Abstract
Chronic myeloid leukemia (CML) is a hematopoietic neoplasm characterized by the Philadelphia chromosome (Ph) and the BCR/ABL oncoprotein. In chronic phase (CP) CML, leukemic cells display multilineage differentiation and maturation capacity. The BCR/ABL inhibitor imatinib exerts profound antileukemic effects in these patients and is considered standard frontline therapy. However, not all patients show a long-lasting response to this drug. Rather, resistance to imatinib has been recognized as an emerging clinical problem in CML. While CML stem cells exhibit intrinsic resistance against imatinib and thus survive therapy, one or more stem cell-derived subclones may acquire additional hits over time, so that an overt relapse occurs. A major triggering hit in such subclones are BCR/ABL mutations. For these patients, novel multikinase inhibitors targeting BCR/ABL such as nilotinib, dasatinib, bosutinib, bafetinib, as well as Aurora kinase inhibitors have been developed and shown to exert antileukemic effects in imatinib-resistant CML. In addition, hematopoietic stem cell transplantation (HSCT) remains an important treatment option for drug-resistant patients. The decision concerning second- or third line therapy, selection of drugs, and HSCT, is usually based on the presence and type of BCR/ABL mutation(s), phase of disease, other disease-related factors, and patient-related variables including age and co-morbidity. The current article provides an overview on current diagnostic and therapeutic strategies for patients with drug-naïve and drug-resistant CML.
Introduction
Chronic myeloid leukemia (CML) is a stem cell-derived myeloid neoplasm in which leukemic cells retain multilineage differentiation and maturation capacity over a certain time period. Cytogenetically, the disease is characterized by the Philadelphia chromosome (Ph) that results from the reciprocal translocation t(9;22).1–3 The specific fusion-gene product, BCR/ABL, is a 210 kDa-sized cytoplasmic oncoprotein that contributes essentially to the pathogenesis of CML.3–5 The clinical course in CML is divided into a chronic phase (CP), accelerated phase (AP), and a blast phase (BP) that behaves as an acute leukemia.6–8
During the past decades, several effective treatment approaches have been introduced for patients with CML.8–15 An effective cytoreductive agent that was used as standard treatment in the past, is interferon-alpha.14,15 An approach with curative potential is hematopoietic stem cell transplantation (HSCT) which can be offered to a subgroup of patients.9–11 HSCT is associated with enhanced disease-free survival, but also with transplant-related mortality and transplant-related morbidity.9–11
Today, imatinib (Gleevec®) is prescribed as first-line therapy in most patients with freshly diagnosed CML. This BCR/ABL tyrosine kinase inhibitor (TKI) produces major cytogenetic responses in a majority of these patients.12–15 In addition, imatinib exerts anti-CML activity in patients refractory to interferon-alpha, and is even effective in some patients with AP or BP.12–18 However, unfortunately, CML stem cells display intrinsic resistance to imatinib.19–21 As a consequence, imatinib can suppress leukemia cell growth for prolonged time periods but is usually not capable of eradicating the malignant clone.21–23 Thus, life-long imatinib appears to be required in responding patients, even when a major molecular response is obtained.21,22 In addition, even as slowly cycling cells, leukemic stem cells (LSC) can acquire further hits in initially small-sized subclones, and later, when these hits lead to clonal expansion and enhanced proliferation, frank resistance and thus an overt relapse develops.
21
In many cases, such stem cell-derived subclones exhibit
During the past few years, a number of novel BCR/ABL TKI have been developed and used in preclinical and clinical trials.34–40 Two of these ‘second generation’ TKI, dasatinib and nilotinib are now approved for treatment of patients with imatinib-resistant or imatinib-intolerant CML. Other new agents such as bosutinib (SKI-606), bafetinib (INNO-406), and the Aurora kinase inhibitors, are currently being tested in clinical studies. In several instances, targeted drugs are combined with other treatment approaches or HSCT. All in all, substantial progress has been made in the diagnosis, management, and therapy of CML in recent years. In the current article, optimal management and therapy of CML is discussed, with special focus on established and novel TKI. In addition, the article provides an overview on forthcoming new agents and future strategies to improve antileukemic therapy in freshly diagnosed and drug-resistant CML.
Investigations in Freshly Diagnosed Patients
Blood examinations at diagnosis include a complete blood count with microscopic evaluation of leukocyte subsets, BCR/ABL, serum chemistry, and HLA-typing. The demonstration of BCR/ABL p210 confirms the diagnosis CML. Initial staging includes a bone marrow (BM) examination, an ultrasound of the spleen and a chest X-ray. Depending on the overall situation and symptoms, further staging investigations such as a computed tomography, or neurologic tests may be required. In patients with primary BP, immunophenotyping of blast cells by flow cytometry (lymphoid versus myeloid) is mandatory. 41 A thorough investigation of the basophil compartment should be performed in each case, especially in suspected AP. Morphologic assessment of basophils in blood- and BM smears is regarded standard, although immature basophils may escape counting, especially in AP.42–44 Biochemical and immunohistochemical markers of basophils may be useful to overcome this problem.45–49 Cellular blood histamine levels are elevated at diagnosis in CML and correlate with basophil numbers. 48 An interesting parameter is the serum tryptase level which is a marker of immature basophils in CML, and may correlate with (imminent) AP and prognosis. 49 Therefore, whole blood histamine- and serum tryptase levels, although not considered routine tests in CML, might be of interest and should be assessed at diagnosis when the assays are available. Screening for BCR/ABL mutations at diagnosis is presently not recommended.
A BM biopsy and aspiration should be performed in all patients at diagnosis. The BM histology and immunohistochemistry are of importance and may reveal fibrosis or multifocal accumulations of CD34+ blasts.49,50 If blast cells are CD34-negative, KIT is used as a progenitor-marker.
49
In addition, the histologic evaluation of the BM is useful to assess the morphology and numbers of basophils and megakaryocytes as well as the microvessel density.46,47,49 Immunohistochemical evaluation of the BM in CML is of particular importance when a dry tap or non-representative smear was obtained (example: BM fibrosis). The aspirate smear is used to perform morphologic studies, including the percentage of blasts, promyelocytes, and basophils, and to screen for signs of dysplasia. The aspirate is also used to perform karyotyping and fluorescence in situ hybridization (FISH).51–53 The karyogram should be based on at least 20 metaphases and should report the percentage of Ph+ cells and, if present, additional chromosome abnormalities (ACA), which are prognostic and often seen in AP.54,55 Chromosome abnormalities in other (Ph-) cells (OCA) may also be detected,
56
but such (sub)clones usually become detectable (develop) later during (not before) therapy. BCR/ABL transcripts are quantitated by real-time PCR (qPCR) using
Parameters indicative of an accelerated phase (AP) of CML.

In these patients, the criteria for AP are not (yet) fulfilled, but findings and symptoms point to imminent AP. These patients may be regarded as potential candidates for higher doses of imatinib (600 mg daily) or a second generation TKI such as nilotinib, similar to AP patients.
Serum tryptase >15 ng/ml.
BM fibrosis may also be regarded as sign of acceleration. 50
In several classification proposals including the WHO, 140 ACA at diagnosis is not an established sign of AP.
Frontline therapy (treatment-naïve patients)
Imatinib is still considered standard frontline therapy in patients with CP, independent of patient-related factors. The initial standard dose in CP in adults is 400 mg per day p.o., independent of body weight.12,65 In patients with AP and BP, the recommended dose of imatinib ranges between 600 and 800 mg daily.12,17,18 In all patients and disease phases, the dose may be adjusted (recommended maximum: 800 mg/day) during treatment with imatinib, based on responses and the plasma trough level.66–68 In particular, it has been described that a lower trough level of imatinib (<650 μg/ml) is associated with a lower probability of achieving a complete cytogenetic response (CCyR).67,68 In patients with a CCyR, life-long treatment is recommended, since (at least subsets of) neoplastic stem cells display intrinsic resistance.19–21 An unresolved question is whether imatinib therapy can be stopped in the few patients who reach a complete molecular remission without detectable BCR/ABL (in BM cells) by nested PCR, which may be indicative of complete suppression or even eradication of most CML subclones. However, most CML patients with CCyR relapse after treatment interruption.22,23 Whether this will also be the case in CP patients treated with nilotinib or dasatinib upfront remains at present unknown, but should be addressed soon in clinical trials. It also remains unknown whether and when nilotinib or/and dasatinib can be regarded standard first line drug therapy in CML. Based on the strong activity and superior clinical effects, it appears that at least in AP and BP, initial therapy with nilotinib or dasatinib, with or without consecutive HSCT, may be regarded a reasonable upfront approach, although a consensus recommendation is not yet available. In CP patients, response rates obtained with nilotinib and dasatinib as frontline therapy clearly exceed response rates obtained with imatinib.69,70 In addition, responses to second generation TKI may occur more rapidly.69,70 However, long term response data and long term side effect profiles for nilotinib and dasatinib are not available for all patients’ groups yet. If such evaluation shows a favourable outcome and no safety issues remain, these new TKI (one or both) must be regarded as standard frontline treatment in CML.
Prognostication and follow up during the First 18 Months
As CML stem cells are resistant towards imatinib and probably also to nilotinib and dasatinib, MRD monitoring is an essential approach in the follow up in all patients. Established assays for MRD monitoring in CML are cytogenetics (percent of Ph+ metaphases in the BM) and FISH (percent of Ph+ interphases) as well as assessment of BCR/ABL transcript levels by qPCR in the peripheral blood.57–59,71–75 FISH is usually performed when results from conventional cytogenetics are unclear or unavailable. In addition, certain ACA/OCA-related abnormalities (example: deletion of derivative chromosome 9) are only detectable by FISH.51–53 During the first year, the BCR/ABL transcript burden is quantified at least every 3 months, and cytogenetics at least every 3–6 months. After the first year, the frequency of investigations depends on the responses achieved. The response is suboptimal when a CCyR is not achieved within 12 months or/and a major molecular response (MMR, defined as a 3-log reduction of BCR/ABL mRNA levels) is not achieved within 18 months. In patients with stable CCyR and completely suppressed BCR/ABL or almost completely suppressed transcript levels (4 log reduction), it may be appropriate to reduce the frequency of investigations to 6- or even 12-month intervals. In these patients a nested PCR can be performed to confirm the depth of remission.
76
In daily practice, the levels of BCR/ABL are determined by real-time PCR and are expressed as the ratio of BCR/ABL mRNA relative to a control gene (in percent) such as
A forthcoming approach may be to quantify CML subclones bearing BCR/ABL mutants by mutation-specific PCR.79,80 With this approach, clinically relevant mutants like T315I may be detected early in disease evolution,79,80 which may have clinical implications and may assist in planning optimal treatment (examples: switch to alternative TKI or urgent search for HSCT donor). These assays must show sufficient precision and sensitivity to detect BCR/ABL mutants in small-sized subclones and the expansion of such subclones over time. Notably, the clinical impact of subclones that remain small over time is usually unknown (an exception may be T315I). The current recommendation is to screen for BCR/ABL mutants as soon as BCR/ABL transcripts increase substantially from baseline MRD levels in repeated tests. The most common approach to screen for
Long-Term Follow-Up
Assays and laboratory investigations in the monitoring and long-term follow-up depend on cytogenetic and molecular responses that can be achieved over time. In patients with stable continuous CCyR and a complete molecular response, qPCR analyses should be performed every 6–12 months and BM investigations in case of a suspected relapse. In all other patients, shorter time intervals are recommended, depending on the levels of BCR/ABL transcripts, the dynamics in changes of BCR/ABL transcript levels over time, cytogenetics, and the overall risk profile. In high risk patients, a BM investigation should be performed at least once a year.
Diagnostic Approaches and Assays in Imatinib-resistant Patients
Appropriate criteria for responses to imatinib and other TKI, including a ‘suboptimal response’ and a ‘treatment failure’ have been provided by the European Leukemia Net (ELN).81,82 These recommendations should be applied in all patients. In those with suspected ‘treatment failure’ and thus suspected drug resistance, all diagnostic tests including a complete (re)staging with re-biopsy of the BM (histology, morphology, chromosome analysis, FISH), BCR/ABL mRNA levels, and analysis of
Management and Therapeutic Algorithm for Imatinib-resistant Patients
Resistance against imatinib (or intolerance) is a major challenge in the management of CML. In a substantial number of patients,
Predicted effectiveness (PE) of BCR/ABL inhibitors in CML patients bearing various imatinib-resistant BCR/ABL mutants. *
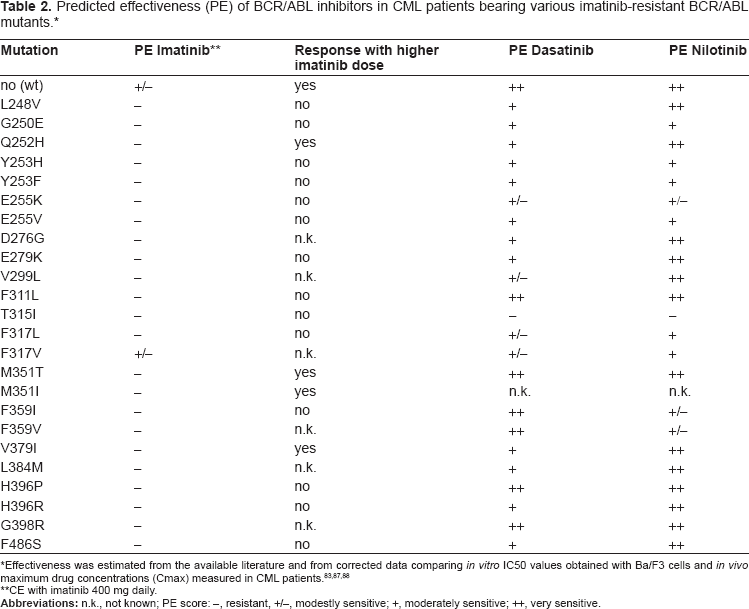
Effectiveness was estimated from the available literature and from corrected data comparing
CE with imatinib 400 mg daily.
After re-staging and after having explored patient adherence (compliance), the risk profile, and all treatment options, the first question is whether the patient may benefit from imatinib dose escalation. Factors that would argue for an attempt to increase the imatinib dose are good tolerability (no adverse side effects), absence of signs of AP or BP, absence of ACA/OCA, no signs for central nervous system (CNS) involvement, no imatinib-resistant
The following factors would argue for allogeneic HSCT: young age, absence of co-morbidity, donor available, ‘good’ HLA-match, primary resistance, rapid disease progression, BCR/ABL T315I or other BCR/ABL mutants known to be resistant to all TKI, ‘poor-prognosis-ACA’, or a substantial increase in blasts. A rapid increase in blasts may argue for HSCT, but may require ‘debulking’ prior to HSCT, which can usually be performed using conventional chemotherapy (e.g. patient with BCR/ABL T315I or Ph-negative clone) or by using a novel TKI, if the mutant is responsive.95,96
The following factors would argue for long-term treatment with dasatinib or nilotinib: patient is not eligible for HSCT (because of age, co-morbidity or other features) or is not prepared to take the risk, presence of BCR/ABL mutants that will respond well to one of these drugs, no relevant ACA/OCA, no BP and no signs of rapid progression (Table 3). A most important ‘pro’ for continuous treatment with dasatinib or nilotinib is a complete response (CCyR, MMR) to these drugs after 3 and 6 months.
When dasatinib or nilotinib should be avoided, interrupted, or switched based on the occurrence of side effects. *
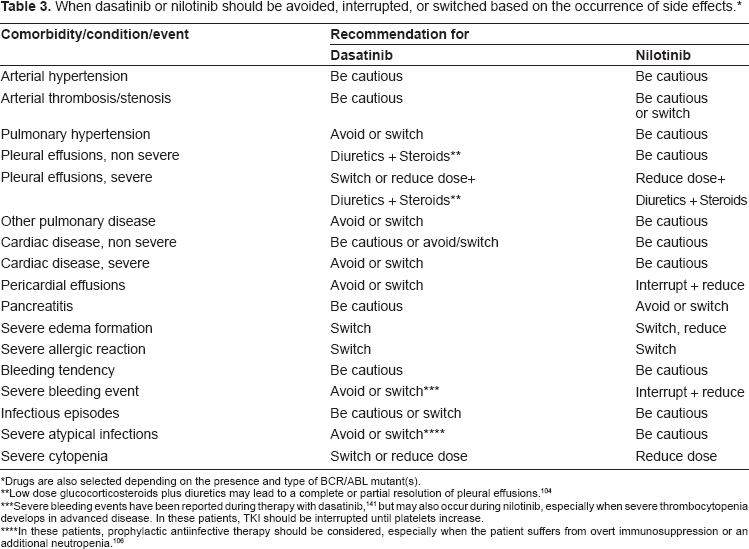
Drugs are also selected depending on the presence and type of BCR/ABL mutant(s).
Low dose glucocorticosteroids plus diuretics may lead to a complete or partial resolution of pleural effusions. 104
Severe bleeding events have been reported during therapy with dasatinib, 141 but may also occur during nilotinib, especially when severe thrombocytopenia develops in advanced disease. In these patients, TKI should be interrupted until platelets increase.
In these patients, prophylactic antiinfective therapy should be considered, especially when the patient suffers from overt immunosuppression or an additional neutropenia. 106
Selection of Patients for Either Nilotinib or Dasatinib
An important decision is which of the two available second-generation TKI (dasatinib or nilotinib) to use in case of resistance or intolerance to imatinib. This decision should primarily be based on the type of
Another important question is whether sequential treatment with all three TKI is an appropriate manoeuvre. Here, it should be mentioned that cross-resistance against dasatinib and nilotinib has been described, but does not occur in all patients. Thus, it may be a reasonable approach to switch from one second-generation BCR/ABL TKI to another in case of resistance.97–100 An important aspect is that subclones bearing drug-resistant BCR/ABL mutants can ‘occur’ by selection at any time during treatment. In most cases these mutants may be pre-existent before start of therapy.97–101 Therefore, it is important to screen repeatedly for BCR/ABL mutants in TKI-resistant patients and those in whom BCR/ABL levels increase in repeated tests during therapy (suspected or imminent resistance). In some of these patients, multiple drug resistant BCR/ABL mutants are selected and it may happen that combinations of TKI are required to suppress all relevant subclones bearing
The standard start dose of dasatinib in imatinib-resistant CML is 100 mg once daily. 102 In patients with rapidly progressing BP or highly aggressive disease, the dose can be escalated (with caution) to 140 mg daily. It is important to determine cardiac and pulmonary parameters carefully before and during treatment with dasatinib as side effects including pericardial and pleural effusions are quite common, especially when the drug is applied at 140 mg daily.103–106 In addition, these patients may be at a higher risk to develop atypical infections and weight loss, especially in an advanced phase of CML. 106 Dasatinib is prescribed on a continuous basis unless the patient is prepared for HSCT or develops resistance, and the same holds true for nilotinib. Nilotinib is prescribed at 400 mg twice daily per os in patients with imatinib-resistant CML.107,108
Side effects of nilotinib have also been described, especially an elevation in bilirubin and elevation in pancreatic enzymes.107,108 A slight elevation in bilirubin is quite commonly seen, whereas severe side effects (overt pancreatitis) are rather uncommon. An unresolved question is whether the suppression of KIT and other key kinases by nilotinib and dasatinib may result in long term side effects. Likewise, nilotinib-treated patients (unpublished) and dasatinib-treated patients 106 often have undetectable levels of serum tryptase suggesting that not only CML basophils have been eliminated but also growth and function of normal mast cells are markedly suppressed.
Special Clinical Situations in Imatinib-resistant Patients
A CNS relapse may not only develop in patients with Ph+ ALL, but also in patients with CML, especially in bi-phenotypic or lymphoid BC, but sometimes even in patients with myeloid BC.109–114 In most patients, a systemic relapse is also found, whereas a local isolated CNS relapse is uncommon.109–114 In most cases, local and systemic treatment is started in parallel. Local therapy usually consists of irradiation and/or intrathecal cytostatic drugs like Ara-C, glucocorticoids, and MTX.109–114 Regarding systemic therapy, it is important to know that imatinib does not cross the blood-brain barrier in sufficient quantities,115–117 whereas dasatinib reportedly enters the CNS quite effectively.114,118 Therefore, patients with a CNS relapse should be considered for treatment with dasatinib. Since every CNS relapse must be regarded a high-risk situation, patients should always be considered for allogeneic HSCT, if possible.
Another important situation is the occurrence of OCA, i.e. an additional chromosomal abnormality in a Ph-negative (sub)clone.56,119–125 Such type of evolution may occur during imatinib, but may also happen in patients treated with second generation TKI.119–125 Clinically, Ph-negative (sub)clones may be completely silent and may remain small without any tendency to transform into a myelodysplastic syndrome (MDS) or AML over months. In other cases, however, rapid transformation to frank MDS or AML is recorded.56,119–125 For these patients, alternative or additional therapy has to be considered. In young patients who are fit and eligible, HSCT may be an appropriate therapy. In other patients, drug combinations might be considered, especially when it appears that the BCR/ABL+ portion of the disease is completely suppressed by the TKI. In these patients, the Ph-negative component may be treated as if no CML had been diagnosed (e.g. as secondary AML), and the Ph+ disease-portion as if no other (sub)clones were found. However, it has to be borne in mind that all these are (highly) experimental therapeutic manoeuvres, and no standard therapy is available for these patients (exception: HSCT).
An interesting observation is that some patients may have a complete loss of CCyR with predominant Ph+ hematopoiesis, but completely normal and apparently stable blood counts. A BCR/ABL mutant is frequently found in these patients. This type of ‘relapse’ may reflect a pre-leukemic phase in the development of CML, in which (a) BCR/ABL-bearing subclone(s) has/have replaced normal hematopoiesis and thus become(s) predominant (with or without
Resistance against Dasatinib or/and Nilotinib
Although many patients with imatinib-resistant CML can be kept in CCyR by dasatinib or nilotinib, relapses may occur. The situation in such a patient is quite difficult and treatment options are limited. Important questions to be asked then are: i) is the patient eligible and thus a candidate for allogeneic HSCT, ii) what BCR/ABL mutants are detectable and what BCR/ABL mutants are currently suppressed (had been detected in the past) by the TKI administered thus far, iii) is the T315I mutant of BCR/ABL detectable, iv) is resistance based on other (BCR/ABL-independent) factors such as pharmacologic determinants. As mentioned above, it may be reasonable to switch from one TKI to the other based on testing for
Prevention and Management of Side Effects of Dasatinib and Nilotinib
Nilotinib displays a favorable side effect profile, and is considered to have the potential to become standard frontline therapy in CML in the near future. However, in some patients, substantial side effects such as an increase in pancreatic enzymes have been reported.107,108 Most side effects rapidly resolve after dose-reduction or treatment interruption. If this is not the case or side effects appear again, it is appropriate to switch to another TKI (dasatinib) if possible. A side effect of nilotinib with unknown clinical significance is the continuous complete suppression of wild type (wt) KIT tyrosine kinase, 126 and thus mast cell growth and function, which is evident clinically by a suppressed serum tryptase level (unpublished observation). Since mast cells are considered to be key regulators of (cardio)vascular repair mechanisms (by providing histamine, heparin, and uncomplexed tissue type plasminogen activator),127–129 current projects in our center focus on possible effects of nilotinib on cardiovascular parameters and cardiovascular events in long term-treated patients.
A major side effect of dasatinib is the development of pleural and pericardial effusions.101,102–106 It is important to consider this side effect and to perform repeated examinations (chest X ray, echocardiography) in long term-treated patients. Pleural and pericardial effusions are quite common in patients with advanced CML treated with dasatinib at 70 mg twice daily.104–106 In patients treated with dasatinib at 100 mg once daily, side effects may be less common.
102
Nevertheless, even in these patients, severe pleural and pericardial effusions may develop (personal observation). An important aspect is that pleural effusions may develop more frequently in advanced, drug resistant CML, and often during or shortly after an infectious or allergic episode.
106
All these observations suggest that the immune system may be involved in effusion formation. Once diagnosed, pleural effusions are best managed by administering low dose glucocorticosteroids and diuretics.101,104–106 If such co-medication remains ineffective, pleuracenthesis is performed and treatment has to be reconsidered, i.e. dasatinib has to be dose-reduced or stopped or switched to an alternative drug. Weight loss and infections as well as the development of skin tumors have also been reported in patients treated with dasatinib at 140 mg daily.
106
Unfortunately, even opportunistic infections were recorded.
106
These side effects may be explained by a transient suppression of various immune cells in these patients.
106
In fact, it has been described that dasatinib blocks T cell receptor-dependent activation of T lymphocytes and IgE-receptor-dependent activation of blood basophils
Cytopenias have been reported in patients treated with nilotinib and dasatinib. This ‘side effect’ usually develops in patients with advanced CML and may simply result from the eradication of CML cells in patients in whom normal stem cells are markedly suppressed and thus need time to recover and to regenerate normal hematopoiesis. The general strategy is to continue nilotinib or dasatinib in these patients at the same dose, provided that i) a molecular response is obtained, ii) no severe event (major bleeding, septicemia) occurs, and iii) cytopenia is transient and resolves after several weeks. In these patients, TKI-induced cytopenia is managed by supportive therapy including transfusions if necessary. If no adequate response is obtained or/and cytopenia becomes life-threatening, the TKI must be stopped.
Combination Treatment Strategies and Novel Anti-CML Drugs
An increasing number of novel targeted drugs potentially effective in CML, have been developed in recent years.30–40 The most promising drugs may be multikinase inhibitors that are effective against most or all mutants of BCR/ABL. Some of these drugs also block BCR/ABL-downstream or BCR/ABL-independent signaling molecules.34–40,136,137 Examples for such TKI are bosutinib (SKI-606), bafetinib (INNO-406), or the Aurora-Kinase inhibitors (Table 4).34–40,136–139 Especially the novel multi-kinase inhibitors that also target BCR/ABL T315I, may represent ideal drugs and drug partners in CML. Most of these drugs are currently tested in clinical trials. A short overview of some of these emerging new drugs is shown in Table 4. For the future, it might be a logical approach to combine most potent targeted drugs early in the course of disease, in order to prevent selection and outgrowth of imatinib-resistant subclones and thus to increase the rate of continuous CCyR.136,137 Another interesting approach may be to combine these targeted drugs with established agents such as interferon-alpha. Finally, it has to be pointed out that several of these drugs can be combined with a transplantation strategy. Likewise, in high risk patients (AP, BP, T315I mutant) who are young and have a suitable donor, HSCT may still be the primary choice of treatment, even if an initial response was obtained with a TKI. In this regard it should also be pointed out that HSCT remains the only established CML-stem cell-eradicating and thus curative approach for patients with CML.
Some of the novel emerging anti-CML agents and their targets.
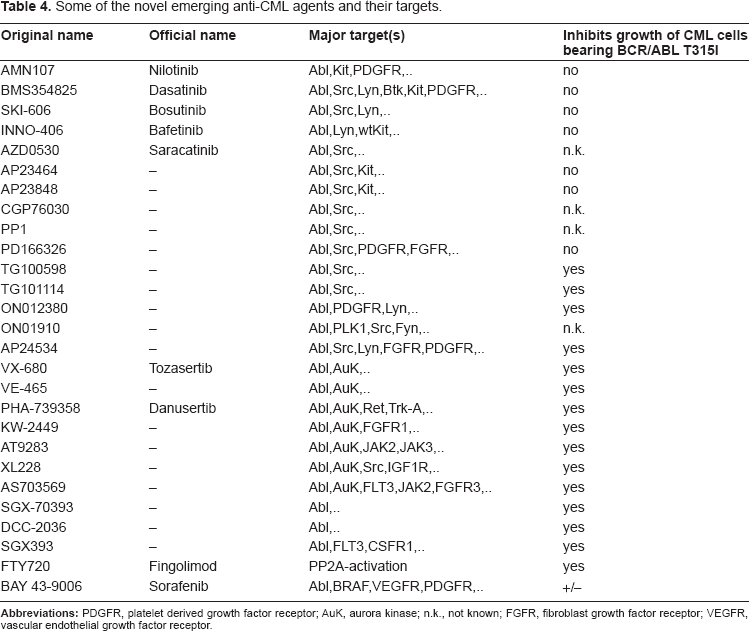
A novel elegant but nontrivial approach may be to eradicate leukemic stem cells in CML by using novel targeted drugs and certain drug combinations. 21 In fact, leukemic stem cells in CML may display a unique composition of antigens, some of which are involved in the mechanism of intrinsic resistance. Interesting targets and target pathways in CML stem cells include certain surface molecules (IL-3 receptor alpha chain, MDR-1, CD33, CD44, or KIT = CD117), the PI3 kinase pathway, the Sonic Hedgehog, Wnt and the β-catenine pathway. Currently, targeted drugs are examined for their potential to induce apoptosis in CML stem cells in preclinical models, and to eradicate these cells if possible. Some of these agents are already applied in clinical trials in patients with TKI-resistant CML. Another approach is to prime CML stem cells (often quiescent cells) with cytokines such as IL-3 or G-CSF, or to overcome intrinsic resistance of CML stem cells by applying MDR-1 inhibitors together with TKI or combinations of TKI in order to increase intracellular drug concentrations. Whether these new agents and approaches swill indeed improve therapy by combining molecular targeting and CML stem cell-targeting concepts, remains to be elucidated.
Concluding Remarks and Future Perspectives
With the advent of novel targeted drugs and second-and third generation TKI, the prognosis in CML may improve significantly during the next few years. Although imatinib is still considered frontline therapy in freshly diagnosed CML, novel more potent BCR/ABL inhibitors have been developed, and most of them are also active in imatinib-resistant CML. Two of these drugs, nilotinib and dasatinib, are approved for the treatment of imatinib-resistant or imatinib-intolerant patients. Currently, both drugs are evaluated as frontline therapy in patients with freshly diagnosed CML. More inhibitors are currently being developed in preclinical studies and clinical trials. Some of these novel TKI-type inhibitors, like the Aurora kinase inhibitors, may even be effective against the T315I mutant of BCR/ABL. The hope for the future is that such novel drugs, when applied in combination, will further improve treatment outcome and survival in CML. For younger patients who are resistant to all available drugs and have a suitable donor, allogeneic HSCT remains a solid alternative treatment option.
Disclosures
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other journal and has not been published elsewhere. P.V. received research grants from Novartis and Bristol-Myers Squibb as well as speaker's honorarium (company-sponsored symposia) from both companies.
