Abstract
Tremor is the most common movement disorder encountered during daily practice. Most forms of tremor are currently managed with drugs. However, the response of tremor to pharmacotherapy is variable and a combination of agents is often required. Surgery is considered when the response is not sufficient. In particular deep brain stimulation (DBS) is now playing a key-role, especially for refractory Parkinson's disease. DBS is also a well established therapy for essential tremor resistant to pharmacological therapy. New therapies are emerging, not only in the field of pharmacological agents, but also in the domain of bioengineering. They are presented in this review article.
Keywords
Introduction
Definition
Tremor is a rhythmic shaking of a body part. 1 Tremor is the most common movement disorder encountered during daily practice, with its incidence and prevalence increasing as the population ages. 2 Although tremor is not life-threatening, it causes functional disability and social inconvenience, disturbing daily life activities. 3 More than 65% of the population with upper limb tremor presents serious difficulties in performing activities of daily living (ADL).
Clinical description
Tremor is clinically defined as an oscillatory movement. Tremor may be observed at rest and/or during action. The terminology of action tremor encompasses postural, kinetic and intention tremor. 1 Rest tremor occurs while the body segment is maintained at rest and may disappear with action. Its frequency is typically in the 3–6 Hz frequency range. 4 Rest tremor is usually asymmetrical, starting distally in the arms. It often resembles the “pill rolling” movement. Rest tremor may increase with mental stress (i.e. counting backwards) or contralateral voluntary motion. Postural tremor occurs in body parts during the maintenance of a posture, such as holding a cup, and is triggered by maintaining a position against gravity. 4 The frequency of postural tremor is usually between 4 and 12 Hz. Tremor appearing during limb movement is called kinetic tremor. The so-called intention tremor (cerebellar tremor) worsens when approaching a target. The frequency is between 2 and 5 Hz in most cases, although frequencies up to 7 Hz can be encountered. Addition of inertia tends to improve kinetic tremor. 4 Midbrain tremor (also named Holmes’ Tremor 1 ) combines rest, postural and kinetic tremor. Isometric tremor occurs when a voluntary muscle contraction is opposed by a rigid stationary object. 4
Main disorders associated with tremor
Tremor may be generated by the central and/or peripheral nervous system (CNS or PNS). The main disorders associated with postural, rest and kinetic tremor are summarized in Table 1. The most common cause of rest tremor is idiopathic Parkinson's Disease (PD). Numerous disorders are associated with postural tremor. Essential Tremor (ET) is a typical example. Tremor occurring in cerebellar patients is mainly a kinetic tremor. Table 2 focuses on the extrapyramidal syndromes associated with tremor.
Main disorders associated with tremor. 5
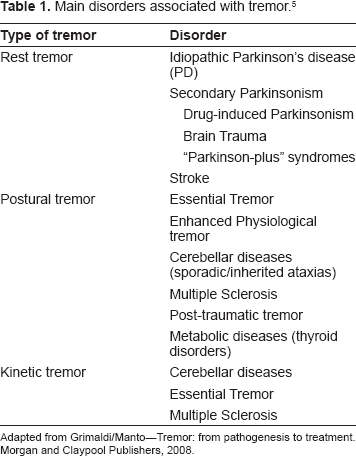
Adapted from Grimaldi/Manto–-Tremor: from pathogenesis to treatment. Morgan and Claypool Publishers, 2008.
Extra-pyramidal syndromes associated with tremor. 5

Adapted from Grimaldi/Manto–-Tremor: from pathogenesis to treatment. Morgan and Claypool Publishers, 2008.
Current Treatments of Tremor
Pharmacological treatment
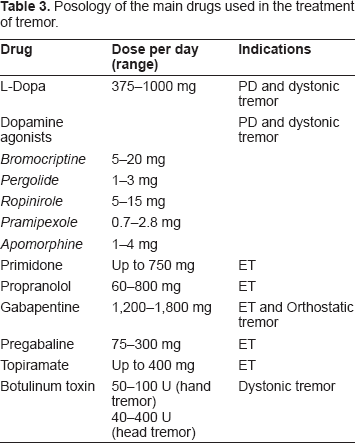
Tremor is first managed using drugs (Table 3). Levodopa, anticholinergic medications and beta-blockers such as propranolol are effective drugs for rest tremor. 6 Primidone and propranolol have similar efficacy in reducing the magnitude of hand postural tremor. 4 Kinetic tremor may respond to beta-blockers, primidone and anticholinergic medication in case of ET. 6
Posology of the main drugs used in the treatment of tremor.
Parkinson's disease
Levodopa is still recognized as the “gold standard” for symptomatic treatment of PD. 7 However, its clinical efficacy declines after a few years and the so-called “long-term levodopa syndrome” may develop progressively. 7 Despite the efforts of scientists, the optimal use (mode of administration and dose adjustment) of levodopa in the different stages of the disease tailored to individual needs has not yet been achieved. 7
Dopamine agonists, such as pramipexole and ropinirole, are probably the most effective tremorolytic drugs among all dopaminergic treatments available and should be considered in newly diagnosed PD patients exhibiting tremor in absence of cognitive impairment. 8 Improvement of tremor has also been reported with other dopamine agonists, including pergolide and bromocriptine. Dopamine agonists are also useful in advanced PD patients exhibiting a tremor refractory to levodopa and anticholinergics. 9 Rotigotine–-a non-ergot dopamine agonist transdermal patch-has demonstrated clinical efficacy, alongside a good tolerability profile. The drug is indicated for use in early (USA and Europe) and advanced (Europe only) PD. 10 The advantages of the rotigotine patch include immediacy of effect onset, constant drug delivery, and ease of use via application of a once-daily adhesive patch. An interesting feature is constant drug delivery, avoiding the pulsatile dopaminergic stimulation, which might participate in the development of motor complications. 10 Further studies focusing on tremor are required. Apomorphine (administered by the subcutaneous route 11 or using puffs) is used in advanced forms of PD and is considered less invasive than the administration of levodopa/carbidopa by continuous duodenal infusion. The latter technique requires a percutaneous endoscopic gastrostomy. 12 Clozapine -an “atypical” neuroleptic agent producing fewer extra pyramidal side effects- has a good efficacy in PD patients with rest tremor. Regular blood studies are required in order to monitor the potential agranalocytosis. 13
Given some pre-clinical evidence that the cannabinoid receptor type 1 agonist (CB1) benefits tremor, the potential therapeutic effects of targeting the cannabinoid system in basal ganglia disorders are under investigation. 14
As a supplement to conventional medical treatments in patients with PD, relaxation techniques such as the relaxation guided imagery (RGI) and relaxing music have been suggested. In a recent study patients reported an improvement lasting 2–14 hours. 15
Essential tremor
First line therapy for ET remains based on propranolol or primidone. 16 Primidone is significantly superior to placebo in reducing the magnitude of hand postural tremor. Its efficacy is similar to propranolol. 17 Topiramate (up to a maximum of 400 mg/day) is effective in the treatment of moderate to severe ET. Tremor reduction is accompanied by functional improvements, such as writing or speaking.18,19 Improvement of ET has also been reported with lower doses of topiramate (50 mg/day). 20 Levetiracetam has not been demonstrated to have a noticeable effect on ET. 21 A recent study using Pregabalin showed significant improvements in terms of accelerometry recordings and action tremor limb scores on the Fahn-Tolosa-Marin rating scale. 22 Few studies with zonisamide-approved as an adjunctive treatment for epilepsy-have shown partial benefits on tremor. 23 Adverse events include decreased concentration/cognition, constipation, nocturia, abdominal pain/diarrhea, altered taste, imbalance, and sedation.24,25 Zonisamide seems to be promising in the treatment of ET patients exhibiting head tremor. 26 The barbiturate T2000 is also under study for ET. 27 Sodium oxybate–- the sodium salt of gamma hydroxybutyrate-has shown interesting effects in preliminary studies. 28 Other new drugs, including 1-octanol,29,30 olanzapine 31 and carisbamate, are currently being investigated. 32 According to the recommendation of the American Academy of Neurology (AAN), botulinum toxin may be proposed for upper extremity ET (Level B of evidence). 33 The injections have to be repeated as the effects wear off. However, about 20% of patients develop immunity to the toxin. Although ET often responds to ethanol, this cannot be recommended as a regular treatment for obvious medical reasons. Lacosamide, which enhances slow inactivation of voltage-gated sodium channels, has shown antitremorgenic activity in rodents. 34 Further studies are warranted.
Dystonic tremor
Several drugs are active to reduce dystonia. Anticholinergics, benzodiazepines, baclofen, levodopa/dopaminergic agonists are widely used. Botulinum toxin injections may be particularly active for dystonic tremor of neck/upper limbs.
Osthostatic tremor
Gabapentin is the most effective treatment for Orthostatic Tremor (OT). Gabapentin is the first-line therapy, reducing both tremor/postural instability and improving quality of life. 35 Clonazepam alone or in combination with gabapentine or primidone may decrease unsteadiness. 36 Levodopa can also provide symptomatic relief in primary OT. 37
Cerebellar diseases
Some ataxic patients with cerebellar cortical atrophy exhibit a benefit from gabapentin at high doses. Clonazepam may improve tremor, although the improvement is often mild. Most patients with cerebellar degeneration do not report improvement in activities of daily life. Tremor associated with ataxia due to vitamin E deficiency (AVED) responds to supplementation.38,39 The effects of ondansetron (a 5HT3-antagonist) on cerebellar dysfunction has been studied. The drug provides no benefit or only a minor reduction of cerebellar tremor.40–42 The kinetic tremor associated with Wilson's disease responds to zinc and chelators such as trientine. Although neuroprotective agents such as NMDA antagonists might slow down the progression of the neurodegenerative process, their tremorolytic activity remains undefined.
Non-pharmacological treatments
When drugs fail or the clinical response is partial, patients may benefit from non-pharmacological procedures such as lesioning surgery (thalamotomy, pallidotomy, subthalamotomy), gamma-knife (γK) and deep brain stimulation (DBS).
DBS
The main targets of DBS are thalamic ventralis intermedius nucleus (Vim), subthalamic nucleus (STN) and pallidum. The optimal stimulation site is the zone located just below the Vim nucleus. 43 Recent findings support the hypothesis that STN-DBS acts by direct interference with the neurotransmission of basal ganglia networks involved in tremor. 44 Nowadays, DBS has replaced lesioning techniques in most cases. Advantages of DBS are reported in Table 4. DBS procedures replicate the effects of ablative interventions, but do not require a destructive brain lesion. 45 The procedure is indicated in patients with severe resting tremor, unresponsive to conventional medical treatment or with motor complications. DBS is an effective surgical treatment for advanced Parkinson's disease (PD), with significant advantages in terms of morbidity-mortality and quality of life when compared to lesion techniques such as thalamotomy and/or pallidotomy. 46 DBS improves social life. 43 Bilateral STN stimulation is beneficial in the long-term for PD patients, as demonstrated by a 5 years’ follow-up study in 23 patients with advanced PD. 47 Vim DBS modifies multiple neurophysiologic features of ET (amplitude, frequency, regularity, and tremor–EMG coherence). 48 Deep brain stimulation of the VIM is an efficient and safe treatment for ET. Long-term follow-up demonstrates stability in tremor, as well as handwriting improvements. 49 Vim DBS is now a relatively common procedure to treat disabling intention tremor in multiple sclerosis. It remains unclear whether DBS in multiple sclerosis tremor is superior to thalamotomy. Whether DBS improves the quality of life and the activities of daily living is not defined, but some patients report an improvement. 50 Interestingly, postmortem imaging and pathologic findings in a patient with multiple sclerosis who underwent thalamic DBS have revealed focal areas of demyelination within the thalamus, particularly in the region corresponding to the electrical field. These observations suggest that focal demyelination may be induced by the electrical field. This could account for the long-lasting alleviation of tremor in the absence of continued electro-stimulation. 51 A beneficial effect of thalamic DBS for postural/action neuropathic tremor has been described in a patient with severe demyelinating neuropathy. 48 This interesting observation requires confirmation by a large study. Orthostatic tremor 49 and cervical dystonic tremor 54 may also respond to DBS.
Adapted from Grimaldi/Manto–-Tremor: from pathogenes is to treatment. Morgan and Claypool Publishers, 2008.
The activation of adenosine A1 receptors has been proposed recently as a new pharmacological target to replace DBS or improve its efficacy. 55 Indeed, DBS is associated with a marked increase in the release of ATP, resulting in the accumulation of its catabolic product adenosine. Adenosine A1 receptor activation depresses the excitatory transmission in the thalamus and reduces both tremor and DBS-related side effects. Intrathalamic infusion of A1 receptor agonists directly reduces tremor. 55 DBS has proven successful in a number of cases of severe generalized dystonia.
Lesioning surgery
Lesioning therapy (subthalamotomy, pallidotomy or thalamotomy) has also several advantages, including a reduced need for specialized care and close clinical follow-up, improved affordability, and a low infection risk. 59 In ET, thalamotomy should be considered when DBS fails. 60
Gamma-Knife radiosurgery
This technique represents a minimally invasive alternative in the treatment of PD and ET, when patients are not candidates for thalamotomy or thalamic DBS. 61 Long-term follow-up of a group of 158 patients (diagnosis: PD, ET, tremor due to stroke, encephalitis, or cerebral trauma) who underwent MRI-guided radio-surgical Vim thalamotomy, indicates that relief of tremor is well maintained free from radiation-induced complications. 62
Repetitive transcranial magnetic stimulation (rTMs)
rTMS modulates the corticospinal excitability and the effects last beyond the duration of the trains of rTMS. 63 Although it is not entered yet in the field of therapeutics for tremor, motor cortical stimulation has been proposed as a possible therapeutic target for the treatment of action tremor. 64
New Approaches in the Therapy of Tremor
Currents treatments are not effective in approximately 25% of patients. Researchers are continuously looking for new techniques. Promising developments are evolving in the field of biotechnology, wearable sensors and brain-computer interfaces (BCIs).
Orthosis and prosthesis
Inertial loading results in a reduction of postural tremor in ET subjects as confirmed recently. 65 Mechanical loading (by means of orthotic exoskeletons) represents a new method for tremor suppression.2,66 The principle is that a mechanical damper produces an opposing force proportional to velocity. As a result, damping loads attenuate the tremulous component of movement. A significant tremor reduction has been demonstrated in patients with post-traumatic and Multiple Sclerosis cerebellar action tremor. 67 A wearable orthosis applying effective dynamic forces between consecutive segments of the upper limb and changing its biomechanical characteristics has been proposed as a new treatment for ET. 2 The wearable orthosis is able to detect position, rate and acceleration of joint rotation by means of a chip gyroscope. The system makes a real-time estimation of tremor and consequently selects a cut-off frequency below the frequency of tremor. Evaluation of this orthosis in patients suffering from ET reveals that this technique is a non invasive alternative to medication and DBS. 2 Evaluation of a patient presenting a disabling ET resulted in a drastic decrease of tremor power. 68 However, the approach of mechanical suppression of tremor by means of orthotic devices presents limitations mainly due to the physical interaction between the exoskeleton and the human limb.68,69 The transmission of forces through soft tissues plays an important role in the efficiency of tremor suppression. There is a physical limitation for tremor suppression through wearable devices due to force generation (size and power consumption of the actuators) and transmission through soft tissues. Currently, there is no actuator technology fulfilling the cosmetic and aesthetic requirements of the patients (low weight, hiding the device under the clothes), as well as for functional requirements (torque, bandwidth). Bulky exoskeletons cannot be envisioned as a solution because of the risk of social exclusion.
Brain-machine interfaces and assistive therapy
Brain-computer interfaces (BCIs) enable users to control devices using electroencephalographic (EEG) signals from the scalp or single-neuron activity from inside the brain. These signals include slow cortical potentials, P300 potentials, mu/beta rhythms recorded from the scalp, and cortical neuronal activity recorded by implanted electrodes. 70 BCIs aim is to control limbs as smoothly as possible. The electrical activity recorded from the brain cortex is translated to command signals driving prosthetic limbs and computer displays. By adding somatosensory feedback to this control, more complex behaviors can be controlled. 71
BCIs could provide an alternative communication and control technology in the daily life of people severely disabled by degenerative disorders of the brain such as ALS (Amyotrophic Lateral Sclerosis). 72 Invasive and noninvasive BCIs show also a potential for communication in locked-in syndrome and movement restoration in chronic stroke. 73
Cincotti and colleagues have proposed a prototype which integrates several assistive technologies including a BCI system. 74 An assistive technology system (“the Tremor Control System”), consisting of a motion-filtering software program that supports multiple interfaces, has shown significant improvements in performance on the PC-tests when compared to a standard PC-mouse. Patients with marked intention tremor seem to benefit from this assistive technology. 75
Fuzzy logics
Fuzzy logics is an emerging field for tremor research and therapy. The purpose is the creation of a predictor for the dynamic characteristics of the tremor. 76 The benefits of the fuzzy fused tremor parameters rely on more complete and accurate assessment of the functional impairment and on improved feedback for rehabilitation, based on the fused parameters of the tremor. Such movement prediction is strictly required during the control of tremor by electrical stimulation. 76 The benefits of the neuro-fuzzy predictor are a good prediction capability, versatility, and apparently a high robustness to individual variations of the tremor. 76 A dynamic recurrent neuro-fuzzy system has been proposed to provide context-awareness for wearable intelligent agents/assistants that can be applied in any field of rehabilitation. 77
Surface Functional Electrical Stimulation (sFES)
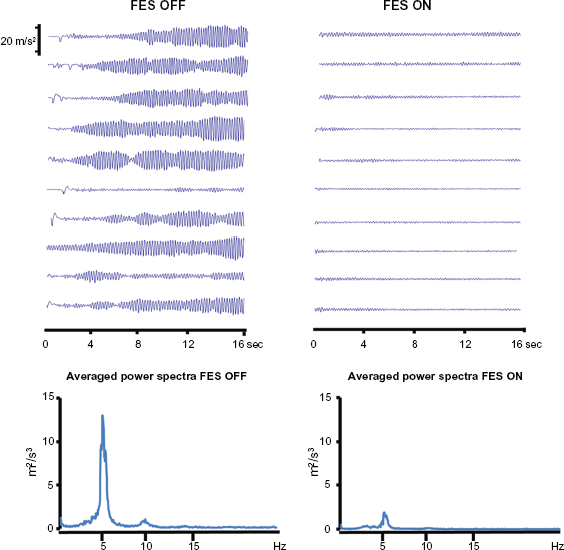
Surface stimulators for muscle exercise are commonly used in rehabilitation clinics and homes. The group of Prochazka has demonstrated the possibility of suppressing pathological tremors using a closed-loop functional electrical stimulation (FES) to activate the tremorogenic muscles out-of-phase. 78 The evaluation of the FES technique for tremor suppression in patients affected with ET, parkinsonian tremor, and cerebellar tremor associated with multiple sclerosis, revealed a significant attenuation of tremor. 79 The system is designed to attenuate movements in the tremor frequency range, without significantly affecting slower, voluntary movements. Recently, a group of researchers from the Nanyang Technological University, Singapore proposed a theoretical framework for a FES control system aiming to suppress forearm tremor. Accelerometer and EMG signals are extracted by a two-stage filtering algorithm, which separates intended motion from tremor. The signal is then used as a control input in the FES controller to stimulate reciprocally the biceps and triceps muscles 80 and to actuate the antagonist of the trembling muscle in anti-phase with respect to the tremor signal. 81 A European project is currently evaluating the feasibility of combining FES with a BCI (http://www.iai.csic.es/tremor). Preliminary results confirm that FES can reduce action tremor in ET (Fig. 1).
Conclusion
Numerous developments are occurring in the field of therapies of tremor. They extend beyond the traditional methods based on medications and surgery. However, detailed studies are required to validate these emerging tools as effective alternative to conventional therapies. While new treatments are emerging, our understanding of the pathogenesis of tremor and the genesis of oscillatory processes in the nervous system is still growing quickly. This knowledge will impact on the development of future therapies.16,82 Combination of so-called “old” and new therapies are likely to occur in the next decade.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
Footnotes
Acknowledgments
G. Grimaldi is supported by the EC (grant ICT-2007-224051). M. Manto is supported by the FNRS Belgium. We gratefully acknowledge the support of the members of the Tremor consortium, especially Aalborg University, UNA, CSIC-IAI and Technaid. We thank Alison Headley for manuscript editing.

