Abstract
Chronic obstructive pulmonary disease (COPD) is a disorder of the airways characterized by inflammation and airflow limitation. It is a major cause of morbidity and mortality globally and its prevalence is on the rise. COPD is predominantly a disease of the older age group. As the average life-span increases world-wide, and the burden of COPD increases, correct recognition and optimal management of this disease has become essential in the elderly. In the proceeding article, changes in the respiratory system associated with normal aging, as well as with COPD are reviewed. Important implications of aging in COPD management are also discussed.
Introduction
Chronic obstructive pulmonary disease (COPD) is a preventable, treatable and, unfortunately, under-recognized condition. It is characterized by airway inflammation and scarring, parenchymal destruction causing progressive poorly reversible airflow obstruction, and multi-organ system involvement.
COPD is a major health burden both in the US and worldwide. It is currently the 4th leading cause of death in USA. Globally, its prevalence continues to rise and it is estimated that it will become the 3rd leading cause of morbidity and mortality in the world by the year 2020. The annual economic cost of COPD (in healthcare costs and lost productivity) is immense and was projected to be around 49.9 billion dollars in 2010 in the US.1–6
COPD is predominantly a disease of the older age group. Prevalence increases significantly with age: from ~1% for the general population to 10% in those aged 40 and above; in individuals aged 65 and above, the prevalence is even higher (14.2% to 19%). As life expectancies rise across the world, and the burden of the disease increases, recognition of COPD and its optimal management has become essential in the elderly.5,6 In the following text, the authors aim to review the pathophysiology, diagnosis and treatment of COPD, with particular focus on aspects of management that are specific to the elderly. Salient results and implications of recently conducted trials relevant to COPD, as well as ongoing research, will also be discussed.
Factors Associated with Aging that Impact COPD and Its Management
Respiratory system structure and function
Aging affects structure, function, and control of the respiratory system. Both the lungs and the thoracic cage undergo changes that affect respiratory function.7–9 The net effect of these changes is an overall decrease in expiratory flow and oxygenation with advancing age.
Lung parenchyma, airway diameter and oxygenation
The elastic property of the lungs is the major determinant of maximal expiratory flow and diminishes with aging.7–9 The bronchiolar diameter decreases and alveolar ducts enlarge as a result of the reduced elasticity. These changes result in decreased expiratory flow and decreased surface area for gas exchange, respectively. Further, due to the loss of recoil, airways in dependent portions of the lung close at higher volumes, such that more airways are closed during all or part of the respiratory cycle. The lower portions of the lung are better perfused at all ages, but higher closing volumes with age increase ventilation-perfusion mismatch and account for the age related declining Pao2 (oxygen pressure).
Thoracic cage (including the respiratory muscles)
In contrast to lungs, the chest wall stiffens with aging and its compliance decreases. 10 Costochondral cartilages become calcified, and intercostal muscle contraction causes less chest expansion. Changes in lung and chest wall compliance result in diminished static and dynamic compliance of the overall respiratory system. This leads to an increase in the work of breathing which is further compounded by increased anterior-posterior diameter of the chest and kyphosis of the thoracic spine with advancing age. 1 In addition, the strength and endurance of respiratory muscles diminishes with age, especially above 55 years of age. 11 By age 65, inspiration normally depends on abdominal muscles, which is only partially effective in opening airways in the seated or supine position, as complete airway expansion occurs only in the upright (standing) position. While the alveoli are not fully expanded, they continue to be perfused and extract alveolar gases, which produce alveolar collapse and atelectasis.
The muscle and thoracic wall changes may also affect clearing of the airway in the mucus hyper secreting state seen in COPD. Cough becomes less vigorous with aging and, because of the greater closing volumes, is unable to clear certain portions of the lungs. Mucociliary clearance becomes slower and less effective. 12
Additionally, pulmonary functional reserves diminish in the elderly. In non-smoking men, FVC decreases between 0.15 and 0.3 L/decade while FEV1 decreases by 0.2 to 0.3 L/decade. These changes are smaller and more gradual in women. Total lung capacity (proportional to height) does not change significantly with age; however, the residual volume increases because of higher closing volume.
Ventilatory control
Control of ventilation is modestly compromised with age, with decreased responses to hypoxemia, hypercapnia, and mechanical loading (such as breathing through a small-diameter endotracheal tube). With exercise training, many of these changes normalize; the implication is that central or peripheral receptor hyporesponsiveness are due to deconditioning, and not due to an intrinsic change with aging. 13
Changes in the respiratory system in COPD
Current evidence suggests that persistent pulmonary inflammation drives the disease process in COPD. 14 Inflammatory cells infiltrate the airways, pulmonary parenchyma and vasculature. Enhanced activity of cytokines, oxidative stress and metallo-proteinases set in motion a process that leads to structural damage in the lungs and causes systemic manifestations (such as cachexia, skeletal muscle wasting, as well as worsening of co-morbid conditions eg, heart disease, osteoporosis, anemia, depression and diabetes).15,16 The presence of an irritant, most commonly cigarette smoke, appears to be integral in the amplification of this inflammatory cascade to pathological levels.
The airflow limitation seen in COPD appears to be related to airway narrowing and structural changes associated with inflammation and repeated injury and healing. Goblet cell metaplasia and mucociliary dysfunction, leading to exudative plugs in the terminal bronchioles, also contribute to decreased airflow. Parenchymal destruction causes abnormalities of gas exchange with resultant hypoxia and hypercapnia.
Currently available treatments focus on relief of airway constriction (bronchodilators) and decreasing airway inflammation (corticosteroid and PD4 inhibitor). However, so far no therapy has been shown to slow the progression of disease or significantly alter the inflammatory process in COPD. 17
Comorbid conditions
The typical elderly patient with COPD often has multiple co-morbid conditions. Medical problems that have a higher prevalence in the older age groups are frequently encountered in such patients. Diseases related to smoking, the predominant causative factor for COPD, are also commonly seen. Furthermore, multi-organ system involvement can occur due to the pro-inflammatory state and systemic manifestations associated with COPD.16,18 Cardiovascular diseases, malignancies, musculoskeletal and psychiatric pathologies (cachexia, osteoporosis, muscle dysfunction; depression and anxiety) are often present in many COPD patients. The physician needs to be cognizant of the above when managing an elderly individual with COPD in order to successfully tailor the treatment plan to the individual.
Pharmacokinetics and pharmacodynamics
Various changes occur with aging that affect the metabolism and effects of medications. The volume of distribution of drugs changes in the elderly–-due to a combination of a decrease in lean muscle mass and an increase in total body fat–-leading to higher serum levels of water soluble medications and longer half-lives of fat soluble drugs. A decrease in oxidative metabolism by the cytochrome p450 system results in delayed clearance of medications degraded by this pathway. Renal clearance can be variably affected, leading to unpredictable levels of drugs that are excreted renally.19–21
The physician needs to exercise caution when using COPD medications in the elderly because of the above mentioned changes. Systemic medications, such as theophylline and systemic steroids and antibiotics, have the highest potential for adverse effects. Inhaled agents, however, can also have significant side effects and need to be monitored.21,22
Polypharmacy
Elderly patients are often on complicated therapeutic regimens for multiple medical problems. Drug-drug interactions with major adverse events are a significant concern with initiation of any medication in this age group. Drugs that affect the hepatic metabolism of other medications (especially regimen containing statins, antibiotics and antacids) need to be closely monitored. Organ specific side effects related to a particular medication are another cause for alarm. Examples include cardiovascular side effects with theophylline and beta adrenergic bronchodilators; ocular, metabolic, musculoskeletal, and psychotropic effects of steroids; gastrointestinal and genito-urinary problems with anticholinergic agents. 23
Diagnostic Considerations
The mainstay of COPD diagnosis is pulmonary function testing as the clinical presentation is quite non-specific. The overwhelming majority of affected patients have a significant smoking history (often 20 pack years or more of tobacco use). Common symptoms include dyspnea, especially with exertion and chronic productive cough and wheezing. In the setting of a significant history of tobacco use, the above symptoms are quite suggestive of COPD. However, as some or all of these findings can be seen with other pathologies (such as cardiovascular disease, asthma, malignancy, and medication use–-eg, cough with ACE inhibitors), objective evidence with pulmonary function testing is required to establish the diagnosis.
Presently, two different criteria can be used for COPD diagnosis on spirometry: post bronchodilator FEV1/FVC ratio of <70% (Global Initiative for Obstructive Lung Disease or GOLD Guideline), or post bronchodilator FEV1/FVC ratio of <5% lower limits of normal (LLN) for reference population (American Thoracic Society/ European Respiratory Society). The drawback with the fixed FEV1/FVC -ratio of <70% is that its use can lead to over-diagnosis of COPD, especially in the never-smoking, asymptomatic elderly population.24,25–27
However, the post bronchodilator FEV1/FVC fixed ratio cut-off has important prognostic implications. Elderly with FEV1/FVC ratios above the LLN but below the fixed 0.70 limit, have an increased risk of death and COPD-related hospitalizations when compared to asymptomatic age-matched individuals with normal lung function. 28 At present, the Global initiative for Chronic Obstructive Lung Disease (GOLD) guideline, recommends using the fixed ratio criterion for diagnosing COPD. 24
Besides diagnosis, pulmonary function testing is also used for rating the severity of COPD (see Table 1). 24 FEV1 is an easily measured, reproducible parameter that has reasonable predictive utility for morbidity and mortality in COPD. Use of the post bronchodilator FEV1 is recommended for severity determination of COPD. The BODE index is a recent tool that incorporates four different prognostic factors, including FEV1 (the other measures are body mass index, severity of dyspnea, and exercise capacity on a 6 minute walk test) and is currently the most reliable predictive model for outcome/mortality in COPD patients. 29 However, it has not been thoroughly validated in an elderly population.
Severity staging of COPD (post Bronco-dilator FEV1/FVC < 0.70) and treatment.

Adapted from: Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. 2010. 2
Other prognostic factors, such as the presence of hypoxia (with and without exertion), evidence of right heart failure, pulmonary hypertension, and polycythemia, are also used in determining severity of disease and, along with hospitalization with hypercapneic respiratory failure, serve as poor predictors for outcome.30,31 These should be part of the routine periodic evaluation of elderly COPD patients.
Therapeutic Considerations for COPD in the Elderly
Overview
Management of COPD should ideally involve a multi-disciplinary approach. Treatment should focus not only on the management of the disease itself, but also on COPD's complications, as well as its social and psychological consequences. Goals of therapy include alleviation of symptoms, improvement in quality of life, reduction of mortality and morbidity (including exacerbations, emergency room visits and hospitalizations), and preservation of lung functionwhile minimizing side effects of therapy. 24
Prevention
Smoking cessation
Since smoking is the major risk factor for development of COPD, primary prophylaxis focuses on smoking cessation. Even in cases where the diagnosis of the disease has already been established, quitting smoking can arrest the rate of decline of lung function and, hence, slows progression of COPD.24,32
The best success rates for smoking cessation are with programs that use a combination of behavioral support and pharmacological treatment. 18 Behavioral support can vary from as little as a brief counseling session by the physician during a clinic visit to more extensive, frequent periodic visits in a smoking cessation support group. Telephone support is also available via a free nationwide quit line (1-800-QUIT-NOW) in USA.
Various pharmacological agents are available to assist with smoking cessation. First line therapies include nicotine replacement therapy (NRT), bupropion and varenicline. These have been found to be safe and effective for the treatment of nicotine dependence, and have been approved by the FDA for thispurpose. Although, the guidelines published by the US Department of Health and Human Services do not recommend one medication over another, varenicline and combination nicotine replacement therapy (long term nicotine patch plus ad lib nicotine replacement) appear to have the best abstinence rates.33,34
NRT works by alleviating nicotine withdrawal symptoms in smokers. NRT products differ in their route of delivery: transdermal (patch), oral mucosa (lozenges, gums, inhalers) or nasal mucosa (spray). 35 The transdermal patch has a long half-life, slow onset of action and leads to generally stable nicotine levels over the course of 24 hours. Subjects generally have good compliance; however, the main drawback is its inability to counter acute cravings. In contrast, the other nicotine replacement products that are absorbed by the mucosal surfaces, have a rapid onset of action, short half lives and lead to variable nicotine blood concentrations, necessitating repetitive use during the day. However, they do offer flexibility for the subject's nicotine withdrawal symptoms. Typically, treatment regimens include use of transdermal patch (usually starting at its highest dose of 21 mg/day for 4–6 weeks; individuals <45 kg in weight or those who smoke less than 10 cigarettes a day are advised to begin with the 14 mg/day dose) for at least 8 weeks, with a gradual taper; supplementation with other short acting NRT products can be used as needed to alleviate nicotine cravings. In general, NRT is safe and has minor side effects (gastrointestinal complaints, palpitations, insomnia, and skin irritation with the patch, especially if applied to the same spot daily); no increase in the incidence of myocardial infarctions or death, or depression/anxiety was observed in a recent meta-analysis. 36 Both bupropion and varenicline counteract nicotine at the nicotinic acetylcholine receptors in the central nervous system (CNS). Varenicline is a partial agonist, while bupropion is an antagonist at these receptors. Bupropion also blocks the dopamine and norepinephrine reuptake pumps in the CNS, neurotransmitters that are intricately involved in the reward system associated with nicotine dependence. Multiple trials have demonstrated the efficacy of these medications in smoking cessation; comparisons between the two drugs have shown superior sustained abstinence rates with varenicline. 18 Varenicline is used alone, whereas bupropion can be used alone or in combination with NRT (combination therapy has been shown to have better success than bupropion alone). The clinician needs to exercise caution when using these medications–-especially in patients with comorbid depression–-as there is an increased risk of suicidality reported with both. 37 The FDA placed a black box warning to this effect for both medications in 2009. Further studies are currently under way to evaluate the safety of varenicline. Lastly, bupropion can lower the seizure threshold and is, contraindicated for use in patients with seizure disorders.34,37,38
Vaccinations
It is strongly recommended that COPD patients receive yearly flu vaccines and be current on their pneumococcal vaccine. Viruses and bacteria, especially S. pneumoniae and influenza virus, are responsible for the majority of acute exacerbations in COPD patients and are associated with significant morbidity and mortality. 39 Vaccinations against these pathogenic microorganisms are hence advised. Influenza vaccine, in particular, has been shown to significantly decrease the rate of hospitalization and death in COPD patients. 40 Pneumococcal vaccine decreases the incidence of the possibly fatal invasive disease; it should be given at the time of diagnosis of COPD and should be repeated once at age 65 years or after 5 years–-whichever is later. 41 There is no need for a repeat dose if the initial pneumococcal vaccine is given at or above age 65.
Non-Pharmacological Therapy (LTOT, Pulmonary Rehab, LVRS, Pulmonary Transplant)
Supplemental oxygen
Supplemental oxygen, along with smoking cessation, is so far the only proven medical therapy to improve survival in selected COPD patients. Continuous oxygen therapy (15 hours/day, including at night) is recommended for patients with severe significant resting hypoxemia.42,43 The indications for supplemental oxygen in COPD are given in Table 2. 44
Indications for continuous oxygen use in patients with COPD. 44
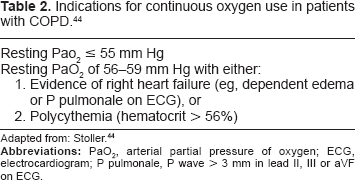
Adapted from: Stoller. 44
Pulmonary rehabilitation
Pulmonary rehabilitation is an evidence-based, comprehensive, cost-effective, multi-disciplinary program that encompasses individual patient assessment, education, exercise training, nutritional counseling, and psychosocial support.45,46 It has been shown to confer significant improvement in health-related quality of life in patients with moderate to severe COPD, including improvement in subjective dyspnea, fatigue, emotional function and patients’ perception of disease control. 47 It also decreases health care utilization and number of hospitalized days in these patients.
Surgical Interventions Bullectomy/lung volume reduction surgery/lung transplant
Patients who remain symptomatic despite optimal medical management are candidates for surgery. As surgery can be associated with significant morbidity and mortality, careful patient selection is necessary. Currently, there are three viable surgical procedures available for COPD. Two of these, bullectomy and lung volume reduction surgery (LVRS), aim to improve lung mechanics and redress the balance between diseased and unaffected parenchyma; the third option, lung transplantation, replaces the damaged lung(s) with healthy donor tissue.
The parenchymal destruction in COPD is associated with loss of elastic recoil. Hyperinflation occurs, placing the diseased lungs at a mechanical disadvantage and leads to increased work of breathing. 48 Bullectomy and LVRS decrease hyperinflation by removing damaged lung parenchyma that–-due to its loss of recoil-had expanded, compressed and adversely affects normal lung tissue. Lung volume reduction also improves the radial traction on terminal bronchioles, thus decreasing airflow obstruction, as well as optimizes diaphragmatic positioning and function. Both procedures are useful in patients who have non-uniform distribution of parenchymal damage.49–51
Bullectomy can be considered when a large air filled bulla (ideally, ≥1/3rd of hemithorax, with significant compression of the rest of the lung, and an FEV1 of >50%), is encountered on chest radiology in patients with significant dyspnea; it is also indicated in the setting of recurrent pneumothoraces, bleeding or infection. 29 Resection can be performed even in asymptomatic patients if the lesion is classified as giant (>50% of hemithorax). Extensive co-morbid conditions, bilateral bullous changes and significant emphysema in the contra-lateral lung are contraindications to the procedure. Bullectomy is associated with short-term improvement in lung function, dyspnea and quality of life.
The National Emphysema Treatment Trial examined the role of LVRS in treatment of COPD patients. Certain high risk patients fare poorly after LVRS with a significantly increased mortality rate compared to medical therapy. LVRS in this group (FEV1 ≤ 20% and either DLCO ≤ 20% or homogenous distribution of emphysema) is hence contraindicated. However, highly selected patients with emphysema appear to have a survival benefit, along with improvement in functional status and dyspnea, with use of LVRS. This group includes patients with severe obstruction (FEV1 < 45%), but whose FEV1 is >20%, heterogenous distribution of disease (predominantly upper lobe involvement), with a low exercise tolerance after pulmonary rehabilitation. 30 Other sub-groups of emphysema patients, including patients with low exercise tolerance and non-upper lobe predominant disease, also appear to obtain benefit in exercise tolerance and dyspnea with LVRS. 52 It should be noted that LVRS may be associated with detrimental outcomes in advanced age (>75 years of age) and hence should be advocated with caution in elderly.53,54
Lung transplantation confers a benefit in exercise tolerance and improves quality of life in patients with advanced COPD. It is controversial whether there is a significant survival advantage associated with transplant. 49 However, as with LVRS, risks of complications in the elderly are higher with lung transplantation and generally, most centers do not consider subjects >65 years of age for listing for lung transplant.
Pharmacotherapy of COPD
Standard maintenance therapy (Table 3)
Commonly used medications for COPD and their potential side effects.
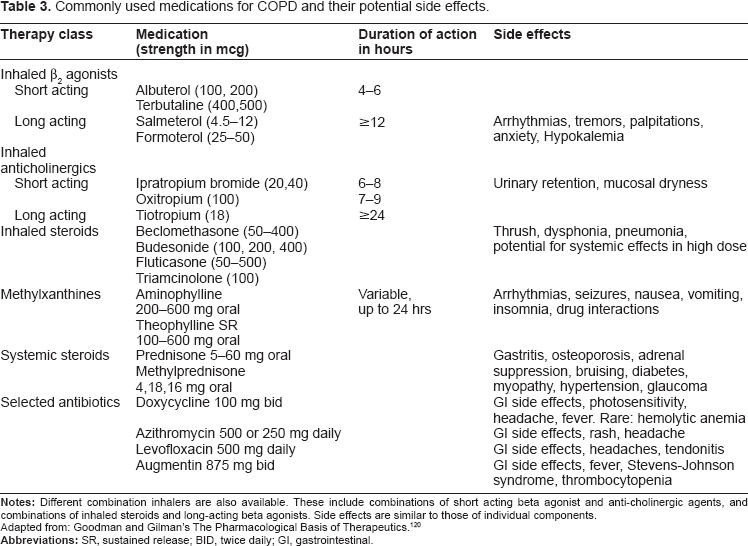
Maintenance pharmacological treatment improves quality of life and decreases the frequency of acute exacerbations in patients with COPD. As needed short acting bronchodilator therapy are recommended for mild disease. As the disease becomes more severe, long acting bronchodilators become a mainstay of therapy. Inhaled corticosteroids are also useful in patients with severe disease with recurrent acute exacerbations (See Table 1). 1 Long-term oxygen therapy improves survival and quality of life in patients with severe disease and hypoxia (see above).
Short acting inhaled bronchodilators
Both anti-cholinergic and β2 agonists cause significant bronchodilation and improvement in expiratory flow rate and are the mainstay of short term symptomatic relief in patients with COPD.55–57 Dyspnea also improved with use of β2 agonists. 57 These effects are dose-related; higher doses, especially with β2 agonists, are associated with increasing frequency of side effects. 55 Side effects are generally mild and well tolerated. 57 However, anti-cholinergic therapy appears be more efficacious in causing persistent bronchodilation than the short acting β2 agonists. 58 Combination therapy with both bronchodilators also causes significant airway dilation.59–61
Long acting inhaled bronchodilators
Long acting bronchodilators–-β agonists (LABA) and the anti-cholinergic inhaler tiotropium–-can be used alone or in combination for moderate or severe COPD. Both appear to have beneficial effects on dynamic hyperinflation as well as dyspnea indices, and are superior to short acting bronchodilators in terms of improvements in quality of life and dyspnea.62–65 Tiotropium, in particular, displays significant benefits, including decreased frequency of exacerbations, cost-effectiveness, decreased hospitalizations and length of stay in COPD patients.66,67 A recently conducted large scale double-blind placebo controlled trial (UPLIFT) showed significant improvement in quality of life, lung function and exacerbations with tiotropium use in patients with moderate to severe COPD–-regardless of the other respiratory medications used by the subjects. 68 At the present time, it is unclear whether use of anti-cholinergic agents is associated with significant adverse cardiovascular events. 69 Until this question is adequately answered; tiotropium should be used with caution, especially in patients with cardiovascular disease.
As with short acting agents, combination therapy with both tiotropium and long acting beta agonists appears to have greater benefit (with improvement in spirometric indices, quality of life and symptoms) than monotherapy alone.70,71
Inhaled corticosteroids (ICS) and combination therapy (ICS/LABA)
ICS are indicated for therapy in patients with severe COPD (FEV1 < 50%) who have recurrent exacerbations (eg, 3 over the course of the previous 3 years). Treatment with ICS in this group of patients decreases the frequency of exacerbations and is associated with improved health status; conversely cessation of ICS leads to frequent exacerbations and worsening of health measures in such patients.72–74 Combined ICS and long acting beta agonist therapy appears to be more beneficial than ICS alone. Patients on combination therapy have fewer exacerbations, improved lung function, symptoms and health status when compared to treatment with the individual components alone.75–78 A recent post-hoc analysis of the TORCH trial suggests that combination therapy with ICS/LABA may decrease the rate of decline of lung function. 79 There is no significant change in mortality with treatment with ICS and ICS/LABA; however, use of both is associated with an increased likelihood of pneumonia.80,81
Methylxanthines
Bronchodilator effects with theophylline are seen only at high doses. However, low dose sustained theophylline, even in the absence of significant bronchodilation, was shown to decrease the frequency of exacerbations and improve quality of life in COPD in one small trial. 82 The authors would urge physicians to use caution when prescribing this medication due to its potential for significant side effects, even at therapeutic levels, in the elderly. 22 Metabolism and degradation of this medication is impaired significantly with age especially in the presence of comorbid conditions and other drugs.
Therapy for acute exacerbation
The course of COPD is punctuated by acute episodic worsening in the majority of patients, especially those with severe disease. These exacerbations are characterized by deterioration in the patients’ status with aggravation of dyspnea, cough and/or sputum beyond that seen with day to day variation, and necessitate a change in their medication regimen.83,84 Exacerbations are associated with a decrease in quality of life, worsening of symptoms and lung function, and increased mortality.83–86 Often, patients’ symptoms and airflow take weeks to recover; in some cases recovery is incomplete.87,88
The most common etiologies of COPD exacerbations are respiratory tract infections–-both viral and bacterial–-and environmental pollution.89–91 In up to a third of the cases, however, the exact cause remains unknown.92,93 Other conditions that may masquerade or complicate an exacerbation of COPD–-eg, cardiac pathologies, pulmonary emboli–-should be looked for and treated if indicated.94,95
The optimal setting of treatment is determined by the severity of the patient's symptoms, baseline lung function and co-morbid medical conditions. Patients who develop respiratory acidosis and require ventilator support have a significant associated mortality risk. Physicians should have a low threshold to hospitalize patients who have severe baseline pulmonary impairment and/or significant co-morbidities. Management includes use of bronchodilators, antibiotics and steroids, depending on the severity of exacerbation. 95
Bronchodilators during acute exacerbation
Bronchodilator therapy as covered in maintenance therapy is continued during exacerbations. Short acting bronchodilators, however, are used more frequently to provide symptomatic relief and improve airflow. 95 SABAs are used as first line therapy and are used in higher doses and frequency than for maintenance treatment during acute COPD exacerbations. Short acting anti-cholinergic agents are used as second line treatment, if adequate response is not achieved with SABAs. The role of methylxanthines is controversial; clinical benefit during exacerbations has not been shown consistently and is, at best, modest.96–98 Coupled with the potential for serious side effects with the intravenous formulation (the route used for the drug during exacerbations),98,99 the above factors limit use of methylxanthines only to refractory cases with no significant improvement with SABAs and anti-cholinergic agents.
Antibiotics
Antibiotics are recommended for an exacerbation if there is an increase in sputum purulence along with one or more cardinal symptom (increase in sputum volume, or increase in dyspnea) or if the exacerbation is severe enough to warrant ventilatory support (invasive or non-invasive). 2 Antibiotics have been shown to enhance recovery from exacerbation and reduce airway inflammatory markers. Antimicrobial therapy is directed against bacteria that are most commonly isolated during exacerbations–-S. pneumoniae, H. influenza, and M. catarrhalis (although atypical pathogens, such as Chlamydia and Mycoplasma have also been identified).100–102 Typically, macrolides, doxycycline or cephalosporins are used initially; fluoroquinolones or amoxicillin/clavulanic acid are generally reserved for treatment failures.25,103 Institutionalized patients may also need coverage for gram negative bacteria, including Pseudomonas species, and may require fluoroquinolones, in combination with another antibiotic, as first line therapy. 25
Corticosteroids
Systemic steroids curtail the duration of exacerbation, shorten hospital stay, decrease treatment failure and relapse, and improve lung function and hypoxia.104–108 However, they are associated with significant side effects. 107 They should be used for acute COPD exacerbations in all hospitalized patients, and should be considered for outpatient treatment of exacerbations if baseline FEV1 is <50%. 2 The exact strength and duration is controversial. Generally, prednisolone of 30–40 mg per day or its equivalent, is used for 7–10 days.105,106,108 Prolonged duration of treatment is not associated with greater efficacy and is associated with a higher risk of side effects. 109
Special considerations
Compliance to therapy can often be suboptimal in the elderly. Advanced age, especially in association with complicated medical regimens, is a predictor for poor adherence to therapy. Physical and cognitive handicaps (eg, arthritis, poor memory and vision), along with improper technique of usage of inhalers, can further compound matters. The physician needs to keep these factors in mind when implementing his or her management strategy for the patient. 110 Prescribed regimens should be simplified as much as possible and written down in large fonts for the patient. Inhaler technique should be assessed and, if necessary, corrected during every clinic visit. Spacer or breath actuated devices, dry powder inhalers or nebulizers may be needed in patients who continue to have difficulty mastering inhalation technique and may also be useful for those with problems with co-ordination due to neurological or musculoskeletal conditions. Patients also need education with regards to identification and avoidance of triggers that may worsen airflow, and in identifying warning signs of worsening disease that may warrant assessment in a medical facility.
Conditions that worsen control of COPD should also be managed. Beta blockers and ACE inhibitors, which may worsen cough associated with chronic bronchitis, may need to be discontinued. Gastro-esophageal reflux disease and allergic rhinitis should be treated with appropriate medications. Life-style changes (head of bed elevation, small frequent meals, avoidance of oral intake 2 hours before bedtime) may be required as part of the management of GERD.
Lastly, end of life discussions should be an integral part of management of patients with advanced disease. Factors that help identify such patients include severe disease with poor baseline lung function, hypoxia, heart failure, cachexia, hospitalization with respiratory failure, significant co-morbid conditions, and age >70 years.30,111 Patients with two or more of the above risk factors are more likely to have a poor outcome if admitted to the ICU and placed on mechanical ventilation. Early discussion about end of life planning, directives and palliative care should be initiated in these patients. Concomitant psychiatric disorders such as anxiety and depression are common and often undiagnosed in these patients and may influence their decision making; these should be identified and treated prior to starting the above dialogue. 112 Typically, patients are quite receptive to discussions with their physician about their disease, prognosis, and end of life planning. 113
Other therapies
The cyclic nucleotides, adenosine mono-phosphate (cAMP) and guanosine mono-phosphate (cGMP), are intracellular second messengers that suppress the activity of inflammatory cells in the airway. They also mediate the relaxation of airway smooth muscles through the activation of protein kinase A and B. However, both cyclic nucleotides are unstable due to the presence of the phosphodiesterase enzymes (PDEs), which hydrolyze and degrade them. So far, 11 genetically distinct phosphodiesterase isoenzymes have been identified. Amongst the isoenzymes, PDE4 is highly selective for cyclic AMP and represents the major cyclic AMP metabolizing enzyme in all immunocompetent cells. Inactivation of cAMP by PDE4 results in a pro-inflammatory state. PDE4 has a ubiquitous role in most phases of the inflammatory process in COPD. It is expressed in many airway cells involved in the pathogenesis of COPD, including neutrophils, macrophages, T cells, and endothelial cells. PDE4 inhibition increases the level of cAMP, which in turn suppresses release of pro-inflammatory mediators, augmentation of the discharge of anti-inflammatory mediators, and bronchodilation. Further, PDE4 inhibitors suppress the pro-inflammatory chemokinesTNF-alpha and GM-CSF. 114
Since airway and systemic inflammation is now thought to play a major role in the pathogenesis of COPD, recent research has focused on anti-inflammatory therapy, including PDE4 inhibitors and macrolides. The selective PDE4 inhibitor roflumilast has been recently approved for the treatment of COPD. A randomized placebo-controlled clinical trial showed modest improvement in prebronchodilator FEV1 and reduction in episodes of acute exacerbations of COPD with roflumilast. The major side effects related to use of Roflumilastare gastrointestinal symptoms including diarrhea (9%), nausea (5%) and weight loss (12%). 115
Macrolides are another newer generation of medications currently understudy for COPD treatment. They belong to a family of agents that affect the 50 S ribosomes of both prokaryotic and eukaryotic organisms. 116 These agents have anti-inflammatory, antifungal and antineoplastic properties. Macrolides have been well studied in the treatment of Bronchiolitis Obliterans and have been shown to reduce airway inflammation. A recent trial evaluated the use of macrolides in COPD and showed a significant reduction in COPD exacerbations with 12 month use of erythromycin 250 mg bid compared to placebo. 117 A current NIH sponsored study aims to evaluate the effect of azithromycin in the treatment of stable COPD. The result of the study is still pending at this time.
Future directions
COPD is a heterogeneous disease. Its presentation can vary from one extreme of pure emphysema, the classically described “pink puffer”, to the other extreme of chronic bronchitis or “blue bloater”. Prior research enrolled all COPD comers and thus, was not able to distinguish between the diverse phenotypes that may respond differently to various interventions. Fortunately, current research focuses on separating various phenotypes that may respond to an intervention differently from the stand point of efficacy and/or safety. This issue is even more important in the elderly as the efficacy and/or safety of the interventions may differ significantly from the younger patients. Further, identifying the genetic predisposing factors is underway in a large National Institute of Health sponsored study. 118 The study not only will help to differentiate various genotypes and phenotypes, but also will help to identify new therapeutic targets. Potential therapy includes agents that target oxidants, leukotreines, adhesion molecules, chemokines and cytokines, proteinases, fibrosis, mucus hypersecretion and lung regeneration. 119 Research and development of many such new treatments are already under way. Although, development of these therapeutic targets into approved medication for use may take a while, time and funding is in favor of the fight against COPD. The flow of resources form federal, private and pharmaceutical sources will help accelerate discovery and production of new therapeutic targets.
Conclusion
COPD is a significant, under-recognized, worldwide health problem that is common in the elderly. Its diagnosis is complicated by a lack of recognition of symptoms by the patients and by the absence of a consensus on accurate diagnostic criteria in this population. As no cure is currently available, 119 prevention with smoking cessation is an important strategy to tackle this disease. Management consists of both pharmacological and non-pharmacological measures. Co-morbid conditions, drug interactions and other special considerations need to be addressed when treating COPD in the elderly. Ongoing research will help to identify subsets of COPD patients with more favorable response to interventions.
Disclosure
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
