Abstract
B1 or Tositumomab was the first B-cell specific antibody to be discovered and it targets the CD20 antigen. The potential therapeutic importance of this discovery in targeting CD20, however remained unrealised until the mid 1990's when Tositumomab was radiolabelled and the 131I Tositumomab radioimmunotherapy (RIT) regimen (Bexxar™) developed. The 131I Tositumomab regimen is completed within one to two weeks and consists of a tracer dose of the radioimmunoconjugate followed by the therapeutic dose 7 to 14 days later. Each infusion of 131I-tositumomab is preceded by an infusion of a pre-dose of 450 mg “cold” or unlabeled tositumomab. 131I Tositumomab has demonstrated remarkable clinical activity in patients with relapsed follicular lymphoma with high response rates and durable remission even in patients with disease that is refractory to chemotherapy and rituximab antibody therapy. Recent new data has provided new insights into the potential mechanisms of the antibody and targeted radiation effects and these as well as the safety and efficacy of this novel therapy in follicular lymphoma are reviewed.
Introduction
Following the advent of monoclonal antibody (mAb) technology in the 1970's, there was a great expectation that mAb would provide effective targeted therapy for cancer, however it took nearly 30 years for mAb to fulfil that early promise as effective anti-cancer therapeutics. 1 The first B-cell specific antibody B1, later renamed Tositumomab, was discovered in 1981 by Nadler and colleagues. 2 The B1 or Tositumomab was found to target the B cell specific antigen that we now know as the CD20 antigen. The potential therapeutic importance of this discovery in targeting CD20 however remained unrealised until the late 1990's when Rituximab, another mAb directed against the CD20 antigen, became the first mAb to be approved by the US Food and Drug Administration (FDA). This provided not only a significant milestone in the history of targeted therapy but rituximab has revolutionized the treatment of a wide range of B cell malignancies when combined with chemotherapy, leading to significant improvement in outcome.3–6
Radioimmunotherapy (RIT) is a conceptually appealing approach for cancer treatment whereby the conjugation of radioisotope to mAb enables the delivery of targeted radiotherapy in addition to the specific cytotoxic effects of the mAb. Over the last few years RIT has been shown to demonstrate vastly superior clinical responses to unlabelled anti-CD20 mAb7–13 A wide variety of different mAb, delivery schedules, radioisotopes, and doses of radioactivity have been used in RIT and have resulted in impressive durable responses following the US FDA approval of two RIT drugs namely 90Y Ibritumomab tiuxetan (Zevalin®; Cell Therapeutics, Seattle, WA; Bayer Schering Healthcare, Berlin, Germany) and 131I-Tositumomab (Bexxar®, GlaxoSmithKline, Philadelphia, PA). This review focuses on the safety and efficacy data of 131I-Tositumomab.
Mechanisms of Action of 131I Tositumomab
Predose
In order to improve the biodistribution of radiolabelled mAb in RIT, a pre-dose of “cold” or unlabeled anti-CD20 mAb is given prior to the therapeutic dose of the anti-CD20 radioimmunoconjugate.11,14 For the established 131I-tositumomab regimen, 450 mg unlabeled pre-dose of tositumomab is given prior to 35 mg of 131I-tositumomab. 14
The pre-dose is considered to prolong the circulating half-life of the radiolabelled mAb, block “non-specific” binding sites (e.g. circulating and splenic B-cells) and result in increased tumor retention of the labelled mAb. Buchsbaum et al investigated whether a pre-dose of anti-B1 improved the delivery of a subsequent radiolabelled mAb to tumor using in vivo preclinical human xenograft models. 15 The anti-B1 (anti-CD20) pan-B-cell mAb, reactive with human B-cell lymphomas, but not with host mouse B-cells was used. A pre-dose of unlabelled anti-B1 was found to significantly increase the tumor-uptake of the subsequent radiolabelled anti-B1 by blocking the B1 specific receptor sites, although this improvement in tumor targeting appeared to plateau at the highest pre-doses of unlabeled anti-B1.
More recently the use of predose in the rituximab era has been questioned by Sharkey and colleagues. 16 In contrast to the pioneering studies outlined above the vast majority of patients who are currently suitable for RIT will have received rituximab. Therefore an important question in current clinical practice is whether the predose of cold mAb is required for example, when 131I-Tositumomab is given as consolidation therapy after rituximab containing chemotherapy. This issue comes into sharper focus, if as suggested by Sharkey and colleagues, repeated doses of rituximab may prevent subsequent binding of radiolabeled anti-CD20 antibody to tumor sites, potentially compromising tumor targeting and clinical efficacy.
Sharkey and colleagues cite recent preclinical evidence supporting the view that rituximab might if given in higher enough doses block the binding of the anti-CD20 radioimmunoconjugate in a xenograft model.17,18 The animal models used in these studies were human Burkitt lymphoma cell lines inoculated into nude mice. In such xenograft models there is no cross-reaction of the predose targeting the human CD20 and the host normal B-cell reservoir. As such in these xenograft models there is a finite antigen sink that is entirely limited to the volume of the small human tumors in these models and it is not surprising that this can be saturated with large enough doses of rituximab. Preclinical models with syngeneic tumors appear preferable to address this “proof of principle” questions, 18 but ultimately confirmatory clinical studies should be done.
Currently there is a lack of evidence from clinical studies that prior rituximab does in fact compromise subsequent anti-CD20 based RIT. In stark contrast to the preclinical data, recently published clinical data addressing this question found that induction therapy with rituximab significantly increases the effective half-life of subsequent RIT with 131I-rituximab and that high serum levels of rituximab after induction therapy correlate with increased effective half-life of the 131I-rituximab with impressive therapeutic efficacy. 19
Tositumomab antibody effector mechanisms
With regard to mAb induced tumor cell killing, four potential mechanisms are thought to play a role and these include, (1) complement dependent cytotoxicity (CDC), (2) antibody dependent cellular cytotoxicty (ADCC) via the recruitment of immune effector cells, (3) direct induction of growth inhibition and/or cell death and (4) stimulation of host-adaptive immunity. 20 Large amounts of experimental data support the three former mechanisms in anti-CD20 mAb immunotherapy, although robust evidence for the latter is currently lacking. In particular, several lines of evidence indicate that rituximab operates through conventional effector mechanisms involving complement and immune effector cells. 21 More recently, data has emerged demonstrating that type I anti-CD20 mAb such as rituximab and 1F5 require presence of their Fc domains for optimal therapeutic activity in human lymphoma xenograft models. 22
Cragg et al have previously shown that anti-CD20 mAb may be sub-divided into either (Type I) rituximablike or (Type II) which are Tositumomab like according to their linked activity in a number of in-vitro assays. For example, rituximab and other Type I mAb redistribute CD20 into Triton X-100 insoluble membrane rafts, correlating with their ability to engage complement effectively and cause target cell lysis. 22 In contrast Type II mAb such as Tositumomab do not redistribute CD20 membrane rafts, but are generally potent at inducing cell death in target cells.23,24 Importantly, these differences appear to translate to the in vivo mechanisms employed by these mAb, at least in xenograft tumor models. 22
Binding of anti-CD20 mAb to lymphoma cells in vitro has been shown to induce modest levels of cell death presumably via signalling through the CD20 molecule. 25 A range of signalling events may be induced following ligation of CD20, 23 including activation of the mitogen-activated protein kinase (MAPK) cascade. 26 The Ras–Raf–MEK–ERK1/2 pathway is an evolutionary conserved pathway that is involved in the control of many fundamental cellular processes that include cell proliferation, survival, differentiation, cell death, motility and metabolism. 27 Although commonly thought of as a component of proliferation and survival pathways, ERK1/2 signalling has been associated with apoptotic signalling in immature B cell lymphoma 28 and diffuse large B cell lymphoma cells. 29 Similarly, it has been suggested that the overall balance of MAPK activity may determine B cell fate depending on the kinetics of activation and maturation state of the cell.30,31
Intrigingly, the MAPK cascade is also triggered by radiation and other DNA damaging agents32,33 where it may be necessary for DNA double strand break repair by homologous recombination. 34 Given the observation that both anti-CD20 mAb and irradiation (IRR) can trigger MAPK activation the impact of combining radiation with Type I (rituximab) or type II (B1/Tositumomab) anti-CD20 mAb has been investigated. Interestingly increased cell death was observed with Tositumomab combined with IRR but not with rituximab. This additive cell death was found to be MAPK/ERK kinase signalling dependent and could be reversed with pharmacological MEK inhibitors as well as siRNA targeting MEK1/2. Although Bcl-2 over-expression resulted in resistance to IRR induced apoptosis, it had no impact on the cell death induced by Tositumomab plus IRR, suggesting a non-apoptotic cytoplasmic form of cell death. This data provide new clinically relevant insights into how RIT with Tositumomab may causes additive cell death that can overcome apoptosis resistance. 35
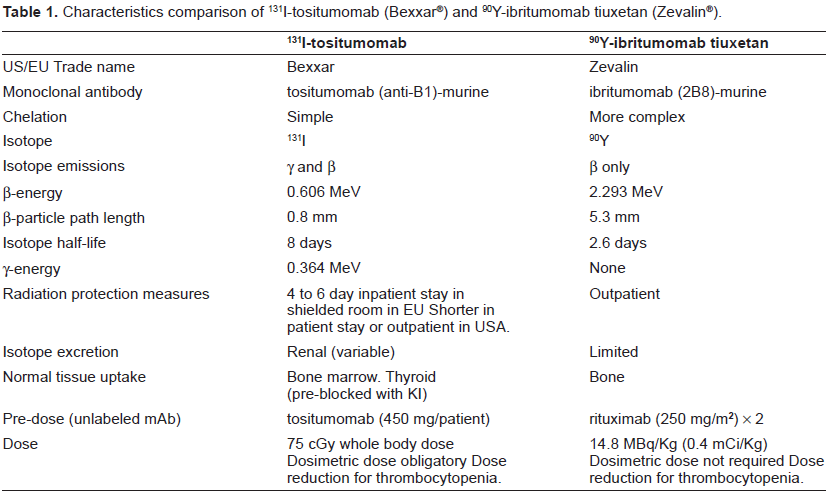
The 131I-tositumomab regimen is completed within one to two weeks and consists of a tracer dose of the radioimmunoconjugate followed by the therapeutic dose 7 to 14 days later. Each infusion of 131I-tositumomab is preceded by an infusion of a pre-dose of 450 mg “cold” or unlabeled tositumomab and the therapeutic regimen is outlined in Figure 1. Whole body gamma camera imaging is performed three times over the week following the trace labelled infusion to calculate the whole body half-time and the dose required for the therapeutic infusion to deliver a 65 to 75 cGy whole body dose (WBD) (usually 3700 to 5550 MBq (100 to 150 mCi). Dose adjustments to 65 cGy are made for a baseline platelet count of 100 000/mm3 to < 150 000/mm3 and for obesity. These characteristics and differences to the competitor drug 90Y Ibritumomab tiuxetan are highlighted in Table 1.
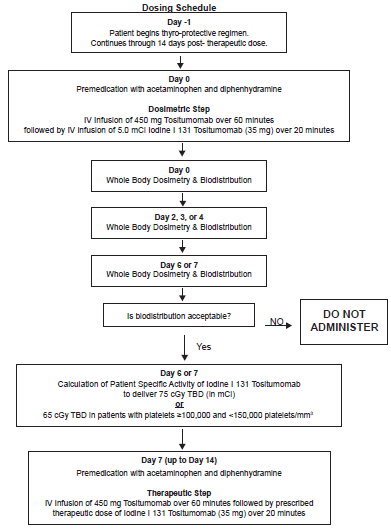
Treatment regimen for 131I-tositumomab.
Characteristics comparison of 131i-tositumomab (Bexxar®) and 90Y-ibritumomab tiuxetan (Zevalin®).
Kaminski and colleagues initially conducted a series of trials at the University of Michigan using 131I-tositumomab, in the treatment of relapsed FL.11,36 In a pivotal study, 60 extensively pre-treated patients were given a single administration of 131I-tositumomab. 37 Disease responses were compared to their previous responses to chemotherapy for follicular or transformed FL. A PR or CR was observed in 39 patients (65%) after 131I-tositumomab, compared with 17 patients (28%) after their last qualifying chemotherapy (P < 0.001). 131I-tositumomab therapy was shown to provide greatly superior relapse free survival compared to the last qualifying chemotherapy. Since 1990 well over 1000 patients with “low grade” and transformed lymphoma have been treated with 131I-tositumomab. Long-term follow-up data was published by Fisher et al. 38 who performed an integrated efficacy analysis of five clinical trials of 131I-tositumomab in 250 patients with relapsed or refractory follicular, or transformed FL and the durability of these responses are discussed below. Impressive response rates have also been seen in patients that were refractory to rituximab which were subsequently treated with 131I-tositumomab. In another important study high efficacy was seen in a heavily pretreated (median number of 4 prior therapies) group of follicular lymphoma patients who were refractory to rituximab, and 59% were chemo-refractory, Importantly the ORR (65%) and CR (38%) rates were not significantly associated with prior rituximab response. A median PFS of 24.5 months was seen for the responders. Patients with follicular grade 1 or 2 histology and tumors ≥ 7 cm achieved very high OR (87%) and CR (57%) rates. 39 A further analysis was performed which included 230 patients treated with 131I-tositumomab. Durable CR were noted with similar frequency in patients with rituximab-refractory disease (28%) and rituximab naïve patients all of which had chemo-refractory disease (23%). With a median follow-up of 4.6 years, 75% of patients with durable CR continue in complete remission. 40
Frontline Treatment of Follicular Lymphoma Using 131I Tositumomab
Given the high efficacy and relatively low toxicity of RIT in relapsed disease (the licensed indication), it was logical to investigate the use of RIT in first line treatment of follicular lymphoma.
The use of 131I Tositumomab as initial treatment for follicular lymphoma was investigated in a single centre phase II trial with a total of 76 patients. 41 Patients all had stage III or IV disease and less than 25% bone marrow involvement. They received a dosimetric dose of 131I Tositumomab followed by therapeutic dose of 131I Tositumomab a week later. The patient group was then assessed for response rates, progression free survival and toxicity. The bone marrow was assessed at 6, 12, and 25 weeks for rearrangement of the BCL2 gene and myelodysplastic syndrome. Serial serum samples were also assessed for human anti-mouse antibodies. The OR was 95% and 75% of patients had a complete response. After a median follow up period of 5.1 years, the PFS for all patients was 59%, with a median PFS of 6.1 years. Of those who had a CR, 80% showed molecular response, and 70% had a progression free survival for 4.3-7.7 years. The regimen was well tolerated, with no patients requiring haematological support, and no cases of myelodysplastic syndrome were reported. 63% patients developed human anti-mouse antibodies, and those with levels higher than five times the lowest level of detection had a 35% PFS, compared with 70% in the remaining patients (P = 0.003).
Contribution of Targeted Radiation to Clinical Response
The contribution of targeted radiation to the overall responses seen with 131I Tositumomab has been addressed using dosimetric methods 42 and also in a randomized study which compared treatment outcomes for unlabeled tositumomab (pre-dose) and 131I-tositumomab to an equivalent total dose of unlabeled Tositumomab. The randomized study involved 78 patients with refractory/relapsed low-grade NHL and an ORR of 55% versus 19% (P = 0.002) with a CR 33% versus 8% (P = 0.012) in 131I-tositumomab versus unlabeled tositumomab groups respectively was reported. 43 The median duration of the ORR was not reached for 131I-tositumomab versus 28.1 months for unlabeled tositumomab. The median duration of CR was not reached in either arm and the median time to progression was 6.3 versus 5.5 months (p = 0.031), respectively. Although hematological toxicity was more severe and non-hematological AEs were more frequent after 131I-tositumomab than after tositumomab alone, there were no serious infectious or bleeding complications. The frequency of developing human anti-mouse antibodies (HAMA) was similar in the two arms at 27% (131I-tositumomab group) versus 19% (tositumomab alone group), respectively. This study demonstrated that although unlabeled tositumomab showed single agent activity, the conjugation of iodine-131 to tositumomab significantly enhanced the therapeutic efficacy and improved responses were observed in the cross over part of the study whereby patients who had failed to respond to unlabelled Tositumomab subsequently responded to 131I Tositumomab.58
Defining Whether a Tumor Radiation Dose-response Exists with 131I Tositumomab
The Michigan group has concluded that there could be a radiation dose-response for 131I-tositumomab, 42 however this remains controversial and opinions vary.44,45 Goldenberg and colleagues for examples commented that as RIT has two potentially therapeutic arms, namely radiation and mAb mechanisms, poor radiation targeting does not exclude a good therapeutic response from the mAb. 45
Durability of Clinical Remissions after 131I Tositumomab
Perhaps the most impressive finding to emerge from these maturing data using RIT in FL, is the remarkable duration of response enjoyed by some patients. Fisher and colleagues performed an analysis of the long term follow-up of patients treated with 131I-tositumomab. 38 The ORR was 47%-68%, with CR rates of between 20% and 38%. At a median follow-up of 5.3 years, 5-year PFS was 17% and eighty-one (32%) of 250 patients had a time to progression of greater than or equal to 1 year. These were termed the durable response population. For durable response population, 44% had not progressed at greater than or equal to 2.5 to greater than or equal to 9.5 years, with a median duration of response of 45.8 months and impressively the median duration of those patients who had achieved a CR, had not been reached. Interestingly many of the patients who enjoyed these long durable responses had many poor prognostic characteristics, including bone marrow involvement (41%), bulky disease > or = 5 cm (49%), and transformed histology (23%). Forty-three percent of the patients had been treated with more than four prior therapies and 36% had not responded to their most recent therapy.
Side effect profile of 131I Tositumomab
Non-hematologic toxicities following treatment with 131I Tositumomab are relatively uncommon and generally minor. The most common, clinically significant, non-hematologic toxicities are shown in Table 2 and a comparison made between 131I Tositumomab and 90Y Ibritumomab. Myelosuppression is the primary toxicity with neutropenia and thrombocytopenia occurring several weeks after treatment and recovering to pre-treatment levels after 2-3 months. Among 995 patients treated with 131I tositumomab, higher number of prior therapies, lower baseline blood counts, prior fludarabine, and extent of bone marrow involvement with lymphoma were significant predictors of grade 3-4 hematologic toxicity. 46
Toxicity.
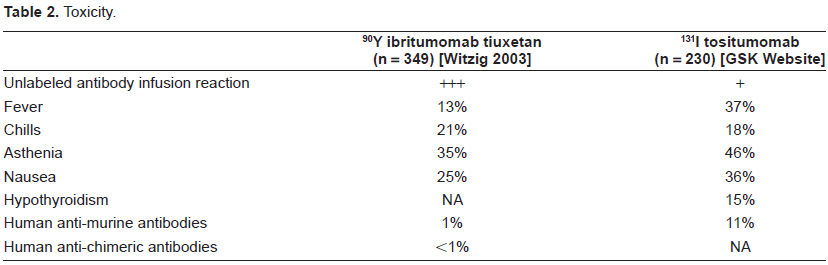
Although concern has been raised about the risk of these patients subsequently developing therapy-related myelodysplastic syndrome and/or acute myelogenous leukemia (tMDS/tAML), the actual reported incidence has been relatively low and not substantially different from that expected among similarly treated patients that have not received radioimmunotherapy. Among 995 patients with relapsed or refractory, indolent, follicular, or transformed B-NHL, who received a single dose of 131I tositumomab, with a median follow-up of 6 years from diagnosis and 2 years from radioimmunotherapy, tMDS/tAML was reported in 35 (3.5%) for an annualized incidence of 1.6%. 46 In fact, in an independent blinded review of the peripheral blood and marrow before and after treatment with 131I tositumomab, only 23 of 985 patients were confirmed to have developed tMDS/tAML after receiving 131I tositumomab for an overall crude incidence rate of 2.3% per year and an annualized incidence of 1.1% per year.
A total of 1071 patients were enrolled in 7 studies: 995 with relapsed/refractory low-grade NHL, +/-transformation (median, 3 prior regimens [range,1-13 regimens]) and 76 patients with previously untreated low-grade follicular NHL. After a median follow-up of 6 years from diagnosis and 2 years from RIT for previously treated patients, and 4.6 years from RIT for previously untreated patients. tMDS/tAML was reported in 35 (3.5%) of 995 patients (annualized incidence, 1.6%/y [95% confidence interval,1.0%-2.0%/y]), and 52% of the tMDS/tAML diagnoses of tMDS/tAML were confirmed in a blinded review (annualized incidence of 1.1%/y [95% confidence interval,0.7%-1.6%/y]). Of the 25 cases, 10 patients (40%) were diagnosed with tMDS/tAML prior to receiving radioimmunotherapy; 2 (8%) had no pathologic or clinical evidence to support such a diagnosis; and 13 (52%) were confirmed to have developed tMDS/tAML following RIT. This incidence is consistent with that expected on the basis of patients’ prior chemotherapy for NHL. 47
The risk of tMDS and tAML appear to be slightly higher when RIT is given after fludarabine based therapies for both 131I Tositumomab 48 and also for 90Y Ibritumomab. 49
Using 131I Tositumomab as Consolidation Therapy after Initial Chemotherapy
Although the results of using 131I Tositumomab are very encouraging and this treatment approach appears an effective alternative treatment option for some patients, future more widespread application is likely to involve integrating RIT into chemotherapy treatment protocols. The current challenge for clinical investigators is to determine the optimal approach of integrating RIT into chemotherapy schedules. Promising data has emerged using 131I Tositumomab as consolidation therapy following either abbreviated or full course chemotherapy. Three Phase II studies have investigated the role of consolidation RIT using 131I after chemotherapy and these are summarised in Table 3.
Phase II trials using 131I Tositumomab as consolidation therapy after initial chemotherapy.
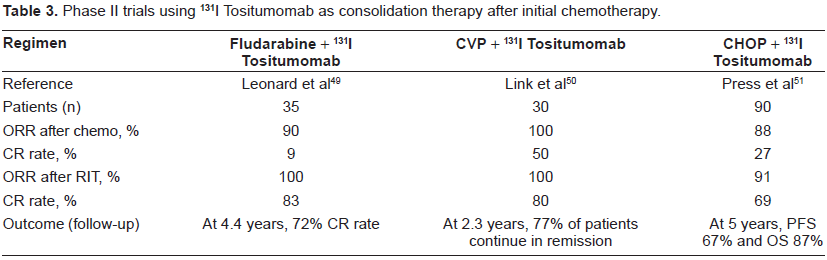
A phase II open-label, multicenter study assessed the efficacy of 131I tositumomab after CVP chemotherapy in 30 patients with previously untreated advanced stage follicular NHL. Six cycles of CVP were given three weeks apart, followed by 131I tositumomab therapy within 56 days. The primary endpoint was overall response. Secondary endpoints were complete response (CR), duration of response (DR), progressionfree survival (PFS), and toxicity. 100% pts responded to CVP chemotherapy. 50% of these achieved CR. Following RIT, 9 out of the 15 patients with a partial response converted from PR to CR, giving a complete response rate after combined chemotherapy and RIT of 80%. After a median follow-up of 2.3 yrs, the median PFS had not been reached, and 77% had duration of responses between 0.6 and 3.4 yrs. Grade 4 neutropenia and thrombocytopenia occurred in 33% and 23% patients respectively, however there were no serious infections. There were no reported cases of development of human anti-mouse antibodies. 50
A further single center phase II trial evaluated thirty-five patients who received an abbreviated course of fludarabine (three cycles given 5 weeks apart), followed 6 to 8 weeks later by 131I tositumomab. The study measured response rates, DR, PFS, OS and toxicity. The patients’ serum was also analysed for human anti-mouse antibodies prior to and after treatment. When assessed after the fludarabine phase of treatment, 89% of patients responded, and 9% achieved CR. After consolidation with 131I tositumomab, OR was 100% and CR was seen in 86%. The median progression-free survival (PFS) after a median follow-up of 58 months had not been reached and was predicted to be longer than 48 months. 5-year estimated progression free survival was 60%. The PFS was shown to be significantly associated with baseline FLIPI (Follicular Lymphoma International Prognostic Index) status (P = 0.003). 83% patients with more than 25% bone marrow involvement at baseline achieved adequate bone marrow response to receive standard-dose 131I tositumomab and 77% patients with baseline bone marrow Bcl-2 positivity had molecular remissions at one year. 29% patients experienced neutropenia after Fludarabine, and this was increased to 74% after RIT. Only 6% patients developed human anti-mouse antibodies (HAMA). 49
The largest of these Phase II studies was conducted by the Southwest Oncology Group (SWOG) and involved 90 patients who received 6 cycles of CHOP. If the initial response to chemotherapy was a PR or CR, this was followed 4-8 weeks later by 131I Tositumomab. All patients had previously untreated advanced stage follicular lymphoma, 23% had masses more than 10 cm at presentation and 62% were stage IV. In total 81 patients received CHOP followed by tositumomab. After a median follow up of 5.1 years, OR was 91% with 69% achieving CR. 5-year PFS was 67% and overall survival (OS) rate was 87%. The 5-year estimates of OS and PFS were each 23% better than the corresponding result for patients treated on previous SWOG protocols with CHOP alone. Regarding toxicity, 12% had grade 4 and 35% grade 3 toxicities, mainly neutropenia and thrombocytopenia during treatment 51 (Table 2).
These highly promising results prompted the SWOG to perform a randomized, multicenter phase III study which is comparing CHOP followed by 131I Tositumomab to 6 cycles of R-CHOP (SWOG 0016/CALG 50102). This study has now completed recruitment and the results are eagerly awaited. These results will be particularly interesting given the recent publication of the FIT study (First line indolent Trial) which provides compelling evidence for the efficacy of 90Y Ibritumomab after induction chemotherapy. In this study patients who were randomized to receive 90Y Ibritumomab tiuxetan (Zevalin®); gained a greater than 3 year improvement in PFS in CR patients and around 2 years improvement seen in the PR patients. 52 However the majority of patients in the FIT study did not receive rituximab containing regimens as part of their initial treatment, which makes it difficult to know what the benefit might have been in the group receiving rituximab containing chemotherapy. The SWOG study will inform us regarding the comparative efficacy of induction treatment with immuno-chemotherapy versus chemotherapy followed by 131I Tositumomab RIT.
Integrating 131I Tositumomab into Treatment Algorithm for Follicular Lymphoma
The high response rates and durable remissions achieved with 131I Tositumomab make this treatment an attractive treatment option for many patients with relapsed FL. The duration of responses seen in patients who achieve a CR are compelling and suggest that the unique mechanism of action of 131I Tositumomab provides a valuable treatment modality in follicular lymphoma. Furthermore these impressive responses are achieved with a treatment that is completed within a week, is very well tolerated and that has minimal predictable and manageable non myelotoxicity toxicity.
There can be no doubting that 131I Tositumomab is highly active drug, arguably the most active “single drug approach” thus far developed for the treatment of Non-Hodgkin lymphoma. However uncertainty, reluctance, and indifference remain in many parts of the hemato-oncology community as to when and how best to integrate RIT into clinical practice. Having been investigated in clinical trials in a few centers in the EU, 53 the drug is no longer available. The algorithm shown in Figure 2 was first suggested in 2004 at the American Society of Hematology educational meeting. Here it was suggested that RIT should be used in first relapse after a rituximab-chemotherapy combination. 54 The difficulty in interpreting this recommendation is the heterogeneity of the duration of remission for patients who relapse after rituximab containing chemotherapy. Certainly for those that relapse within 12-18 months after completion and older patients unsuitable or unwilling to have further chemotherapy, this appears to be an entirely reasonable treatment approach to FL. For younger patients the recommendation would be that consideration is given to progenitor stem cell collection at the time of initial remission in selected patients and thus transplantation is not excluded at a later date (see algorithm). Although data is emerging to suggest RIT can be given after transplantation this is inevitably at lower doses than those for the licensed indication.
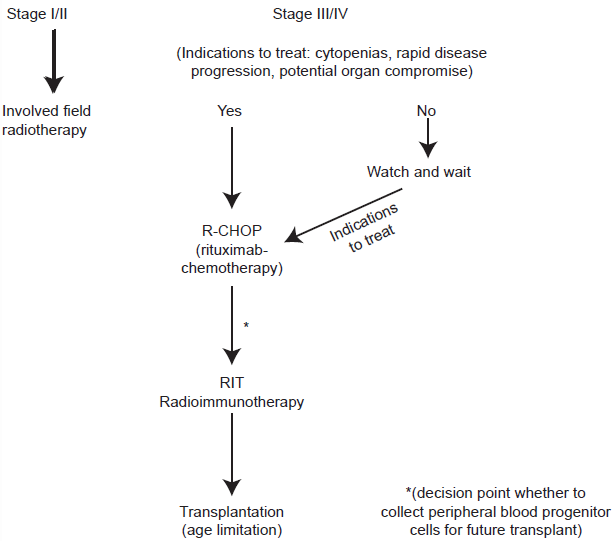
A proposed treatment algorithm for follicular lymphoma patients after relapse first relapse following a Rituximab-chemotherapy combination. 51
In summary the exquisite sensitivity of follicular lymphoma to low doses of radiation makes a targeted radiation approach to the management attractive. Recent experimental and clinical work suggests that 131I Tositumomab is effective secondary to the combination of targeted radiation and antibody effector mechanisms, with both appearing to be critical for long term clearance of tumor. Further work is required to enhance our understanding of the mechanisms of action underlying durable remissions of years that some patients enjoy. In particular whether these impressive durable responses induced by 131I Tositumomab are secondary to a prolonged host immune response. There has been much progress made over the last decade with the development of RIT in the treatment of follicular lymphoma and 131I Tositumomab is arguably one of the most active drug ever to have been developed for the treatment of this malignancy. For those physicians who have significant experience in using it, there is almost universal acclaim of the unique mode of action and undoubted clinical efficacy it possesses. However the drug remains grossly underused within the US and is not available within the EU, which brings into question its long-term financial viability. In an era where immuno-chemotherapy has substantially improved outcome it perhaps easy to become complacent that such an effective treatment such as 131I tositumomab is not required in the treatment algorithm of follicular lymphoma. For those with low risk FLIPI disease (Follicular Lymphoma International Prognostic Index) that achieve complete remission with rituximab containing regimens that may be so. However this is an extremely heterogeneous disease, multiple courses of anthracycline based chemotherapy carry a high early and late toxicity and there are and will continue to be increasing number of patients with chemotherapy and rituximab refractory disease. For the latter groups the unique mechanisms of action of RIT have resulted in high activity with durable remissions in both chemotherapy and rituximab refractory disease.38,39 Perhaps what is required is a re-examination of 131I tositumomab in the treatment of follicular lymphoma and an integration of this unique approach in the treatment alogorithm of more patients groups with follicular lymphoma.
Disclosure
The authors report no conflicts of interest.
