of liraglutide to glimepiride blunted the weight gain observed in the group treated with glimepiride plus rosiglitazone (p < 0.0001) [Marre et al. 2009]. In a randomized, controlled trial comparing liraglutide with insulin glargine, liraglutide decreased weight by 1.8 kg compared with a weight gain of 1.6 kg in the insulin glargine group (p < 0.0001) [Russell-Jones et al. 2009]. In combination with metformin and rosiglitazone, weight loss with liraglutide was 2 kg at the 1.8 mg dose and 1 kg at the 1.2 mg dose [Zinman et al. 2009]. In a recent 20-week trial, 546 people with obesity were randomly assigned to any one of four doses of liraglutide, placebo, or orlistat. Patients on liraglutide lost significantly more weight than those on placebo and orlistat [Astrup et al. 2009]. The weight loss appears to be associated with reduction in body fat. Studies with dual X-ray absorptiometry and computerized tomography have shown that weight reduction with liraglutide is accompanied by significant decreases in subcutaneous and intra-abdominal fat [Jendle et al. 2009].
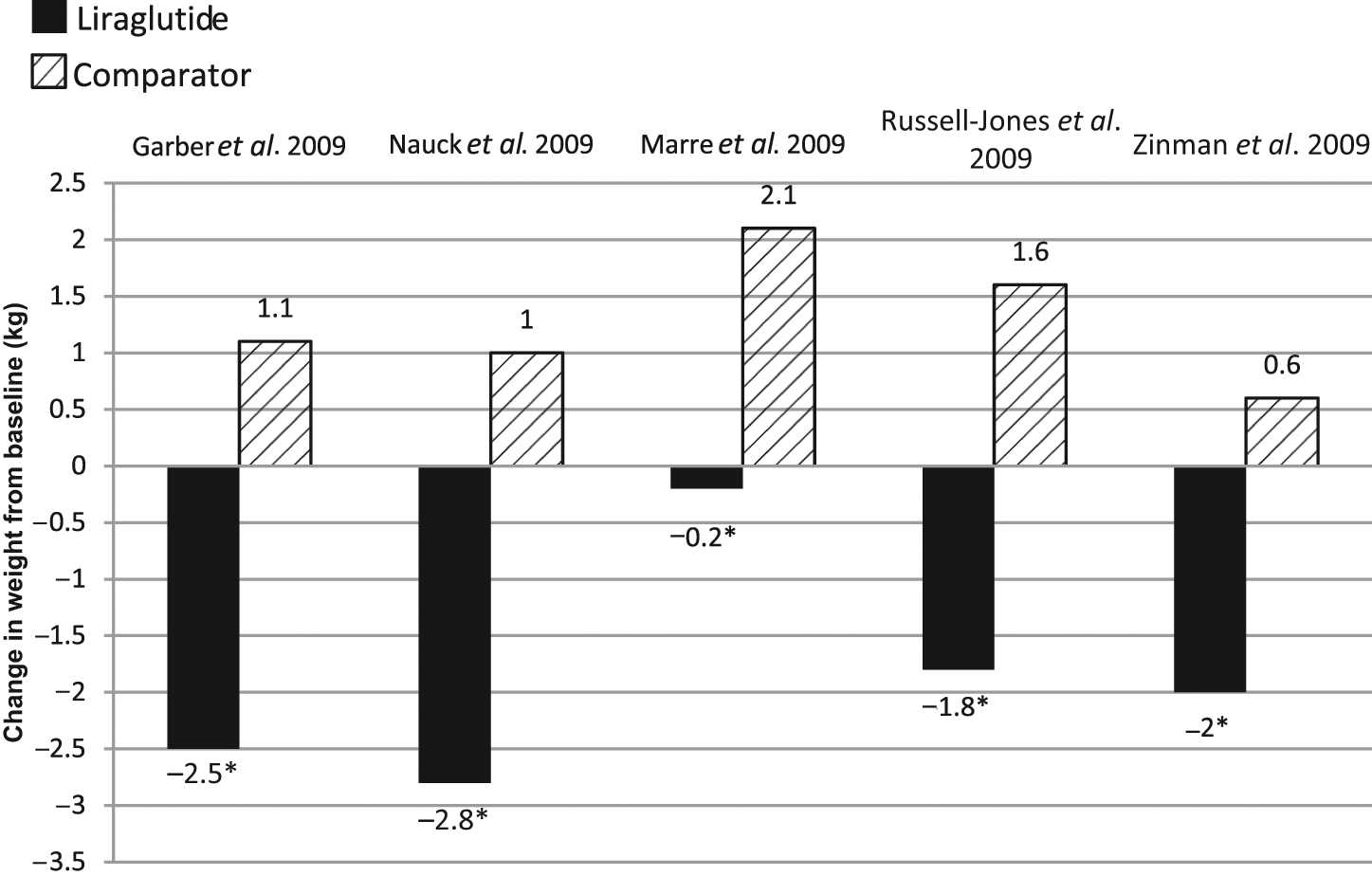
Weight reductions with liraglutide from selected clinical trials.
Effects on blood pressure
Increased sympathetic activity, endothelial dysfunction, and impaired natriuresis have been linked to hypertension in patients with type 2 diabetes [Bell, 2003]. Clinical trials with GLP-1R agonists have shown modest though sometimes significant reduction in blood pressure (Table 1). Although the mechanism for blood pressure reduction is not clear, weight loss, natriuretic, vasodilatory, and direct central effects may play a role [Yu et al. 2003; Barragan et al. 1994; Bojanowski and Stempniak, 2000].
Effects of glucagon-like peptide 1 agonists on selected cardiovascular risk factors.
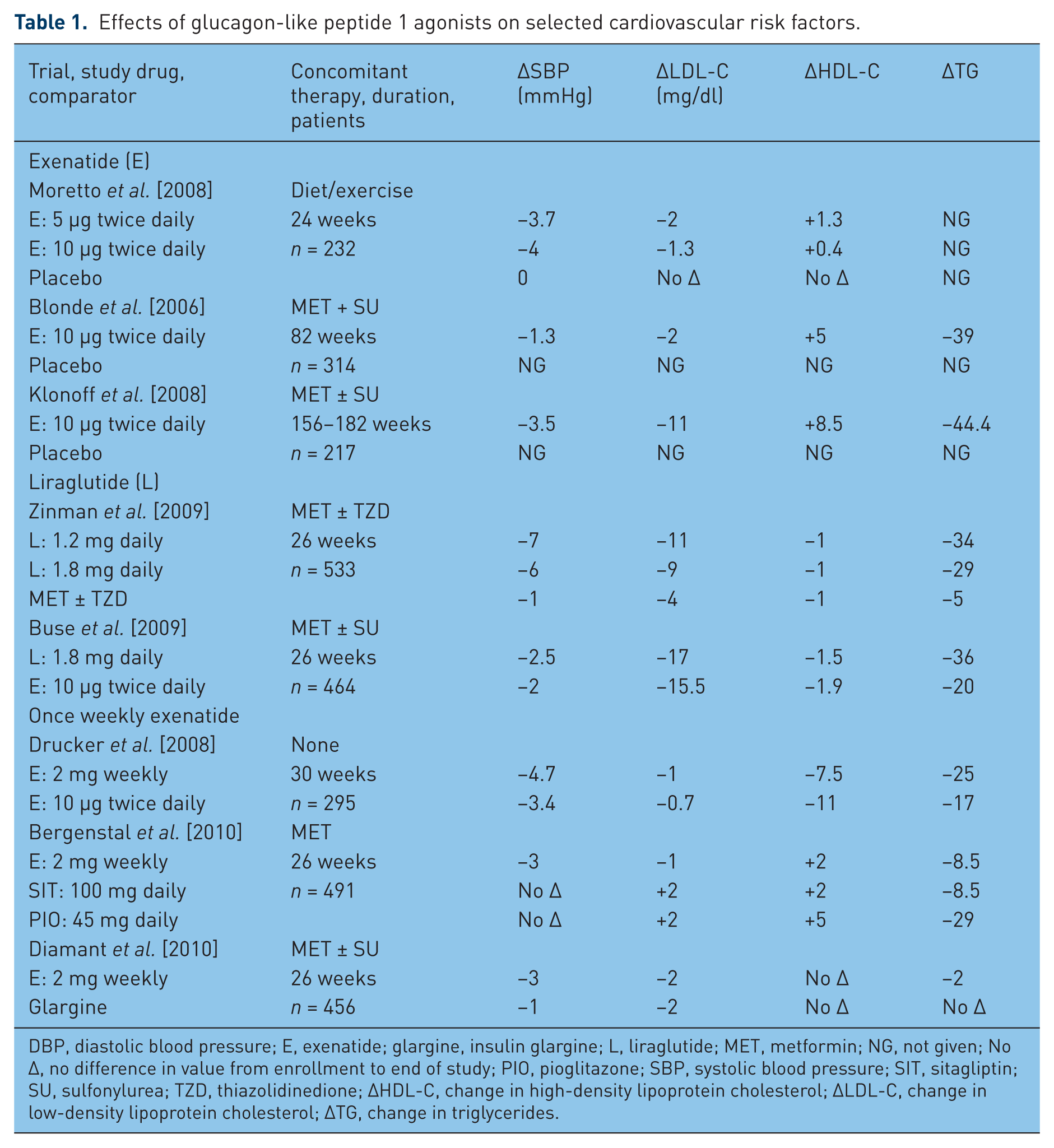
DBP, diastolic blood pressure; E, exenatide; glargine, insulin glargine; L, liraglutide; MET, metformin; NG, not given; No Δ, no difference in value from enrollment to end of study; PIO, pioglitazone; SBP, systolic blood pressure; SIT, sitagliptin; SU, sulfonylurea; TZD, thiazolidinedione; ΔHDL-C, change in high-density lipoprotein cholesterol; ΔLDL-C, change in low-density lipoprotein cholesterol; ΔTG, change in triglycerides.
In a 24-week study that included 232 treatment-naïve patients, exenatide (5 or 10 µg twice daily) produced significant reductions in systolic blood pressure (SBP) (up to −3.7 mmHg, p = 0.037) and diastolic blood pressure (DBP) (up to −2.3 mmHg, p = 0.046) compared with placebo [Moretto et al. 2008]. Added to metformin or a thiazolidinedione (TZD), a 12-week placebo-controlled, randomized trial showed that treatment with exenatide twice daily produced a trend toward reductions in mean 24 h, daytime, and nighttime SBP compared with placebo. There was no effect on DBP [Gill et al. 2010]. In a pooled analysis of data from six randomized, placebo-controlled or insulin-controlled trials involving 2171 patients with a follow up of 24–52 weeks [Okerson et al. 2010], treatment with exenatide resulted in a significantly greater reduction in SBP compared with placebo (difference of −2.8 mmHg; p = 0.0002) and versus insulin (difference of −3.7 mmHg; p < 0.0001). The reduction in blood pressure was highest in patients with SBP of 150 mmHg or higher. No significant differences in DBP were observed in the exenatide versus placebo group (−0.7 mmHg versus −0.2 mmHg, p = 0.21) or the exenatide versus insulin group (−1.6 mmHg versus −0.8 mmHg, p = 0.16) [Okerson et al. 2010]. Clinical trials comparing once-weekly with twice-daily exenatide [Drucker et al. 2008], sitagliptin or pioglitazone [Bergenstal et al. 2010], and insulin glargine [Diamant et al. 2010] have also demonstrated beneficial effects on SBP reduction. No significant changes in DBP were observed in any treatment. Evidence from long-term studies (up to 3.5 years) indicates that the blood pressure lowering effect of exenatide may be sustained [Blonde et al. 2006; Klonoff et al. 2008].
Decreases in SBP have also been demonstrated with liraglutide [Buse et al. 2009; Marre et al. 2009; Nauck et al. 2009; Garber et al. 2009; Zinman et al. 2009; Russell-Jones et al. 2009]. When compared with glimepiride, liraglutide produced a 2–3 mmHg reduction in SBP reduction [Nauck et al. 2009]. In a 26-week study of liraglutide in combination with metformin plus a TZD, there was a 5–7 mmHg reduction of SBP, but no change in DBP [Zinman et al. 2009]. A meta-analysis of the Liraglutide Effect and Action in Diabetes (LEAD) trials showed SBP reductions of between 1.9 and 4.5 mmHg associated with liraglutide [Marre et al. 2009; Nauck et al. 2009; Garber et al. 2009; Zinman et al. 2009; Russell-Jones et al. 2009; Buse et al. 2009]. No change in DBP was observed. Interestingly, blood pressure reductions were evident within the first 2 weeks of starting liraglutide and preceded weight loss; suggesting that GLP-1R agonists lower blood pressure through effects on the vasculature even prior to significant weight loss.
Effects on heart rate
In addition to blood pressure, heart rate also contributes to CV risk. In patients with suspected or established CVD, an increased heart rate has been associated with an increased risk of overall and CV mortality, an effect that is more pronounced in patients with diabetes [Diaz et al. 2005]. Clinical trials have found that the use of liraglutide is associated with statistically significant increases in heart rate ranging from 1.5 to 4 beats per minute [Marre et al. 2009; Garber et al. 2009; Zinman et al. 2009; Russell-Jones et al. 2009]. In a 12-week study that directly examined the effect of exenatide on heart rate, Gill and colleagues found a nonsignificant increase of 1.5 beats per minute with exenatide therapy compared with placebo (p = 0.41) [Gill et al. 2010]. Lastly, after 26 weeks heart rate was found to increase by 4 beats per minute with once weekly exenatide compared with insulin glargine (p < 0.05) [Diamant et al. 2010].
Effects on lipids
Diabetic dyslipidemia is characterized by elevated triglyceride (TG) and reduced high-density lipoprotein cholesterol (HDL-C), while total cholesterol (TC) and low-density lipoprotein cholesterol (LDL-C) levels may be elevated or remain normal [Howard and Howard, 1994]. Treatment goals are aimed at raising HDL-C and lowering TG and LDL-C [ATP III, 2001]. Standard antidiabetic agents have variable effects on the lipid profile. Metformin lowers LDL-C and TG, while pioglitazone has been associated with increases in HDL-C and reduction in TG compared with placebo [Buse et al. 2004b]. High postprandial TG and free fatty acid (FFA) levels have been associated with insulin resistance and atherogenicity [Patsch et al. 1992; Karpe et al. 1994; Tushuizen et al. 2005; Bell et al. 2008].
In an early study in 14 healthy subjects, Meier and coworkers studied the effects of GLP-1 infusion on postprandial lipid levels [Meier et al. 2006]. A single intravenous infusion of GLP-1 completely abolished the postprandial increase in TG levels. Selected trials showing the lipid effects of GLP-1R agonists are presented in Table 1. In monotherapy, exenatide showed no significant changes in lipid parameters compared with placebo [Moretto et al. 2008]. Placebo-controlled trials have evaluated lipid changes when exenatide was added to metformin alone [DeFronzo et al. 2005], sulfonylurea alone [Buse et al. 2004], or metformin plus sulfonylurea [Kendall et al. 2005]. At week 30, no significant differences in lipid parameters were observed in these studies for either the exenatide group or placebo in terms of TC, LDL-C, HDL-C, or TG. Nevertheless, in an 82-week open-label extension of these studies, patients receiving exenatide 10 μg twice daily experienced a 16% reduction in TG levels (mean 38.6 mg/dl from baseline; 95% CI −55.5 to −21.6), and a 12% increase in HDL-C (mean 4.6 mg/dl from baseline; 95% CI 3.7–5.4) [Blonde et al. 2006]. Interestingly, the authors noted a definite correlation between decrease in body weight and improvements in the lipid profile. The greatest decreases in TG and increases in HDL-C occurred in patients who lost the most weight [Blonde et al. 2006].In a subset of patients treated with exenatide for approximately 3.5 years, Klonoff and colleagues reported a 12% reduction in TG (mean change 44.4 mg/dl; p = 0.0003), a 5% reduction in TC (mean change 10.8 mg/dl; p = 0.0007), a 6% decrease in LDL-C (mean change 11.8 mg/dl; p < 0.0001), and HDL-C increased significantly by 24% (mean change 8.5 mg/dl; p < 0.0001) [Klonoff et al.2008].
Analysis of liraglutide trials have demonstrated reductions in TG, LDL-C, and free fatty acids [Buse et al. 2009; Marre et al. 2009; Nauck et al. 2009; Garber et al. 2009; Zinman et al. 2009; Russell-Jones et al. 2009]. One study randomized 533 patients in a double-blind, placebo-controlled parallel-group trial to liraglutide 1.2 mg, 1.8 mg once daily, or placebo plus metformin and rosiglitazone [Zinman et al. 2009]. There were significant reductions in LDL-C and TG versus placebo, while slight but nonsignificant changes in HDL-C were observed (Table 1). In a 26-week randomized, parallel-group, open-label trial Buse and associates evaluated the effects of exenatide versus liraglutide [Buse et al. 2009]. Patients receiving metformin ± sulfonylurea were randomly assigned to receive supplementary liraglutide 1.8 mg once daily (n = 233) or exenatide 10 µg twice daily (n = 231). Reductions in TG were significantly greater in the liraglutide group (p = 0.048). Both treatment groups had a similar reduction in LDL-C. HDL-C levels were slightly reduced in both groups (Table 1).
In a randomized, open-label, noninferiority study, exenatide once-weekly versus twice daily showed insignificant changes in lipid parameters [Drucker et al. 2008] (Table 1). Bergenstal and colleagues, in a 26-week study, compared once weekly exenatide with sitagliptin or pioglitazone. Patients receiving once-weekly exenatide experienced a mean 8.5 mg/dl reduction in TG compared with reductions of 8.5 mg/dl in the sitagliptin and 29 mg/dl in the pioglitazone group (p = 0.062). No significant changes in LDL-C were observed in any group [Bergenstal et al. 2010]. In an open-label, 26-week, randomized trial, once-weekly exenatide was compared with insulin glargine in patients receiving metformin ± sulfonylurea [Diamant et al. 2010]. Patients receiving once weekly exenatide experienced a 2 mg/dl nonsignificant reduction in TG and LDL-C compared with no change in the insulin glargine group [Diamant et al. 2010].
Effect on endothelial function
In patients with type 2 diabetes, vascular endothelial dysfunction plays a key role in atherosclerosis. It is linked to increased protein glycosylation and impaired nitric oxide (NO) production [Dunn et al. 2006; Tabet and Rye, 2009]. Advanced glycated end products can cause endothelial damage. There is emerging evidence to show that GLP-1 has beneficial effects on the endothelium. In diabetic rats, prolonged exposure to GLP-1 or exendin-4 improved endothelial dysfunction, relieved vasoconstriction [Ozyagan et al. 2005; Yu et al. 2003; Golpon et al. 2001], and induced an endothelial-dependent relaxation of pulmonary artery vessel tone [Golpon et al. 2001]. In human studies, GLP-1 infusions significantly increased flow-mediated vasodilation (FMD) in the brachial artery, 3.1% ± 0.6% versus 6.6% ± 1.0%; p < 0.05 [Basu et al. 2007]. FMD correlates with endothelial dysfunction in the coronary circulation. The exact mechanism by which GLP-1 improves endothelial dysfunction is unclear. Some investigators observed a vasodilatory effect of GLP-1 independently of NO, indicating a direct action of GLP-1 at GLP-1 receptors in vascular smooth muscles [Nystrom et al. 2005; Green et al. 2008]. Others postulate that GLP-1 may have an impact on reducing glycosylation and improving NO production through improvement in hyperglycemia [Ban et al. 2008]. Regardless of the mechanism, GLP-1-related vasodilation and improvement in endothelial function may inhibit atherosclerosis and provide benefits in improving tissue perfusion.
GLP-1R agonists may have beneficial effects on endothelial function. In a double-blind, randomized crossover study, a single dose of exenatide 10 μg improved postprandial endothelial dysfunction as assessed by arterial tonometry (p = 0.0002) [Koska et al. 2010]. Beneficial effects have also been shown on various inflammatory markers and adhesion molecules linked to endothelial dysfunction and CV risk. In cultured human vascular endothelial cells, liraglutide significantly decreased the expression of tumor necrosis factor α, a proinflammatory cytokine, as well as the prothrombotic vascular cell adhesion molecule 1, and plasminogen activator inhibitor type 1. This finding suggests a beneficial effect of liraglutide on improving endothelial dysfunction. A 14-week study of liraglutide in 165 patients with type 2 diabetes showed a nonsignificant, dose-dependent reduction of high-sensitivity C-reactive protein (hs-CRP), a marker of increased CV risk [Courreges et al. 2008]. Furthermore, a meta-analysis of liraglutide trials showed a significant 23% reduction in hs-CRP from baseline [Plutzky et al. 2009]. Similar reductions in hs-CRP have also been reported with exenatide. In a recent randomized study, 128 patients with type 2 diabetes were treated with either exenatide or glibenclamide and followed for 12 months. Compared with glibenclamide, patients receiving exenatide experienced significant reductions in hs-CRP (−0.6 mg/liter; p < 0.001) [Derosa et al. 2010]. In the glibenclamide group, hs-CRP, 2.0 mg/liter at baseline, was reduced to 1.8 mg/liter (p = NS). In another study, treatment with exenatide produced a mean 34% decrease in hs-CRP from baseline [Viswanathan et al. 2007]. Kendall and colleagues found a significant reduction in hs-CRP from a baseline of 3.2 mg/liter to a mean of 1.35 mg/liter over 82 weeks. The effect was more pronounced in patients at high risk as indicated by a baseline hs-CRP level of more than 3 mg/liter. In this subgroup, exenatide was associated with a 50% reduction in hs-CRP [Kendall et al. 2006]. These data suggest that GLP-1R agonists may have the potential to improve endothelial function and CV risk.
Effects on the myocardium
GLP-1 appears to provide cardioprotective effects during times of ischemia. Animal studies have shown that 10–30 min infusions of GLP-1 during acute ischemia limits infarct size, improves cardiac motion, enhances coronary blood flow, and improves contractility as well as ventricular dysfunction [Bose et al. 2005; Nikolaidis et al. 2005; Timmers et al. 2009; Zhao et al. 2006]. Postischemic administration of GLP-1 was also found to enhance coronary blood flow [Ban et al. 2008]. Further, animals pretreated with liraglutide prior to an induced ischemic event also showed benefit. The infarct size was significantly reduced in the liraglutide-treated animals versus controls (p = 0.02) and cardiac function was significantly increased (p < 0.05), leading to improved cardiac output and survival [Noyan-Ashraf et al. 2009]. The mechanism by which GLP-1R agonists provide cardioprotection in ischemia is not well understood. Various theories have been proposed, including improved glucose utilization during ischemia, as well as direct action at cardiac GLP-1 receptors resulting in improved cardiac contractility and expression of cardioprotective proteins [Noyan-Ashraf et al. 2009]. This theory is supported by findings showing that the ischemic benefits of GLP-1 are blunted by the administration of a GLP-1R antagonist [Noyan-Ashraf et al. 2009; Bose et al. 2005]. Antioxidative effects of GLP-1 [Timmers et al. 2009] and vasodilatory actions through non-GLP-1R mediated pathways have been suggested [Ban et al. 2008, 2010]. Researchers have also identified a GLP-1 breakdown product, GLP-1(9-36), that has shown beneficial effects on postischemic recovery and vasodilation [Ban et al. 2008, 2010]. Given the differences in resistance to DPP-4 degradation among available GLP-1R agonists, this finding may have clinical implications.
In human studies, GLP-1 infusion during ischemic conditions appears to hold promise. In patients with left ventricular ejection fraction (LVEF) less than 40% and undergoing primary percutaneous coronary intervention (PCI), a 72 h continuous infusion of GLP-1 significantly improved LVEF (29 ± 2% to 39 ± 2%; p < 0.01) with concomitant improvements in cardiac wall motion and left ventricular systolic function post PCI [Nikolaidis et al. 2004]. These favorable outcomes remained detectable even several weeks after hospital discharge. Similar benefits in terms of myocardial function were observed in patients receiving GLP-1 infusion before and after coronary artery bypass graft surgery (CABG). Compared with the control group, patients with preserved left ventricular function (LVEF ≥ 35%) who received short-term GLP-1 infusions started 12 h before and continued for 48 h after the procedure experienced better glycemic control, required 45% less insulin, less inotropic and vasoactive agents, and experienced less arrhythmia [Sokos et al. 2007]. It is postulated that GLP-1 may prevent or blunt the risk of reperfusion injury syndrome, characterized by impaired contractility, diminished perfusion, thrombotic and inflammatory changes, and cardiac cell damage after blood flow restoration following periods of ischemia [Miura, 1990]. Reperfusion syndrome has been associated with increased rates of post-infarct left ventricular dysfunction and heart failure leading to high mortality rates.
Although most of the research to date has focused on cardiac ischemia, a small number of trials have evaluated the effect of GLP-1 infusions in patients with heart failure. As previously described, continuous GLP-1 infusion over 72 h to patients with acute MI and LVEF less than 40% significantly improved LVEF [Nikolaidis et al. 2004]. In a small trial with 12 patients who had New York Heart Association (NYHA) class III/IV heart failure, 5 weeks of GLP-1 infusion produced significant improvements in LVEF (p < 0.01), maximal ventilation oxygen consumption (p < 0.001), and 6 min walk distance (p < 0.001). Quality of life, as measured by the Minnesota Living with Heart Failure quality of life score, also improved significantly (p < 0.01) [Sokos et al. 2006]. However, interpretation of these findings is limited by the small sample size and nonrandomized design of the trial. Further research is required to confirm the observations.
Safety and tolerability
GLP-1R agonists appear to have a favorable safety profile, with a low risk of hypoglycemia. The most frequent side effects are gastrointestinal intolerance, particularly nausea [Amori et al. 2007], occurring in up 57% of patients treated with exenatide [Moretto et al. 2008], and 29% in liraglutide-treated subjects [Garber et al. 2009]. The nausea is generally transient, subsiding over time in most patients. Starting with low initial doses and slow upward titration over 3–4 weeks may reduce the intensity of nausea. In postmarketing reports acute pancreatitis has been observed in association with exenatide and liraglutide [Parks and Rosebraugh, 2010]. The FDA adverse events reporting system (AERS) found that, compared with other therapies, the incidence of pancreatitis was six times higher among patients taking exenatide [odds ratio (OR) 10.68; 95% CI 7.75–15.1; p < 10–16) [Elashoff et al. 2011]. However, bias in spontaneous reporting cannot be ruled out. Other reviews of the FDA AERS database have reported a lower risk of pancreatitis with exenatide compared with other therapies (OR 1.76; 95% CI 1.61–1.92; p < 0.001) [Raschi et al. 2011]. Furthermore, retrospective cohort studies and experimental data have not been able to find an association between exenatide use and pancreatitis [Dore et al. 2011; Garg et al. 2010; Tatakiewicz et al. 2010]. Although a true causal association has not been confirmed, GLP-1R agonists should not be used in patients at risk of developing acute pancreatitis. Patient education regarding the signs and symptoms of pancreatitis is highly important. If a patient develops acute pancreatitis while using a GLP-1R agonist, the drug should be discontinued. In animal studies, GLP-1R agonists have been associated with an increased risk of medullary thyroid cancer [Parks and Rosebraugh, 2010; Bjerre Knudsen et al. 2010; Amylin Pharmaceuticals, 2011]. However, the doses used in these studies were many times higher than those recommended in humans and clinical trials have not shown an increase in thyroid cancer with GLP-1R agonist use. Presently, the FDA does not recommend routine monitoring of calcitonin levels for patients taking GLP-1R agonists, although caution is necessary. GLP-1R agonists should not be used in patients with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2.
Incorporating glucagon-like peptide 1 receptor agonists in clinical practice
Metformin remains a logical first-line agent for treating patients with type 2 diabetes, provided there are no active contraindications such as renal insufficiency. The 2009 recommendations of the American Association of Clinical Endocrinologist/American College of Endocrinology consensus panel suggests that patients with HbA1c levels of 7.6–9.0% are treated with dual therapy with metformin plus, in order of preference, GLP-1R agonists, DPP-4 inhibitors, glinides, or sulfonylureas [Rodbard et al. 2009]. GLP-1R inhibitors are preferred over DPP-4 inhibitors because of greater effects on lowering postprandial glucose and potential for greater weight loss. For patients requiring triple therapy, metformin, a GLP-1R agonist, and a third oral agent such as sulfonylurea, TZD, or glinide are recommended. The American Diabetes Association (ADA)/European Association for the Study of Diabetes (EASD) and the National Institute for Health and Clinical Excellence also recommend GLP-1 agonists as add-on therapy in patients unable to control their hyperglycemia with metformin and lifestyle changes [Nathan et al. 2009; Adler et al. 2009; National Institute for Health and Clinical Experience, 2010]. In the 2009 ADA/EASD guidelines, the use of GLP-1R agonists is considered later in the algorithm, after initiation with metformin followed by the addition of sulfonylureas and basal insulin.
Although various agents have been used as add-on therapy to metformin, undesirable side effects limit their use. Sulfonylureas and insulin have been associated with a weight gain of approximately 2 kg for every 1% reduction in HgbA1c [Kahn et al. 2006], and in addition, hypoglycemia is of particular concern. The safety of sulfonylureas in patients with CVD or at high risk for CV events remains unclear due to effects on cardiac preconditioning [Cleveland et al. 1997]. The TZDs have been associated with fluid retention that can exacerbate heart failure. Patients with significant heart failure should not use TZDs, while asymptomatic patients at risk of heart failure should be closely monitored for signs of heart failure. The GLP-1R agonists, in addition to lowering blood glucose, have beneficial effects related to weight loss, low risk of hypoglycemia, improve CV risk factors, and have a favorable safety profile. In combination therapy, GLP-1R agonists have been shown to offset the weight gain associated with sulfonylureas, TZDs, and insulin.
An individualized approach to treatment is important. Given their low risk of hypoglycemia and ability to promote weight loss, GLP-1 agonists are the preferred add-on agents for certain patients, including those with a BMI greater than 35 kg/m2 and those who are at risk of hypoglycemia. A potential advantage of early initiation of GLP-1R agonists is their ability to preserve β-cell function, which may provide a more durable and sustained effect on maintaining glycemic control than that observed with sulfonylureas or metformin [Brubaker and Drucker, 2004; Drucker and Nauck, 2006]. In light of these findings, it is possible that exenatide and liraglutide will be used earlier and in a wider variety of patients than currently recommended by treatment guidelines. In patients with some pancreatic function remaining, GLP-1R agonists may be used as add-on therapy to other oral agents, in preference to a direct move to insulin therapy. Studies have shown that GLP-1R agonists provide blood glucose control similar to insulin [Heine et al. 2005; Nauck et al. 2007; Russell-Jones et al. 2009], and improved glucose control when exenatide was added to basal insulin [Buse et al. 2011] along with weight loss and low risk of hypoglycemia.
Differences among the GLP-1R agonists are likely to influence drug selection. The once-daily dosage of liraglutide and convenience of once-daily administration at any time of day, regardless of meals, favors liraglutide over exenatide. Patients with renal impairment may also be candidates for liraglutide therapy as pharmacokinetic considerations suggest less risk of drug accumulation with liraglutide compared with exenatide. Direct comparisons of once daily liraglutide or once weekly exenatide and twice daily exenatide have shown that the longer acting GLP-1R agonists have a greater impact on fasting glucose and HbA1c, whereas twice daily exenatide provides greater reductions in postprandial hyperglycemia [Buse et al. 2009, 2010]. These differences give clinicians the ability to individualize GLP-1R agonist therapy to patients with predominantly postprandial or fasting hyperglycemia. Treatment cost for exenatide ($US350/month), liraglutide ($156–624/month depending on dose), and Once-weekly exenatide ($390/month) is also an important consideration [Thomson Reuters, 2012].
Unresolved questions
Whether the beneficial effects of GLP-1R agonists on the myocardium, endothelium, vasculature, and various markers of CV risk will translate into decreased CV outcomes is not known. Analysis of data from a large healthcare insurance claims database showed that despite higher rates of coronary disease, obesity, hypertension, and hyperlipidemia at baseline, patients treated with exenatide had a lower risk of experiencing a CV event compared with those treated with other antidiabetic agents (HR 0.81; 95% CI 0.68–0.95; p = 0.01) [Best et al. 2011]. Further, patients treated with exenatide had lower rates of CVD-related hospitalization (HR 0.88; 95% CI 0.79–0.98; p = 0.02) and all-cause hospitalizations (HR 0.94, 95% CI 0.91–0.97; p < 0.001) relative to patients treated with other glucose-lowering agents [Best et al. 2011]. However, the retrospective nature of the study and the potential for incomplete data capture inherent to claims database analyses limit the interpretation of these findings. A meta-analysis of clinical trials aimed to investigate the CV safety of GLP-1R agonists identified 20 trials including 6490 and 3995 patients in the GLP-1R agonist and comparator groups respectively. Overall, the use of exenatide or liraglutide was not associated with an increased or decreased risk of major CV events compared with control groups (OR 0.74; 95% CI 0.5–1.08; p = 0.12). However, when only placebo-controlled trials were included, a significant reduction in CV events was observed with GLP-1R agonist therapy (OR 0.459; 95% CI 0.255–0.862; p = 0.009) [Monami et al. 2011].
Despite growing evidence regarding the potential cardiovascular benefits of GLP-1R agonist therapy, large, randomized, blinded clinical trials with hard CV endpoints have not been performed. Most human studies have been small, and have focused on surrogate endpoints. The findings need to be confirmed by prospective, randomized CV outcomes trials. Several ongoing trials, designed to evaluate the cardiovascular outcomes and safety of GLP-1R based therapies, are in various stages of progress (Table 2). Data from these trials will provide valuable insight regarding the potential role of GLP-1R agonists in the treatment of patients with diabetes and CVD.
Ongoing clinical trials evaluating cardiovascular outcomes with glucagon-like peptide-1 agonists
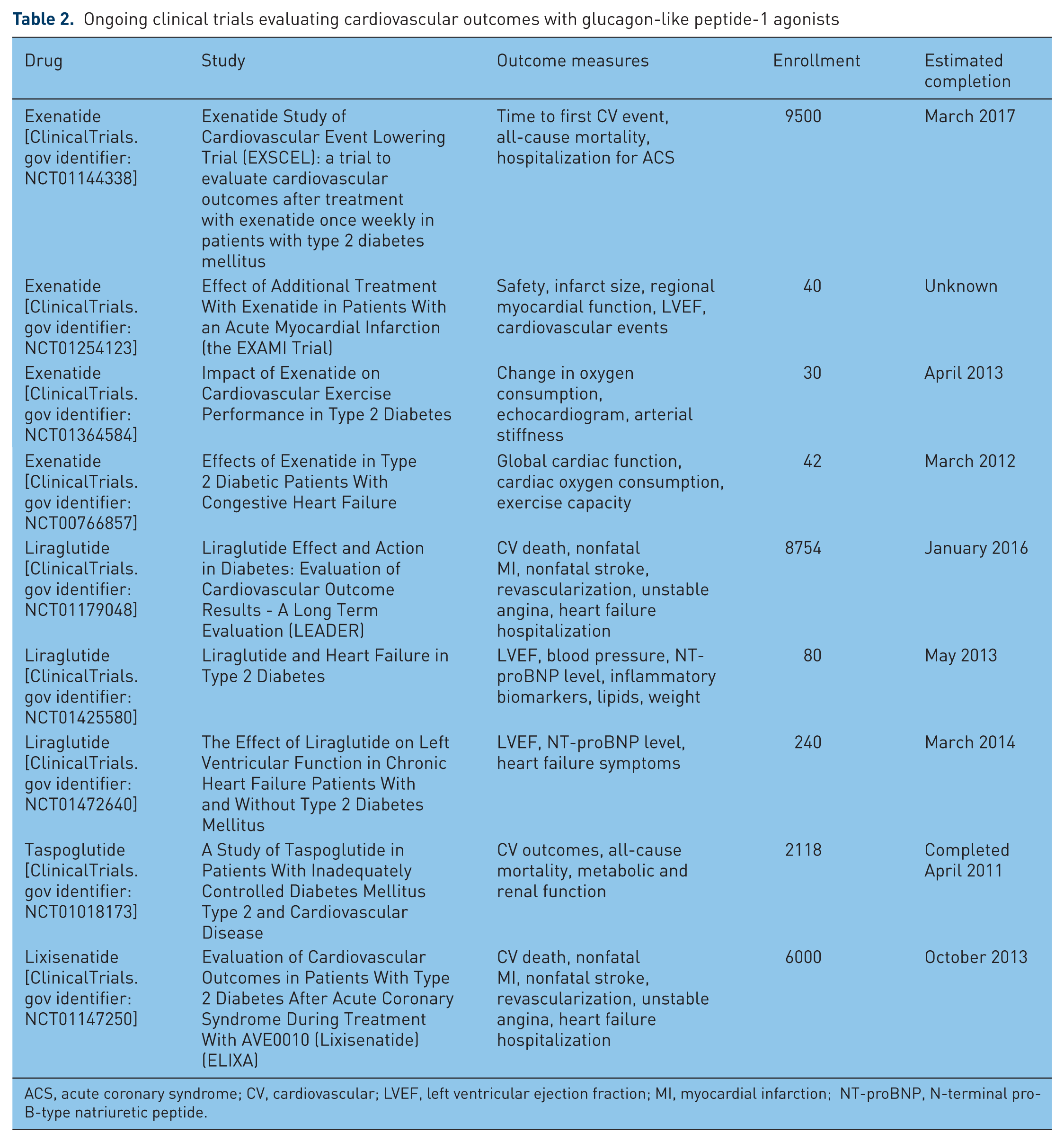
ACS, acute coronary syndrome; CV, cardiovascular; LVEF, left ventricular ejection fraction; MI, myocardial infarction; NT-proBNP, N-terminal pro-B-type natriuretic peptide.
Summary
A growing body of evidence suggests that GLP-1R agonists have beneficial CV effects beyond glycemic control. Studies have demonstrated that, in addition to lowering blood glucose, GLP-1R agonists exert a number of CV effects that include reduction in blood pressure, improved endothelial function, improved myocardial function, and beneficial changes in dyslipidemia. Whether the preliminary findings translate into decreased CV outcomes needs to be confirmed by large randomized placebo-controlled trials. GLP-1R agonists also hold promise in overcoming the limitations of current glucose-lowering agents that cause weight gain and hypoglycemia. By offering weight reduction and reduced risk of hypoglycemia in addition to glycemic control, the GLP-1R agonists provide an effective therapeutic option for patients with type 2 diabetes.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that they have no conflicts of interest regarding this review.
