Abstract
Pegaptanib sodium is a polyethylene-glycolated, 28-nucleotide RNA aptamer that binds selectively to vascular endothelial growth factor (VEGF)165 but not smaller isoforms. Preclinical studies identified VEGF165 as an especially potent promoter of ocular neovascularization and inflammation. Following the pivotal clinical trials demonstrating the efficacy of intravitreal pegaptanib in treating all angiographic subtypes of neovascular age-related macular degeneration (NV-AMD), pegaptanib became the first anti-VEGF therapy to receive regulatory approval for this condition. In view of the importance of VEGF in a variety of tissues, including the cardiovascular system and the retina, concerns have been raised as to the risks that might accompany VEGF inhibition. It is thus of particular note that pegaptanib has proved to have a favorable safety record in treating NV-AMD, with no ocular or systemic safety signals having emerged over more than 4 years of clinical studies. Accordingly, in addition to its use as a single agent, pegaptanib has demonstrated promise in combinatorial regimens that employ nonselective anti-VEGF agents as an initial treatment followed by maintenance therapy with pegaptanib. Pegaptanib has also shown encouraging preliminary results in the treatment of diabetic macular edema, proliferative diabetic retinopathy, and macular edema secondary to retinal venous occlusive conditions.
Keywords
Introduction
Age-related macular degeneration (AMD) is the leading cause of blindness in the elderly population in Western countries.1–3 Although the dry, or nonexudative, form comprises approximately 85% of cases, 4 90% of vision loss results from the neovascular form. 5 In neovascular AMD (NV-AMD), damage to the retina is primarily caused by choroidal neovascularization (CNV), the growth of new blood vessels originating in the choroid. These vessels penetrate Bruch's membrane, and as they proliferate they lead to the disruption of retinal structures, loss of photoreceptors, and, in the final stages, a fibrovascular scar. 6 Long before the structural damage is evident, vision loss can result from leakage and hemorrhage owing to the permeable and fragile nature of the aberrant neovasculature.6–9
Until recently, the only available therapies for NV-AMD were laser-based ablative approaches, including laser photocoagulation 10 and photodynamic therapy with verteporfin. 11 In addition to being relevant for only a small fraction of AMD patients, both of these approaches are of limited efficacy and carry the potential for unwelcome side effects.12,13 Treatment of NV-AMD has been transformed over the past decade, however, largely due to research into the molecular and cellular mechanisms mediating physiological and pathological neovascularization. This work has culminated in the identification of a central role for vascular endothelial growth factor (VEGF) in NV-AMD and to the development of targeted molecular therapies.
Two intravitreally administered agents, pegaptanib sodium 14 and ranibizumab, 15 are currently approved for the treatment of NV-AMD. Bevacizumab, which is approved for oncologic use in combination with chemotherapy, is often used as an “off-label” treatment for NV-AMD. 16 Pegaptanib, the first of these agents to become available for clinical use, is an RNA aptamer while ranibizumab is a monoclonal antibody fragment and bevacizumab is a full-length monoclonal antibody. This review will focus on the development of pegaptanib and its use in the treatment of AMD.
Mechanism of Action
Pegaptanib is a 28-nucleotide RNA aptamer (Fig. 1) 17 that interacts with the heparin-binding domain of VEGF165 18 and higher molecular weight VEGF isoforms while sparing smaller isoforms, including VEGF121. 17 Pegaptanib was developed through the application of the Systematic Evolution of Ligands by Exponential Enrichment (SELEX) process for isolating highly specific aptamers19,20 using VEGF165 as a target molecule and including chemical substituents to the component RNA nucleotides to improve stability and nuclease resistance.17,21,22 In addition, the 5‘-attachment of polyethylene-glycol enhanced its bioavailability. 22 The binding of pegaptanib to the heparin-binding domain 18 prevents VEGF from interacting with its receptors, VEGF receptor 1 23 and receptor 2. 24 A steric interference mechanism may be responsible for preventing interaction of the receptor binding domain with these receptors, whereas other proposed mechanisms underlying pegaptanib's therapeutic activity include capturing of soluble VEGF165, and blocking the interaction of VEGF165 with neuropilin-1. 18 In keeping with the isoform-specific nature of this binding, studies of the in vitro action of pegaptanib established its inhibition of endothelial cell proliferation induced by VEGF165 but not by VEGF121. 24
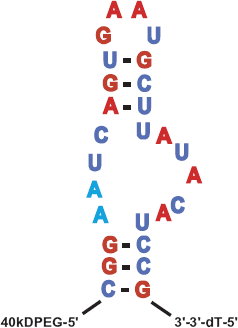
Pegaptanib, a 28-nucleotide RNA aptamer with a 5‘-attachment of polyethylene-glycol that enhances bioavailability (with permission from Ng et al 17 ).
The inactivation of VEGF inhibits two key actions that are of major importance for the vision loss in NV-AMD. Not only is VEGF a powerful promoter of both physiological and pathological angiogenesis, 25 but it is also the most potent known promoter of vascular permeability. 26 The inhibition of VEGF-induced vascular permeability by pegaptanib has been demonstrated in vivo.22,27 VEGF-induced occludin trafficking and disruption of endothelial cell tight junctions have been shown to be important elements in the induction of vascular permeability. 28 VEGF165, but not VEGF121, was found to promote endothelial cell migration and delocalization of tight junction proteins; these effects were prevented by the addition of ranibizumab, demonstrating the significance of VEGF in these processes. 29
The importance of VEGF in ocular neovascular diseases was first suggested in the 1990s, with clinical studies demonstrating that VEGF was elevated in the vitreous of patients with a variety of ocular neovascular conditions.30,31 These investigations were further reinforced by a series of preclinical studies that established that these elevations were both necessary and sufficient for experimentally induced ocular neovascularization.14,17 In preclinical studies of ischemic neovascularization, the VEGF165 isoform proved to be especially pathogenic, with expression levels elevated compared to VEGF121. 32 VEGF165 was found to be significantly more potent at inducing inflammation than VEGF121, resulting in greater expression of intercellular adhesion molecule-1 and chemotaxis of monocytes, both of which are important in promoting ocular neovascularization. 33 In addition, intravitreal administration of pegaptanib was as efficient at preventing pathological neovascularization as a fusion protein that inactivated all VEGF isoforms. 32 These findings ultimately paved the way for the clinical trials that established the efficacy of pegaptanib in treating the pathological neovascularization associated with AMD.
Pharmacokinetic Profile and Metabolism
Studies of pegaptanib pharmacokinetics in monkeys established that the terminal plasma half-life following intravitreal administration was 94 hours 34 while it was only 9.3 hours after intravenous administration, 35 implying that clearance from the eye is the rate-limiting step in pegaptanib elimination. Biologically active pegaptanib was detected for 28 days following intravitreal administration of a single bilateral injection of 0.5 mg/eye in monkeys; no toxicities were observed. 34 Studies in rabbits established that pegaptanib is eliminated primarily in the urine as the parent drug and its metabolites, following the action of endonucleases and exonucleases. 36
In a dedicated pharmacokinetic/safety study, 147 patients with NV-AMD received either 1 or 3 mg intravitreal injections every 6 weeks for 54 weeks (3 and 10 times the approved dose, respectively). Mean maximal plasma concentrations ranged from 20 to 24 ng/mL in patients receiving a 1 mg injection. Pegaptanib remained detectable (with a limit of detection of 8 ng/mL) for 1 week, 37 and the mean terminal half-life was 10 days. There was no evidence of plasma accumulation after repeat doses nor did any patient have detectable antipegaptanib antibodies.
Clinical Efficacy
Following encouraging phase 1 27 and phase 2 38 studies, the pivotal VEGF Inhibition Study in Ocular Neovascularization (V.I.S.I.O.N.) trials 39 were initiated and ultimately led to clinical approval of pegaptanib. These were two concurrent multicenter, randomized, sham-controlled, dose-ranging trials, including 1208 subjects with all angiographic subtypes of AMD, with lesions measuring ≤12 disk areas. Subjects were randomized to four treatment groups, one to receive sham injections and the others to receive intravitreal pegaptanib (0.3, 1, or 3 mg) every 6 weeks for 54 weeks. Photodynamic therapy with verteporfin was allowed at the investigator's discretion for patients with predominantly classic lesions. The primary end-point was the proportion of subjects losing <15 letters of visual acuity (VA). Secondary endpoints included mean changes in VA, proportions gaining ≥0, 5, 10, or 15 letters, proportions losing ≥30 letters, and proportions progressing to legal blindness (VA 20/200 or worse) in the study eye.
Overall, 1186 subjects received at least one treatment (mean: 8.5 of a possible 9 injections) and were evaluated at week 54. All three pegaptanib groups met the primary endpoint; proportions losing <15 letters were 55% for sham, 70% for 0.3 mg (P < 0.001), 71% for 1 mg (P < 0.001), and 65% for 3 mg (P < 0.03). Further analyses were confined to the 0.3 mg pegaptanib dose since there was no additional benefit to higher doses. Compared to sham, 0.3 mg pegaptanib met all secondary endpoints, including mean change in VA (-7.95 versus -15.05 letters, P < 0.05; Fig. 2), proportions gaining ≥0, 5, 10, or 15 letters (P < 0.05 in each case), proportions losing ≥30 letters (10% versus 22%, P < 0.001), and proportions progressing to legal blindness (38% versus 56%, P < 0.001). 39 A treatment benefit was provided irrespective of angiographic subtype, sex, age, iris color, or baseline VA. 40
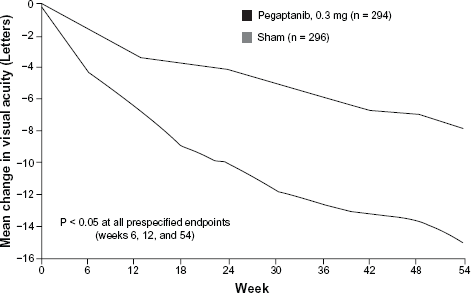
Mean change in visual acuity at week 54 in patients who received 0.3 mg of intravitreal pegaptanib or sham injections in the pivotal V.I.S.I.O.N. trials (with permission from Gragoudas et al 39 ).
In the second year of the study, subjects were rerandomized, with those receiving pegaptanib in year 1 randomized either to continue the same dose or to discontinue pegaptanib. Treatment was continued for 48 weeks. Subjects from the sham group in year 1 were randomized either to sham again or to one of the three pegaptanib doses. In all, 1053 subjects were included in the randomization protocol, with 943 evaluated at week 102. 41 With respect to the primary endpoint, of those subjects continuing 0.3 mg pegaptanib during the second year, 59% lost <15 letters compared to 45% of those continuing sham during the second year (P < 0.05). Subjects continuing pegaptanib for 2 years were also more likely to gain vision and less likely to progress to legal blindness. 41 There was a 45% relative benefit in mean change in vision at the end of 102 weeks in subjects receiving 0.3 mg of pegaptanib compared with subjects receiving usual care (P < 0.01; Fig. 3). 42
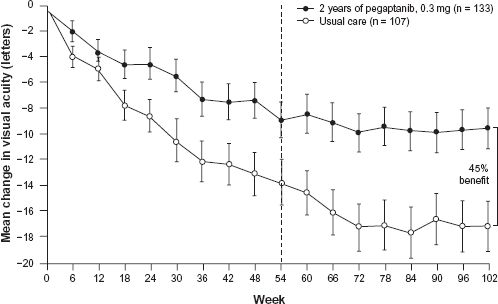
Mean change in visual acuity though week 102 in patients who received 0.3 mg of intravitreal pegaptanib sodium or sham injections (usual care) in the pivotal V.I.S.I.O.N. trials (with permission from Ng and Adamis 41 ).
Further subgroup analysis of the 1-year V.I.S.I.O.N. data also suggested that pegaptanib treatment was likely to be more effective if lesions were treated early in the course of the disease. 43 Two different definitions of early lesions were employed based on criteria such as lesion size, absence of scarring or atrophy, baseline VA, absence of lipid, lack of prior photodynamic therapy, and absence of classic CNV. In these two groups, 80% and 76% of patients receiving 0.3 mg pegaptanib met the primary endpoint of a <15-letter loss compared to 57% and 50% for the respective sham-treated patients (P = 0.05 and 0.03, respectively). These findings of pegaptanib superiority compared to sham injections were paralleled by significantly better outcomes in terms of changes in mean VA, proportions gaining ≥3 lines of VA, and the likelihood of avoiding severe vision loss. 43
Since the completion of the V.I.S.I.O.N. trials, 39 several studies have examined the utility of pegaptanib as a single agent in treating AMD and in many instances have reported results superior to those of the pivotal trials. In a retrospective study involving 90 patients with newly diagnosed NV-AMD, with a mean follow-up of 9.1 months, the loss of <3 lines of VA was seen in 90% of patients receiving pegaptanib while 20% had a gain of ≥3 lines. 44 Another retrospective study involving 111 patients with a mean follow-up of 31 weeks found that 62% of patients with small lesions lost ≤3 lines compared to 46% of patients with large lesions; the percentage with a >3-line gain was similar between those with small and large lesions (15%). 45 In a prospective case series of 56 patients with occult lesions, with 0.3 mg pegaptanib given every 6 weeks for a year, 79% lost <15 letters of VA, with 9% having a VA gain of ≥15 letters. 46 Finally, a pan-European, multicenter, retrospective study evaluated 253 patients receiving first-line treatment with 0.3 mg pegaptanib, most of whom were given three initial doses administered at 6-week intervals with additional treatment as required; a mean of 4.4 injections was administered. 47 At 24 weeks, the proportion losing <15 letters of VA was 93.3%, with 10.7% gaining ≥15 letters (compared to 6% in the V.I.S.I.O.N. trials); the majority of patients did not receive injections between 24 and 52 weeks, resulting in the deterioration of VA for many patients.
Safety
VEGF has a wide range of both ocular and systemic physiological effects. This section reviews the potential concerns for VEGF inhibition and also safety studies relevant to pegaptanib.
Ocular and systemic safety concerns related to VEGF inhibition
VEGF is a pleuripotent growth factor, acting in a wide variety of physiological processes including maintenance of the microvasculature, 48 homeostasis of the cardiovascular system, 49 and the survival of a wide variety of neuronal types. 50 Inhibition of VEGF thus has the potential for generalized systemic effects as well as impacts on tissues in the immediate local environment of the eye.
The choriocapillaris and the neural retina are the tissues most closely examined for potential effects of VEGF inhibition. Polarized secretion of VEGF from the retinal pigment epithelium (RPE) is believed to be important in maintenance of the choriocapillaris 51 while intravitreal injections of bevacizumab in monkeys were found to lead to occlusion of the choriocapillaris by thrombocytes and leukocytes as well as to the loss of choriocapillaris fenestrations. 52 It has recently been reported that in patients with geographic atrophy secondary to AMD, loss of the RPE is accompanied by degeneration of the adjoining choriocapillaris; these findings are consistent with the hypothesis that the choriocapillaris is dependent on RPE-secreted VEGF. 53
With respect to the retina, preclinical data have indicated a protective role for VEGF in maintaining retinal neurons in ischemic conditions. In rat experiments, intravitreal injections of both VEGF121 and VEGF165 were able to prevent retinal cell apoptosis; however, inhibition of all VEGF isoforms by a nonselective antibody against VEGF resulted in a loss of retinal ganglion cells whereas pegaptanib, which is specific for VEGF165, did not. 54 Similar findings with respect to the importance of VEGF in supporting retinal neural cells have been reported using transgenic techniques in mice. 55 Intravitreal injection of bevacizumab was found to induce mitochondrial damage and activation of apoptotic pathways in rabbit photoreceptors. 56 In a subsequent study, both intravitreal pegaptanib and bevacizumab induced greater apoptotic activity than saline when injected at doses equivalent to those employed clinically; yet, bevacizumab had a dose-dependent effect that resulted in greater cell death at the higher doses than pegaptanib. 57
Concerns for the systemic impact of VEGF inhibition have focused primarily on the circulatory system, including its potential role in the development of collateral vessels in the context of cardiovascular disease. 49 The importance of VEGF for the growth and maintenance of blood vessels reflects its various effects on endothelial cells, including its actions as a mitogen, 58 chemoattractant,59,60 and survival factor. 61 The systemic administration of the VEGF antagonists bevacizumab62,63 and VEGF-Trap,64,65 when used as an adjunct in chemotherapy or for the treatment of NV-AMD, has been associated with an increase in hypertension and proteinuria,62,64 bleeding, 62 thromboembolic events,62,63 and delayed wound healing.62,65
When administered intravitreally, systemic exposure would be expected to be greatly reduced since much lower doses are used; however, systemic exposure will not be completely eliminated. 66 This is especially true given the fragile and more permeable nature of the aberrant blood vessels, 67 it is thus of note that the effects of intravitreal anti-VEGF antagonists have been reported in untreated fellow eyes. 68 Moreover, any impact on cardiovascular health is of particular concern given the apparent association of AMD with an elevated risk for cardiovascular disease. 69 In retrospective pooled analyses of the efficacy trials with ranibizumab, intravitreal treatment for NV-AMD has been associated with an increased risk of nonocular hemorrhagic events 70 and cerebrovascular accidents 71 compared to sham-injected groups. In the dedicated SAILOR trial comparing 0.5 and 0.3 mg doses of ranibizumab, there was a trend of higher stroke incidence in patients in the 0.5 mg group (1.2% versus 0.7%, respectively), and this trend was maintained among those patients with a prior history of stroke; these differences were not statistically significant, though. 72
Safety studies with pegaptanib
The safety of pegaptanib has been examined extensively, not only in dedicated studies but also in the context of the pivotal V.I.S.I.O.N. trials. In a systemic safety study in dogs and rabbits involving intravitreal pegaptanib doses up to 10 times the approved clinical dose as well as bolus intravenous pegaptanib injections, no clinical, ophthalmologic, pathologic or cardiovascular abnormalities were observed. 73 Similarly, no systemic signals were detected in the safety/pharmacokinetic trial in which patients treated intravitreally for AMD received up to 10 times the clinical dose for 1 year. 37 There was no evidence of an impact of pegaptanib treatment on blood pressure or urinary protein.
These conclusions have been reinforced by the continuing safety monitoring of subjects in the V.I.S.I.O.N. trials in which patients treated with pegaptanib have been followed for more than 4 years without the emergence of serious ocular or systemic safety signals.74–76 In addition, a retrospective analysis of fundus photographs from the V.I.S.I.O.N. trials found no evidence of pegaptanib-induced harm to retinal neurons or the RPE. 77
Injection-related adverse events such as endophthalmitis (0.16% per injection), traumatic lens injury (0.07% per injection), and retinal detachment (0.08% per injection) occurred infrequently in the V.I.S.I.O.N. trials.39,74 In a subsequent report from a clinical setting with all three intravitreal anti-VEGF drugs, only three cases of endophthalmitis were observed in more than 10,000 injections. 78 In addition, in a retrospective chart review examining the effects of 102 injections in patients receiving warfarin therapy and pegaptanib injections in clinical practice, there were no immediate postoperative hemorrhagic complications. 79
Elevation in intraocular pressure immediately after injection is another reported injection-related adverse event. In the V.I.S.I.O.N. trials,74,75 the increases occurred on average 30 minutes postinjection and had returned to preinjection levels at the 1-week postinjection assessment. Subsequent studies have demonstrated transient elevations in intraocular pressure following anti-VEGF agents (generally lasting less than 30 minutes) which require monitoring in only select cases, such as in patients with small eyes or preexisting glaucoma.80–83
RPE tears following intravitreal injection of pegaptanib 84 and other intravitreal anti-VEGF agents 85 have been described; most of these tears occurred in eyes with preexisting RPE detachments. A large RPE detachment basal diameter and vertical height were found to be significant risk factors for developing an RPE tear. 86 In some rare cases, serious allergic reaction has been reported soon after injection36,87 including an anaphylactoid response in one instance and lip swelling with urticarial rash in another. 88 As these are reported voluntarily in the postmarketing phase of pegaptanib use,36,87 a direct relationship to pegaptanib or any of the various medications administered as part of the injection preparation procedure, or to other factors, has not been established in these cases. 87
Combination Studies with Pegaptanib
Because of complementary mechanisms of action of different agents, a rationale exists for combined approaches to target different processes in NV-AMD that may optimize efficacy and/or safety. In view of the potential systemic effects of long-term VEGF inhibition, several groups have reported encouraging results on the use of a nonselective-VEGF inhibitor as an “induction” or “booster” agent in the treatment of patients with NV-AMD, with a selective anti-VEGF agent as the principal maintenance therapy.
In the first of these studies, Hughes and Sang 89 employed 1.25 mg bevacizumab as the inductive agent followed by nine injections of 0.3 mg pegaptanib injections administered at 6-week intervals, with assessments performed at 54 weeks. Bevacizumab booster injections were administered as required based on a criterion of a 15% increase in lesion size as measured by fluorescein angiography; only 4 of 20 patients required a second injection of bevacizumab. The mean Snellen VA improved from 20/200 to 20/80 at 54 weeks, and mean central point retinal thickness on optical coherence tomography (OCT) decreased from 341 μm to 191 μm, with all patients showing an improvement. Furthermore, 95% of the patients gained ≥1 line of VA, with 45% of patients gaining ≥3 lines. 89
In a small retrospective study involving 17 eyes receiving pegaptanib injections every 6 weeks, booster injections of either 0.5 mg ranibizumab or 1.25 mg bevacizumab were administered if patients demonstrated no improvement or a decrease in VA, or if there was an increase in retinal thickness on OCT. 90 Over a 12.1 month follow-up, 47% of eyes gained ≥3 lines VA; the mean number of administered injections was 7.8 for pegaptanib, 1.4 for bevacizumab, and 0.9 for ranibizumab.
In the largest of these studies, a phase 4, prospective, open-label study enrolled 568 patients who received induction therapy with a variety of agents, including ranibizumab, bevacizumab, or multiple agents, and including different combinations of pegaptanib, ranibizumab, bevacizumab, photodynamic therapy, corticosteroids, and transpupillary thermotherapy. 91 Subjects received a mean of 2.6 induction treatments over a median of 15 weeks, with pegaptanib given every 6 weeks for 48 weeks. Mean VA improved during induction from 49.6 ± 21.9 letters to 65.5 ± 15.3 letters and at 54 weeks was 61.8 ± 18.9 letters; the mean center point thickness was 191 ± 54 μm postinduction and 211 ± 70 μm at 54 weeks. Overall, approximately 50% of subjects (283/568) required booster treatments with 46% (130/283) of them requiring only a single injection. The protocol was well tolerated, with adverse event data similar to those observed during the V.I.S.I.O.N. trials.39,74
In addition to the induction-maintenance regimens, other combination approaches have been assessed. In a study involving 22 eyes of 16 patients, Liggett et al 92 used triple therapy (i.e. high-dose triamcinolone, photodynamic therapy, and pegaptanib) for the treatment of CNV and reported an improvement and/or stabilization in VA, particularly when used as first-line therapy. Finally, Bennett et al 93 used intravitreal injections of pegaptanib in combination with moxifloxacin in 65 patients (80 eyes) every 6 weeks; after a 13-month median follow-up, results were superior to those observed in the V.I.S.I.O.N. trials, with 97% of treated eyes losing <15 letters of VA and 80% having stable or improved VA.
Place in Therapy for Pegaptanib
Clinical studies, including the pivotal V.I.S.I.O.N. trials,39,41 have demonstrated that intravitreal pegaptanib provides clinical benefit in the treatment of NV-AMD and that better results may occur if lesions are treated early in the disease process. These studies have established that pegaptanib has a favorable profile, which may reflect its underlying molecular mechanisms of action in that only VEGF165 and larger isoforms are inactivated. Given the comorbidities that are often present in the predominantly elderly patient population with NV-AMD and the potential systemic consequences of long-term VEGF inhibition, pegaptanib may be an especially promising candidate for use as maintenance therapy in conjunction with nonselective VEGF therapies. In addition, pegaptanib is currently being investigated as a treatment for ocular diseases other than NV-AMD and has shown preliminary efficacy in the treatment of diabetic macular edema94,95 and proliferative diabetic retinopathy 96 as well as macular edema secondary to central retinal vein occlusion 97 and branch retinal vein occlusion. 98
Conclusions
The development and success of anti-VEGF therapies have validated the concept of targeting the underlying molecular mechanisms of ocular neovascular diseases. The availability of a variety of anti-VEGF agents further increases the flexibility of ophthalmologists in developing treatment strategies. Pegaptanib has shown favorable efficacy in treatment of NV-AMD and is being investigated for treatment of other ocular neovascular diseases as well. Protocols using induction with a nonselective agent followed by pegaptanib maintenance seek to take advantage of the proven long-term safety profile of pegaptanib.
Disclosure
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author reports no conflicts of interest.
Footnotes
Acknowledgment
Editorial support, including assistance in the manuscript development and styling for the journal, was provided by Lauren Swenarchuk, PhD, of Zola Associates and was funded by Pfizer Inc.
