Abstract
Type 2 diabetes mellitus (T2DM) is a complex multifactorial disease which affects the length and quality of life by severe chronic complications. Chronic hyperglycemia, which is the main alteration in T2DM, is strictly related to microvascular complications (such as retinopathy and nephropathy) and neuropathy, whereas large vessel atherosclerosis is also dependent on lipid and hemostasis abnormalities, arterial hypertension and other known cardiovascular risk factors. An early intervention to control hyperglycemia and to prevent deterioration of β-cell function is considered mandatory in patients with T2DM to minimize the risk of chronic complications. Recently, the availability of new pharmacological agents with different targets, including the activation of the incretin system has allowed the proposal of more effective strategies for early treatment of metabolic alterations in patients with T2DM. This commentary will focus on the role of new oral agents influencing the incretin system and the putative advantages of their co-administration with metformin, an old, effective anti-hyperglycemic agent also able to exert beneficial actions on arterial vessels, reducing the risk of macrovascular-related events. The vasoprotective role of metformin is largely independent of its hypoglycemic action, and has been ascribed to pleiotropic effects.
Commentary Strategy and Selection Criteria
Searches for original articles and review from 1985 to August 2009 focusing on metformin and dipeptidyl peptidase-4 (DPP-4) inhibitors were performed in MEDLINE and PubMed electronic databases. The search terms were: metformin, diabetes, insulin resistance, obesity, incretin glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic hormone (GIP). All papers identified were English–language, full–text papers and were selected on the basis of relevance and novelty; a priority was given to those published in peer-reviewed journals, from the past 5 years without excluding older publications commonly referenced or highly regarded.
Introduction
The International Diabetes Federation estimated that in 2007 the number of patients affected by type 2 diabetes mellitus (T2DM) reached 240 million worldwide, i.e. more than 5% of the adult population. 1 This multifactorial and progressive disease, caused by a combination of insulin resistance and β-cell dysfunction with decreased insulin secretion,2,3 is associated with long-term complications, mainly involving blood vessels and peripheral nerves, which affect the length and quality of life. 4
Clear evidence indicates that chronic hyperglycemia plays a major pathogenic role in the development of microvascular complications (such as retinopathy and nephropathy) and neuropathy 5 whereas large vessel atherosclerosis, which is responsible of coronary, cerebral and lower limb atherothrombotic damage, also depends on lipid and hemostasis abnormalities, arterial hypertension and other known cardiovascular risk factors. 6
In patients with T2DM early interventions to control hyperglycemia and to prevent deterioration of β-cell function are considered pivotal to minimize the overall risk of developing chronic complications. 7
A therapeutic approach to achieve this target includes intervention regarding life-style and the use of metformin as a first-line pharmacological strategy; 8 however, this approach and the combination with sulphonylureas in non–responsive patients seldom achieve and maintain optimal glycemic control.9,10
Recently, however, this scenario has changed due to the availability of newer families of pharmacological agents (either injectable or oral) with different molecular targets and specific indication to control hyperglycemia and progression of β-cell dysfunction, as extensively reviewed by Corsino and Coll. 11
The Incretin System and its Role in Glycemic Control
The endogenous incretin system, which accounts for an increased insulin release after an oral glucose load in comparison with intravenous administration, 12 is represented by the neuroendocrine hormones glucagon–like peptide-1 (GLP-1) and glucose–dependent insulinotropic hormone (GIP), small peptides of 30 and 42 amino acids, respectively, which are released from the L and K cells of the gastrointestinal tract, as a response to intraluminal glucose.13,14
Both hormones cause a glucose–dependent increase in insulin secretion; GLP-1 influences also insulin biosynthesis under hyperglycemic conditions, inhibits glucagon release,14,15 and is associated with increased satiety, possibly because it reduces the rate of gastric emptying. 14
Indirect benefits on the cardiovascular system are recognized to incretins depending on weight reduction over time, improvements in arterial blood pressure and circulating lipid pattern, and reduction of inflammatory markers and insulin resistance.16,17
The incretin effects on insulin secretion, however, are blunted by degradation through the ubiquitous serine protease dipeptidyl peptidase-4 (DPP-4). 14
Type 2 diabetic patients have reduced circulating levels of GLP-1 but retain the ability to respond to this hormone: therefore, it is possible to improve glycemic control through administration of exogenous GLP-1 analogs (exenatide, liraglutide) 18 or molecules that prolong the activity of endogenous GLP-1 and GIP by circumventing or reducing the rate of degradation by DPP-4.18–20
DPP-4 inhibitors
DPP-4 inhibitors are small molecules absorbed from the gastrointestinal tract, which decrease the activity of the enzyme over 80%.18–20 They enhance meal-related circulating levels of incretins close to the physiological range, increasing their effects on glucose–dependent stimulation of insulin secretion and suppression of glucagon output. 18 The effects on gastric emptying retardation and appetite reduction and weight loss are still controversial. 18
As extensively reviewed by Corsino and Coll, two DPP-4 inhibitors, i.e. sitagliptin and vildagliptin, are available and investigated in clinical trials, and several others (saxagliptin, alogliptin, and denagliptin) are under evaluation. 11
Sitagliptin, the first orally bioavailable selective DPP-4 inhibitor approved by the Federal Drug Administration (FDA), exhibits rapid absorption and elevated selectivity for DPP-4 in comparison with other DPP enzymes;18,19 through this mechanism both acute and chronic studies showed that it preserves the circulating levels of intact GIP and GLP-1 following meals and reduces blood glucose levels without significant increases in hypoglycemic events.18,19 Clinical phase III trials demonstrated a reduction of glycated hemoglobin A1c (HbA1c) levels ranging 0.79% to 0.94%, in relationship to the employed doses. 19
Sitagliptin is moderately bound to plasma proteins and does not exhibit clinically relevant interactions with drugs that use the cytochrome P450 system or p–glycoprotein transport system.
Vildagliptin is the second DPP-4 inhibitor approved in Europe; similar to sitagliptin, it is rapidly absorbed by the gastro-enteric tract with maximal plasma concentrations within 1–2 hours; 20 its pharmacokinetic properties support a once daily dosing regimen. 20
The drug interactions of vildagliptin are limited: in particular, in clinical trials significant interactions with pioglitazone, metformin, or glibenclamide have been excluded.
The pharmacology of metformin
The glucose–lowering effect of guanidine derivatives, i.e. phenformin, buformin and metformin, was observed starting from the 1950s. Phenformin and buformin were withdrawn from the pharmacopoeia in the early 1970s owing to their ability to induce lactic acidosis and their association with an increased cardiac mortality.
Metformin (N,N-dimethyl biguanide), a less lipophilic biguanide, was proved safer and, after about 20 years of use in Europe, was approved for use in the USA in 1995. 21
After single dose oral administration, metformin is absorbed in the upper intestinal tract within 6 hours with an absolute bioavailability ranging from 40% to 60%. 22 Co-administration with food slightly decreases and delays metformin absorption. 22
Metformin has a negligible binding to plasma proteins, 22 accumulates in the gastrointestinal tract, salivary glands and kidney, 22 does not undergo hepatic metabolism, 22 and, due to its elevated hydrophilia, is almost totally excreted, unchanged in the urine by glomerular filtration and tubular secretion. 22 Renal clearance of metformin is approximately 3.5 times greater than creatinine clearance and plasma half–life is approximately 6.2 hours. 22
Both intestinal absorption and renal excretion of metformin are dependent on at least 2 organic cation transporters (OCT), namely OCT1 and OCT2; OCT1 mediated transport is the major mechanism for metformin to entry into hepatocytes and enterocytes, whereas OCT2 is the major renal metformin transporter. 23
As extensively reviewed, metformin exerts little effects on blood glucose concentrations in normoglycemic subjects;22,24 its glucose-lowering action in T2DM is dependent on increased insulin sensitivity in liver and skeletal muscle in the presence of circulating insulin and of adequate β-cell function;22,24,25 in particular, metformin reduces hepatic glucose output, 25 increases insulin stimulated glucose uptake by skeletal muscle and adipocytes,22,24,25 and decreases the rate of intestinal glucose absorption. 26 Other extrahepatic actions of metformin include increased glucose transport and utilization by skeletal muscle due to the improvement in non–oxidative glucose metabolism,22,24 and an influence on the incretin system either through increased secretion of GLP-1, or inhibition of DPP-4 activity. 27
The United Kingdom Prospective Diabetes Study (UKPDS) firstly showed that in obese type 2 diabetic patients metformin improved macrovascular outcomes, including all–cause death, and myocardial infarction in a subgroup of obese patients with newly diagnosed T2DM 28 more than sulphonylureas or insulin. 28 This observation identified metformin as the first–line treatment of T2DM for reducing the risk of macrovascular events and induced investigations into the mechanisms involved in its vascular protective effects beyond its glucose lowering action.
The vasoprotective role of metformin is largely independent of its hypoglycemic action and has been ascribed to pleiotropic effects.
As recently reviewed, metformin has shown to exert beneficial actions on some mechanisms involved in atherothrombosis, including a reduction in insulin resistance, an improvement in circulating lipid patterns, a reduction of a pro–inflammatory status, a correction of pro–thrombotic tendencies and an overall positive influence on vascular reactivity.29–31 Experimental and clinical evidences indicate a decrease of advanced glycation end product (AGEs) formation and of oxidative stress as potential actions of the vascular effects of metformin, with improvement of endothelial function both in the macro and micro vasculature.29,30
Combination Therapy with Metformin and Dipeptidyl Peptidase Inhibitors
Rationale
In the natural history of T2DM, the development of impaired glucose tolerance and finally of hyperglycemia occurs gradually over many years. Pancreatic β-cells, which are initially able to respond to increased insulin resistance by increasing insulin secretion to maintain normoglycemia, progressively reduce their function.2,3 Metformin, which exerts neutral or protective effect on β-cell function, is recommended by guidelines as the first-line therapy in T2DM, 8 but presents a high rate of failure after relatively few years. Therefore, an intensification of the therapy by combination of agents with complementary mechanisms of action and lacking of pharmacological interferences is an almost mandatory step in the treatment of T2DM patients.
As evidenced in Figure 1, the hypoglycemic effects of metformin and incretins are expected as additive, the actions on β-cell function as beneficial (differently from combination between sulphonylureas and metformin), the risk of hypoglycemic events as reduced and the effect of body weight as neutral.
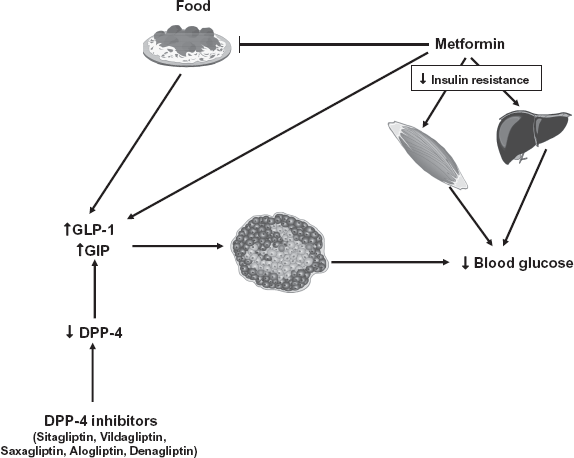
Mechanisms involved in the hypoglycaemic effects of metformin and dipeptidyl peptidase-4 (DPP-4) inhibitors.
An additional aspect of this combination therapy derives from the evidence that metformin per se increases plasma levels of GLP-1. 27
Finally, recent evidence indicates some incretin mediated beneficial effects on vasculature, which could be additive to those of metformin (Table 1).
Vasoprotective mechanisms ricognized to incretins and metformin.

Clinical studies
Results of trials on glycemic control in T2DM patients utilizing the combination between metformin and DPP-4 inhibitors are available as recently reviewed for vildagliptin 32 and sitagliptin. 33 The evidence firstly indicated that the addition of a drug active on the incretin system improves the deterioration of glycemic control in patients treated with metformin as monotherapy.32,33 Furthermore, additive effects on HbA1c levels were shown with combination therapy in comparison with either drug alone.32,33
Conclusion
Current American Diabetes Association (ADA) and European Association for the Study of Diabetes (EASD) treatment guidelines recommend metformin as the first–line drug in the treatment of T2DM as monotherapy, 8 and indicate its combination with other anti–hyperglycemic or with hypoglycemic agents, including sulphonylureas, glitazones, and insulin, as further therapeutic options in patients who fail to reach therapeutic targets. 8 The combination between metformin and DPP-4 inhibitors has to be considered as an effective therapeutic choice for patients with T2DM not adequately controlled by monotherapy due to the complementary and possibly additive effect on metabolic control and, probably, on vascular risk.
Disclosures
The authors report no conflict of interest in this work.
Footnotes
Acknowledgements
This study was partially supported by a grant from Italian Ministero dell'Istruzione, Università e Ricerca (MIUR) within the project; The molecular basis of insulin resistance and their importance in the pathogenesis of the alterations of the vessel wall. (Local Coordinator: Giovanni Anfossi, National Coordinator: Amalia Bosia) and by 2 grants from Regione Piemonte (years 2004 and 2006).
