Abstract
Etravirine (ETR) is a novel non-nucleoside reverse transcriptase inhibitor (NNRTI) designed for binding to HIV reverse transcriptase in spite of most of the mutations which normally confer resistance to the other drugs of the same class. Evidences from clinical trials and post-marketing drug use provide guidance for the use of the drug in NNRTI-experienced patients. However, despite the raised genetic barrier, progressive loss of ETR susceptibility is seen as resistance mutations accumulate and a certain degree of cross-resistance with first-generation NNRTI occurs however. Moreover, most of the experiences in using ETR come from the setting of co-administration with darunavir/ritonavir, whilst little is known about its use with other companions. A full knowledge of all available evidences on the drug is therefore needed, in order to optimize the use of ETR in clinical practice.
Keywords
Introduction
Etravirine (ETR), formerly known as TMC-125, is a novel non-nucleoside reverse transcriptase inhibitor (NNRTI) developed by Tibotec (Janssen-Cilag) for the treatment of HIV-1 infection. It has been designed with a conformational “flexibility” that allows for binding to HIV reverse transcriptase in spite of most of the mutations which normally confer resistance to the other drugs of the same class. The expanded activity spectrum of this new drug may open up the possibility of consecutive NNRTI treatment. However, the optimal use of ETR in NNRTI-experienced patients still needs to be defined, as evidences have started to emerge from clinical trials and post-marketing drug use.
Methods
The present article is based on a comprehensive review of published studies on ETR. Literature searches of the PubMed/MEDLINE databases were undertaken to identify pertinent articles. The major search terms included: Etravirine, TMC-125, TMC125, TMC 125, ETV, ETR and R165335. Searches were limited to English language. In addition, websites of the major international congresses on HIV were browsed in order to identify relevant unpublished studies.
Pharmacology and Mode of Action
Etravirine is a di-aryl-pyrimidine derivative that was identified by using a screening strategy which involved several NNRTI-resistant strains in the study of structure-activity relationship, and an assessment of metabolic stability early in the discovery process, thereby establishing a structure-metabolism relationship. 1 Like other NNRTIs, it is a non-competitive inihibitor that binds allosterically HIV-1 reverse transcriptase in a lipophilic pocket of the p66 palm domain, about 10 å away from the catalytic site. The polyciclic molecule of ETR consists of three aromatic ring structure with single bonds between the rings (Fig. 1). This provides torsional flexibility, which allows the inhibitor, through accomodation by small conformational adjustments, to bind reverse transcriptase with substitutions that change the shape or electrostatic conditions of the binding site and confer resistance to the other currently available NNRTI. Etravirine can adapt to changes in the NNRTI-binding pocket in several ways: it can bind in at least two conformationally distinct modes; within a given binding mode, torsional flexibility (“wiggling”) permits access to numerous conformational variants; the compact design of ETR permits significant repositioning and reorientation (translation and rotation) within the pocket (“jiggling”). 2 Switching between different conformations, accomodating aminoacid changes within the binding pocket, does not involve a significant energetic cost. 3
Etravirine molecular structure.
As a consequence of ETR binding, reverse transcriptase structure is altered, thus leading to a reduction in catalytic enzyme efficiency. The inihibition of ETR, like other NNRTI, is specific for HIV-1, because structural differences in the lipophilic pocket of HIV-2 reverse transcriptase confer inherent drug resistance.
Pharmacokinetics, Metabolism and Drug Interactions
Etravirine is currently available as 100 mg tablets in a solid dispersion formulation, which significantly improved oral bioavability and reduced inter-patient absorption variability, as compared with the former granulo-layered tablet formulation used in Phase II studies. 4
When administered at the registered dosage of 200 mg twice daily in HIV-1 infected volunteers, mean Cmin, Cmax and AUC0-12h were 184.7, 451.3 and 3,713 ng/ml, respectively. The median tmax was 4 hours after dosing (ranging from 2 to 8), 5 whilst the mean (± SD) drug half-life was approximately 40 (± 20) hours. 6 The prolonged half-life of ETR permits twice-daily dosing with small fluctations in plasma levels in the dosing interval.
When administered 400 mg once daily in healthy volunteers, the daily sistemic exposure to ETR was the same as that obtained with twice-daily dosing, although Cmin was 25% lower and Cmax 40% higher than in twice-daily administration. 7 Whether this could permit the use of ETR once-daily is unclear, since data on efficacy and safety are lacking.
The oral bioavailability of ETR is significantly influenced by food intake and the drug should always be administered after a meal. In a study involving 12 healthy volunteers, administration of ETR in a fasted state resulted in 51% lower mean exposure compared with dosing after a standard breakfast. 8 The drug has low solubility and over 99% is protein bound.
Etravirine is metabolized by cytochrome P450 (CYP) isozymes 3A, 2C9 and 2C19, with subsequent glucuronidation of the metabolites. M8 and M12 have been shown to be the most abundant metabolites in plasma, whilst more than 80% of ETR is excreted unchanged in the faeces with minimal (<1.2%) renal excretion. 6 No dose adjustments are necessary in patients with renal or mild to moderate hepatic dysfunction. 9
Currently available data suggest that ETR is an inducer of CYP3A and a weak inhibitor of CYP2C9, CYP2C19 and P-glycoprotein.10–11
Due to its effect on this important metabolic pathway, pharmacokinetic interactions between ETR and other co-administered medications have been explored.
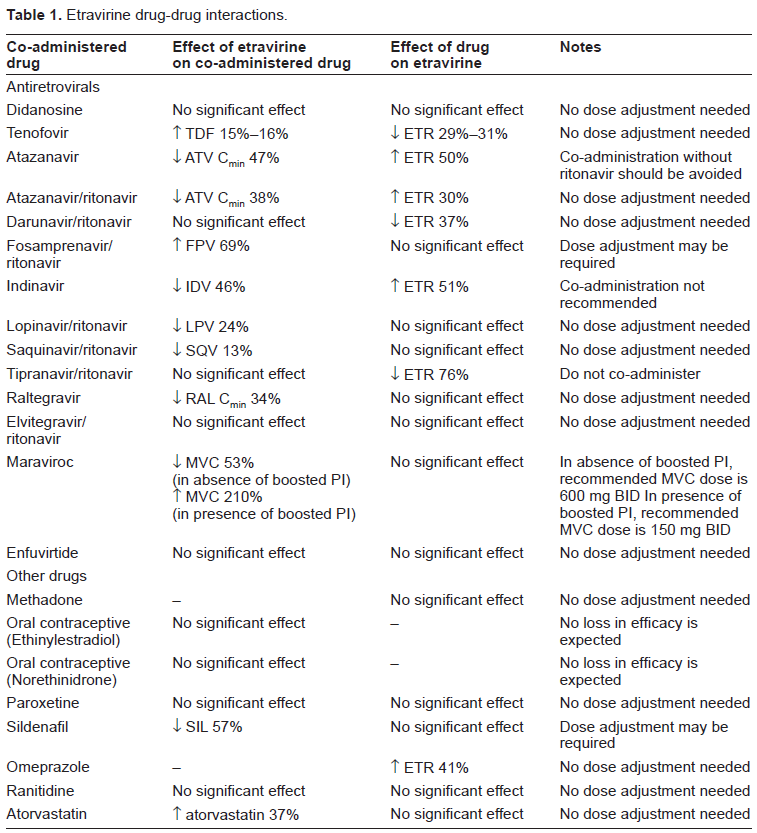
Etravirine can reduce protease inhibitors (PI) concentrations. However, when protease inhibitors are “boosted” with low-dose ritonavir, no dose adjustment is needed. Fosamprenavir makes exception, because its plasma concentration is increased by 69% when co-administered with ETR and dose adjustment may be needed. Boosted PI may, in their turn, affect ETR plasma concentration. These interactions are generally considered not to be clinically significant, with the exception of tipranavir/ritonavir, whose co-administration with ETR is contraindicated.10,12 Etravirine has been demonstrated to reduce maraviroc concentration by roughly 50%, so that dose increase of maraviroc to 600 mg twice-daily is indicated in the absence of boosted PI. These and other drug-drug interactions with medications commonly used in HIV-positive patients are summarized in Table 1.10,12–22
Etravirine drug-drug interactions.
Etravirine Efficacy Studies
The efficacy of ETR as an antiretroviral agent was first demonstrated in vivo in the TMC125-C208 study. 23 Among 12 treatment-naïve subjects, treated for 1 week with ETR monotherapy, the median virion clearance rate constant and the median decline in HIV-1 RNA were 0.68 per day and 1.92 log10 copies/ml, respectively. This dramatic viral load drop was similar to that observed in 11 naïve subjects, treated with a five-drug, triple-class antiretroviral regimen. 24
The first demonstration of ETR activity against NNRTI-resistant virus came from a proof-of-concept study, conducted by Gazzard and colleagues. 25 In this open-label phase IIa study, 16 males infected with HIV-1 with documented phenotypic resistance to NNRTI and at least 1 NNRTI-associated mutation were switched from a first-generation NNRTI to ETR for 7 days and maintained on the nucleoside back-bone unchanged. The HIV-1 viral load decay was 0.13 log10 copies/ml per day and a median decrease of 0.89 log10 RNA copies/ml (range, −1.71 to −0.18 log10 copies/ml) was documented at day 8. Seven out of 16 patients (44%) obtained a viral load decrease of >1 log10 in 1 week.
Two large dose-finding phase IIb studies were conducted. TMC125-C223 study was a multicenter, open-label, partially blinded, randomized clinical trial aimed at evaluating the efficacy and safety of ETR in highly treatment-experienced patients infected with multi-drug resistant HIV-1, defined as a virus harbouring ≥ 1 NNRTI-associated mutation and ≥3 major PI mutations. 26 One hundred ninty-nine subjects were randomized 2:2:1 to receive optimized background regimen (OBR), including 2 NRTIs and/or lopinavir and/or enfuvirtide, plus ETR 400 mg twice daily, OBR plus etravirine 800 mg twice daily or standard of care OBR. The rate of discontinuation by 24 weeks was much greater in the control group (95%) than in ETR groups (26.3% and 24.1% for ETR 400 mg and ETR 800 mg, respectively). Among controls, 75% discontinued because of virological failure compared with 6.3% and 5.1% of the ETR 400 mg and 800 mg groups, respectively. The mean viral load change from baseline to week 24 was greater in etravirine groups (−1.04 and −1.18 log10 copies/ml) than in the control group (−0.19 log10 copies/ml), as well as the proportion of subjects with a viral load decrease of 1.0 log10 copies/ml (36.3% and 41.8% for etravirine and 7.5% for controls). The proportion of patients achieving undetectable viral load (<50 copies/ml) after 24 weeks of treatment was 21.3% and 17.7% in etravirine groups and 7.5% among controls. Undetectable viral load was maintained at week 48 by 88.2% and 87.5% of patients who had achieved an undetectable viral load at week 24. 27 Basing on results on surrogate markers, it was estimated, using independent prediction methods, that the inclusion of ETR in the salvage regimen had reduced the risk of progression to AIDS/death by 31%-39%. 28
Another randomized, placebo-controlled, phase IIb, clinical trial (TMC125-C203) failed to show a clear advantage of ETR over placebo in multi-experienced HIV-1-infected patients. 29 In this study, ETR was administered with an OBR at 3 twice-daily doses. In stage 1 of the trial, 166 patients received ETR (400 mg or 800 mg twice daily) or placebo. In stage 2 of the trial, 74 patients received ETR (800 mg or 1200 mg twice daily) or placebo. Notably, the study was limited by important drawbacks. Intergroup disparities in the OBR, originated by an amendment which allowed the use of more active drugs (particularly ritonavir-boosted lopinavir and enfuvirtide), had the potential to affect the efficacy results. Moreover, the authors excluded from the analysis 21%-23% of the results of the stage 1 of the study, because of inaccuracy in viral load assessments. In stage 1, no statistically significant difference in plasma viral load reduction after 24 and 48 weeks of treatment was noted between ETR and placebo group. An additional antiviral effect of ETR was noted only for patients who had either a virus with >2 primary PI mutations or were not receiving an active PI in the OBR, and only in the group who received ETR 800 mg twice daily. Conversely, during stage 2, the plasma viral load reduction at week 24 was statistically significantly greater in patients who received ETR, than in those who received placebo, and a trend towards superiority was shown at week 48. A statistically significant benefit in CD4 cell recovery was also observed during stage 2 with both etravirine doses, compared with placebo.
Initial enthusiasms on ETR were halted by the unexpected results of the TMC125-C227 trial, presented at the 8th International Conference on HIV Drug Therapy, held in Glasgow in 2006, and recently published.30–31 Etravirine was compared to a PI in patients who were failing a first generation NNRTI-based therapy. The trial was stopped by Data and Safety Monitoring Board because of significant failure rate in ETR group already evident at week 8. Many factors may account for this finding. First of all, the trial was conducted mainly in resource-poor countries, where the absence of routine viral load monitoring could have led to the accumulation of multiple resistance NRTI and NNRTI mutations. Secondly, in a post-hoc multivariate analysis of baseline resistance data for the ETR group, a reduced virological response was observed at week 12 in patients who had any of the following baseline characteristics: at least one detectable NNRTI resistance-associated mutation (RAM); the presence of Y181C mutation (in combination with other NNRTI RAMs) but not K103N; baseline ETR fold change in EC50 equal to or greater than the arbitrary threshold of 10; and a higher number of baseline NRTI mutations, thymidine analogue mutations or ETR RAMs. Therefore, the enrolment of PI-naïve patients with combined baseline NRTI and NNRTI resistance skewed the virological response in favour of the control PI treatment. Thus, this trial has elucidated the limitation of ETR treatment in combination with only two NRTIs, in the setting of high levels of NNRTI and NRTI resistance.
The benefit of ETR over placebo was clearly established only with two large phase III randomized placebo-controlled clinical trial. The DUET-1 32 and DUET-2 33 studies are two identical clinical trials (except for the geographical location in which they have been conducted), each involving around 600 subjects, in which treatment experienced patients with documented genotypic evidence of NNRTI resistance and at least 3 primary PI mutations were randomly assigned to receive etravirine 200 mg (solid dispersion new formulation, equivalent to 800 mg of the phase II trials) twice daily or placebo, plus an OBR which included ritonavir-boosted darunavir in all cases, 2 investigator-selected NRTIs, and, optionally, enfuvirtide. The proportion of patients with undetectable viral load at week 24 in ETR and placebo groups were 56% vs. 39% in DUET-1 and 62% vs. 44% in DUET-2 study, respectively. In both studies the advantage of ETR was more pronounced in the subgroup of patients with no active drugs in the background regimen, while only slightly added benefit was seen for patients already receiving two or more active drugs (i.e. darunavir and enfuvirtide). Nonetheless, the proportion of responders, either in the ETR or in the placebo group, increased with increasing number of sensitive drugs in the OBR. A more specific analysis considered simultaneous use of enfuvirtide: in DUET-1 the proportion of patients who used enfuvirtide de novo or re-used enfuvirtide was equally distributed between ETR and placebo recipients (24% and 26% de novo and 16% and 16% re-use, respectively). Among patients re-using or not using enfuvirtide, more patients in the ETR group (55%) reached the primary end-point (a viral load below 50 copies/ml at week 24) than those in the placebo group (33%, difference in response rate 22%, 95% CI 13-31; p < 0.0001). The corresponding results in the DUET-2 study were 58% vs. 34%, p < 0.0001. In the subgroup of patients who introduced enfuvirtide de novo, a significant difference favouring ETR over placebo could not be demonstrated.
A higher CD4 cell count increase for ETR was shown in DUET-1, but not in DUET-2 trial. In both studies a clinical benefit of ETR was seen, because the number of CDC category C AIDS-defining illnesses or deaths was lower for patients in etravirine group. However, a statistically significant difference was reached only in a subgroup of patients who re-used or did not use enfuvirtide in DUET-1 study, and only for deaths in the DUET-2 study. Criticism has been expressed on this point, because a unique trial with 1200 subjects could have better shown small differences on critical clinical outcomes. 34 In a pooled analysis of the 24-week results of the DUET studies, a consistent trend towards fewer clinical endpoints (any new AIDS-defining illnesses and/or deaths) in the ETR group was shown, and this trend reached statistical significance in patients not using or re-using enfuvirtide. Moreover, patients receiving ETR were less likely to be hospitalized than those receiving placebo (11% vs. 16%, respectively, p = 0.0031). 35 This translated into significant savings in hospital-related costs (estimated 1.4-2.7 million US dollars for ETR and 2.2-4.1 million US dollars for placebo). 36
Results at 48 weeks confirmed the durability of virological benefit of ETR: among patients achieving undetectable viral load at week 24, 94% in DUET-1 37 and 90% in DUET-2 38 maintained virologic suppression at week 48. Moreover, the pooled analysis demonstrated a significant reduction in AIDS-defining events or death with ETR compared to placebo over 48 weeks (35 [5.8%] versus 59 [9.8%], respectively, p = 0.041). Time to AIDS-defining events or death was shorter for the placebo recipients. 39
Fewer data are available on ETR use in current clinical practice. Some reports derive from single center experiences. Scott et al recently presented a case series of 36 subjects who were enrolled in the ETR expanded access program (EAP) (TMC125-C214 study): 24/36 used darunavir in the OBR, 15/36 used raltegravir, 1/36 maraviroc, 1/36 enfuvirtide; 10/36 used ETR with nuceloside analogues alone. After 24 weeks, 27/36 (75%) had viral load <50 copies/ml. 40 Updated data were presented at the 9th International Conference on HIV Drug Therapy in Glasgow in 2008: 40 the intention-to-treat proportion of virological suppression was 76% at week 48 for patients who switched to ETR with a detectable viral load (n = 25) and 88% for those who switched with undetectable viral load (n = 8). In the ANRS 139 TRIO study, ETR was co-administered with darunavir and raltegravir in a single arm multicenter trial on 103 subjects, in the context of multidrug failure. 41 Unprecedented high rate of virological success of 90% was reported at 24 weeks.
Data on concurrent use of novel antiretroviral compounds are more and more reported. On 53 patients of the Kaiser Permanente cohort who were co-enrolled in the EAPs of etravirine and raltegravir, 50 (94%) had undetectable viral load at week 24. 42 All 3 patients who did not achieve viral load < 75 copies/ml had documented drug non-compliance. Background regimen included darunavir/ritonavir in 83% of cases, thus rendering this case series very close to the TRIO trial. Similar results were reported in a Spanish cohort of 32 patients who were treated with darunavir/ritonavir, etravirine and raltegravir: 30/32 (94%) had undetectable viral load at week 24. Of the two patients who did not achieve viral load success, one patient stopped treatment at week 4 and the other had poor adherence overtime. 43 Data on etravirine EAP for 877 European patients showed 67% proportion of 24-week virological success. The background regimen included darunavir/ritonavir in 69% of cases, raltegravir in 54% and maraviroc in 14%. 44
Etravirine has been used in combination with raltegravir and optimized background regimen in patients failing a darunavir-containing cART: 13 out of 20 patients reached viral load < 40 copies/ml at week 24, and all 20 patients had viral load < 400 at week 24. 45 In a similar setting, ETR has been used in combination with raltegravir and maraviroc: 14/15 patients (93%) had undetectable viral load at week 24, and exhibited an excellent CD4 recovery (adjusted mean 214.8 ± 37.3 cells/mm3. This small data series is the first example of a NRTI- and PI-sparing rescue regimen for heavily pre-treated patients. Whether this promising strategy can provide adequate rates of virological suppression, minimizing the risk of drug toxicities merits to be further elucidated by future studies. 46 Of note, use of ETV in NRTI-sparing regimens has also been suggested in combination with darunavir/ritonavir in a compact, once-daily regimen for first-line treatment. 47 Such an approach, however, needs to be further investigated.
So far, only one case of successful rescue therapy with raltegravir and etravirine in a patient failing to four classes of antiretroviral drugs has been published. 48
Etravirine has been used also in switch strategies for patients with undetectable viral load. A small pilot phase IIIb study has investigated the possibility to switch to a darunavir/ritonavir plus ETR based regimen patients who were already suppressed by a combination containing enfuvirtide. 49 Of the 9 patients who were switched and continued treatment for 24 weeks all the patients maintained viral load below 50 copies/ml.
Etravirine has been used successfully, together with darunavir/ritonavir, tenofovir/emtricitabine and enfuvirtide to prevent mother to child transmission in a woman with a multidrug resistant virus. 50 Interestingly, significant levels of darunavir, ritonavir and ETR were found in cord blood samples. Moreover, the association of darunavir/ritonavir and ETR has been successfully used for treating a baby who vertically acquired multidrug resistant HIV infection. 51
Recently a case of acute HIV meningoencephalitis successfully treated with the addition of ETR to a fully suppressive regimen (on plasma HIV RNA, but not on cerebrospinal fluid viral load) has been reported. 52 An HCV-coinfected woman who had already experienced two episodes of acute meningoencephalitis successfully treated with efavirenz, developed depression and had to stop efavirenz for this adverse event. An attempt with nevirapine was successful in recoverying meningoencephalitis, but caused acute liver cytolysis. Thus etravirine was introduced: it completely resolved the symptoms within 2 weeks, with no recurrence 17 months later, and decreased the cerebrospinal fluid HIV load to undetectable levels.
Resistance Profile
Despite their high potency, first-generation NNRTIs are limited by the low genetic barrier to resistance. Selection of resistance mutations occurs rapidly when these drugs are used suboptimally and the occurrence of any previous NNRTI resistance mutation is likely to reduce subsequent response to the first-generation NNRTI.
The mutations in HIV-1 retrotranscriptase gene (RT) associated with NNRTI resistance have been identified and cluster within the NNRTI binding pocket. The discovery program of ETR have focused on activity against these key mutations. An initial screen for activity against NNRTI-resistant HIV-1 was carried out on a panel of 25 viruses carrying single or double RT substitutions associated with NNRTI resistance. 1 Etravirine showed good activity (defined as EC50 < 5 nM and a fold change in EC50 < 4) against 18 of these viruses. These included mutant viruses carrying K103N, G190A/S and the double mutants K101E + K103N and K103N + Y181C. Etravirine EC50 values of > 10 nM and fold resistance value of > 10 were observed for viruses harboring L100I + K103N, Y181I or F227C.
In vitro selection experiments with ETR demonstrated a delayed time to viral breakthrough as compared to nevirapine and efavirenz. Significant resistance to ETR required at least two mutations to develop. 53 Passage experiments starting with site-directed mutations containing K103N, Y181C or both in combination, resulted in the accumulation of one or more additional mutations. The most significant change in resistance owing to a single additional mutation was the addition of V179F following the passage of Y181C. Other single aminoacid substitutions resulted in modest (< 10-fold) increases in resistance. Selection experiments identified mutations selected by ETR that included known NNRTI-associated mutations L100I, Y181C, G190E, M230L and Y318F and the novel mutations V179I and V179F. According to the researchers, these experiments indicate that ETR possesses a high genetic barrier to the development of resistance in vitro.
Nonetheless, antiviral activity of a potential second-line agent against only an initial signature mutation (or a combination of two mutations) is not enough to support sequential use of NNRTI. This limitation has been convincingly demonstrated with efavirenz following nevirapine treatment failure. 54 Although the presence of a single mutation may not be associated with a signifùicant reduction of the response to ETR in vitro, its presence may lay the mutational foundation for the development of higher levels of ETR resistance in vivo. Therefore, only combination of laboratory and clinical data can help to better identify the resistance profile of ETR.
Data regarding resistance to ETR in vivo are scarce and limited to the setting of co-administration of darunavir/ritonavir. Table 2 shows the three main algorithms used to predict ETR resistance. Correlation between baseline mutations and the virological response to an ETR-containing salvage therapy regimen was first studied in a pooled analysis of the DUET trials. 55 The effect of baseline genotype on virological response (i.e. <50 HIV-RNA copies/ml) to ETR at week 24 was studied in patients not using enfuvirtide de novo, and excluding those who had discontinued for reasons other than virological failure. From an initial list of 44 NNRTI resistance associated mutations, 26 were selected for being present in five or more patients. Among them, 13 signature mutations resulted to be associated with decreased response to ETR. They were: V90I, A98G, L100I, K101E/P, V106I, V179D/F, Y181C/I/V and G190A/S. No single mutation was identified to have a significant impact on virological response to ETR, although V179F, Y181V and G190S had the largest impact. Notably, K103N was not associated with decreased virological response and Y181C, if present as the only ETR resistance associated mutation, was associated with a rate of virological suppression comparable with that observed in the overall group. The patients who experienced the greatest benefit with ETR treatment were those who had <3 ETR resistance associated mutations, therefore isolates with fewer than 3 mutations were considered eligible for ETR. Although simple, this score lacks of an accurate weighing of each single mutation and arbitrarly excludes the clinical impact of mutations under-represented in the study cohort. Moreover, it can only be generalyzed in the setting of darunavir/ritonavir co-administration (or, at least, to the cases of co-administration with an active boosted-PI).
Algorithms for interpretation of ETR genotypic resistance.
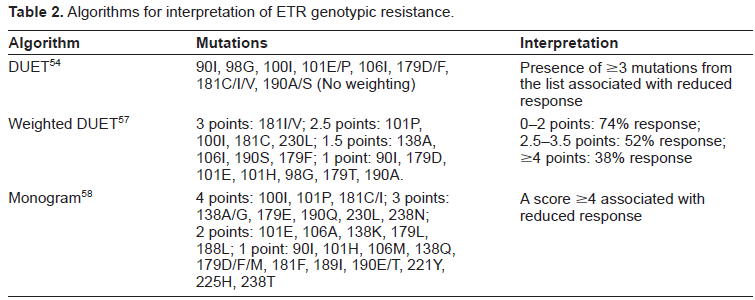
In another post-hoc analysis, a regimen-based resistance interpretation was proposed, suggesting that virological response was more likely to occur in the presence of <3 ETR- and <3 darunavir-resistance associated mutations. 56 The clinical applicability of this finding is though limited. Considering mutations emerging at time of failure on ETR, four NNRTI resistance associated mutations appeared to emerge in at least 10% of the patients in DUET studies. Two of them were already included in the ETR score (V179F and Y181C), whilst the other two (V108I and V179I) were not. 57 Whether these mutations may have an influence on the susceptibility to ETR when present at baseline is unclear.
More recently, a new and updated weighted list of mutations associated with reduced virological response from the DUET studies has been proposed by Tibotec, in order to refine the list and improve the genotype/phenotype correlation. 58 Using a different statystical approach, the former list of 13 mutations was extended to include 4 additional mutations (K101H, E138A, V179T and M230L). Different weight was attributed to each mutation of the list and the new weighted score of 0-2, 2.5-3.5 and > = 4 corresponded to 74%, 52% and 38% virological response rates (Table 2). Another, indipendent scoring system has been proposed by Monogram Bioscience. 59 Correlating genotype and phenotype resistance data, a list of 29 mutations with the potential to affect ETR sensitivity was proposed. Each mutation was then assigned a weight of 4, 3, 2, or 1, with each higher value representing an incremental drop in susceptibility to ETR. A score ≥4 indicated a foreseen reduced response to ETR. Of note, four mutations (L100I, K101P, Y181C and Y181I) already included in the original ETR mutation list merited a weighting factor of 4. Conversely, four mutations from the original ETR mutation list (A98G, V106I and G190A/S) were excluded from this score (Table 2).
Concordance between the different algorithms was found to be weak by a French group, analyzing more than 2,500 RT sequences from a real-life setting. 60 Most of the discordance was due to the differential weights attributed to Y181C/V, L100I, and K101P in the weighted algorithms. It is unclear what algorithm predicts better clinical response to ETR and validation studies of the scores in separate patient populations are urgently needed. Moreover, new mutations with a possible impact on ETR susceptibility are being discovered (e.g. E399D). 61 Thus, the ETR resistance-associated mutation list should be continuously updated.
In any case, several studies analyzed the mutational patterns in cohorts with heavy experience to NNRTI, in order to determine the prevalence of ETR associated mutations.62–64 Overall, presence of at least 1 mutation associated with ETR resistance ranged from around 40% to 60% in patients with previous failure on NNRTI. However, the prevalence of virus harboring > 1 mutation was lower. Increasing number of ETR resistance mutations has been associated with nevirapine use and longer time on failure with NNRTI containing regimen. Moreover, ETR resistance mutation accumulation was demonstrated in patients who continued on failing nevirapine-containing regimen. 62 These data suggest that ETR may retain its activity in most patients with resistance to first-generation NNRTI. However, ETR-resistant virus may have a significant cross-resistance with nevirapine, resulting from, in large part, the selection of Y181C.
As for phenotipic resistance, different cutoffs have been proposed to predict virological response. Values of 3 and 13 (Antivirogram phenotypic assays) and 1.6 and 27.6 (VircoType assay) have been proposed as the lower and the upper cutoffs, signalling reduced and abolished responses, respectively.65,66
Drug Safety and Tolerability
Information on ETR safety and tolerability are mainly derived by large clinical trials and concentrated mostly on neuropsychiatric effects and rash, the two major concerns of efavirenz and nevirapine toxicity.
Neuropsychiatry Adverse Events
In the TMC125-C223 study, the overall the incidence of psychiatric adverse events with etravirine was 10.7%, with no grade 3/4 psychiatric events. 26 The most commonly nervous system events were headache and insomnia, neither of them leading to discontinuation. The TMC125-C203 study was specifically designed and powered to investigate long-term (48 weeks) safety and tolerability with respect to placebo: 29 in this case the overall incidence of neuropsychiatric events was comparable across groups, including placebo.
A similar finding was documented in the DUET trials, where no difference in the incidence of neurpsychiatric adverse events were seen between etravirine and placebo groups.32,33
Skin Rash
In the TMC125-C223 study the incidence of rash was 22.5 and 17.7% in the ETR 400 mg and 800 mg groups, respectively, as compared with 7.5% in the control group, with no dose-related effect. Rashes generally had early onset, were mild to moderate in severity and lasted less than a week on average. Four patients (2.5%) experienced grade 3 rash, while no grade 4 rashes were reported. Six ETR patients (3.8%) discontinued because of rash.
In the TMC125-C203 study the incidence of rash was higher in ETR group, but the difference was not statistically significant (19.5% vs. 12.1%, P = 0.25). There was no relationship between incidence of rash and ETR dose. Etravirine-associated rash was generally mild to moderate in severity, occurred early, was typically transient, and infrequently led to treatment discontinuation. No cases of Stevens-Johnson syndrome, toxic epidermal necrolysis, or erythema multiforme were observed. It is noteworthy, however, that all the patients enrolled in the trial were NNRTI-experienced, but none of them had a history of severe allergic or dermatological reaction leading to discontinuation with any NNRTI. Therefore, it is unclear whether ETR can be administered to a patient who experienced severe allergic reaction to nevirapine or efavirenz.
When compared head-to-head with a PI, ETR was associated with increased risk of skin rash. 30
In the DUET-1 study, rash was observed in 20% of patients in the ETR group and 10% of the patients in the placebo group (p < 0.0001). 32 Most cases were grade ≤2, while only four (1%) patients developed a grade 3 rash. No grade 4 rashes were reported. Rash led to treatment discontinuation in six (2%) patients, and was more frequent in females (34% vs. 18%, p = 0.0192). In the DUET-2 study, rashes were reported in 14% andf 9% of the patients in the ETR group and placebo group, respectively. 33 Only four (1%) patients had grade 3 rash, and seven (2%) discontinued because of rash. A similar occurrence of rash was reported in patients with and without a history of NNRTI-related rash, but numbers are too small to draw any definitive conclusion. The pooled analysis of the DUET trials confirmed the increased risk of rash among ETR recipients, either at 24 weeks 67 or 48 weeks of follow-up. 68
Other Investigated Toxicities
Etravirine has been associated with good gastrointestinal tolerability. In the study TMC125-C203 study, diarrhoea was less likely to occur in ETR-treated patients than in patients receiving placebo (28.7% versus 42.4%, respectively, P < 0.05). The authors speculate that the more frequent use of ritonavir-boosted lopinavir in the placebo group may have contributed to this finding. Indeed, when ETR was compared head-to-head with a PI (TMC125-C227), a lower incidence of gastrointestinal side effects was reported. 30
The effect of ETR on embryo-fetal and pre and postnatal development was assessed in pregnant rats and rabbits. 69 Etravirine showed no adverse effects on embryo-fetal development at any doses tested in rats or rabbits. Moreover, ETR had no effect on offspring development and maturation during lactation or postweaning in rats. In these studies, maternal systemic exposure to ETR was equivalent to the clinical exposure at the recommended therapeutic dose, and was 2-3 fold higher at the beginning of dosing during the early stages of organogenesis.
Electrocardiographic safety was deeply investigated by a clinical trial on 41 healthy HIV-negative volunteers, who received two doses of ETR for 36 days. 70 No effect on QT and QTc interval was reported, and no clinically relevant ECG changes were observed.
Laboratory Abnormalities
In the TMC125-C223 study the most common treatment-emergent grade 3/4 laboratory abnormality with ETR was elevated pancreatic amylase, occurring in 13 patients (8.2%). Only 2 patients permanently discontinued ETR, one for pancreatitis, the other one because of elevated pancreatic and amylase levels. No clinically relevant increase in cholesterol and triglyceride levels was reported. In the DUET studies no differences in nature, frequency and severity of laboratory hepatic and lipid-related parameters between ETR and placebo were reported.
Conclusions and Place in Therapy
The introduction of ETR in the treatment armamentarium of HIV infection has changed our knowledge on the possibility to sequence NNRTI. Whilst broad cross-resistance among first-generation NNRTI has precluded their sequential use so far, ETR is likely to retain (at least in part) its activity after efavirenz and, to a lesser extent, nevirapine failures. The most solid data on ETR use come from the DUET studies, where ETR has been used in combination to darunavir/ritonavir in treatment experienced patients. In this context, the additional value of ETR in suppressing HIV replication has been clearly demonstrated. Therefore, in patients with limited treatment options and who are infected with a virus with at least partial susceptibility to darunavir, co-administration of darunavir/ritonavir and ETR should always be considered.
Like other antiretrovirals, ETR is likely to rapidly lose its activity in the absence of other active drugs in the regimen. Therefore, its use in combination only with a NRTI-backbone after first-line NNRTI failure should be discouraged, particularly when NRTI resistance associated mutation are present. Use of ETR in association with newer classes in patients with multi-class-failure has led to high rates of virological suppression in some uncontrolled studies. However, whether ETR retains its activity also in the absence of a potent boosted-PI, when used in combination with active drugs with lower genetic barriers, is still unclear.
Switch to ETR has been proposed as a simplification strategy in patients using enfuvirtide with suppressed plasma viral load. Although the results of a preliminary study are encouraging, this strategy should be used with caution and under the guidance of previous genotypic resistance testing. Moreover, in this setting switch to raltegravir appear to be more promising.
There are currently different, and to some extent conflicting, data regarding resistance to ETR. None of the current available genotypic resistance score can be considered definitive, because they need to be validated in separate populations. A continuous surveillance is needed to identify possible previously unreported mutations able to reduce response to ETR. Nonetheless, available data may provide guidance for the use of ETR in NNRTI-experienced patients with limited NNRTI resistance. Patients with a history of prolonged failure on NNRTI, particularly nevirapine, are more likely to have a reduced response to ETR. Thus, a prompt discontinuation of a failing NNRTI regimen is essential to minimize the accumulation of resistance mutations and to maximize the chance of subsequent response to ETR.
Disclosure
The authors report no conflicts of interest.
Footnotes
Acknowledgments
We acknowledge Dr. Alessandra Calabresi and Dr. Ilaria Izzo (Clinical of Infectious and Tropical Diseases, University of Brescia) for their kind assistance in the selection of all the relevant literature on ETR.
