Abstract
Type 2 diabetes mellitus (T2DM) is a chronic, progressive metabolic disorder that is associated with long-term microvascular (retinopathy, neuropathy and nephropathy) and macrovascular (myocardial infarction, stroke, peripheral arterial disease) complications. Both the prevalence of T2DM and the cost of its long-term complications have driven the focus and emphasis on treatments aimed at reducing hyperglycemia and controlling hypertension and dyslipidemia while minimizing hypoglycemia and weight gain. Exenatide twice daily, the first GLP-1R agonist approved by the US Food and Drug Administration (FDA) and European Medicines Agency (EMEA), has been shown to reduce hemoglobin A1C, lower fasting and postprandial plasma blood glucose concentrations as well as reduce body weight without causing significant hypoglycemia. However, its current formulation requires twice daily subcutaneous injections and does not provide continuous GLP-1R activation. Therefore, a long-acting release form of exenatide has been developed for use as a once-weekly injection, providing for convenient administration and continuous GLP-1R activation. This review covers the currently published data on this new formulation including mechanism of action, pharmacokinetics, efficacy and comparison trials to other commonly used anti-diabetic agents.
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic, progressive metabolic disorder that is initially characterized by hyperglycemia and associated with long-term microvascular (retinopathy, neuropathy and nephropathy) and macrovascular (myocardial infarction, stroke, peripheral arterial disease) complications. It affects more than 230 million people worldwide and is expected to affect 366 million by 2030. 1 The initial hyperglycemia results from a combination of insulin resistance and relative insulin deficiency. Persistent hyperglycemia as well as co-morbidities such as hypertension, dyslipidemia and obesity contributes to the development of vascular complications and an increased risk of cardiovascular disease.2,3 Intensive glycemic control has been shown to reduce the development of microvascular complications, but its effect on macrovascular complications is less clear. 4 However, despite the introduction of several new classes of anti-diabetic medications, the majority of patients with T2DM do not achieve recommended glycemic targets. 5
Both the prevalence of T2DM and the cost of its long-term complications have driven the focus and emphasis on treatments aimed at reducing hyperglycemia and controlling hypertension and dyslipidemia while minimizing hypoglycemia and weight gain. Two new classes of anti-hyperglycemic medications act on the incretin system, glucagon-like peptide-1 (GLP-1) receptor agonists (incretin mimetics) and dipeptidyl peptidase (DPP-IV) inhibitors (incretin enhancers). Exenatide twice daily, the first GLP-1R agonist approved by the US Food and Drug Administration (FDA) and European Medicines Agency (EMEA), has been shown to reduce hemoglobin A1C by 0.8%–1% in placebo-controlled trials and 1.1%–1.4% in open-label comparator studies.6–12 It lowers fasting and postprandial plasma blood glucose concentrations as well as reduces body weight in a substantial proportion of treated patients without causing significant hypoglycemia. However, its current formulation requires twice daily subcutaneous injections due to a short half-life (t ½ 2.4 hrs) and does not provide continuous GLP-1R activation. Therefore, a long-acting release form of exenatide has been developed for use as a once-weekly injection, providing for convenient administration and continuous GLP-1R activation. This review covers the currently published data on this new formulation including mechanism of action, pharmacokinetics, efficacy and comparison trials to other commonly used anti-diabetic agents.
Mechanism of Action
Exenatide LAR, subsequently designated exenatide once weekly (QW), is a glucagon-like peptide-1 (GLP-1) receptor agonist. GLP-1 is the most notable incretin hormone, an intestinally derived hormone that stimulates glucose-dependent insulin secretion in response to food intake. Not only does GLP-1 enhance nutrient-stimulated insulin secretion via activation of GLP-1 receptors on pancreatic β-cells, it also binds to receptor sites in multiple tissues to inhibit glucagon secretion, slow gastric emptying, increase satiety to decrease food ingestion, decrease body weight and improve endothelial dysfunction. Also, GLP-1 has been shown to have inotropic effects in patients with heart failure, exert cardioprotective actions when administered post-ischemia and may possibly be of neuroprotective significance by limiting hyperglycemic fluctuations in the brain.13–16 Because GLP-1 is predominantly a potentiator of glucose-stimulated insulin secretion, it has little or no effect on insulin release at low or normal glucose levels; thus, there is minimal risk of hypoglycemia when used alone.
Basal and stimulated β-cell function, as well as serum blood glucose levels, can be normalized by 24 hour GLP-1 infusion, 17 demonstrating the potential role of continuous GLP-1 receptor activation in improving glycemic control in T2DM. Exenatide once weekly uses a proprietary technology for long-acting medications developed by Alkermes. The technology encapsulates active medication into polymer-based microspheres made of polylactide co-glycolide (PLG), that are injected into the subcutaneous tissue where they degrade slowly, gradually releasing the drug in a controlled manner to provide continuous therapeutic exenatide levels in plasma. 18
Pharmacokinetics and Metabolism
Exenatide twice daily reaches median peak plasma concentrations in 2.1 hours, has a mean terminal half-life of 2.4 hours and is primarily eliminated by the kidneys. 19 With exenatide once weekly injections, mean plasma concentrations rise gradually over 6 weeks with greater time to peak plasma and steady-state concentrations compared to exenatide twice daily. Based on graphical evaluations of plasma exenatide trough concentrations, plasma exenatide concentrations reached 50 pg/mL, a concentration previously shown to significantly reduce plasma glucose, 20 by week 2 for the 2.0 mg once weekly dose and by weeks 4–5 for the 0.8 mg once weekly dose (Fig. 1). 21 Plasma exenatide concentrations were maintained at concentrations similar to the maximum concentration of 10 μg exenatide twice daily after about 6 weeks (steady-state concentrations of 232 pg/mL with 2.0 mg exenatide once weekly compared with 211 pg/mL after a single injection of 10 μg exenatide twice daily). 22 The steady-state concentration with 0.8 mg exenatide once weekly was 111 pg/mL. After the last injection at week 15, exenatide concentrations decreased steadily to below those considered to have a therapeutic effect by week 21. Pharmacokinetic parameters noted in a study of Japanese patients were similar to findings in the study above which consisted of a primarily Caucasian population. 23 These findings were consistent with expectations as exenatide is a peptide that is administered subcutaneously and therefore would not be subject to polymorphic differences in gut or liver enzyme expression or dietary variations.
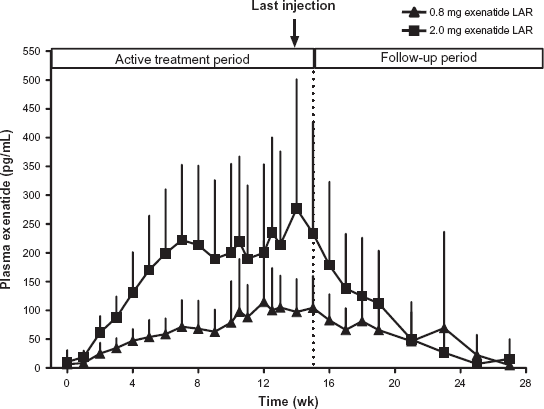
Plasma Exenatide Reached Steady-State Concentrations by ~Week 6. ITT subjects. Exenatide LAR: 0.8 mg N = 16, 2.0 mg N = 15. Mean ± SD. Adapted from Kim D, et al. Diabetes Care. 2007;30:1487–1493. 12
Efficacy
Exenatide once weekly has completed phase 2 and 3 studies, and is currently under review by regulatory authorities. In one 15 week, randomized, placebo-controlled phase 2 study exenatide once weekly (0.8 or 2.0 mg) was administered to 45 patients with type 2 diabetes suboptimally controlled with metformin (60%) and/or diet and exercise (40%). 21 Exenatide once weekly reduced mean A1C levels by 1.4% and 1.7% in the 0.8 and 2.0 mg groups respectively compared to an increase in A1C by 0.4% with placebo (P < 0.0001). Fasting plasma glucose was also significantly reduced and a greater percentage of patients receiving exenatide once weekly were able to achieve an A1C ≤ 7.0% (36%, 86% and 0% in patients receiving 0.8, 2.0 mg exenatide once weekly and placebo, respectively). Only the 2.0 mg/week dosage of exenatide once weekly was associated with body weight reduction (-3.8 kg, P < 0.05), whereas weight was unchanged in both the 0.8 mg/wk dose and placebo. Mild nausea was the most frequent adverse event, occurring in 19%, 27% and 15% of patients in the exenatide once weekly 0.8 mg, 2.0 mg groups and placebo group, respectively.
Comparison Trials
DURATION, which stands for “Diabetes therapy Utilization: Researching changes in A1C, weight and other factors Through Intervention with exenatide ONce weekly”, is an acronym for the overall exenatide once weekly clinical program. It was designed to test the safety and efficacy of exenatide once weekly to other medications in the treatment of type 2 diabetes, results shown in Table 1.
Clinical efficacy.
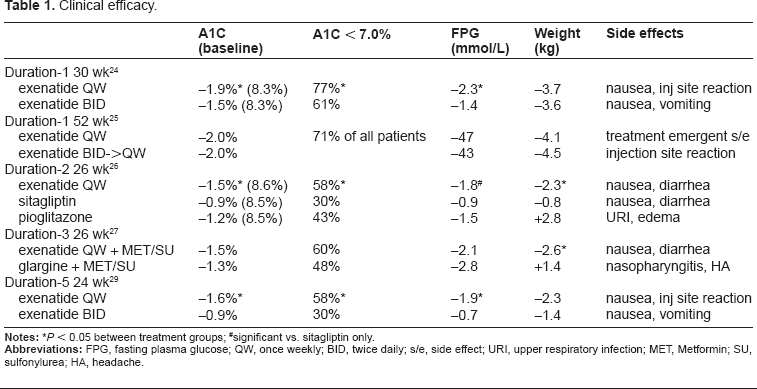
#significant vs. sitagliptin only.
To directly compare the two dosing methods, a 30 week, randomized, non-inferiority study (DURATION-1), was completed using exenatide once weekly 2 mg once weekly versus traditional dosing of exenatide 10 mcg twice daily in 295 patients with uncontrolled type 2 diabetes on one or more oral agents. There were no differences in baseline A1C, fasting blood glucose, BMI or duration of diabetes between the groups. The exenatide once weekly group demonstrated a significantly greater reduction from baseline in A1C (-1.9% vs. −1.5%, P = 0.002) and fasting blood glucose (-42 mg/dl vs. −25 mg/dl, P < 0.0001) compared to the group dosed twice daily. In addition, a greater percentage of patients on the weekly regimen (77% vs. 61%, P = 0.004) achieved an A1C level ≤7%. Weight loss did not differ between the groups and nausea was significantly lower in the group dosed weekly. 24
In a 22 week open-ended extension of the above trial, subjects originally randomized to exenatide once weekly remained on weekly doses and those previously receiving twice daily converted to weekly doses. 25 Both groups experienced durable glycemic improvement and weight loss at the 52 week mark regardless of the original dosing regimen, but patients who switched from the twice daily to the once weekly dosing regimen were noted to have a temporary decline in glycemic control during the conversion. This is thought to be due to the long half-life of the once weekly formulation that may require 4–6 weeks to achieve steady state. At 52 weeks, A1C decreased by 2% and 72% of patients were able to achieve an A1C < 7%. On average, patients lost about 4 kg without any episodes of major hypoglycemia and in those patients not receiving a concomitant sulfonylurea (SU), there were also no episodes of minor hypoglycemia. 25
The DURATION-2 trial directly compared exenatide once weekly (GLP-1 receptor agonist) and maximum approved doses of sitagliptin (DPP-4 inhibitor) and pioglitazone (thiazolidinedione) in patients with T2DM not achieving adequate glycemic control on metformin alone. 26 This 26 week randomized, double-blind, double-dummy, superiority trial consisted of 491 patients with T2DM and baseline A1C 8.5%, fasting plasma glucose of 9.1 mmol/L (164 mg/dL) and weight of 88 kg (194 lbs). 26 Exenatide once weekly produced significantly greater A1C reduction (-1.5%, 95% CI −1.7 to −1.4) compared to sitagliptin (-0.9%, −1.1 to −0.7) or pioglitazone (−1.2%, −1.4 to −1.0). Treatment differences were −0.6% (95% CI −0.9 to −0.4, adjusted P < 0.0001) for exenatide once weekly versus sitagliptin, and −0.3% (−0.6 to −0.1, adjusted P = 0.0165) for exenatide once weekly versus pioglitazone. Significantly more patients achieved A1C targets of 7.0% or less with exenatide once weekly than with sitagliptin or pioglitazone. All treatments improved fasting plasma glucose (−1.8, −0.9 and −1.5 mmol/L with exenatide once weekly, sitagliptin and pioglitazone respectively), although the treatment differences were only significant versus sitagliptin.
Exenatide once weekly resulted in significantly greater weight loss than did pioglitazone by week 1 and sitagliptin by week 4. At week 26, weight loss with exenatide once weekly (−2.3 kg, 95% CI −2.9 to −1.7) was significantly greater than with sitagliptin (−0.8 kg, −1.4 to −0.1) or pioglitazone (2.8 kg, 2.2 to 3.4). Treatment differences were −1.5 kg (95% CI −2.4 to −0.7, adjusted P = 0.0002) for exenatide once weekly versus sitagliptin, and −5.1 kg (−5.9 to −4.3, adjusted P < 0.0001) for exenatide once weekly versus pioglitazone. No episodes of major hypoglycemia occurred and the frequency of minor hypoglycemia was low and similar between treatment groups (n = 2, 1%, n = 5, 3%, and n = 1, 1% with exenatide once weekly, sitagliptin and pioglitazone respectively. The most frequent adverse events with exenatide once weekly and sitagliptin were nausea (n = 38, 24%, and n = 16, 10%, respectively) and diarrhea (n = 29, 18%, and n = 16, 10%, respectively); upper-respiratory-tract infection (n = 17, 10%) and peripheral edema (n = 13, 8%) were the most frequent events with pioglitazone.
In a 26 week, open-label, randomized, parallel study, exenatide once weekly was directly compared to insulin glargine (DURATION-3) in adults with T2DM suboptimally controlled on maximum tolerated doses of metformin or metformin plus sulfonylurea. 27 456 patients were randomly assigned to add exenatide once weekly (2 mg, once-a-week injection) or insulin glargine (once-daily injection, starting dose 10 IU, titrated to target glucose range 4.0–5.0 mmol/L or 72–90 mg/dL) to their oral regimens. At 26 weeks, mean reduction in A1C was significantly greater in the exenatide once weekly group than with insulin glargine (−1.5% vs. −1.3%, treatment difference −0.16%, 95% CI −1.29 to −0.03, P = 0.017). 27 More patients treated with exenatide once weekly than with insulin glargine achieved target A1C less than 7.0% (60% vs. 48%, P = 0.010). Exenatide once weekly was also associated with a decrease in body weight whereas insulin glargine was associated with weight gain (-2.6 kg vs. + 1.4 kg, P = <0.0001). The most frequently reported adverse events were nausea and diarrhea with exenatide once weekly and nasopharyngitis and headache with insulin glargine. Three patients (one taking exenatide once weekly and two taking insulin glargine) had an episode of hypoglycemia that required assistance of another person, but did not involve loss of consciousness or lead to study discontinuation. Minor hypoglycemia was reported in 19 (8%) of exenatide once weekly patients (46 events) compared with 58 (26%) of insulin glargine patients (135 events). Hypoglycemia was reported most often in patients receiving concomitant sulfonylurea; however incidence was consistently lower with exenatide once weekly than with insulin glargine.
The DURATION-4 study examined exenatide once weekly as a monotherapy treatment compared to either metformin, sitagliptin (DPP-4 inhibitor) or pioglitazone (thiazolidinedione). Results of the 26 week, double-blind, randomized, four-arm parallel study were announced in a press release June 15, 2010. 28 820 patients with T2DM uncontrolled on diet and exercise were randomized to either exenatide once weekly (2 mg, once-a-week injection, n = 248), metformin (dose escalated up to 2,500 mg/day, n = 246), pioglitazone (dose escalated up to 45 mg/day, n = 163) or sitagliptin (100 mg/day, n = 163). Exenatide once weekly produced significantly greater reduction in A1C compared to sitagliptin (-1.5% vs. 1.2%, statistical significance not available in press release) and similar to metformin (-1.5%) and pioglitazone (-1.6%). Patients receiving exenatide once weekly, pioglitazone and metformin achieved an average A1C of less than 7% by study end. Exenatide once weekly resulted in significantly greater reduction in body weight than sitagliptin or pioglitazone (-4.5 lbs, −1.7 lbs and +3.3 lbs respectively) and was similar to metformin (-4.4 lbs). There were no major hypoglycemic events in any treatment group and the frequency of minor hypoglycemia was low. The most frequently reported adverse events were nausea and diarrhea with exenatide once weekly, diarrhea and headache with metformin, upper respiratory tract infection, headache, hypertension and edema with pioglitazone, and upper respiratory tract infection and headache with sitagliptin.
The DURATION-5 trial, a randomized, open-label, parallel-group, comparator-controlled, multi-center study, evaluated the glycemic effects, safety and tolerability of exenatide once weekly (2 mg once weekly) compared to exenatide twice daily (5 μg twice daily for 4 weeks followed by 10 μg twice daily) in 252 subjects with T2DM managed with diet and exercise alone or with oral antidiabetic medications. 29 At 24 weeks, exenatide once weekly produced significantly greater changes from baseline compared to exenatide twice daily in A1C (−1.6% ± 0.1% vs. −0.9% ± 0.1%; P < 0.0001) and fasting plasma glucose (-35 ± 5 mg/dL vs. −12 ± 5 mg/dL; P = 0.0008). 29 Reductions in mean body weight from baseline to week 24 were similar between the two groups (−2.3 ± 0.4 kg and −1.4 ± 0.4 kg). Both treatments were generally well tolerated with no episodes of major hypoglycemia in either group. The most common adverse event was transient and predominantly mild to moderate nausea which occurred less with exenatide once weekly (14%) compared with exenatide twice daily (35%). Injection site reactions were more common with exenatide once weekly.
In a 26 week, head-to-head, open-label, superiority study (DURATION-6), 900 patients with T2DM inadequately controlled with diet and exercise in conjunction with metformin, a sulfonylurea, metformin plus pioglitazone, or metformin plus sulfonylurea were randomized to receive either subcutaneous injections of exenatide once weekly (2 mg, once weekly) or liraglutide (forced titration to 1.8 mg, once daily), another long-acting GLP-1 receptor agonist. 30 Results were published in a press release March 3, 2011 and showed a reduction in A1C of 1.3% from baseline for exenatide once weekly compared to a reduction of 1.5% for liraglutide, which did not demonstrate non-inferiority of exenatide once weekly to liraglutide. Gastrointestinal side effects occurred more frequently among liraglutide patients (20% experienced nausea, 11% vomiting and 13% diarrhea) compared to exenatide once weekly (9% experienced nausea, 4% vomiting and 6% diarrhea. Injection side reactions occurred more frequently in exenatide once weekly patients, 10% versus 1% with liraglutide. There were no major hypoglycemic events in either treatment group. 30
Cardiovascular Risk Factors
Other metabolic abnormalities that contribute to the increased cardiovascular mortality seen in patients with T2DM include hypertension and dyslipidemia. While improvement in lipids and markers of cardiovascular risk have been noted to varying degrees with many anti-diabetic medications, exenatide once weekly has been associated with favorable mean changes in all lipid parameters as well as systolic and diastolic blood pressure (Table 2). Exenatide once weekly reduces systolic blood pressure by about 5 mm Hg (2.9–6.2) which is significantly more than sitagliptin and similar to exenatide twice daily.10,24,27 Compared to sitagliptin and pioglitazone, exenatide once weekly is the only medication that lowers total cholesterol (TC), low-density lipoprotein (LDL), triglycerides (TG) and raises high-density lipoprotein (HDL).10,24,26,27 While pioglitazone significantly reduces triglycerides and raises HDL more than exenatide once weekly, TC and LDL are increased with pioglitazone whereas all parameters have shown improvement with exenatide once weekly.
Cardiovascular risk factors.
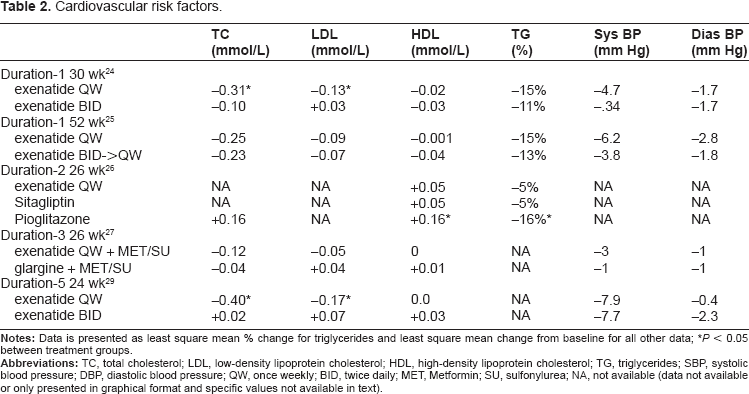
*P < 0.05 between treatment groups.
Other intermediate markers of cardiovascular risk also show improvement with exenatide once weekly including high sensitivity C-reactive protein (CRP), B-type natriuretic peptide (BNP), and ratio of albumin to creatinine. 26 Compared to sitagliptin and pioglitazone, all treatments were associated with significant improvements in CRP, but only exenatide once weekly was associated with significantly improved BNP and ratio of albumin to creatinine. While pioglitazone was associated with a significantly greater improvement in adiponectin and plasminogen activator inhibitor-1 than was exenatide once weekly, it was also the only treatment associated with a worsening of BNP. 26 A statistically significant increase in heart rate of 4 beats per minute from baseline was observed with exenatide once weekly in both the DURATION-3 and DURATION-5 trials which was not associated with cardiovascular or arrhythmia-related adverse events. 29
Safety
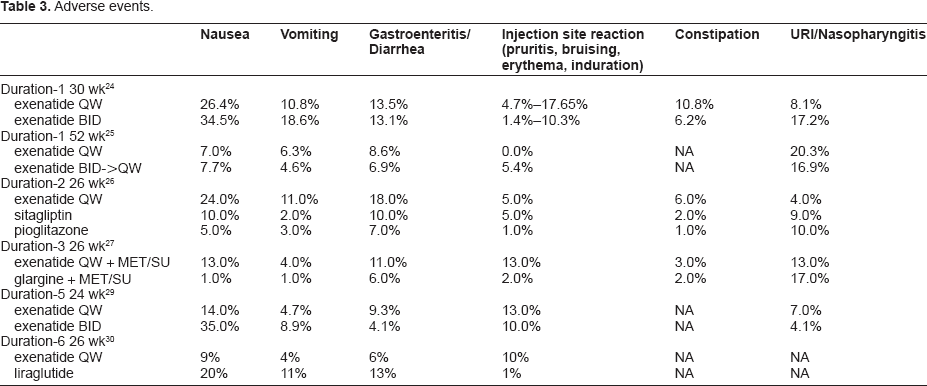
Overall, exenatide once weekly is generally well tolerated, the incidence of adverse events are listed in Table 3. Nausea and injection site reactions were the most frequent adverse events related to exenatide once weekly in the above trials. The nausea was mild to moderate and tended to abate over time. Compared to exenatide twice daily and liraglutide, patients on exenatide once weekly appeared to experience a milder nausea profile which could be due to the more gradual increase in plasma drug concentrations with exenatide once weekly.24,30,31 Injection site pruritis is an effect more commonly reported with injectable sustained release formulations that undergo in-vivo-degradation32,33 and was typically mild in intensity and resolved with continued exenatide once weekly treatment. Only one major episode of hypoglycemia was reported with exenatide once weekly in the DURATION-3 trial. A subject on exenatide once weekly and metformin experienced hypoglycemia requiring assist of another person, but did not experience loss of consciousness or need to discontinue the study medication. The frequency of minor hypoglycemia was low and tended to occur in patients on concomitant sulphonylurea. Pancreatic enzymes were measured in the DURATION-3 and DURATION-5 trials and few patients had either amylase or lipase concentrations raised to more than three times the upper limit of normal (ULN). These changes were generally asymptomatic, with the exception of two patients taking exenatide once weekly. One patient in the DURATION-3 trial was diagnosed with edematous pancreatitis (enzymes < 3 x ULN) and went on to recover fully within 1 day without hospitalization. Another patient in the DURATION-5 study with a history of dyslipidemia was hospitalized with a diagnosis of pancreatitis and the event resolved within 3 days. Exenatide once weekly was discontinued at the time of diagnosis of pancreatitis in both cases.
Adverse events.
As previously reported in studies with exenatide twice daily, anti-exenatide antibodies also developed in patients taking exenatide once weekly in the above trials. While antibody levels were higher in the exenatide once weekly group versus the exenatide twice daily group (74% vs. 48% antibody positive in DURATION-1 and 73% vs. 51% antibody positive in DURATION-5), the reduction in A1C was significantly greater with exenatide once weekly (−1.9% vs. −1.5% in DURATION-1 respectively P = 0.0023 and −1.6% ± 0.1% vs. −0.9% ± 0.1% in DURATION-5 respectively; P < 0.0001).24,29 The majority of patients in each trial had either low (< 1/625) or undetectable antibodies and antibody levels tended to plateau at 6–14 weeks and progressively decline thereafter. Regardless of antibody status, exenatide once weekly showed a significant improvement in A1C from baseline in each trial and when assessed on an individual basis, antibody status was not predictive of treatment response or rates of adverse events in any of the above trials.10,21,24,26,27,29
Long-acting GLP-1 receptor agonists have been shown to activate thyroid C cells in some rodent models, resulting in increased secretion of calcitonin, a marker of thyroid C cell hyperplasia and medullary carcinoma. 34 Calcitonin was measured in both the DURATION-3 and DURATION-5 trials with no mean changes in calcitonin concentrations noted and no events of thyroid neoplasms reported. 29 These results are consistent with observations in primate models and in patients with T2DM treated for 2 years with liraglutide, another long-acting GLP-1 receptor agonist. 34
Quality of Life
Type 2 Diabetes is associated with a significantly diminished health-related quality of life (QOL) for both patients and caregivers.35,36 Insulin therapy, obesity and diabetic complications are particular problems that lead to the impaired QOL seen in these patients. 35 T2DM is a progressive disease that often requires combination therapy and eventually insulin and these regimens are often complicated by side effects including hypoglycemia and weight gain. These complicated regimens and side effects may contribute to further worsening of the already impaired QOL of these patients and may contribute to poor compliance that is often seen in this patient population. One systematic review showed that many patients took much less than their prescribed dose of both oral anti-diabetic medications and insulin. 37 Another study showed that a substantial proportion of patients had difficulty dealing with various elements of chronic disease management, particularly adhering to a strict drug regimen. 38 Since patient satisfaction and tolerability could play an important role in the use and choice of different medications as new diabetes treatment options become available, QOL is an important component to consider.
In DURATION-1, treatment compliance (injections received/injections planned) was 98% with both exenatide regimens, but patients treated with exenatide once weekly reported significant increases in treatment satisfaction from baseline based on results from Diabetes Treatment Satisfaction Questionnaires (DTSQ). 24
To assess treatment satisfaction and weight-related quality of life, a 52-week randomized, multi-center, open-label study was done is 295 subjects with T2DM managed with diet and exercise and/or oral glucose-lowering medications. 39 These patients were randomized to either exenatide once weekly or twice daily during weeks 1–30 and then patients receiving exenatide twice daily were switched to exenatide once weekly for the remaining 22 weeks. Diabetes Treatment Satisfaction Questionnaire–-status (DTSQ-s) and Impact of Weight on Quality of Life–-Lite (IWQOL-Lite) were assessed at baseline, 30 weeks and 52 weeks. At week 30, both groups showed statistically significant improvements in total DTSQ and IWQOL-Lite scores without significant between-group differences (5.17 ± 0.54 versus 3.97 ± 0.53, P = 0.09), but treatment satisfaction did improve more in the exenatide once weekly arm for perceived hypoglycemia frequency (P = 0.03) and willingness to continue current treatment (P = 0.01). The group that switched from exenatide twice daily to exenatide once weekly experienced significantly improved total treatment satisfaction (P = 0.037), treatment convenience (P = 0.003), treatment flexibility (P = 0.012) and satisfaction with continuing treatment (P = 0.048) by week 52. From week 30 to 52, there was also a significantly improved satisfaction with treatment convenience (P = 0.006) and treatment flexibility (P = 0.025) in the group that continued with exenatide once weekly. Total treatment satisfaction and QOL were not affected by the occurrence of adverse events.
In the DURATION-2 trial, weight related QOL, psychological general well being, diabetes treatment satisfaction, and general health status were assessed using the IWQOL-Lite, Psychological General Well-being (PGWB) index, DTSQ and EQ-5D at baseline and week 26. All five domains of weight-related QOL and IWQOL total score were significantly improved with exenatide once weekly and sitagliptin, although there was no significant between-group difference. However, scores with both exenatide once weekly and sitagliptin were significantly improved compared to pioglitazone, which only improved in self-esteem. All domains of the pgWB and the DTSQ total score were significantly improved in all treatment groups, but there was greater improvement in overall treatment satisfaction with exenatide once weekly compared to sitagliptin (difference 1.61, P = 0.0406). 26
In the DURATION-3 trial, patients in both exenatide once weekly and insulin glargine treatment groups had improvements between baseline and end-point in IWQOL-Lite, BES and DTSQ-s total scores, but only patients taking exenatide once weekly had significant improvements on the EQ-5D index. Between–treatment differences in total scores were not recorded, but the exenatide once weekly group did show significant improvement in self-esteem (IWQOL-Lite domain) and usual activities (EQ-5D domain) compared to the insulin glargine group. 27
Place in Therapy
In a revised version of the American Diabetes Association and the European Association for the Study of Diabetes treatment algorithm for type 2 diabetes as well as the American Association of Clinical Endocrinologists and American College of Endocrinology's most recent treatment algorithm for type 2 diabetes, metformin is at the cornerstone of combination therapy, but new classes of drugs are now included to consider after metformin with an emphasis on keeping hypoglycemia and weight gain to a minimum.40,41 In addition to basal insulin, sulphonylurea and thiazolidinedione therapies, new treatments such as glucagon-like peptide 1 (GLP-1) receptor agonists and dipeptidyl peptidase-4 (DPP-4) inhibitors are being considered as add-on treatment to metformin or as first line therapy if contraindications to metformin are present. These newer agents not only improve glycemic control, but also could positively affect some of the metabolic abnormalities associated with type 2 diabetes including obesity, hypertension and dyslipidemia. Since obesity and drug-associated hypoglycemia are continuing concerns for many patients with T2DM, these additional effects are of particular importance.
Exenatide once weekly has shown to be well tolerated by patients and produce greater decrease in A1C than sitagliptin and exenatide twice daily. It also produces similar reduction in A1C as insulin glargine and pioglitazone, but is associated with weight loss whereas insulin glargine and pioglitazone are both associated with weight gain. Exenatide once weekly has also shown to produce similar reduction in A1C and weight loss profile as metformin, making it a reasonable alternative as first line therapy to patients with a contraindication to metformin.
Current Status
A New Drug Application (NDA) for exenatide once weekly (Bydureon) was submitted in May 2009. In October 2010, the FDA issued a complete response (CR) letter requesting a thorough QT (tQT) study to be done using high doses of exenatide in healthy subjects. Of note, in a previous tQT study with exenatide twice daily (Byetta), there was a small positive correlation between plasma exenatide concentrations and changes from baseline in QTcF (corrected QT calculated using Fridericia's formula) interval following a single dose of 10 μg exenatide twice daily (Fig. 2).42,43 The FDA extrapolated from this data to infer the potential for QT prolongation at high plasma concentrations of exenatide, which may occur with the once weekly formulation. In addition, since exenatide is excreted via the kidney, plasma concentrations may be even higher in diabetic patients in whom up to 35% have some degree of renal impairment. While evaluation of corrected QT (cQT) intervals, via comparison of ECGs at baseline and steady-state, did not show any clinically meaningful changes in the DURATION 1 trial, the FDA is requesting a dedicated thorough QT study to be done to test the cQT at supratherapeutic levels of exenatide. 24 The tQT studies with exenatide once weekly are expected to be completed by the end of 2011, which means Bydureon, could have a possible launch around mid-2012. 44
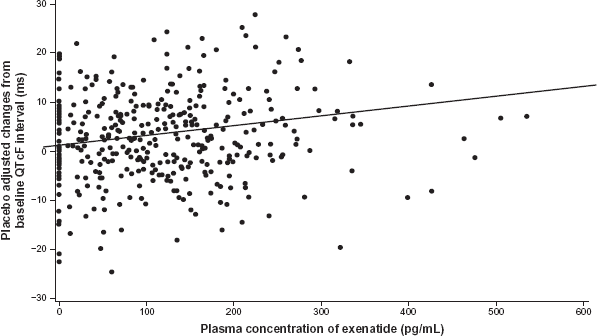
Scatterplot of changes from predose in QTcF interval versus plasma exenatide concentrations following a single10 μg dose. 43
Conclusion
Exenatide once weekly is a new agent for the treatment of type 2 diabetes and has shown promising results when compared to placebo, exenatide twice daily and other anti-diabetic agents. It has demonstrated significant reductions in weight, A1C and cardiovascular risk factors with minimal risk of hypoglycemia. Furthermore, exenatide once weekly has been generally well tolerated and shown to be associated with improvements in patient satisfaction and QOL, which might have a positive impact on patient adherence and compliance. Type 2 diabetes is a complex, progressive disorder with many barriers to treatment, imposing a significant need for more effective and more convenient treatment options. Once weekly exenatide provides both the efficacy and convenience to impact disease modification and encourage adherence, making it a promising treatment option for patients with type 2 diabetes.
Disclosure
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author and peer reviewers of this paper report no conflicts of interest. The author confirms that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgments
The author thanks Dr. Robert Henry (Division of Endocrinology and Metabolism, VA San Diego Healthcare System, University of California, USA) for assistance with editing and data compilation, and the anonymous peer reviewers for their constructive comments on this article.
