Abstract
Chronic myeloid leukemia (CML) is characterized by the reciprocal chromosomal translocation, t(9;22), forming the BCR-ABL oncogene known as the Philadelphia chromosome. The development of imatinib, a small-molecule kinase inhibitor targeted against BCR-ABL, has revolutionized the management of CML and significantly improved the prognosis and outcome and until very recently was the standard of care in patients presenting with newly diagnosed CML. Nilotinib (Tasigna®) is an orally administered kinase inhibitor made by the Novartis Pharmaceuticals Corporation that was rationally designed to bind to the ABL kinase domain of BCR-ABL resulting in enhanced BCR-ABL inhibition. It is well tolerated and has a favourable safety profile. Nilotinib has been shown to be effective in patients who have failed prior therapy with imatinib. Recently a large randomized control trial comparing imatinib and nilotinib has demonstrated that niloitinb is superior to imatinib in the frontline treatment of CML. This review summarizes the preclinical and clinical data supporting the use of nilotinib in the frontline and secondline treatment of CML.
Introduction
Chronic myeloid leukemia (CML) accounts for approximately 20% of adult leukemias. It is driven by the reciprocal chromosomal translocation, t(9;22), forming the BCR-ABL oncogene known as the Philadelphia chromosome. The resultant constitutively activated protein tyrosine kinase leads to activation of multiple signaling pathways, uncontrolled cell proliferation and reduced apoptosis. The development of imatinib, a small-molecule kinase inhibitor targeted against BCR-ABL, has revolutionized the management of CML and significantly improved the prognosis and outcome for this disease.
The landmark IRIS (International Randomized Study of Interferon) study lead to the FDA approval of imatinib for CML in 2001. At 18-months, it showed major cytogenetic response (MCyR) of 87.1%, complete cytogenetic response (CCyR) of 76.2% and freedom from progression (FFP) of 96.7% in patients receiving imatinib versus 34.7%, 14.5%, and 91.5% respectively in the group given interferon alpha plus cytarabine with better tolerability in the imatinib group. 1 At 8 yr follow up, event free survival (EFS) was 81%, OS was 85% and FFP to accelerated phase (AP)/ blast crisis (BC) was 92% in the imatinib group. 2
Imatinib remains the current standard of care in patients presenting with newly diagnosed CML. Unfortunately, the outcome is not optimal in 30% to 35% of patients as they either become resistant or intolerant. In the IRIS study, at 7-yr analysis, 17% of patients who had achieved CCyR had subsequently lost CCyR and at the 8-yr analysis, 5% and 15% of patients had come off imatinib for adverse events/safety and lack of efficacy/progression, respectively.2,3
The most common mechanism of resistance is attributable to kinase domain point mutations which are associated with impaired binding of imatinib to BCR-ABL. In an analysis of 281 patients, 41% had detectable kinase domain mutations. 4 Other postulated mechanisms of resistance include BCR-ABL oncogene amplification, clonal evolution, inadequate plasma concentrations, variations in expression of drug transport proteins such as organic cation transporter-1 (OCT), increased levels of plasma α1-acid glycoprotein and over-expression of downstream signaling molecules, in particular the Src-family kinases (SFKs).5,6
In efforts to improve on the long-term outcomes in CML, there has been development of several second generation tyrosine kinase inhibitors.
Dasatinib is a small-molecule, multitarget kinase inhibitor of BCR-ABL which is 300-fold more potent than imatinib. Unlike imatinib, it binds both the active and inactive conformations of ABL. It has a wider profile of kinase inhibition including c-KIT, platelet derived growth factor receptor (PDGFR), Eph and SFKs.7,8 It effectively inhibits many of the cells lines with imatinib-resistant BCR-ABL mutations with the notable exception of those with T315I mutation. 9 In 2006, dasatinib became the first FDA approved second-generation TKI (tyrosine kinase inhibitor) in CML patients who are imatinib-resistant or -intolerant at a dose of 100 mg once daily in patients with CML-chronic phase (CP) and at a dose of 70 mg twice daily in patients with CML-AP and -BC.
In 2007, nilotinib, an analogue of imatinib was approved at a dose of 400 mg po BID for CML-CP and CML-AP.
Nilotinib
Nilotinib (Tasigna®) is an orally administered kinase inhibitor made by the Novartis Pharmaceuticals Corporation. Unlike dasatinib, which has a completely different structure to imatinib, nilotinib was methodically and rationally designed to create a better topological fit in the ABL kinase domain of BCR-ABL resulting in enhanced BCR-ABL inhition. It is an aminopyrimidine derivative of imatinib, structurally changed to eliminate two energetically unfavorable hydrogen bonds with the replacement of the N-methylpiperazine ring of imatinib by a trifluoromethyl-substituted phenyl group.10,11 Like dasatinib, its spectrum of inhibition also includes c-KIT and platelet derived growth factor (PDGFR). However, unlike dasatinib, it only binds the inactive conformation of ABL and it does not inhibit Src kinases. 10 Nilotinib uptake does not involve OCT. 12 Niltonib's various characteristics makes it 20-fold more potent against BCR-ABL expressing cells with well-documented activity against 32 of 33 imatinib-resistant BCR-ABL mutants. However, like imatinib and dasatinib, nilotinib is unable to overcome the resistance of the T351I mutation. 13
Nilotinib is orally bioavailable and has a half life of approximately 15 hours. It should be taken 2-hours before or 1-hour after meals as food consumption increases its drug bioavailability. It is metabolized by hepatic oxidation reactions, mainly involving the CYP3A4 pathway, therefore has potential to interact with CYP3A4 inhibitors and inducers. 13
Efficacy in Second Line/Relapsed Treatment
A phase II, open-label study evaluated the efficacy and safety of nilotinib in 321 patients with CML-CP who were imatinib resistant or intolerant, MCyR and CCyR were observed in 57% and 41% of the patients respectively and estimated overall survival (OS) was 91% at 18 months. 14 At 24-months follow up, the overall CCyR, major molecular response (MMR), PFS and OS were 46%, 28%, 64% and 87%, respectively. 15 A major molecular response is defined as a three log reduction in BCR-ABL transcripts by quantitative PCR.
In the phase IIIb ENACT (Expanding Nilotinib Access in Clinical Trials) study, designed to evaluate the efficacy and safety of imatinib-resistant and intolerant CML patients outside of a registration program, in 168 patients with CML-CP, overall complete hematologic response (CHR), MCyR and MMR were 54%, 54% and 42%. Median duration of exposure to nilotinib at the time of this analysis was 363 days. 16
The overall CHR, MCyR and CCyR for all 1,422 patients involved in the ENACT study was 43%, 45% and 34% respectively. Subset analysis of this group demonstrated that prior exposure to other therapeutic agents, including interferon alpha, busulfan and cytarabine did not affect nilotinib dose intensity or response rates. An additional subset analysis showed that patients with suboptimal cytogenetic response on imatinib had higher MCyR and CCyR rates with faster times to these milestones. 17
An additional phase II open label study evaluated the efficacy and safety of nilotinib in 136 patients with CML-AP. At 12 months, survival was estimated to be 81% and PFS was estimated to be 57%. MCyR and CCyR were 31% and 19% respectively.18,19
Nilotinib has been shown to be effective in patients with imatinib-resistant and imatinib-intolerant CML-BC. In 136 patients with CML-BC treated in a phase II study, overall survival was 42% at 12 months and 27% at 24 months. At 24 months, nilotinib induced MCyR and CCyR in 38% and 30% respectively of patients with myeloid blast crises and 52% and 32% respectively of patients with lymphoid blast crises.20,21
Nilotinib is effective in patients with CML following imatinib and dasatinib failure. A phase II study, evaluating the safety of nilotinib 400 mg BID in CML-CP, -AP, and -BC who either failed or were intolerant of both imatinib and dasatinib, demonstrated clinical activity in these patients. 22 In a single institution study, long term follow up for 14 patients receiving nilotinib following imatinib and dasatinib failure showed 2 patients achieving CCyR, 1 patient with partial cytogenetic remission (PCyR), 5 patients with MCyR, 4 patients with CHR and 2 with no response. 23
Efficacy in frontline therapy
In a phase II pilot study evaluating the efficacy and safety of nilotinib 400 mg BID as first line therapy for patients with CML-CP, among 51 patients who had been followed for at least 3 mo, CCyR and MMR are 98% and 76% respectively. 24
The GIMEMA CML Working Party investigated the efficacy of nilotinib 400 mg BID in patients with early chronic phase CML in a multicentric phase II trial. In 72 patients, after 2 years, there was only one patient with T315I mutation who progressed to AP. Patients responded very rapidly with CCyR and MMR of 78% and 52% at 3 months. Recently updated data showed CCyR and MMR of 96% and 87% at 18 months.25,26
A lower dose of nilotinib at 300 mg BID is explored in the ICORG (All-Ireland Cooperative Oncology Research Group) phase 2 study. At 3 months, in 5 patients evaluable for response, CHR, CCyR and MMR are 100%, 80% and 60%. 27
In the phase III, randomized, open-label multi-center ENESTnd (Evaluating Nilotinib Efficacy and Safety in Clinical Trials-Newly Diagnosed Patients) study, 846 patients with CML-CP were randomized in a 1:1:1 ratio to receive nilotinib 300 mg twice daily, nilotinib 400 mg twice daily or IM 400 mg once daily. In the original IRIS study, none of the patients who achieved a major molecular response at 12 months with imatinib progressed to accelerated phase or blast crisis. In light of this, attainment of a MMR at 12 months was chosen as the primary end-point in the ENESTnd study. The rate of CCyR by 12 months was a secondary endpoint. At 12-months, CCyR was 80%, 78% and 65% in these three groups respectively and MMR was 44%, 43% and 22%. This similar pattern of response was observed in a subset of patients with high Sokal risk; CCyR in this group was 74%, 63% and 49% and the MMR was 41%, 32% and 17% respectively. No patient achieving a MMR had progression to AP or BC. At the time of the cutoff date, <1% of the patients on nilotinib had progression to AP or BC while progression occurred in 4% of patients on imatinib. 28 Based on this promising data the U.S. Food and Drug Administration (FDA) granted accelerated approval to nilotinib for the treatment of adult patients with newly diagnosed Philadelphia chromosome positive chronic myeloid leukemia (Ph+ CML) in chronic phase (CP-CML) on June 17, 2010. The recommended nilotinib dose for this indication is 300 mg orally twice daily. The 300 mg twice-daily dosing regimen was chosen as its safety profile appeared more favorable than that of the 400 mg twice-daily regimen, while the efficacy appeared comparable.
Efficacy in Various Mutations
In patients with baseline kinase domain point mutations, the in vitro sensitivity of the mutant clone correlates with response.
Baseline mutation data has been evaluated in 288 patients with CML-CP from the phase II nilotinib registration trial. After 12 months of therapy, those with highly in vitro nilotinib sensitive mutations (cellular IC50 ≤ 150 nM) showed the best responses with MCyR, CCyR and MMR of 60%, 40% and 29% respectively, which were equivalent to those without baseline mutations. These include M244V, L248V, G250E, Q252H, E275K, D276G, F317L, M351T, E355A, E355G, L387F and F486S.4,29
The patients who had less sensitive mutations (cellular IC50 201–800 nM) had less favorable responses to nilotinib and none achieved CCyR. These mutations include Y253H, E255K/V, and F359C/V.4,29,30
Patients with the T315I mutation, which has IC50 > 10,000 nM, 13 remain highly resistant to nilotinib.
Safety
Nilotinib is generally well tolerated. The most common hematologic adverse events are neutropenia and thrombocytopenia [Table 1]. The rates of neutropenia and thrombocytopenia were 10% to 12% for patients taking nilotinib in the frontline setting, 27% to 30% in the 2nd line setting, 44% to 51% following imatinib and dasatinib failure, 27% to 30% in patients with CML-AP and 62% to 67% in patients with CML-BC. As would be expected, these cytopenias correlate with disease phase and number of prior lines of therapy.
Hematologic adverse events from nilotinib in various studies.
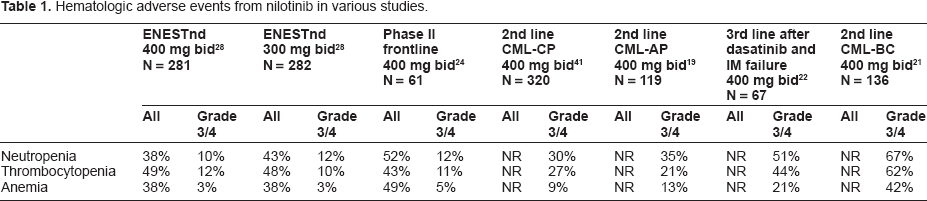
The most common non-hematologic adverse events are rash, headache, nausea, and pruritus [Table 2]. The most common laboratory abnormalities include increased total bilirubin, increased alanine aminotransferase (ALT), increased aspartate aminotransferase (AST), decreased phosphate, increased glucose, increased lipase and increased amylase. The most common grade 3/4 adverse events were neutropenia, thrombocytopenia, increased lipase, increased bilirubin, and increased ALT.
Non-hematologic adverse events from nilotinib in various studies.
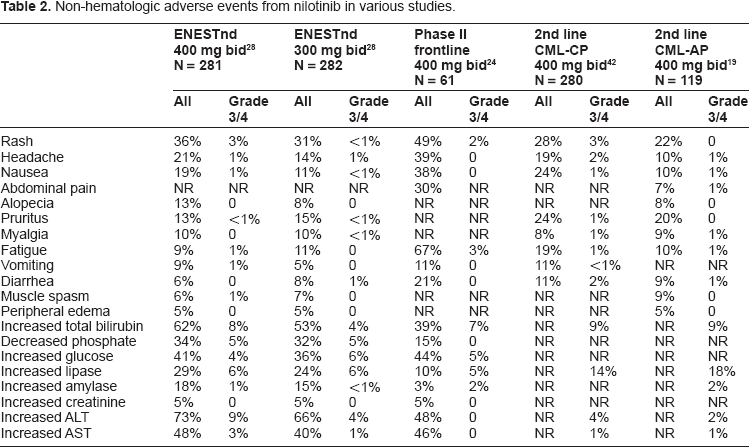
In the ENESTnd study, nausea, diarrhea, vomiting, muscle spasm, edema and grade 3/4 neutropenia and anemia were more common adverse events in the imatinib group. Rash, headache, pruritus, alopecia and laboratory elevations of bilirubin, AST and ALT were seen more commonly in the nilotinib groups. 28
Nilotinib carries a black box warning for QTc interval prolongation and sudden death. No patient in the ENESTnd study had QTc greater than 500 msec. 28 In the phase II frontline studies, one study had 2 patients with QTc greater than 450 msec but none greater than 500 msec. 26 Another study had no patients with grade 3/4 QTc interval prolongation. 24 While recent studies suggest the infrequency of these events, monitoring of electrolytes and EKG remains important.
There is minimal cross-intolerance in patients receiving nilotinib following imatinib-intolerance. Thrombocytopenia was identified as one of the recurring laboratory abnormalities. 31
Dasatinib
Dasatinib has nearly paralleled nilotinib in studies conducted and efficacy.
Dasatinib received its FDA approval and dosing recommendations from the phase II START program studies and the phase III dose-optimization study. The START (Src/ABL Tyrosine Kinase Inhibition Activity Research Trials of dasatinib) program consisted of multiple study arms. START-A examined patients with CML accelerated phase. 32 START-B and -L examined patients in myeloid and lymphoid blast crisis, respectively. 33 START-C examined patients in chronic phase. 34 All these studies demonstrated dasatinib to be effective treatment for imatinib-resistant or -intolerant patients.
The phase III dose-optimization study examined 670 patients with MCyR ranging from 54% to 59% and CCyR ranging from 41% to 45%. 35 Dasatinib has also shown efficacy as third line treatment following imatinib andnilotinib failure. 23 Likenilotinib, dasatinib has been explored in phase II 36 and phase III 37 studies as initial therapy in patients with with CML-CP. Q252H, E255K/V, V299L, F317L, and T315A have been identified to be mutations with less sensitivity to dasatinib. 38
Discussion
Given its tolerability and efficacy, nilotinib has a significant role in the management of CML. Subject to interpretation are the exact definitions of that role. Valid for discussion is the use of nilotinib versus dasatinib. There is a wealth of data to support the use of these second generation tyrosine kinase inhibitors in imatinib-resistant or -intolerant CML however there has never been a direct head to head comparison trial between dasatinib and nilotinib. With fairly similar efficacy, a decision can potentially be influenced by other factors, including mutation status and adverse events profile.
Dasatinib and nilotinib have similar rates of grade 3/4 neutropenia, thrombocytopenia and anemia, favoring nilotinib. Pleural effusions, peripheral edema and bleeding events are significantly higher in dasatinib-treated patients. 38 Hyperglycemia and pancreatic enzyme elevations are higher in nilotinib-treated patients. 39 These variable toxicity profiles can be considered in patients with differing co-morbidities.
An interesting observation was made in a study of health care costs associated with second generation TKIs where the preliminary analysis showed that those treated with dasatinib were associated with higher costs and more frequent health care resource utilization than patients treated with nilotinib. 40
As the data for the frontline use of nilotinib and dasatinib in chronic phase CML matures, there is not only the question of nilotinib versus dasatinib in imatinib-resistant or -intolerant CML patients but the question of whether these agents should become a new standard of therapy as frontline use for newly diagnosed chronic phase CML patients. It is very important to avoid progression to AP or BP CML as both of these are associated with adverse outcomes. Frontline treatment with nilotinib rather than imatinib lessens the risk of progression to these advanced phases. Therefore, nilotinib may emerge as the treatment of choice for newly diagnosed CML. As data is reported on newer therapies such as bosutinib, aurora kinase inhibitors and omacetaxine mepesuccinate, 39 another layer of complexity arises in the treatment of CML.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
