Abstract
Fludarabine, a purine nucleoside analogue, is considered as the most active drug in chronic lymphocytic leukemia. This review summarizes current knowledge concerning the mechanism of action, pharmacological properties, clinical activity and toxicity of fludarabine in the treatment of non-Hodgkin's lymphoma. A literature search on fludarabine for lymphomas was undertaken using MEDLINE databases. Clinical data shows that fludarabine alone and as a component of combination chemotherapy is effective in patients with various types of non-Hodgkin's lymphoma, particularly in follicular lymphoma. The most commonly used combinations are with cyclophosphamide and/or mitoxantrone. Fludarabine-based regimens are highly active in both previously untreated and relapsed indolent lymphomas. The results of the chemotherapy are considerably improved with the addition of rituximab. Fludarabine is generally well tolerated. Myelosuppression and infections, including opportunistic varieties, are the most frequent adverse effects. Indications for fludarabine and treatment regimens for lymphoma patients are discussed.
Introduction
The success of the pyrimidine nucleoside cytarabine in the treatment of acute leukemias prompted development of new purine nucleoside analogues. 1 The most promising agents in this group are deoxycoformycin (pentostatin), fludarabine phosphate and 2-chloro-2‘-deoxyadenosine (2CdA) that cause suppression of DNA synthesis and repair in the tumor cells by distinct enzymatic processes. Fludarabine, synthesized by Montgomery and Hewson in 1969, 2 was first studied in acute leukemia using high doses (150 mg/m2/d for 5-7 days) and found to be excessively toxic to the nervous system. 3 Subsequently fludarabine at lower doses revealed impressive activity in the treatment of indolent lymphoproliferative diseases.4–6 Currently fludarabine is considered as the most active drug in chronic lymphocytic leukemia (CLL) inducing complete remission (CR) in about 30% of previously untreated patients, with an overall response (OR) rate of 70%.7,8
This review summarizes the available data on the fludarabine in the treatment of patients with non-Hodgkin's lymphomas (NHL). A literature search on fludarabine for lymphomas was undertaken to identify published guidelines, meta-analyses, systematic literature reviews and controlled clinical trials using MEDLINE databases. No evidence-based guidelines for the use of fludarabine have been found for the review. Pooling of data of cases from different studies for joint analysis was impractical for the reason that patients with heterogeneous characteristics (age, lymphoma subtype, prognostic factors, previous treatments etc) might have been included. The main goal of this study is to evaluate the majority of published records on fludarabine and to analyze the indications for the administration, the advantages and disadvantages of this drug in order to define its right current place in the treatment of lymphoma patients.
Mechanism of Action and Pharmacokinetics
Fludarabine, a fluorinated analog of adenine, is resistant to adenosine deaminase, the enzyme responsible for inactivation of the parent compound. 9 In clinical practice it is used as a water soluble derivate, phosphate salt (9-β-D-arabinofuranosyl-2-fluoroadenine-5'phosphate). The pharmacokinetics of the drug was studied after i.v. administration by rapid bolus injection and following short-term or continuous infusion. Fludarabine is rapidly dephosphorylated in vivo to F-ara-A showing peak plasma concentrations in a few minutes after the drug infusion. Following bolus injection, a brief 5 minutes early tissue-disposition phase is followed by 1-2 hours intermediate disposition phase and finally a prolonged terminal elimination phase (half-life 10 to 30 hours). 10 After a prolonged intravenous infusion (>30 minutes) the disposition phase is less obvious, exhibiting a biphasic decline in plasma concentration of F-ara-A.11,12 No accumulation of plasma F-ara-A is seen following repeated daily administration. The main route of elimination is renal, 40%-60% of the fludarabine being excreted in the urine. 13
The rephosphorylated product of F-ara-A, triphosphate F-ara-ATP, accumulates intracellulary within several hours (peak concentration at 4 hours) proportional to the dose administered, and has cytotoxic activity. 14 The retention of F-ara-ATP varies between individuals, the half-life ranges from a few hours to several days with a median value of 15 hours. 14 Fludarabine inhibits both DNA and RNA synthesis in the tumor cells by several mechanisms: 1. termination of chain elongation after incorporation into DNA strands, 2. inhibition of DNA and RNA polymerases, 3. inhibition of ribonucleotide reductase, the enzyme responsible for the conversion of ribonucleotides into deoxyribonucleotides, 4. direct activation of the apoptotic mechanisms.15,16 All the mentioned above data regarding the drug pharmacokinetics correlate with clinical results and should be applied in practical recommendations. The long half-life of the active metabolite allows for once-daily dosing. Patients with renal impairment and elderly patients who commonly have decreased creatinine clearance generally do not tolerate usual doses of fludarabine. 17 Fludarabine dose reduction based on creatinine clearance values is needed, while the drug is relatively contraindicated in patients who have less than 30 ml/min. 16 Finally, clinical results in CLL with divided doses administered on 3 or 5 consecutive days were preferable to the same accumulated dose given once weekly, indicating need to maintain therapeutic level for at least 3 days. 18
Non-Hodgkin's Lymphomas
The NHLs comprise a diverse group of lymphoid malignancies with heterogeneous patterns of molecular pathophysiology, histology, behavior and response to treatment. The modern classifications, like the World Health Organization (WHO) lymphoma classification, systematize lymphomas according to the characteristics of morphology, the origin and the state of differentiation of the tumor lymphocytes and specific cytogenetic aberrations. 19 Using clinical criteria, the NHLs can be separated into two large groups: aggressive and indolent (low-grade). Aggressive lymphomas are associated with a rapidly progressive course, short survival if untreated, sensitivity to (immuno)chemotherapy and are treated with curative intent. The most common aggressive lymphoma is diffuse large B-cell lymphoma (DLBCL). Mantle cell lymphoma (MCL) is a distinct aggressive subtype showing low CR rate to therapy, median survival of only 3 to 4 years and few long-term survivors.
Indolent lymphomas constitute 30%-35% of all NHL subtypes. Of these, follicular lymphoma is the most encountered and diagnosed in the vast majority of patients. Follicular lymphoma has been associated with the chromosomal translocation t(14; 18), resulting in overexpression of the bcl-2 oncogene and subsequent inhibition of apoptosis.
The less common subtypes are small lymphocytic lymphoma, nodal and mucosa-associated lymphoid tissue marginal zone lymphomas and lymphoplasmacytic lymphoma. Low grade lymphomas usually present with few symptoms, diffuse lymphadenopathy with or without splenomegaly and frequent bone marrow involvement and follow a long course. These lymphomas have tendency for good response to treatment but inevitable subsequent relapses. Low-grade lymphomas appear to be incurable with currently available treatments that rarely change the natural history, and death ultimately results from progression to more aggressive disease or infection. The overall median survival is 6 to 10 years. 20 The therapeutic approach to indolent lymphomas is controversial and ranges from “watch-and-wait” in asymptomatic patients or palliative chemotherapy to front-line myeloablative chemotherapy followed by autologous hematopoietic cells rescue in high-risk patients. In order to determine need for chemotherapy in follicular lymphoma, most clinicians use Follicular International Prognostic Index (FLIPI) score that assesses the risk of progression based on clinical stage, age, number of involved nodal sites, and hemoglobin and LDH levels (Table 1). 21 The use of more active chemotherapeutic agents and novel therapeutic tools has recently produced complete responses and prolonged disease-free survival (DFS) intervals in most patients giving hope to survival curve improvement in indolent lymphomas. 22 The means that have achieved the best results include monoclonal antibodies (including radioimmunotherapy) and purine analogs, namely, fludarabine.
Follicular lymphoma international prognostic index. 21
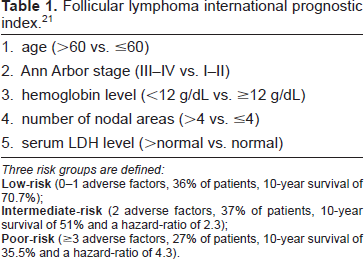
Three risk groups are defined:
Fludarabine Monotherapy
The initial study evaluating fludarabine in lymphoma was published by Leiby et al in 1987. 23 In this study a loading dose of 20 mg/m2, followed by a continuous 48-hour infusion of 30 mg/m2/d, was administered to 26 patients with refractory disease. There were one CR which lasted for 9+ months and 7 short partial responses (PR) (OR 32%). Toxicity was acceptable and consisted mainly of myelosuppression.
Most patients enrolled in the early studies had advanced lymphoma refractory to previously given chemotherapy or progressed after response. Generally a schedule of fludarabine in a dosage of 20 to 30 mg/m2/day for 5 consecutive days and repeated every 3 to 5 weeks was applied. The responses, usually partial, have been achieved in approximately 50% of patients with indolent lymphomas and lasted up to one year (Table 2).4,24,25
Noncomparative studies evaluating fludarabine monotherapy in non-Hodgkin's lymphoma.
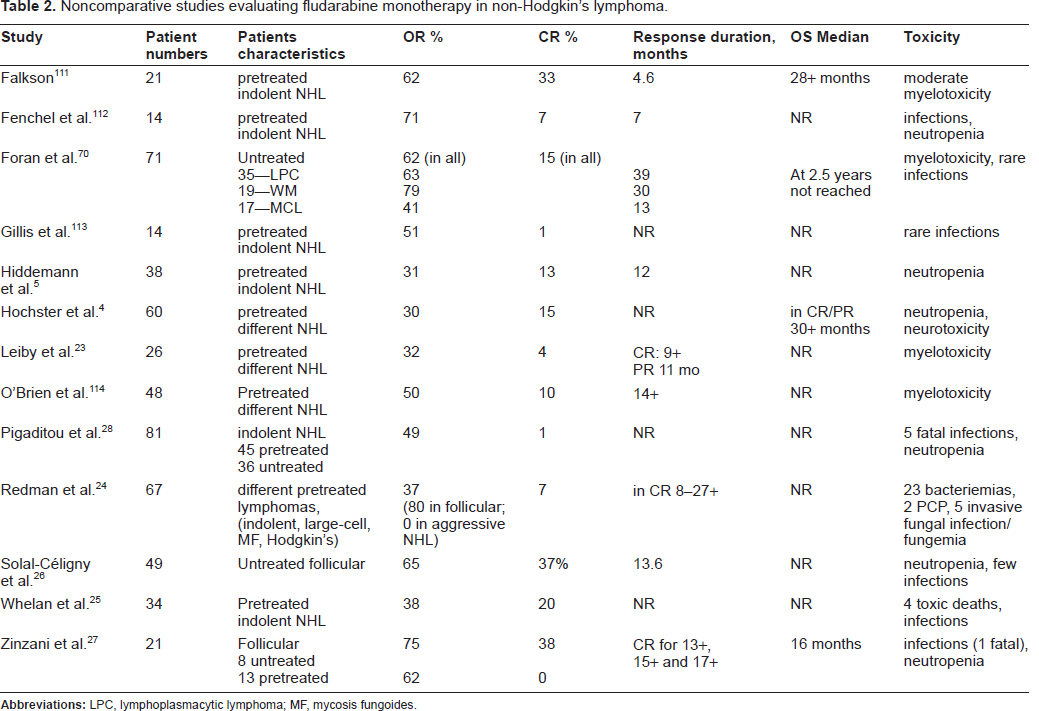
The results in aggressive NHL were disappointing showing low 0%-14% rate of objective responses.4,23
The French cooperative group investigated fludarabine as first-line treatment in patients with advanced follicular lymphoma. Although the treatment was prematurely stopped because of toxicity in nine patients, the response rate was relatively high: for 49 assessable patients the OR rate was 65% with CR rate of 37%; whereas the median time to progression was 13.6 months. 26 An Italian phase II trial used fludarabine monotherapy in 8 untreated and 13 previously treated stage 4 indolent lymphoma patients. The results were better in treatment-naïve (3 CR and 3 PR) than in pre-treated patients (0 CR and 8 PR). 27
The encouraging data demonstrated that fludarabine is capable to eradicate minimal residual disease (MRD) measured by polymerase chain reaction analysis (PCR) for bcl-2 in blood and bone marrow in some patients with follicular lym-phoma.28–30 Although, there are controversies about its significance, the disappearance of MRD after therapy has been associated with significantly improved outcome. 31 The MRD status may serve as good marker for remission quality, DFS and probably overall survival (OS).
Comparative Trials
The Canadian investigators randomly compared the safety and efficacy of fludarabine and cyclophosphamide, vincristine, and prednisone (CVP-regimen) in patients with recurrent low-grade lymphoma after previous response to alkylator-based therapy. 32 Although there were no significant differences in the response rates, the patients in the fludarabine arm had slightly improved median progression-free survival (PFS) and OS and better social function scores compared with patients treated with CVP. An OR rate of 64% (9% CR), a median PFS time of 11 months, and a median OS time of 57 months were demonstrated in the fludarabine group. Two patients died of fludarabine-related toxicities: invasive fungal infection and late development of Stevens-Johnson syndrome.
Recently the European Organization for Research and Treatment of Cancer (EORTC) Lymphoma Group reported long-term follow-up results of a phase III trial comparing fludarabine monotherapy to CVP-regimen in 381 previously untreated patients in advanced stages of indolent lymphoma. The responses rates were significantly improved in the fludarabine arm (70% OR and 38% CR vs. 52% and 15%, respectively, with CVP). 33 However, only nonsignificant trend for survival extension was noted: the 5-year OS in the “wait and watch” cohort treated by fludarabine was 65% while in the “intention to treat” cohort was 76% (vs. 56% and 62%, respectively, in the CVP arm). Fludarabine-treated patients developed more commonly granulocytopenia, although it did not translate to more infections.
In contrast, the GELA study comparing fludarabine to CHVP (cyclophophamide, doxorubicin, etoposide and prednisone) plus interferon in high-risk elderly patients with follicular lymphoma found better 2-year survival in the combination group. 34 Nevertheless, fludarabine in this study was given in much extended course (12 cycles within 2 years) that seems not optimal for elderly patients.
Combination (Immuno) Chemotherapy
The rationale for combining various chemotherapeutic agents with or without monoclonal antibodies is based on the different cytotoxic mechanisms of drugs, synergistic cytotoxicity in vitro and different adverse effects. An important additive cytotoxic activity of cyclophosphamide, anthracyclines or mitoxantrone and rituximab has been demonstrated when combined with fludarabine in vitro.35,36 Fludarabine inhibits repair of cyclophosphamide-induced cross links of the DNA. 37 Fludarabine also down-modulates the complement-resistance proteins CD55 and CD59, thereby potentially making cells more vulnerable to rituximab-induced complement-mediated lysis. 38 In addition fludarabine might (paradoxically) stimulate natural killer (NK) cell activity contributing to intensified tumor cell destruction by rituximab via antibody-dependent cell-mediated cytotoxicity (ADCC). 39 In turn, rituximab down-modulates IL-10 and bcl-2, making tumor lymphoid cells susceptible to fludarabine-induced apoptosis. 40 As a result, the combination of fludarabine and rituximab revealed cytotoxic activity on a lymphoma cell line resistant to either drug alone. 38
Combination with Cyclophosphamide
Cyclophosphamide, the most frequently used alkylating agent in the treatment of lymphomas, was first described as therapy for diffuse large-cell lymphoma in 1975 and subsequently as a part of the CHOP regimen (cyclophosphamide, doxorubicin, oncovin, prednisone) became standard treatment for patients with aggressive lymphomas.41,42 CHOP-regimen induces very high (>90%) response rates in patients with indolent lymphoma and does not compromise subsequent stem cell transplantation (SCT) feasibility. 43 However, the fact that this treatment is not able to change the natural history of indolent lymphomas or to eradicate MRD in follicular lymphomas, as documented by the persistence of cells bearing the bcl-2 gene rearrangement, has encouraged the investigation of cyclophosphamide in combination regimens with newer agents, such as fludarabine, mitoxantrone and rituximab.
The investigators from the Eastern Cooperative Oncology Group (ECOG) developed a regimen of cyclophosphamide and fludarabine (FC) (Table 3). 44 In this trial fludarabine (20 mg/m2, days 1-5) was administered with an escalating dose of cyclophosphamide (600-1.000 mg/m2, day1) in 27 previously untreated patients with indolent lymphomas. A 100% OR and 89% CR rates were achieved after a median number of six cycles. At a median follow-up of 61 months, 12 of these patients remain disease-free, and 5-year OS was 66%. Infectious complications including fungal sepsis, lobar pneumonia, Pneumocystis carinii pneumonia (PCP) (one of each) and three cases of herpes zoster virus (HZV) were reported.
Noncomparative studies evaluating fludarabine-based combination regimens in lymphoma.
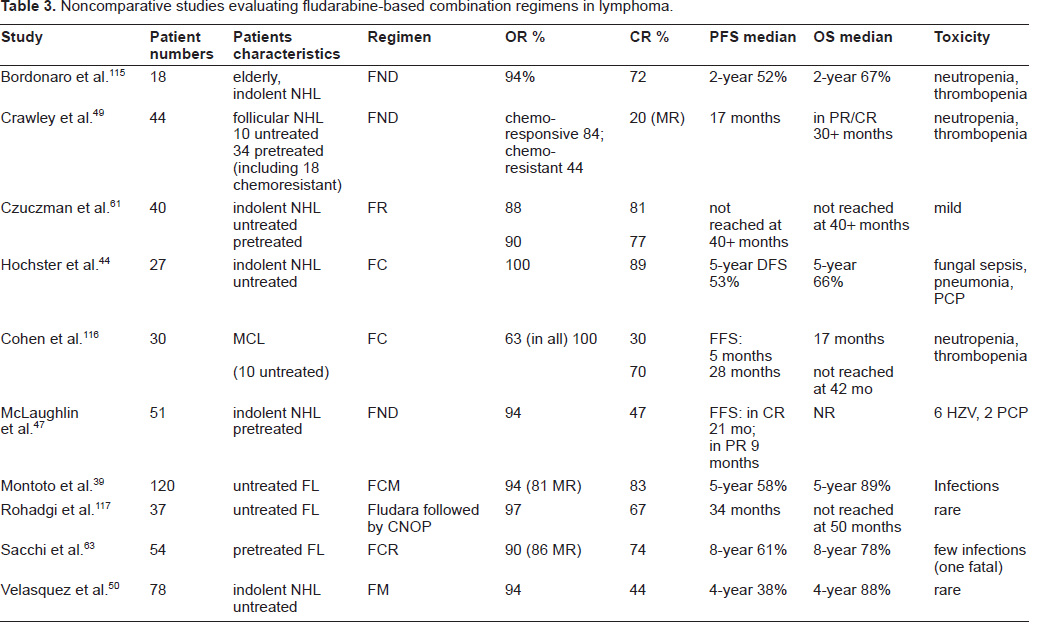
Flinn et al from the John Hopkins Oncology Center using the FC regimen and myeloid growth factor support, achieved similar results in 20 untreated patients with follicular lymphoma: 92% OR rate with 60% CR. 45 A number of these patients were further autotransplanted; it is important to note that an autologous graft suitable for transplant was obtained in all patients seeking transplant.
Combination with Mitoxantrone and Anthracyclines
Mitoxantrone, an anthracenedione compound, has clinical activity similar to anthracyclines and is associated with less cardiac and gastrointestinal toxicity. 46 Response rates of 27%-67% have been reported in the management of indolent lymphomas, but generally the responses were partial. 46
A FND regimen, combined fludarabine (25 mg/m2/d on days 1-3), mitoxantrone (10 mg/m2 on day 1) and dexamethasone (20 mg/d on days 1-5), was first described by McLaughlin et al from the M.D. Anderson Cancer Center (MDACC). 47 In this phase II trial, 51 patients with relapsed or refractory indolent lymphoma achieved an OR rate of 94% including 47% CR. The median duration of the CR was 21 months. Infections occurred in 12% of treatment cycles, including one fatal event caused by Mycobacterium avium-intracellulare, 6 cases of dermatomal HZV and two PCP.
Comparative trials evaluating fludarabine-containing therapy.
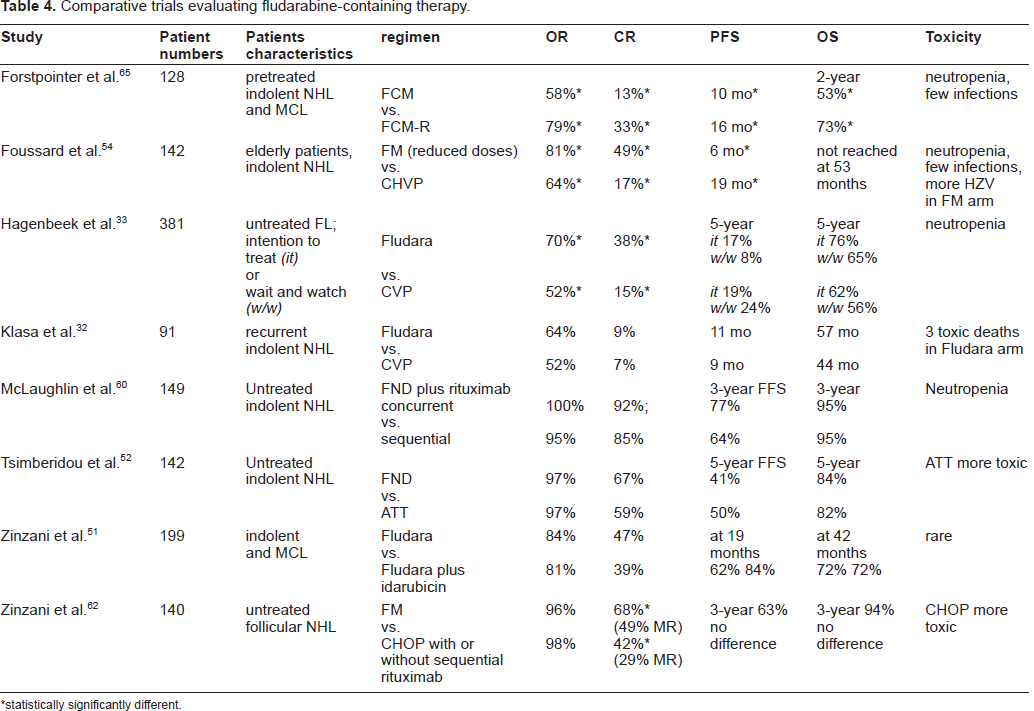
statistically significantly different.
Other study has found similar clinical results with FND in previously treated patients. 48 Crawley et al demonstrated that the FND regimen is capable to induce molecular remission even in chemoresistant follicular lymphoma. 49
The Southwest Oncology Group (SWOG) in the U.S. conducted a trial of fludarabine and mitoxantrone as initial treatment in 78 indolent (in majority, follicular) lymphoma patients. This regimen produced OR rate of 94% and CR of 44%. The 4-year PFS of 38% and OS of 88% were favorable comparing to SWOG's prior experience with CHOP and ProMACE/MOPP (prednisone, methotrexate, doxorubicin, cyclophosphamide, etoposide, mechlorethamine, vincristine and procarbazine) regimens. 50 The toxicity was mild and transient.
Italian investigators designed a comparative trial of fludarabine (25 mg/m2 for 3 days) in combination with idarubicin (12 mg/m2 day 1) vs. fludarabine alone (at same daily dosage for 5 days). 51 Following 6 monthly cycles 199 untreated patients with indolent lymphoma or MCL were assessed. The combination chemotherapy resulted in improved duration of response, although the response and OS rates as well as the rate of complications (a few in both groups) were comparable. Patients with follicular lymphoma had better CR rate (60% vs. 27%) and longer duration of response in comparison to MCL patients.
The MDACC group compared FND chemotherapy with intensive alternating triple therapy (ATT) that included 4 cycles of each CHOP-Bleo (CHOP and bleomycin), ESHAP (etoposide, methylprednisolone, cytarabine and cisplatin) and NOPP (mitoxantrone, vincristine, prednisone, procarbazine) regimens in a study on untreated patients with indolent lymphoma; maintenance with interferon/dexamethasone was given for one year in both treatment arms. The OR, OS and PFS rates were similar for FND and ATT. 52
In order to decrease myelotoxicity, Rohadgi et al used sequential therapy with 3 cycles of fludarabine as a single agent followed by 6-8 cycles of CVP with mitoxantrone (CNOP). The treatment was administered to 37 previously untreated patients with advanced follicular lymphoma and produced OR rate of 97% and CR rate of 67%. At median follow-up of 50 months, more than a half of responders were free of progression. Nevertheless, two patients died of treatment-related events (disseminated HZV and secondary leukemia). 53 With the same intention to reduce toxicities, Foussard et al investigated lower doses of fludarabine in combination with mitoxantrone as firstline treatment and compared the results with cyclophosphamide, doxorubicin, vindesine and prednisone (CHVP-regimen) in 142 elderly patients with indolent lymphoma. 54 The FM therapy resulted in superior remission rates (OR 81% and CR 49% vs. 64% and 17%, respectively) and median failure-free survival (FFS) (36 months vs. 19 months). No difference was found between the study arms in terms of OS, however the lack of differences might be explained by efficient salvage treatment that often included fludarabine after failure of the CHVP. Despite common neutropenia, few infections (but more cases of HZV) were diagnosed in the FM arm.
The concomitant use of fludarabine, cyclophosphamide and mitoxantrone (FCM) in previously untreated patients induced excellent results of 94% OR, 83% CR and 81% molecular response rates. 30
Rituximab
Rituximab, a chimeric mouse/human antibody directed against CD20, is an important therapeutic option for patients with B-cell lymphomas. In preclinical studies, rituximab lysed CD20+ cells via human-complement, ADCC and by inducing apoptosis. 55 Rituximab as a single agent has significant clinical activity in follicular lymphomas, producing a clinical response rate of 73% and a molecular remission rate of 62%.56,57 When rituximab was combined with CHOP, the objective responses have been improved to >90% and CR was achieved in 50%-60% of the patients. 58 This immunochemotherapy resulted in longer DFS times and according to recent data probably improves also OS rates in patients with indolent lymphomas when compared with those who receive chemotherapy alone. 59 An additional advantage of rituximab resides in its good tolerability.
Investigators from the MDACC prospectively studied the FND regimen with rituximab given concurrently or sequentially as initial therapy in patients with stage IV small lymphocytic or follicular lymphomas. 60 Responding patients further received interferon-α2b as maintenance therapy for a year. At a median follow-up of 30 months, 92% patients receiving concurrent treatment achieved CR and 89% molecular remission, whereas the results were inferior (85% and 68%, respectively) post the sequential schedule. 60 The 3-year FFS and OS were statistically not different (77% and 95% for concurrent vs. 64% and 95% for sequential therapy, respectively). Although there was a slightly higher incidence of grade 3-4 neutropenia with concurrent treatment, there were no more infections.
The most used fludarabine-containing regimens.*

In all regimens:
repeated every 28 days.
dose adjustment for patients with renal failure (Table 6).
oral fludarabine round dose to nearest 10 mg.
for the first cycle, rituximab is recommended to administer on a day before chemotherapy. If the total WBC > 30 × 10 9 /L rituximab could be omitted from the first cycle of treatment to avoid risk of massive cytokine release syndrome. Rituximab can be incorporated in subsequent cycles of treatment regardless of the level of the WBC count.
Another phase II study investigated the efficacy of fludarabine and rituximab combination (FR) in 40 treatment-naïve or relapsed patients with indolent lymphomas that were enrolled on intent to treat basis. 61 High and similar response rates were seen in treatment-naïve (88% OR and 81% CR) and previously treated (90% OR and 77% CR) patients. The median duration of response was not reached at 40 months. Out of 16 patients who were bcl-2–positive in bone marrow aspiration samples, 14 (88%) converted to negative after therapy and showed stable molecular response on repeated PCR assays for up to 4 years.
Italian investigators performed a landmark study to randomly compare the efficacy of fludarabine-mitoxantrone (FM) and CHOP regimens. 62 Patients who achieved partial response (PR) or complete clinical but not molecular response were further treated with rituximab. Of 140 assessable patients, the OR rates for FM (96%) and CHOP (98%) were similar, but the rate of CR was higher in the FM arm (68% vs. 42%; p = 0.003). The rate of molecular response was also higher with FM (47% vs. 29%; p = 0.03). After rituximab, the number of patients who had experienced both clinical and molecular CR was higher in the FM arm (71% vs. 51%; p = 0.01). In the overall study population, rituximab treatment led to significantly higher clinical CR (86% vs. 57%) and combined CR and molecular response rates (61% vs. 29%). There were no differences in the PFS and OS times; the estimated survival rate for the entire study population at 3 years was 94%. Hematological toxicities were manageable in both arms, with no cases of treatment-related death or severe infection. CHOP was associated with significantly higher rates of grade 3-4 vomiting, alopecia, peripheral neuropathy and constipation. The results of this trial showed the advantage of the FM regimen over CHOP chemotherapy in terms of clinical and molecular responses, better tolerability, and also the benefit from the addition of rituximab that probably erased differences in the outcome.
Dose modifications in patients with renal dysfunction.
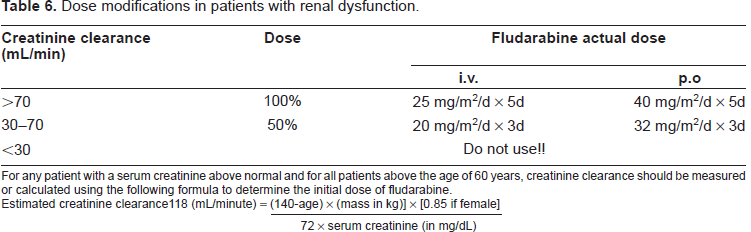
For any patient with a serum creatinine above normal and for all patients above the age of 60 years, creatinine clearance should be measured or calculated using the following formula to determine the initial dose of fludarabine.
Estimated creatinine clearance118 (mL/minute) =
Another Italian multi-center study investigated FCR regimen that was found as the most promising treatment in CLL.63,64 Following 4 cycles of FCR, excellent rates of response (74% CR and 84% molecular remission) were obtained among 54 patients with recurrent follicular lymphoma. At 45 months of follow-up the median OS and DFS had not been reached.
The German Low Grade Lymphoma Study Group prospectively compared FCM plus rituximab (FCM-R) with FCM alone in 128 patients with relapsed or refractory follicular, lymphoplasmacytic or mantle cell lymphoma, mostly post CHOP therapy. 65 The addition of rituximab significantly improved response rates: 79% OR and 33% CR in FCM-R arm as compared with 58% OR and 13% CR, respectively, for FCM alone (p = 0.01), with similar advantage in a subgroup analysis of follicular lymphoma (OR 94% vs. 70%) and MCL (58% vs. 46%, respectively). The R-FCM was significantly superior concerning PFS (p = 0.038) and OS (p = 0.003). There were no differences in the side effects in both study arms. This was the first prospective randomized trial to demonstrate that combined immunochemotherapy in patients with relapsed indolent and mantle cell lymphomas is superior to chemotherapy alone, both in terms of response and survival rates. The further trial of the German Group revealed significant prolongation of response period by rituximab given as maintenance therapy in patients with relapsed or refractory indolent lymphoma and MCL, either or not they were previously treated by rituximab. 66
Radioimmunochemotherapy
The radionuclide applied in the radioimmune therapy has additive cytotoxic effect. Better results are obtained in patients with low tumor burden and non-severe bone marrow involvement. Leonard et al provided tumor mass reduction by an abbreviated course (3 cycles of fludarabine), followed by radioimmunotherapy with unlabeled anti-CD20 monoclonal antibody tositumomab and finally radiolabeled anti-CD20 monoclonal antibody iodine 131 tositumomab. 67 In this study on 35 patients with untreated follicular lymphoma fludarabine alone induced 89% response and 9% CR, and the full regimen—100% response with 86% CR. Ten out of 13 cases with baseline bone marrow bcl-2-positivity demonstrated molecular remission. The estimated 5-year PFS rate was 60%.
Fludarabine in Different Lymphoma Subtypes
The response rates were generally higher in patients with follicular lymphoma than in those with other subtypes, with the exception of follicular large-cell lymphoma (histological grade 3) or transformed follicular lymphomas. The results with fludarabine were worst in the diffuse large-cell lymphoma. 9 MCL is characterized by especially low CR rate to therapy and bad prognosis. 68 Fludarabine monotherapy demonstrated limited efficacy in MCL inducing 33% PR rate with short (4 to 8 months) responses in a study on 15 patients, in majority pre-treated. 69 In previously untreated patients the results were more encouraging (OR rates of 40%-70% with 30% CR),50,70 nevertheless, still not satisfactory. A number of small studies have investigated the efficacy of fludarabine-combined regimens in patients with MCL, usually as a subset of the entire cohort. The combination with mitoxantrone, idarubicin or cyclophosphamide produced OR rate of 60%-80% and CR rate of 20%-40% in either untreated or recurrent MCL patients.44,46,50 Although rituximab as a single agent in MCL revealed limited efficacy, in the setting of immunochemotherapy the results were significantly better compared to chemotherapy alone. 64
Several reports were dedicated to fludarabine therapy in lymphoplasmacytic lymphoma and its counterpart, Waldenstrom macroglobulinemia (WM), a clinicopathologic syndrome characterized by monoclonal immunoglobulin IgM production. Fludarabine salvage therapy induced response in a third of patients resistant to previously administered treatments.71–73 Despite a low rate of qualitative remissions, fludarabine provided useful palliation in this incurable disease. Furthermore, in a comparative trial on 90 patients with first relapse or with primary refractory WM, fludarabine was superior to cyclophosphamide-doxorubicin-prednisone (CAP) regarding response rates (30% vs. 14% PR) and duration (19 months vs. 3 months). 74 Fludarabine has been further studied as front-line treatment of WM: a European multicenter phase II study on 20 patients showed OR in 79%, with a median PFS of 40 months. 70 However, another large SWOG trial on 182 patients with symptomatic or progressive WM showed lower response rate (36% with only 2% CR). A serum beta2m level of ≥3 mg/L was the most significant adverse prognostic factor for survival in this study. 75
The success of fludarabine combinations in other lymphomas prompted similar studies also in WM.76,77 The best response rate of 95% was achieved by fludarabine and rituximab in 43 untreated or with <2 prior therapies patients. 77 The median PFS was 51 months for all patients and 77 months for those previously untreated. However, relatively high rate of short term toxicities (6 pneumonias including 2 fatal) and long-term complications (3 cases of transformation to aggressive lymphoma and 3 cases of MDS/AML) should be evaluated in further studies.
High response rates (OR 91% and CR 53%) were achieved in marginal zone lymphoma in a Greek trial evaluating FM combination followed by rituximab consolidation. At 39 months of median follow-up, 74% patients were alive and disease-free. 78
Finally, successful treatments with fludarabine in T-cell lymphomas, including mycosis fungoides, peripheral T cell lymphoma and angioimmunoblastic lymphadenopathy were occasionally reported.79–82
Toxicity
Myelotoxicity and Infection
The main toxic events in fludarabine treatment are associated with myelosuppression manifested by transient granulocytopenia, and immunosuppresion related to profound decrease in lymphocytes. 83 The depletion in CD4 cells may persist for more than a year, while other lymphocyte populations recover more rapidly. The risk for infectious complications increases during the periods of treatment-related granulocytopenia, and the wide spectrum of infections includes bacterial and various opportunistic organisms. The most encountered are Streptococcus pneumoniae, Staphylococcus and Haemophilus influenzae, whereas gram-negative bacteria are common reason for hospitalization. Early studies with fludarabine have been associated with very high rate of complicated granulocytopenia and infections. For example, in 76 patients with relapsed lymphoma Redman et al diagnosed 23 episodes of fever of unknown origin, 23 cases of bacteremia, 3 of systemic candidiasis, 2 of PCP, one of each aspergillosis pneumonia, Torulopsis glabrata infection and disseminated cytomegalovirus, and 8 patients with localized HZV. 23 Poor performance status, elderly age, advanced or refractory lymphoma, concurrent corticosteroids and prolonged leukopenia predispose to infections. Restriction of concurrent corticosteroids, appropriate prophylactic use of cotrimoxazole and in special cases also acyclovir and antifungal drugs, prophylaxis by myeloid growth factors, doses reduction for elderly patients prevent many infectious events. However, these recommendations are mostly empiric and may be excessive for low-risk patients. PCP prophylaxis might not be required at least in younger patients receiving first-line chemotherapy. 84
Finally, cases of hepatitis B virus reactivation and acute hepatitis were diagnosed following fludarabine therapy. 85 Patients who are positive for HBsAg or HBcoreAg should be treated with lamivudine for the entire duration of chemotherapy and for 6 months afterwards and monitored with liver function tests and hepatitis B virus DNA.
Neurotoxicity
Recommended doses of fludarabine for treatment of lymphomas very rarely cause neurological effects. Nevertheless, paralysis, neuropathy, blurred vision, seizures or headache were occasionally reported. 86 Progressive multifocal leucoencephalopathy (PML) associated with John Cunningham virus was described in lymphoma patients treated by fludarabine.63,87 Elderly patients are more prone to neurotoxicity.
Impaired Stem Cell Collection
Younger patients with indolent lymphoma commonly become candidates for stem cell autotransplantation. It was shown that fludarabine may impair collection of peripheral blood progenitor cells destined for autotransplant in acute leukemia and CLL. 88 This observation could complicate the decision to choose this drug for patients who may require autotransplant in the future. However, at least a part of unsuccessful stem cells mobilizations in the reported lymphoma cases was performed in unsatisfactory circumstances: either too late in the course of lymphoma, or too soon after fludarabine treatment and/ or with myeloid growth factors alone. 89 The data collected by European Blood and Marrow Transplantation (EBMT) CLL subcommittee on 41 patients who had previously been treated by fludarabine, was encouraging. 90 The numbers of CD34+ cells collected at least 2 months elapsed between the last dose of fludarabine and the start of mobilizing therapy, was satisfactory. More CD34+ cells were collected in the patients who were treated with fewer than six courses of fludarabine compared to ≥6 courses and in patients who had received less chemotherapy before the mobilization. All 16 patients who were autotransplanted, engrafted.
The collection of stem cell early enough in the course of disease, the delay in stem cell collection for at least 2-3 months after fludarabine administration and the combination of chemotherapy and myeloid growth factors for stem cell mobilization can improve the quality of stem cell harvest.
Secondary Malignancy
There is data suggesting that fludarabine may increase the risk of secondary malignancies, particularly when combined with other DNA-damaging agents, such as alkylators or topoisomerase II inhibitors.91–93 The estimated rate for MDS/secondary AML reach 3%-4% for lymphoma patients treated with fludarabine-based chemotherapy given as first-line treatment and up to 10% for previously heavily treated patients. The complex cytogenetic aberrations and abnormalities of chromosomes 5 and/or 7 were common. The clinicians should discuss this issue with their (particularly younger) patients before the treatment administration.
Other Events
Fludarabine induces little significant nausea, vomiting and alopecia. Tumor lysis syndrome associated with fludarabine treatment has been documented, usually in CLL and occasionally in lymphoma patients, so prophylactic measures should include hydration and allopurinol. 94 Fludarabine has been implicated in the development or worsening of autoimmune hemolytic anemia (AIHA) and thrombocytopenia (ITP) in CLL patients. 95 The administration of fludarabine needs caution when hemolytic anemia or acute thrombocytopenia have been diagnosed. The combination of fludarabine and cyclophosphamide and/or rituximab appears to reduce the incidence of autoimmune cytopenias. 96 Finally, transfusion associated graft-versus-host-disease after fludarabine treatment has been noted, so the use of irradiated blood products for transfusion following treatment is mandatory. 97
Oral Fludarabine
The oral formulation of fludarabine phosphate has been available since early 2000s. The 10 mg tablet contains an immediate-release form of the drug. Pharmacokinetics studies demonstrated that a once-daily oral dose of 40 mg/m2 corresponded to i.v. exposure of 25 mg/m2. 98 F-ara-A reaches peak plasma concentration following an oral dose in a 1 1/2 hour. The bioavailability of the oral drug ranges from 30% to 80% between patients, while the intra-individual variations in bioavailability are low and not affected by meal. 99
A phase II multi-center study was conducted in Japan to assess the efficacy of oral fludarabine in 46 patients with indolent lymphoma (41 follicular) and 6 with MCL, previously treated by chemotherapy with or without rituximab. 100 The OR rate in the cohort of the indolent lymphoma patients was 65% with 30% CR, the responders showed median time to treatment failure of 12 months. Only one patient with MCL experienced an objective response, the others 5 had stable disease through the treatment period.
The comparison of oral fludarabine alone or in combination with cyclophosphamide to results in similar patients treated by intravenous administration, showed same clinical efficacy. Similar tolerability profile was also found, except more gastrointestinal adverse events for the oral formulation.101,102
Fabbri et al used reduced dosages of oral fludarabine (40 mg/day) and oral cyclophosphamide (150 mg/m2/day) (for 4 days and 4 cycles) in untreated elderly patients with indolent lymphoma. 103 The OR rate was 84% (CR 40%) with median PFS of 20 months.
In conclusion, the oral formulation of fludarabine is effective and attractive for oncologists who do not need the hospital day care facilities and also for patients who benefit from home conditions.
An Italian trial used this drug together with mitoxantrone and radioimmunotherapy (yttrium-90 radiolabeled antibody zevalin) in untreated patients with advanced indolent lymphoma. 104 When treatment accomplished, 55 out of 57 patients achieved CR, an estimated 3-year PFS was 76% and OS 100%.
Discussion regarding the Choice of Treatment for Lymphoma Patients
Clinical evidence from studies in patients with lymphoma suggests that fludarabine monotherapy is at least as effective, if not better, than other chemotherapy such as CVP and CHOP-regimens in follicular and other types of indolent lymphomas and carries acceptable toxicity rate. At the contrary, fludarabine is not suitable for large-cell lymphoma. In comparative studies, fludarabine combined with cyclophosphamide and/or mitoxantrone improved the remission rate and duration when compared with single-agent fludarabine in both previously treated and untreated patients with indolent lymphoma. Finally, it is currently agreed that immunochemotherapy approach produces superior results to chemotherapy alone, whereas rituximab serves as the most effective and safe monoclonal antibody for the combinations. The question that still remains unresolved, which is the most effective combination chemotherapy regimen to use along with rituximab? The main candidates are CHOP and fludarabine-based regimens. Unfortunately, there is no proof that any of the known chemotherapy regimen determines prolonged overall survival. As a result, no established recommendations for treatment of choice exist in indolent lymphoma. In the near future the results of the ongoing European intergroup study PRIMA (Primary Rituximab and Maintenance) that evaluates differences in the efficacy between different upfront R-chemo regimens (R-CVP, R-CHOP, R-FCM), as well as the potential impact of rituximab maintenance may shed light on this issue.
Currently the U.S. Guidelines from the National Comprehensive Cancer Network (NCCN) recommend various options as the first-line treatment of follicular lymphoma including: CVP and CHOP-regimens, fludarabine as a single agent or fludarabine-based regimens, all of them combined with rituximab; radioimmunotherapy or rituximab as a single therapy. 105 Rituximab maintenance after first-line therapy is recommended for further investigation following favorable results in pretreated patients.66,106
For the elderly patients rituximab alone or a single alkylator agent may be selected. For the subsequent treatments bendamustine, chemoimmunotherapy as described for initial therapy, radioimmunotherapy, autologous or allogeneic SCT in selected patients can be used. Fludarabine as a component of combination therapy is also a recommended option for first-line therapy in small lymphocytic and marginal cell lymphoma and as second-line in MCL.
Data from the National LymphoCare survey in the United States demonstrated that lack of specified recommendations weakens the tailoring of the most efficient treatment program. Currently only few patients with follicular lymphoma are treated on clinical trials, whereas the clinicians use diverse regimens based on their personal preference. 107 Approximately half (49%) of them utilized chemoimmunotherapy, the others—“wait and watch” strategy (19%), rituximab monotherapy (13%), chemotherapy alone (4%) or radiation alone (5%). The CHOP-R and CVP-R regimens were favored (78%) when immunochemotherapy was administered, while combination of fludarabine-based regimen with rituximab was selected in 11%. The survey showed also significant differences in physicians’ preferences in various medical centers and regions suggesting absence of a universally recognized policy. For example, initial watch and wait was used in 31% of patients in the Northeast but in 13% in the Southeast, whereas fludarabine was used in 18% of patients in the Southwest and only 3% in the Northeast. M. Czuczman entitled his article “Controversies in follicular lymphoma: “Who, what, when, where, and why?” (Not necessarily in that order!)”, and emphasized that achievement of complete hematological and molecular remission has been associated with improvement in outcome. 108 His main practical conclusion: “suboptimal therapy in = suboptimal results out”.
What are the therapy preferences in our Hematology center? The goal of therapy for the younger patients with follicular lymphoma, requiring chemotherapy, is to achieve the best qualitative response and thus to prevent as possible relapse of the disease. We generally choose combination of fludarabine, cyclophosphamide and rituximab (FCR) as one of the most active regimens that demonstrates the highest clinical and molecular response rates. In cases with massive organomegaly we usually choose CHOP-R regimen because of possibility of hidden transformation of the disease. As known fludarabine-containing regimens are poorly effective in cases with histologic transformation as well as in follicular large-cell lymphoma. In these settings, anthracycline-containing combinations are superior.109,110 The first best response to chemotherapy we use for autologous stem cell harvest (Fig. 1), that can be utilized in a subsequent relapse. We believe that with cautions that were discussed above, successful stem cell harvesting can be performed in most patients in remission.
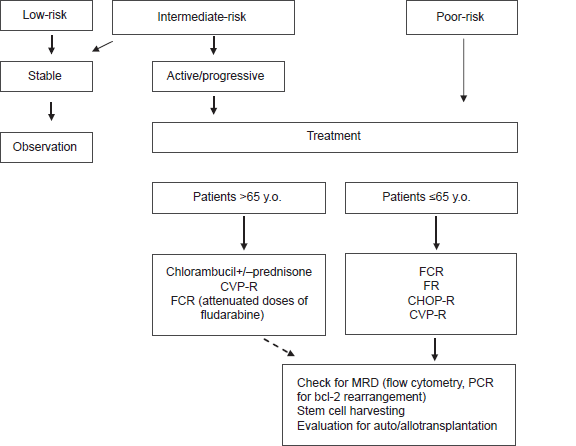
The proposed management of patients with follicular lymphoma.
In the elderly patients in addition to the decrease of tumor mass, the main goal of treatment is the maintenance of a good quality of life, rather than complete eradication of clinical and laboratory evidence of disease. Factors influencing survival in elderly population, apart from lymphoma parameters and prior chemotherapy, include performance status, underlying comorbidities, socioeconomic status and availability of social support. Chlorambucil (with or without prednisone) as an oral, well-tolerated and inexpensive drug is a reasonable first-line option in fragile patients. Combination of rituximab with COP or fludarabine in lower doses, which appears to be still active and has fewer associated toxicities than used in standard dose, may be an alternative. We administer fludarabine in a half of the standard cycle dosage and discontinue therapy when a satisfactory clinical response is achieved.
In conclusion, immunochemotherapy based on fludarabine-including regimens exhibits a high efficacy in the treatment of indolent and particularly follicular lymphoma.
Authors’ Declaration
The article is original, has not been published in any other journal, is not under consideration by any other journal and does not infringe any existing copyright or any other third party rights.
We are the sole authors of the article and have full authority to enter into this agreement and in granting rights to Libertas Academica are not in breach of any other obligation.
The article contains nothing that is unlawful.
The authors have no competing financial interests.
To our and currently accepted scientific knowledge all statements in the article are true.
Footnotes
Appendix
Clinical trials using fludarabine currently conducted at the National Cancer Institute (available at: www.cancer.gov).
Phase III randomized study of fludarabine phosphate and cyclophosphamide with versus without rituximab in patients with previously untreated mantle cell lymphoma.
Rituximab after induction chemotherapy (R-CHOP vs. R-FC) followed by interferon maintenance versus rituximab maintenance in mantle cell lymphoma.
Phase III randomized study of first-line therapy comprising chlorambucil versus fludarabine phosphate in patients with previously untreated Waldenström macroglobulinemia, splenic lymphoma with villous lymphocytes, or non-IgM lymphoplasmacytic lymphoma.
Multicentric study, three randomized arms (R-CVP vs. R-CHOP vs. R-FM) for patients with stage II-IV follicular lymphoma [in Italy].
Phase I–II study of rituximab, fludarabine, and cyclophosphamide followed by radioimmunotherapy comprising rituximab and yttrium-90 ibritumomab tiuxetan in patients with relapsed indolent, mantle cell, or transformed CD20-positive B-cell non-Hodgkin's lymphoma.
Phase I–II study of haploidentical allogeneic aldesleukin-activated natural killer cells and aldesleukin following chemoimmunotherapy comprising rituximab, fludarabine, and cyclophosphamide in patients with CD20-positive relapsed non-Hodgkin lymphoma or chronic lymphocytic leukemia.
Rituximab, fludarabine, mitoxantrone, dexamethasone (R-FND) plus zevalin for high-risk (untreated) follicular lymphoma.
Bortezomib (Velcade) with standard chemotherapy (FMD-R) for relapsed or refractory follicular lymphoma.
