Abstract
Objective:
The present study aimed to assess the patient preference and tolerability of oral dipeptidyl peptidase-4 inhibitor (vildagliptin)
Methods:
This 24-week, randomized, multicenter, crossover study, patients with type 2 diabetes mellitus inadequately controlled on metformin monotherapy with hemoglobin A1c (HbA1c) ⩾6.5% and ⩽9.0% were randomized in a crossover manner to receive either vildagliptin/metformin single-pill combination (SPC) 50/1000 mg twice daily (
Results:
Patient preference at week 24 was similar, with 51.7% (
Conclusions:
In this pilot study, although both vildagliptin and liraglutide therapies were preferred similarly by the patients and showed effective control of glycemia over 12 weeks, vildagliptin was associated with fewer adverse events and was preferred more by elderly patients.
Keywords
Introduction
Incretin-based therapies such as dipeptidyl peptidase-4 (DPP-4) inhibitors or glucagon-like peptide-1 (GLP-1) analogs increase plasma concentrations of active GLP-1 and exert antihyperglycemic effects in a glucose-dependent manner [Scheen, 2013]. Although these therapies are safe and effective in lowering hemoglobin A1c (HbA1c) without the risk of hypoglycemia and weight gain, they vary in administration route, efficacy, and side effects.
Adherence to type 2 diabetes mellitus therapy is vital for effective blood glucose management. Oral antihyperglycemic drugs such as DPP-4 inhibitors are being increasingly used in the management of type 2 diabetes mellitus because of their efficacy and patient preference perceived by the physician [Inzucchi, 2002]. Previous studies have shown that patients with type 2 diabetes mellitus prefer additional oral antihyperglycemic drugs over injectables [Hayes et al. 2006; Khan et al. 2009]. However, there are contradictory findings for patient preference between the oral DPP-4 inhibitor sitagliptin and injectable GLP-1 analog liraglutide in other studies [Dibonaventura et al. 2010; Pratley et al. 2010].
Vildagliptin, a DPP-4 inhibitor, improves glycemic control with a low risk of hypoglycemia and is well tolerated in a wide patient population with type 2 diabetes mellitus [Keating, 2014]. Liraglutide, a GLP-1 analog, is indicated subcutaneously once daily as an add-on to metformin or sulfonylurea in the management of type 2 diabetes mellitus [Novo Nordisk, 2013]. The present crossover study aimed to assess whether patients with type 2 diabetes mellitus inadequately controlled with metformin monotherapy preferred treatment with an oral, single-pill combination (SPC) of vildagliptin/metformin
Methods
Study design
This 24-week, randomized, open-label, crossover, multicenter study was conducted across eight centers in Germany in patients with type 2 diabetes mellitus inadequately controlled with metformin therapy (2000 mg for ⩾12 weeks). After a screening period of 1 week, eligible patients were randomized to receive either vildagliptin/metformin SPC (50/1000 mg twice daily) or liraglutide/metformin (0.6 mg [weeks 0–1] followed by 1.2 mg [weeks 2–12] subcutaneously once daily/1000 mg orally twice daily) for 12 weeks (period I). After period I, the patients were crossed over to receive either liraglutide or vildagliptin while continuing metformin treatment for the next 12 weeks (period II; Figure 1). Eligible patients were randomized by third party using an interactive voice response (IVR) system that automates the random assignment of treatment sequences in 1:1 ratio. As patients were followed only telephonically after randomization, at week 0 they were being instructed by their study investigator to take the prescribed study drug correctly and emphasized the importance of compliance for patients’ safety and study validity. The patients were asked to contact their investigator if they were unable to take the prescribed study drug for any reason. All dosages prescribed and dispensed to the patient and all dose changes during the study were recorded on the Dosage Administration Record Case Report Form. Moreover, dose adjustments (except for liraglutide titration) and/or interruptions were not allowed in the study.

Study design.
Study population
Patients aged 18–80 years with type 2 diabetes mellitus inadequately controlled with metformin (2000 mg) for ⩾12 weeks before randomization, HbA1c ⩾6.5% and ⩽9.0%, and body mass index 19–35 kg/m2 were eligible to participate in this study. The key exclusion criteria included fasting plasma glucose (FPG) levels ⩾15 mmol/l; previous treatment with any antihyperglycemic drug (except metformin) within 3 months before study entry; history of type 1 or secondary forms of diabetes; congestive heart failure (New York Heart Association Class III or IV); myocardial infarction, stroke, or transient ischemic attacks within 6 months before study entry; or unstable angina within 3 months before study entry. Patients with any of the following laboratory abnormalities were excluded from the study: alanine transaminase or aspartate transaminase >3 times the upper limit of normal, total bilirubin >2 times the upper limit of normal, or glomerular filtration rate <60 ml/min/1.73 m2 (measured by Modification of Diet in Renal Disease [MDRD] formula).
Study end points and assessments
The primary endpoint was the proportion of patients preferring oral vildagliptin compared with injectable liraglutide after 24 weeks of treatment. Patients were asked the following two-choice question: ‘Based on your personal experience with both, [vildagliptin] and [liraglutide] plus metformin in the study, which form of medication would you prefer to take in the future?’. Patients were also asked to specify the reasons for their preference and required to select them from a list of reasons provided in the questionnaire [Treatment Satisfaction Questionnaire for Medication (TSQM)-9 questionnaire (official German Translation)] [Bharmal et al. 2009]. Physicians’ preference between oral vildagliptin and injectable liraglutide was also recorded at week 24. The secondary efficacy endpoints, FPG and HbA1c, were recorded at baseline and weeks 12 and 24. Safety assessments including body weight, adverse events (AEs), and serious AEs were monitored and recorded throughout the study.
Sample size and statistical analysis
A sample of 50 patients provided a power of >90% to exclude a preference rate ⩽50% for those preferring vildagliptin/metformin SPC, if the true preference rate was 75%. Demographic characteristics were presented using summary statistics on the safety set. Primary efficacy and patient and physician preferences were analyzed in all randomized patients who completed the preference questionnaire at the end of the study (efficacy set). Safety analyses were performed on all randomized patients who had received at least one dose of the study medication during at least one study period and had at least one safety assessment after baseline (safety set). The primary endpoint was tested using the exact Clopper–Pearson method at 95% confidence intervals (CIs). The secondary efficacy endpoints, FPG and HbA1c, were analyzed using descriptive statistics on the safety set. Treatments were compared using the analysis of variance model, with treatment, period, and patient as factors. Post hoc analysis for patient preference was also conducted in elderly patients (⩾65 years) in the safety set by using descriptive statistics.
Ethics and good clinical practice
The study was conducted in compliance with the Declaration of Helsinki and Good Clinical Practice guidelines and was reviewed and approved by the independent ethics committee or institutional review board of the study centers. Written informed consent was obtained from each patient before randomization. The study was registered at EudraCT (no. 2011-003818-16) and ClinicalTrials.gov [ClinicalTrials.gov identifier: NCT01518101].
Results
Patient disposition and baseline demographics
Of 93 patients screened, 62 were randomized (treatment sequence: vildagliptin–liraglutide [
Patient disposition (safety set).

Refers to the number of patients who received each treatment at least once.
Refers to the last treatment before completion or discontinuation; the patient who discontinued due to an adverse event discontinued only period I but completed period II.
Safety set consisted of all patients who received at least one dose of the study medication during at least one study period and had at least one safety assessment after baseline.
The baseline and demographic characteristics are summarized in Table 2. Overall, the mean patient age was 60.3 years and 22 patients (35.5%) were aged ⩾65 years. The patients were predominantly White (98.4%) and 53.2% were women. The mean body weight was 90.3 kg and the mean HbA1c was ~7.3%. The mean duration of type 2 diabetes mellitus was 7.5 years and that of metformin therapy was 4.4 years.
Patient demographics and baseline characteristics (safety set).
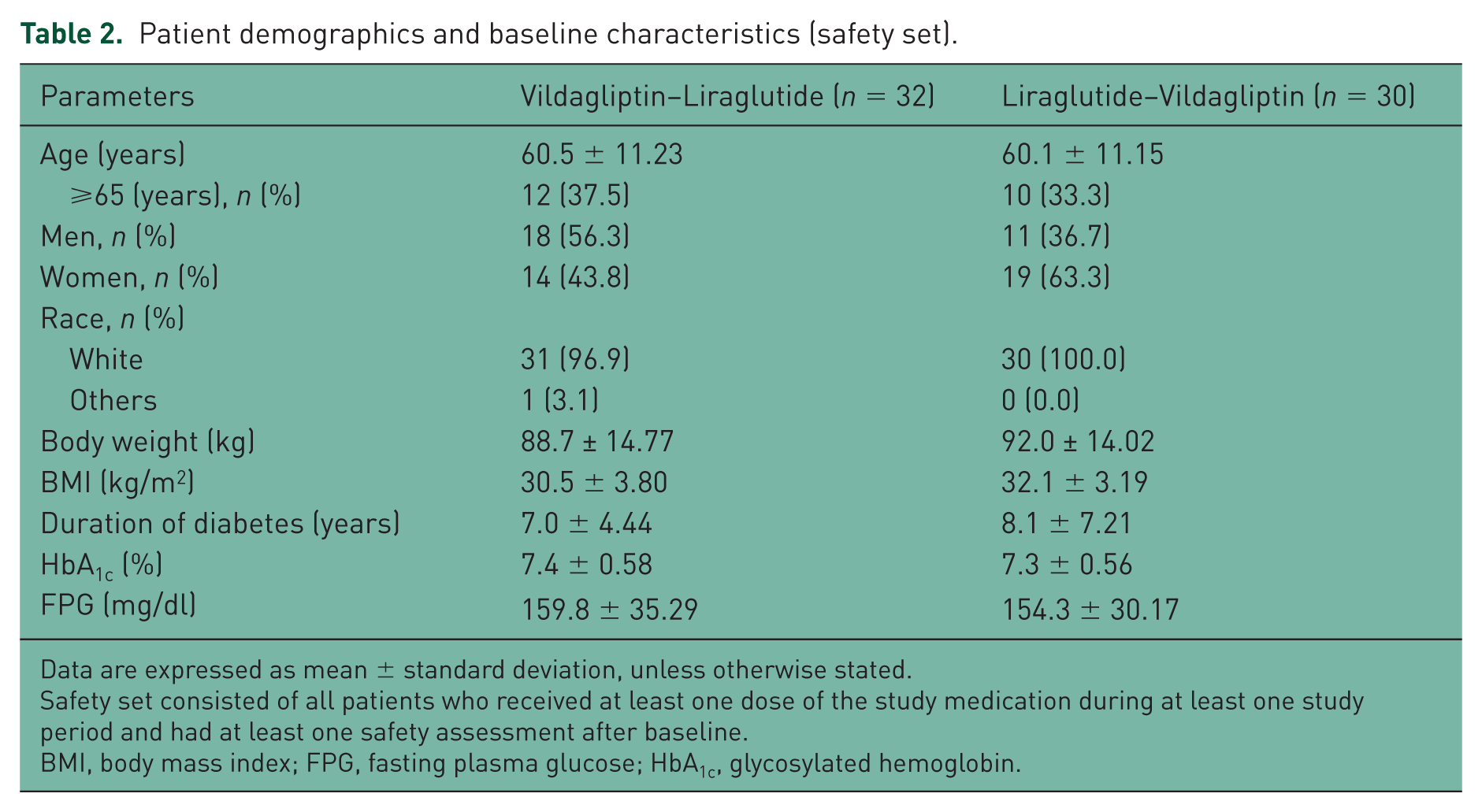
Data are expressed as mean ± standard deviation, unless otherwise stated.
Safety set consisted of all patients who received at least one dose of the study medication during at least one study period and had at least one safety assessment after baseline.
BMI, body mass index; FPG, fasting plasma glucose; HbA1c, glycosylated hemoglobin.
Treatment preference
Of the efficacy-evaluable population (

Patient preference for treatment at week 24.
The reasons for treatment preferences by patients are presented in Table 3. Among the patients who preferred vildagliptin, 71% (
Reasons for patient preference at week 24 (efficacy set).

Efficacy set consisted of all patients who completed the patient preference questionnaire at the end of the study.
In contrast, the preference for vildagliptin (
Physicians and patients showed similar preference for oral treatment: 55% physicians (

Physician preference for treatment at week 24.
Secondary efficacy
The lowering of mean [±SD] FPG (−21.5 [± 39.38]
Safety
Of 62 AEs reported in 28 patients, 46 were with liraglutide, mainly due to more gastrointestinal and metabolism and nutrition disorders, compared with 16 AEs with vildagliptin (Table 4). All AEs were mild to moderate in severity, except 1 AE (common cold) that was considered severe in a liraglutide-treated patient. Two patients experienced one serious AE while receiving liraglutide (cholecystolithiasis [day 80] and moderate coronary heart disease [day 168]), although neither of these events were considered drug-related. No deaths were reported during the study. Two patients withdrew treatment with liraglutide because of safety reasons (1 patient had abnormal laboratory values and another had moderate diarrhea, which was considered to be drug-related, on day 3). The mean [±SD] change in body weight was −0.1 [2.45] kg with vildagliptin compared with −2.2 [2.64] kg with liraglutide after 12 weeks treatment, with a between-treatment difference of −2.3 [3.90] kg (
Safety and tolerability (safety set).

The columns vildagliptin and liraglutide refer to the last treatment received before the onset of an AE.
Safety set consisted of all patients who received at least one dose of the study medication during at least one study period and had at least one safety assessment after baseline.
AE, adverse event; SAE, serious adverse event; SOC, system organ class.
Discussion
A therapy that improves compliance and adherence is beneficial to achieve treatment goals. Similarly, simple-to-use drugs and patient preference could also affect treatment adherence. In this study, patient preference for vildagliptin and liraglutide was similar. However, 71.0% and 67.8% of patients who preferred vildagliptin ranked administration route and side effects as important and very important, respectively, compared with 45.0% and 41.4% of patients who preferred liraglutide, respectively, suggesting that oral formulations and tolerability are concerning issues for certain patients.
Although both GLP-1 analogs and DPP-4 inhibitors have similar pharmacological approaches, their routes of administration (subcutaneous
Patients preferred the treatment which is most convenient/flexible, noninjectable, and had fewer physical and emotional side effects [Hayes et al. 2006]. In the present study, patients’ preference for vildagliptin and liraglutide were similar. In contrast, the results of internet survey conducted between 2008 and 2009 in patients from the United States and Europe (
The FPG and HbA1c reductions were greater with liraglutide than with vildagliptin. However, vildagliptin had a better tolerability profile, particularly with respect to the incidence of gastrointestinal and metabolic disorders. Although the preferences for both medications were similar, the use of a single questionnaire at the end of the study (instead of after each study period) may also have introduced recall bias. In addition, study limitations such as small sample size, short duration, and crossover design without a washout period may not allow clear assessments of the efficacy.
The results from this pilot study indicated similar patient preference for oral therapy, vildagliptin and injectable therapy, liraglutide. Although the preference for vildagliptin was greater in elderly patients, and it was associated with fewer AEs, this data should be interpreted cautiously.
Footnotes
Acknowledgements
The authors would like to thank the patients and staff who participated in this study. All authors participated in the development and writing of the paper and approved the final manuscript for publication. The authors take full responsibility for the content of the paper and thank Anuja Shah, PhD and Madhavi Dokku, PhD (both from Novartis Healthcare Pvt Ltd, Hyderabad, India) for assisting in writing of the manuscript, collating comments from all authors, and editing the final manuscript.
Declaration of Conflicting Interests
JL has received fees for consultancy advisory boards, education talks, and speaker fees from AstraZeneca, Bayer, Bristol-Myers Squibb, Eli Lilly & Co., Merck Sharp & Dohme, Novartis, Novo Nordisk, and Sanofi, outside the submitted work. ED and MD are employees of Novartis Pharma GmBH and as such may be eligible for the company’s stock and stock-options, outside the submitted work.
Funding
The study was funded by Novartis Pharma GmbH, Germany. The study sponsor participated in the study design, data collection, data review, data analysis, and writing of the report, and the decision to submit the article for publication.
