Abstract
Dysequilibrium in calcium and phosphate metabolism with development of secondary hyperparathyroidism (SHPT) is common in patients with chronic kidney disease (CKD) stage III and IV. Dietary phosphate restrictions and calcium based oral phosphate binders have not been effective in all subjects with SHPT, and soft tissue and vascular calcifications with an increased risk of cardiovascular death related are known consequences. Treatment with the calcimimetic Cinacalcet (Cc) has contributed to a better calcium and phosphate control in patients given hemodialysis treatment. In this retrospective study we present our experience with Cc given to ten (one year) or five (two years) patients with CKD stage III and IV and SHPT not suitable for surgery. With conventional therapy target levels of intact parathyroid hormon (iPTH) are seldomly reached the reason why an iPTH value < 300 ng/l was considered acceptable. Levels of iPTH decreased significantly after 3 months of Cc treatment and remained at the lower level. Plasma ionized-Ca (Ca) concentrations decreased initially but remained above 1.00 mmol/l in all but one patient. Phophate (P) levels increased to 1.41 ± 0.09 mmol/l (mean ± SE) leaving the Ca × P product unchanged. While patients with high iPTH needed high Cc doses up to 90 mg/day, some of the patients required very low doses 4.5-20 mg/day in order to achieve a decrease in iPTH levels. Only one patient reported gastric pain needing dose reduction and other adverse effects were not found. No changes in QT-time were observed. We experienced that Cc treatment was promising to control SHPT and stabilized the Ca-P balance in patients with CKD stage III and IV. Dosing may be challenging and laboratory values should be controlled often (monthly) as these patients may have variable response to Cc treatment. Due to the minimal knowledge about its effect on morbidity and mortality in the predialytic population further controlled studies are needed to confirm its efficacy and safety.
Introduction
Disturbances in calcium (Ca) and phosphate (P) metabolism is accompanied with significant secondary hyperparathyroidism (SHPT) in patients with chronic kidney disease (CKD) in the stage III to IV, 1 and have been linked to tissue and cardiovascular (CV) calcifications with increased mortality.2,3 Increased levels of intact parathyroid hormone (iPTH) correlated significantly with CV death 4 and CV mortality proportionally with the elevation of iPTH levels. 5 In spite of restriction of dietary P intake and treatment with oral P-binders which may provide some help in the predialytic population many patients continue to present with elevated iPTH. Elevated P levels and decreased serum Ca levels together with vitamin-D deficiency is often present.
In the presence of low serum Ca and acceptable P levels oral treatment with calcitriol, alphacalcidol, or paricalcitol could be used to lower iPTH levels and eventually prevent renal osteodystrophy. Unfortunately, treatment of SHPT with vitamin-D analogues may cause hyperphosphataemia and hypercalcaemia leading to soft-tissue and vascular calcifications.
According to the K/DOQI guidelines for bone metabolism and disease in chronic kidney disease 6 target levels for iPTH should be within 35-70 pg/ml in the CKD stage III and below 110 pg/ml in the stage IV. In spite of huge efforts, these targets seem difficult to achieve in many patients with CKD in stage III and most patients in stage IV.7–9
Reports on the use of Cc in patients with CKD in predialytic stage III and IV were recently published.10,11 Encouraged by the first report from Chatyran et al 11 we used Cc in selected subjects with SHPT which over a long period of time were unable to reach the targets given in the K/DOQI guidelines. They were either resistant to or had only weak response to the previous therapy with phosphate restriction in the diet and use of calcium-based phosphate binders and vitamin-D analogues. They had either refused parathyroid surgery (PTx) or PTx was considered to risky from a clinical point of view. We report on our experience with Cc given to 10 patients with SHPT due to CKD in the predialytic stage III or IV.
Calcimimetics
Calcimimetics (CMs) are a new class of compounds developed to control the release of PTH by modulation of the calcium sensing receptor (CasR). The CasR belongs to the G protein-coupled receptor superfamily cloned from bovine and human parathyroid cells. 12 CasR is also abundant in the kidney. 13 Activation of the CasR results in an increased mobilization of intracellular Ca (for review see ref 14 ). Type-I CMs are inorganic cations, polyamines or aminoglycosides (i.e. mg2+, Gd3+, neomycin, spermine) that bind to the extracellular part, the Ca-binding site of the CasR, in the absence of extracellular calcium.14,15 Type-II CMs are compounds that bind to regions within the membrane-spanning domain and differently from type-I CMs they are dependent on extracellular calcium. 16 NPS-568 was the first type-II CM to be clinically tested 14 and Cinacalcet (Cc) was the first compound to be clinically used to treat dialysis patients with SHPT. Cc decreased iPTH levels and the Ca × P-product independently of concomitant additional vitamin-D therapy 17 and also reduced the need for parathyroid surgery (PTx). Further, Cc reduced the amount of fractures and the rate of CV hospitalisation in dialysis patients. 18 The two trials the CONTROL study 19 and the OPTIMA study 20 evaluated the effects of Cc with the goal to reduce the dose of vitamin D 19 and to reach the target for iPTH 20 in patients with end-stage renal failure (ESRF).
Methods and Subjects
This retrospective clinical observational study was approved by the ethical committee of the Central Finland Health Care District Hospital of Jyväskylä, Finland, and lasted from January 2006 to the end of September 2008. Following careful and thorough information on development of SHPT, PTx surgery, and the use of Cc in this setting an oral informed consent was obtained from all paients.
Three female and seven male patients, aged 53-88 years (mean age 74 years), with CRF in stage III and IV and with SHPT resistant to previous treatment were given Cc. Previous treatment consisted of P restriction in the diet, oral Ca-based phosphate binders, vitamin D-analogues, or a combination of these compounds. In spite of this treatment all patients had developed SHPT with serum iPTH levels far above the recommendation for their degree of CRF (stage III and IV). All patients had either denied or were found to be at considerable risk for PTx and consequently none of them had undergone PTx prior to Cc treatment. Follow-up was carried out by a nephrologist at our department of nephrology.
The diagnoses leading to CKD were diabetes mellitus type II nephropathy (DMN) in 5, hypertension with nephrosclerosis (HNS) in 2, with combined DMN and HNS in four. One patient had congenital agenesis of the left kidney combined with an anatomic anomaly of the right renal artery which originated from the right iliac artery which simultaneously also had a significant artery stenosis. Another patient had a single kidney after a left-sided nephrectomy due to malignancy. The last patient had non-specific defined chronic glomerulonephritis (GN).
Target iPTH levels are seldomly reached with conventional therapy 6 the reason why it was not aimed for these targets and an iPTH value < 300 ng/l was considered acceptable. Our selected patients had a mean iPTH level of 634 ± 113.8 ng/l (ref. value < 60 ng/l; Table1). Plasmaionized calcium(ion-Ca)concentration was kept above 1.00 mmol/l and plasma P level below 1.50 mmol/l. Active questionnaire for possible side effects was recorded, and if any adverse effect was observed the Cc dose was either reduced or discontinued.
Some selected laboratory data found 1-2 months before and at start of Cc treatment.
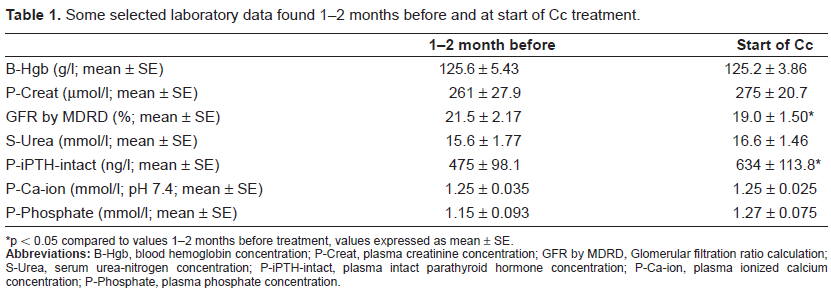
p < 0.05 compared to values 1-2 months before treatment, values expressed as mean ± SE.
Laboratory
Blood for laboratory tests was collected before, at start, and every month after initiation of Cc treatment. Laboratory examinations were performed using our hospital routine test methods. Blood samples were collected between 7 to 10 o'clock at mornings and most of the analyses were performed the same day. Samples for iPTH were stored at -20 °C and analyzed weekly. Urine samples were not collected. Some selected laboratory results one or two months prior to and at initiation of Cc treatment are presented in Table 1. Glomerular filtration rate (GFR) was estimated using the Modification of Diet in Renal Disease (MDRD) calculation. 21 Specific markers of bone turnover were not investigated. Two patients had got a Pace-maker several years earlier, and electrocardiograms (EKG) had been taken previously in all subjects.
Treatment Doses
Cinacalcet
The Cc tablets, administered from our hospital, were adviced to be taken with the main meal between 14 and 16 o'clock. The initial daily dose was 30 mg increasing to 60 mg, or 90 mg daily (the highest dose used) after 1-3 months depending on the clinical evaluation, plasma ion-Ca, P, and iPTH levels. Patients with a tendency of hypocalcemia togther with low iPTH level (<50 ng/l) had the Cc dose reduced by 1/3 of the previous given dose. Patients with low ion-Ca concentration (ion-Ca < 1.00 mmol/l) and low iPTH levels (<80 ng/l) had Cc suspended during two months. These subjects were given increased doses of oral calcium carbonate and vitamin D-analogues. Discontinuation of Cc resulted in recurrence of high iPTH levels (>280 ng/l) within the next two months and Cc was reintroduced with a dose half of that given before suspended.
Vitamin D-analogues
All patients had vitamin D-analogue treatment. Initially alfacalcidol (AC) was mostly used. The mean daily AC dose was initially 0.25 μg in 7 patients, 0.50 μg in two, and one had 1.0 μg daily. Patients with decreasing ion-Ca concentration below 1.00 mmol/l and acceptable P values were given a higher dose of AC or were converted to paricalcitol (PC) which was the case in 4 patients. In these 4 patients PC treatment (1 μg daily dose) was judged by the nephrologist to be a better alternative. At the end of one year of observation the AC dose was 0.25 μg/day in 3 subjects, 0.5 μg/day in 3, and 4 patients were given PC 1 μg/day.
Calcium carbonate
Calcium carbonate was used as an oral phosphate binder in all subjects and was given in variable doses throughout the year. At the end of the observaion the dose was 0.5 g/day in 3 subjects, 1.0 g/day in 4, and 1.5 g daily in 3 patients.
Additional Medicaments
Systolic and diastolic blood pressure was in control with anti-hypertensive treatment using beta-blockade (7/10), angiotensin II receptor blockade (6/10), calcium channel blocker (5/10), imidazole receptor agonist (4/10), and 9 out of 10 patients had addition of a diuretic. Four patients were treated with a combination of 5 different antihypertensive compounds, 1 patient had three different drugs, 4 others had 2 compounds, and 1 patient had only one antihypertensive drug. Eight patients had cholesterol lowering medicaments (CoA-reductase enzyme inhibitors).
Statistical Analyses
All analyses for statistical evaluations were performed using SPSS software (SigmaPlot 8.0, 2002, software for Windows; SPSS Inc., Chicago IL). All values were compared to values before initiating Cc treatment. The Student's t-test was used for paired analyses, and a p < 0.05 was considered statistically significant. All data and laboratory results are expressed as mean ± standard error (mean ± SE) if not otherwise indicated.
Results
Clinical course and side effects
Two of the 10 patients developed end-stage renal failure and had to start hemodialysis treatment after 9 and 12 months of observation, respectively. Two other patients died from reasons not related to treatment after 12 and 14 months, respectively of which one patient with diabetic nephropathy died from acute myocardial infarction and another, also with diabetic nephropathy, died from septicemia caused by staphylococcus aureus from a cutaneous infection. A third patient had been observed during 21 months only. Due to the small number of patients that continued Cc treatment longer than 12 months the statistical evaluation beyond 12 months of observation was considered of no value.
Minimal amounts of untoward effects due to Cc treatment were recorded during the two years of observation and only one patient had to reduce the dose due to gastric pain after taking Cc. Following reduction of the Cc dose this untoward effect did disappear. Hypocalcemia (ion-Ca < 1.00 mmol/l) and low iPTH levels (< 80 ng/l) was found temporarily in two patients which was corrected with an increased dose of calcium carbonate and vitamin-D analogue (AC). Later, these two patients had recurrence of elevated iPTH levels with need for reintroduction of Cc.
Kidney function
Compared to values at start a significant decrease in GFR, expressed as MDRD, was observed after 9 months (p < 0.01) which continued up to 12 months (p < 0.001) after initiating Cc treatment. Simultaneously an increase in plasma creatinine (p-creat) concentration was found after 9 months of observation (p < 0.005 compared to start) which also increased further at 12 months (p < 0.002). Thereafter the kidney function remained almost stable (Fig. 1). Likewise, urea-nitrogen concentration (s-urea) increased after 9 months (p < 0.005) and 12 months (p < 0.01) of observation. Urea levels returned to those previous values found before Cc treatment after 15 months (Fig. 1).
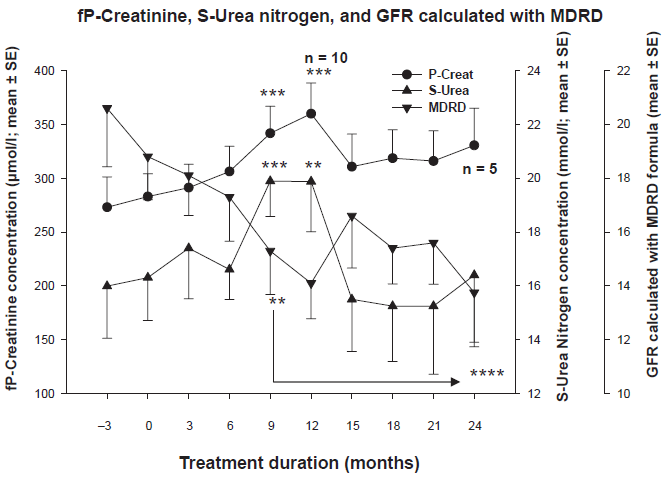
A significant reduction of glomerular filtration rate (GFR) calculated with the MDRD formula together with an increase in plasma Creatinine concentration with no change in plasma Urea-nitrogen was observed during the cinacalcet treatment period. All values expressed as mean ± SE. (*p < 0.05; **p < 0.01; ***p < 0.005; ****p < 0.001) were compared to start of Cc treatment.
iPTH levels
The initial iPTH concentrations prior to Cc treatment were high and as expected with a wide span of variation. After three months of Cc treatment the iPTH level fell by 47.1% which was statistically significant (p < 0.003) compared to start of Cc treatment (Fig. 2). Thereafter iPTH levels remained about 45 to 50% and at the most 59.6% (p < 0.001) lower than pre-treatment levels (Fig. 2). Cc treatment was temporarly discontinued in two subjects due to low iPTH values (<80 ng/l). Later Cc was reintroduced in both at a lower dose. This unexpected response on iPTH levels was observed within 6 to 9 months of Cc treatment at a dose of 30 mg/day. Their initial iPTH concentration was 530 ng/l and 573 ng/l decreasing to 34 ng/l and 67 ng/l, respectively. Following discontinuation of Cc treatment their iPTH levels increased to 288 ng/l and 359 ng/l, respectively and then remained between 100-170 ng/l in both after reintroduction of Cc. The discontinuation of Cc in these above mentioned two subjects resulted in a temporarily increase in mean iPTH levels which again returned to lower levels after reintroduction of cinacalcet (Fig. 2).
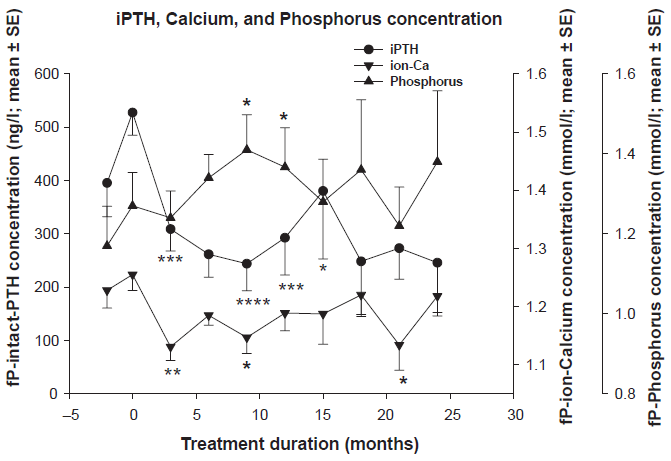
Significant reduction in intact-parathyroid hormone (iPTH) and a slight decrease in plasma calcium (fP-ion-Ca) concentration while no change in plasma phosphorous concentration was observed during cinacalcet treatment. All values (expressed as mean ± SE (*p < 0.05; **p < 0.01; ***p < 0.005; ****p < 0.001) were compared to start of Cc treatment.
Calcium and phosphate
The plasma ionized calcium (ion-Ca) concentration decreased significantly after 3 months of Cc treatment (p < 0.01). Calcium-supply and increaased dose of vitamin-D analogue slowly corrected the ion-Ca concentration. In spite of initial low ion-Ca levels normalization was achieved after 9 months and after 12 months of Cc treatment no difference compared to start could be found. The ion-Ca level remained stable during the rest-observation period (Fig. 2). Plasma P concentration (pP) was significantly increased to 1.41 ± 0.09 mmol/l (mean ± SE) at 12 months (p < 0.05) but remained within normal range later in the study (Fig. 2). The Ca × P product remained unchanged during the whole Cc treatment period.
Other tests
Serum albumin, blood haemoglobin, plasma sodium, potassium, liver, alkaline phosphatase and pancreatic enzyme concentration together with all other laboratory tests remained unchanged during the Cc treatment period. Comparison of EKG registrations taken before and during Cc treatment did not unveil any increase in QT-times. Two patients had a Pace-maker fitted many years before introduction of Cc treatment.
Cinacalcet doses
A large variation was observed with regard to the Cc doses needed to achieve a clinical response and a decrease in iPTH levels. The impression remained that high doses, 90 mg daily, were needed in the subjects with very high basic iPTH concentrations (iPTH > 300 ng/L). Oppositely some of the patients with low initial iPTH needed only small doses (30 mg 1-3 times weekly) and titrating of Cc was difficult in these patients needing low doses. The most common given Cc dose was 30 to 60 mg/day (6 patients), 2 patients required 90 mg/day, 1 patient had 17.15 mg/day, and one patient needed only 4.3 mg daily (the lowest dose).
Discussion
The development of the new calcimimetic compound Cc has dramatically changed the perspectives of treating CKD related secondary hyperparathyroidism. Beside its use in the treatment of SHPT in dialysis patients 22 Cc was also effective to treat persistent SHPT with hypercalcemia after kidney transplantation, 23 primary hyperparathyroidism, 24 and might eventually be used as first-line treatment for calciphylaxis. 25 Further, in spite of high iPTH levels Cc was successfully used to treat parathyroid carcinoma related hypercalcemia after parathyroidectomy. 26
This simple retrospective clinical observational study demonstrated that Cc could be safely used to treat patients with uncontrolled SHPT (judged by high levels of iPTH) in CKD stage III and IV. The iPTH target of the treated subjects in this study was set somewhat higher (<300 ng/l) than that of the recommendations given in the K/DOQI guidelines. Whether Cc should be used to treat SHPT in non-dialysed patients with CRF in stage III and IV is a matter of debate and less obvious than that for dialysis patients,27–29 however. The experience achieved in this study with Cc is in conformity with previous reports with regard to iPTH, calcium and phosphate levels.10,11 Calcium × phosphate product remained stable and measurable iPTH levels decreased significantly during Cc treatment. As plasma calcium concentration tended to fall with Cc treatment, additional calcium carbonate and vitamin D analogues could be used more frankly resulting in an even better control of iPTH levels. All this results were achieved without an increase in plasma phosphate levels. Measurements of urinary calcium, phosphate, and specific markers for bone turnover are recommended in patients given Cc, unfortunately not performed in this retrospective study.
Like observed by others, 7 our patients had developed SHPT at an early stage of CKD and in spite of dietary phosphate restrictions the iPTH level was much higher than that recommended for patients with CRF in the predialytic stage III and IV. Our patients had a GFR ranging from 12 to 26 ml/min/1.73 m2 and the iPTH level varied from 291 to 731 ng/l which was far above the values recommended in the guidelines. 6 Moreover, all subjects had been resistant to previous medication consisting of calcium-based phosphate binders and vitamin D-analogues. As none of the patients agreed to parathyroidectomy no other treatment could be offered it was considered that these subjects required treatment with a calcimimetic. We found that like the two previous reports on early CM intervention10,11 that Cc treatment effectively decreased the level of iPTH in all subjects with few and acceptable side effects.
It was not an option to reach iPTH levels given by the guidelines 6 in this study, and with a target of iPTH concentration of <300 ng/l, this was almost always reached in all the Cc treated subjects. Only few patients reach targets of the K/DOQI guidelines even using combination of several compounds without addition of Cc, and even with a calcimimetic such targets will be difficult to reach. Only 2 patients had a higher phosphate level than that recommended (<1.49 mmol/l) for patients in CKD stage III. Cinacalcet-induced “hungry bone” syndrome 30 was not observed.
All parameters remained stable after 12 months of Cc treatment. This was caused by the death of two subjects with ESRD and the transfer of 3 subjects with previously very bad kidney function to dialysis treatment after 12 months of Cc treatment. The deaths occurring during the observation (see results) were not related to Cc treatment and plasma ionized calcium concentrations were never low in these two subjects. Moreover, other abnormalities than those related to acute myocardial infarction (case 1) and septicemia (case 2) were not found. Progression of the CKD to end-stage disease and need for renal replacement treatment occurred in three subjects, which could be expected in this patient population, and was considered not related to Cc treatment.
Conclusions
Experiences using Cc in stage III and IV grade of CRF have been very sparse. In our cases we found that Cc was effective in lowering iPTH levels. Dosing of Cc should be carefully followed since low iPTH levels may develop, as shown here, and some patients may require only very small doses to achieve response with regard to iPTH levels. Cessation of Cc will increase iPTH levels almost to pretreatment levels the reason why we felt that the Cc treatment dose should be reduced stepwise and not be discontinued in the case of low iPTH levels. Cc might be added when iPTH levels are increased in spite of treatment with calcium-based phosphate binders and vitamin-D analogues and parathyroid surgery is regarded to risky. Patients with SHPT due to CKD grade III and IV, which for different reasons are not suitable for parathyroidectomy, should be offered treatment with the Cc, thus with regular control of plasma iPTH, phosphate, calcium and urinary calcium excretion.
Disclosure
The authors report no conflicts of interest.
