Abstract
Postoperative ileus (POI) in patients undergoing abdominal surgery is associated with significant morbidity. In 2008, alvimopan (Entereg®) was approved by the Food and Drug Administration (FDA), and is the only available POI therapy in the United States for patients undergoing bowel resection. Data from preclinical studies demonstrate that alvimopan and its primary metabolite, ADL 08-0011, behave as potent μ. opioid receptor antagonists. In animals, alvimopan and ADL 08-0011 attenuate opioid agonist-induced reductions in gastrointestinal (GI) transit. Higher doses of alvimopan are required to inhibit opioid-induced analgesia as a result of its inability to penetrate the central nervous system (CNS). ADL 08-0011 is also peripherally selective, although to a lesser degree than alvimopan. In multiple species, including humans, alvimopan has low oral bioavailability, while ADL 08-0011, following its generation by human gut microflora, is more readily absorbed and achieves higher exposures. Three Phase 2 and five Phase 3 clinical trials have been conducted to investigate the efficacy and tolerability of alvimopan in patients undergoing bowel resection. An additional Phase 3 study was conducted in hysterectomy patients. In the majority of the studies, statistically significant, and clinically meaningful, acceleration of GI recovery has been demonstrated. Consistent with animal data, alvimopan has no effect on opioid agonist-induced analgesia in healthy human subjects and POI patients. Clinical experience to date in POI patients indicates that alvimopan is well tolerated when used according to its approved dosing regimen (12 mg b.i.d. for up to 7 days). In this article, the preclinical and clinical properties of alvimopan are reviewed.
Introduction
Although largely associated with major abdominal and gynecological surgeries, postoperative ileus (POI), defined as a temporary delay in the recovery of normal gastrointestinal (GI) function, can present itself following other types of surgical interventions. Postoperative gut dysmotility is claimed to last up to 24 hours in the small intestine, 24 to 48 hours in the stomach, and 48 to 72 hours in the colon, although its duration can be significantly longer. While it is believed by many surgeons that some degree of POI is a normal and essential physiological response to abdominal surgery, POI, particularly when prolonged and severe, is associated with significant morbidity.1–3 POI is characterized by pain, abdominal distention or bloating, nausea and vomiting, accumulation of gas and fluids in the bowel, and delayed passage of flatus or stool. The consequences of POI can be significant: delayed mobilization and recovery from surgery, delayed oral absorption of drugs and nutrients, increased risk of postoperative nausea and vomiting, increased length and cost of hospitalization, decreased quality of life due to pain and discomfort, and increased risk of developing pulmonary infections. 9 The delayed recovery of GI motility following surgery is the principal cause of extended hospitalization. It is estimated that in the United States, POI accounts for a total hospital cost of $1.46 billion annually. 5 POI is a multifactorial condition; risk factors for POI include abdominal surgery, surgical technique, prolonged opioid-induced analgesia, use of inhaled anesthetics, pre-existing GI disease, and physical inactivity either before or after surgery. 6
Until recently, POI treatment was limited to nasogastric suction, increased fluid intake, early feeding, and ambulation, although evidence of the benefits of such approaches is limited. 2 Indeed, it is now recognized that nasogastric suction, one of the more common postoperative interventions, causes significant patient discomfort, is associated with increased occurrence of atelectasis, gastric reflux and pneumonia, and may actually prolong POI.7,8 The use of less invasive surgical procedures, such as laparoscopy is beneficial, when appropriate, as is the use of epidural local anesthetics in place of opioids for analgesia. 2 Opioid receptor agonists, a mainstay for postoperative analgesia, contribute significantly to the occurrence and severity of POI.9,10
The announcement on May 20th 2008, by Adolor Corporation and GlaxoSmithKline that the United States Food and Drug Administration (FDA) had approved alvimopan (Entereg®) capsules to treat patients with POI following partial large or small bowel resection surgery with primary anastomosis was a major advance. Alvimopan ((S)-2-benzyl-3-[(3R,4R)-4-(3-hydroxyphenyl)-3,4-dimethylpiperidin-1-yl]-propionylamino; also known as LY246736 or ADL 8-2698; Fig. 1) is a peripherally-restricted μ opioid receptor antagonist.3,11–13 In this article, the properties of alvimopan are reviewed, with particular emphasis on the clinical experience gained to date in POI therapy.
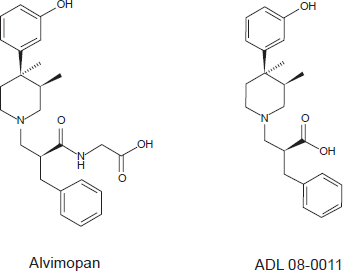
Chemical structures of alvimopan and its primary metabolite, ADL 08-0011.
Mechanism of Action of Alvimopan
The clinical efficacy of alvimopan in POI patients (described below) is attributed to antagonism of opioid receptor agonist-induced activity in the GI tract. Opioid agonists such as morphine and fentanyl delay gastric emptying and reduce small and large intestinal transit largely through agonism of μ opioid receptors.14–18 Activation of μ opioid receptors on neurons of the myenteric and submucosal plexuses, and in the longitudinal and circular smooth muscle of the GI tract, is associated with increased non-productive smooth muscle contractility that disrupts propagating activity.19–23 Inhibition of enteric acetylcholine and non-adrenergic non-cholinergic neurotransmitter release is likely to be an important mechanism underlying opioid agonist-induced reductions in motility.24,25 While there is some evidence that prototypical μ opioid receptor antagonists, such as naltrexone and naloxone, attenuate POI and chronic opioid-induced bowel dysfunction (OBD),26–28 their clinical value in these GI disorders is limited given that these drugs cross the blood brain barrier readily, and thus can attenuate opioid-induced analgesia and provoke an opioid behavioral withdrawal syndrome.28,29 However, it is noteworthy that Targin®, a combination of oxycodone and naloxone, is efficacious in the treatment of moderate to severe pain without significant constipation;30,31 its value in POI therapy remains to be determined.
The mechanism of action of alvimopan has been elucidated preclinically.12,32–34 Data from radioligand binding studies (Table 1) indicate that alvimopan has high affinity for the human μ opioid receptor (Ki values of 0.25 or 0.44 nM).32,35 Alvimopan has significant, but lower, affinity at human δ (Ki values of 2.5 or 10 nM)12,35 and κ opioid receptors (Ki value of 100 nM). 12 It can be concluded therefore that alvimopan is highly selective (by ≥227-fold) for the human μ receptor over the κ subtype, but has a more modest (≥6-fold) μ/δ receptor selectivity. Data from cell-based assays demonstrate that alvimopan is a potent μ opioid receptor antagonist (IC50 or KB values ranging from 0.4 to 1.7 nM). 36 Alvimopan is devoid of any significant positive intrinsic activity (mean values of −5%, −14% and 4% at human μ and δ, and guinea pig κ opioid receptors, respectively, relative to responses evoked by full agonists at each receptor). 32 In the guinea pig isolated ileum, alvimopan is a potent antagonist of morphine, DAMGO or endomorphin-1-induced, and μ opioid receptor-mediated, inhibition of electrically-evoked contractions (pA2 values of 9.6 or 9.7).32–34 The δ and κ antagonist potencies of alvimopan are lower in the guinea pig ileum (pA2 values of 8.7 and 7.8, respectively).33,34 Alvimopan (1 or 10 μM) has no significant affinity for a broad range of non-opioid receptors, ion channels and enzymes at which it has been tested. 12
In vitro binding affinity (Ki values in nM) of alvimopan and its metabolite, ADL 08-0011, at human or guinea pig (#) μ, δ and κ opioid receptors.
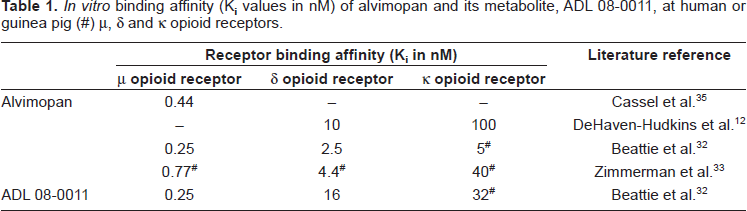
ADL 08-0011 ((S)-2-benzyl-3-[(3R,4R)-4-(3-hydroxyphenyl)-3,4-dimethylpiperidin-1-yl] propionic acid; Fig. 1), the primary metabolite of alvimopan, has at least 60-fold higher affinity for the human μ opioid receptor over human δ and guinea pig κ opioid receptors (Ki values of 0.25, 16 and 32 nM, respectively; Table 1). 32 ADL 08-0011 inhibits responses to endomorphin-1 in the guinea pig ileum (pA2 = 9.6). 32
The in vivo activity of alvimopan has been evaluated in a variety of preclinical models using the mouse, rat, guinea pig and ferret. 12 Of particular relevance to its clinical indication, rodent studies investigated whether alvimopan affected POI.37,38 Alvimopan (1 and 3 mg/kg orally) reduced the severity of ileus (assessed by measurement of 15 Cr transit) in rats when dosed prior to, but not 45 minutes after, laparotomy and intestinal manipulation with or without morphine dosing. 37 Schmidt et al 38 failed to demonstrate a statistically significant effect of alvimopan in a mouse POI model, although attenuation of morphine-induced reductions in GI transit was observed. Indeed, inhibition of morphine or loperamide-induced reductions in GI transit by alvimopan has been demonstrated in several studies with oral doses typically between 0.3 and 1 mg/kg.33,34,38–40 The activity of alvimopan is not limited to a particular region of the GI tract; loperamide or morphine-induced delays in gastric emptying, and small intestinal and colonic transit are attenuated in mice and rats. 12 Alvimopan also prevents morphine-induced inhibition of diarrhea evoked by castor oil or prostaglandin E2 administration in mice.12,41 Alvimopan is much more potent orally in these diarrhea models (ED50 values of 11 or 20 μg/kg) compared to other transit assays, possibly reflecting that in the former a luminal site of drug action plays a more important role, although it is as yet unclear which is the most relevant clinically. Alvimopan (3 mg/kg orally) has no effect on colorectal distension-induced visceromotor responses in its own right, and also following colonic hypersensitivity induced by intracolonic administration of trinitro-benzenesulphonic acid. 39 In the same study, naloxone was active suggesting that central, rather than peripheral, μ opioid receptor-mediated mechanisms are important in regulating colonic sensitivity. Alvimopan also inhibits morphine-induced emesis in ferrets at oral doses of 0.01-10 mg/kg. 12 ADL 08-0011, like alvimopan, is a potent inhibitor of opioid agonist-induced reductions in GI transit in mice (Fig. 2), rats and guinea pigs, consistent with its μ opioid receptor antagonist affinity in vitro.
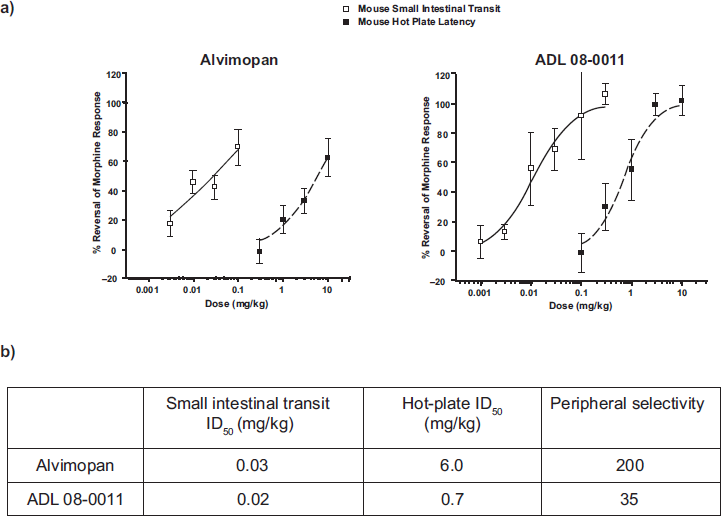
Alvimopan is peripherally selective, and thus does not impair opioid-induced analgesia at doses that affect GI motility. The peripheral selectivity of alvimopan is attributed to its moderately large molecular weight, zwitterionic form and polarity.33,34 In a mouse hot-plate study, 42 subcutaneous dosing of alvimopan and its primary metabolite, ADL 08-0011, inhibits morphine-induced anti-nociception (ID50 values of 6.0 and 0.7 mg/kg, respectively), while lower doses reduce morphine effects on small intestinal transit (ID50 values of 0.03 and 0.02 mg/kg, respectively; Fig. 2). Therefore, the peripheral selectivity (i.e. the ratio of ID50 values for reversal of analgesia and delayed GI transit) in mice is higher for alvimopan than for ADL 08-0011 (ratios of 200 and 35, respectively). Following oral dosing to rats, alvimopan (10 mg/kg) has no effect on morphine-induced analgesia in the hot-plate model while after intravenous dosing the ED50 is 6.1 mg/kg. 12 In mice rendered physically-dependent with morphine, alvimopan (up to 10 mg/kg dosed subcutaneously) fails to provoke a CNS withdrawal response, while naloxone (0.1-10 mg/kg) results in jumping behavior and increased locomotor activity. 42 Data from preclinical studies indicate, therefore, that alvimopan is a potent μ opioid receptor antagonist in vitro and in vivo, and attenuates opioid agonist-induced reductions in GI transit without affecting analgesia or other CNS endpoints.
The Metabolic and Pharmacokinetic Profiles of Alvimopan
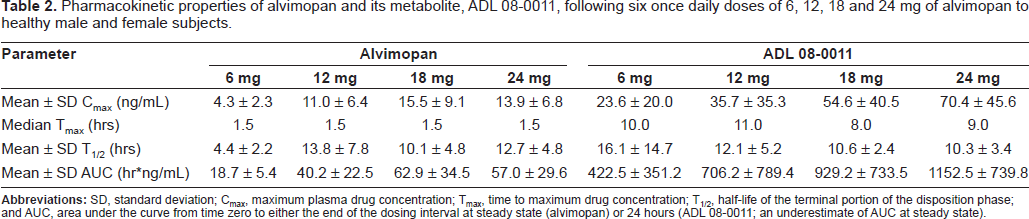
The pharmacokinetic properties of alvimopan and ADL 08-0011 have been studied in a number of species, including humans.12,34,43,44 In vitro data indicate that alvimopan has low solubility and permeability. Alvimopan is not a substrate of cytochrome P450 enzymes, and its plasma protein binding ranges from 70% to 80%. In vivo data demonstrate that alvimopan has low oral bioavailability in humans, rats and dogs (6%, 0.05% and 0.03%, respectively; Entereg® NDA 21-775 FDA advisory panel briefing document). 34 Following oral dosing of alvimopan (200 mg/kg) to rats, peak plasma concentrations are low (20.5 ng/mL). 34 The pharmacokinetics of alvimopan at steady-state, following once daily dosing of 6 to 24 mg, has been determined in healthy human volunteers (Table 2). 43 The mean terminal half-life of alvimopan, after multiple oral doses, ranges from 4.4 to 13.8 hours. Despite its low oral bioavailability, at clinically relevant doses sufficient plasma concentrations (Cmax and AUC values; Table 2) of alvimopan and its active metabolite, ADL 08-0011 are generated in humans. Alvimopan plasma levels at the approved, 12 mg b.i.d. dosing regimen exceed the μ opioid receptor Ki in 95% of subjects for approximately 12 hours. Food has a negative influence on alvimopan exposure (reductions in the rate and extent of absorption of 54% and 18%, respectively). 45
Pharmacokinetic properties of alvimopan and its metabolite, ADL 08-0011, following six once daily doses of 6, 12, 18 and 24 mg of alvimopan to healthy male and female subjects.
The steady-state volume of distribution of alvimopan in humans is estimated to be 30 ± 10 L. Data from whole body autoradiography studies in rats demonstrate that [14C]alvimopan is distributed to most parts of the body, with the exception of the brain and spinal cord. 13 Following oral or intravenous dosing to rats approximately 15% of the dose is secreted into the bile 34 while in humans 65% of alvimopan is eliminated via biliary secretion, with renal excretion accounting for approximately 35% of the total clearance (Entereg® NDA 21-775 FDA advisory panel briefing document).
ADL 08-0011, an amide hydrolysis product, is generated from unabsorbed alvimopan or alvimopan secreted in the bile by human gut microflora. 43 In contrast to its parent, ADL 08-0011 has high oral bioavailability in rats and dogs (53.3% and 80.1%, respectively). In healthy humans, following oral dosing of alvimopan, the systemic exposure of ADL 08-0011 is markedly higher than that of the parent (Table 2).43,46 ADL 08-0011 accumulates upon multiple daily dosing of alvimopan; after 5 days its plasma concentration remains relatively constant for up to 96 hours after the last dose, and then declines with a halflife of between 10 and 20 hours (Entereg® NDA 21-775 FDA advisory panel briefing document). There is an extremely high degree of inter-subject variability in the exposure of ADL 08-0011 following administration of alvimopan (Table 2).43,45 Generation of ADL 08-0011 by human microflora is markedly influenced by ethnicity (43% and 82% lower plasma concentrations in African American and Hispanic subjects, respectively, compared to Caucasians), possibly as a result of dietary differences, while concomitant use of antibiotics or proton pump inhibitors reduces plasma concentrations of ADL 08-0011 by 81% or 49%, respectively.44,45 Exposure of ADL 08-0011 is 51% lower in patients with inflammatory bowel disease compared to healthy volunteers. 45 Unchanged ADL 08-0011 is eliminated in the feces and urine together with a glucuronide conjugate and other minor metabolites. Based on its potent μ opioid receptor antagonist activity, and higher systemic exposure compared to that of alvimopan following oral dosing of the latter, ADL 08-0011 may contribute to the observed clinical efficacy in POI patients (see below). However, the onset of clinical activity following oral dosing of alvimopan appears inconsistent with an absolute requirement for generation of ADL 08-0011, and efficacy is evident in patients receiving antibiotics who have substantially reduced levels of ADL 08-0011. Nevertheless, with repeated dosing, at steady-state, it is likely that ADL 08-0011 contributes significantly to the GI activity attributed to alvimopan.
Clinical Efficacy of Alvimopan in Postoperative Ileus
Phase 1 Studies
Several clinical studies have been completed with alvimopan in healthy human subjects. These studies not only characterized the safety and pharmacokinetic profiles of alvimopan, but also assessed whether, as indicated in the preclinical animal models, the compound could reverse opioid agonist-induced reductions in GI transit without impairing opioid analgesia or miosis (a surrogate for CNS penetration). One study demonstrated that alvimopan (2.4 and 24 mg t.i.d.) completely prevented loperamide-induced delays in colonic transit of radiopaque markers in healthy male subjects. 47 Using the lactulose/hydrogen breath test, another group demonstrated that alvimopan (4 mg orally) inhibited morphine-induced delays in oral-cecal transit time in healthy males and females.48,49 The GI/CNS selectivity of alvimopan was studied by comparing its ability to prevent morphine-induced reductions in the colonic transit of radiopaque markers, with its effects on analgesia (using the cold pressor test) and miosis.13,50 Alvimopan (3 mg t.i.d.) inhibited morphine-induced delays in GI transit time and reductions in stool number and weight without affecting analgesia or pupil diameter. In another study, performed in subjects undergoing third molar extraction, alvimopan (2 mg orally, 60 minutes prior to, and after, surgery) had no effect on analgesia provided by morphine, as assessed by categorical pain and pain relief scores, or visual analog scale, nor did it affect pupil constriction. 49 Therefore, data from initial clinical studies confirmed that alvimopan prevented opioid agonist-induced delays in GI transit without interfering with CNS-mediated activity such as analgesia, and supported its progression to studies in POI patients.
Phase 2 Studies
The preliminary efficacy and safety of alvimopan in POI patients (1 to 12 mg, administered two hours prior to surgery and b.i.d. thereafter for up to 7 days) were evaluated in three Phase 2 studies in subjects undergoing partial colectomy, and simple or radical hysterectomy.51,52 All patients received intravenous, patient controlled analgesia (PCA) with opioids following surgery. While alvimopan had no significant effect on GI recovery in one of the studies, likely due to insufficient statistical power, efficacy was evident in the other two trials at doses ranging from 3 to 12 mg. Analysis of safety data indicated that alvimopan was well tolerated. Based on the Phase 2 efficacy results, larger studies were initiated to investigate the efficacy of alvimopan in patients undergoing bowel resection and hysterectomy.
Phase 3 Studies
Six Phase 3 studies (five in the United States and one in Europe; see Table 3 for patient characteristics) have been conducted with alvimopan in POI patients, and several pooled analyses have been performed.53–60 The primary reason for surgery was rectal or colon cancer.53,60 All were randomized, double-blind, placebo-controlled parallel studies comparing the efficacy of alvimopan to that of placebo in hospitalized patients. The majority of the studies investigating the efficacy of alvimopan in POI patients utilized the time to GI-3 recovery as the primary endpoint—a composite assessment that measured upper (toleration of solid food) and lower (first bowel movement or flatus) GI tract recovery. Secondary endpoints included GI-2 recovery (toleration of solid food and first bowel movement, but not flatus), time to hospital discharge or readiness for such, the incidence of nasogastric tube insertion, duration of hospital stay, and occurrence of hospital readmission due to POI. Patients were considered to have postoperative morbidity related to POI if nasogastric tube insertion, or complications of POI resulting in prolonged hospital stay or readmission within 7 days of actual discharge, occurred.
Characteristics of all Phase 3 POI studies in patients treated with placebo and 12 mg b.i.d. alvimopan (ND = no data).
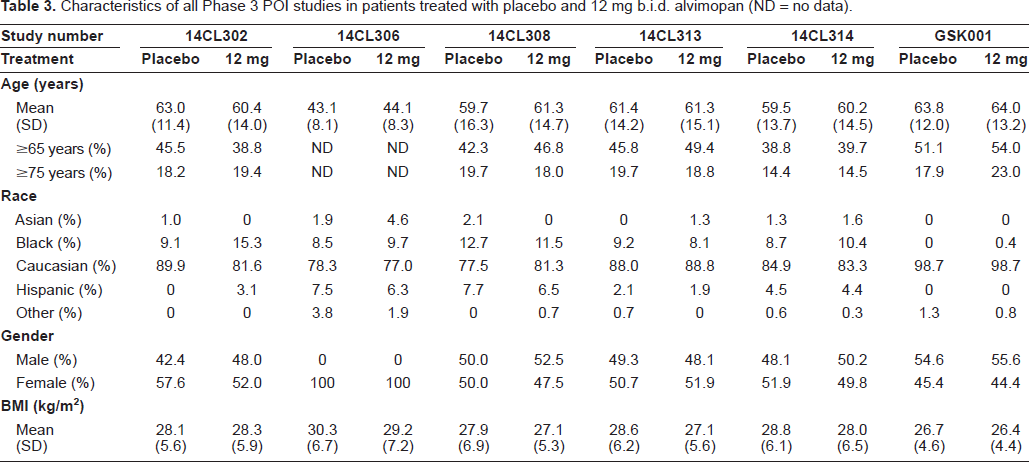
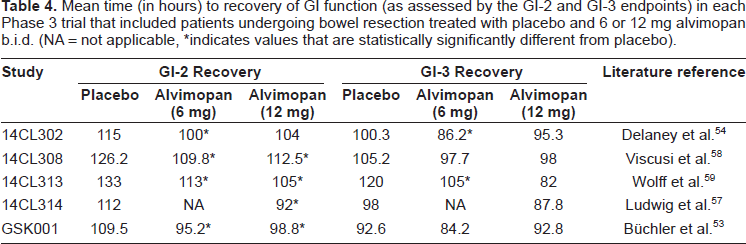
In study 14CL302, 54 patients undergoing partial colectomy or simple or radical hysterectomy were randomized to receive alvimopan (6 or 12 mg) or placebo orally two hours prior to surgery and b.i.d. thereafter until hospital discharge, or for up to 7 days. Alvimopan (6 mg) was associated with a statistically significant reduction in the mean time to GI-3 recovery (the primary efficacy endpoint) by 14.1 hours compared to placebo (Table 4). At the higher dose (12 mg), there was a 7.5 hour reduction in GI-3 recovery, which failed to achieve statistical significance. The hazard ratios, generated using the Cox Proportional Hazards model, for a selection of the efficacy endpoints indicated that alvimopan was associated with a higher probability of achieving GI recovery compared to placebo (Fig. 3). Alvimopan (6 and 12 mg) significantly accelerated the time to readiness for hospital discharge (mean differences of 16 and 14 hours, respectively) while the hospital discharge orders were written, respectively, 14 or 7.2 hours on average earlier than placebo. Time to first bowel movement was significantly accelerated (by 17 and 15 hours, respectively, at 6 and 12 mg alvimopan) relative to placebo, as was the time to tolerance of solid food with alvimopan at 6 mg (by 10 hours on average). There was only a modest difference in the proportion of patients with postoperative morbidity in favor of alvimopan (12 mg) over placebo (Fig. 4). Unlike patients undergoing partial colectomy or radical hysterectomy, alvimopan (6 or 12 mg) had no significant effect on GI-3 recovery time in patients subjected to a simple hysterectomy.
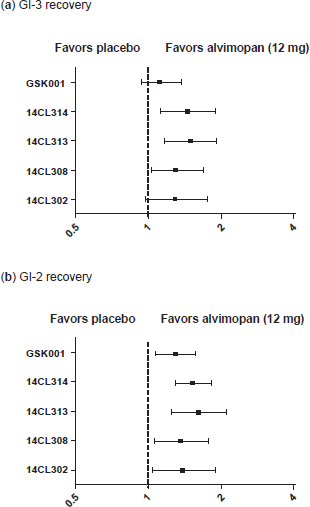
Hazard ratios (with 95% confidence intervals) for (
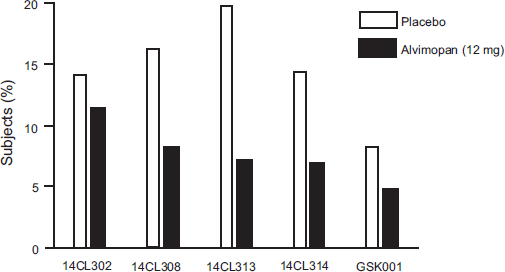
Proportion of patients with postoperative morbidity related to POI in each of the Phase 3 studies performed with alvimopan (12 mg b.i.d.) in patients undergoing bowel resection.
Mean time (in hours) to recovery of GI function (as assessed by the GI-2 and GI-3 endpoints) in each Phase 3 trial that included patients undergoing bowel resection treated with placebo and 6 or 12 mg alvimopan b.i.d. (NA = not applicable, *indicates values that are statistically significantly different from placebo).
Study 14CL306 56 enrolled only patients undergoing simple total hysterectomy. Alvimopan (12 mg b.i.d.) had no statistically significant effect on time to GI-3 recovery, toleration of first solid food, or written hospital discharge order, but did significantly accelerate GI-2 recovery compared to placebo, due primarily to a reduction in the time to first bowel movement (by 22 hours on average). More frequent bowel movements and better bowel movement quality were also noted with alvimopan compared to placebo.
In study 14CL308, 58 alvimopan (6 and 12 mg) or placebo was administered to patients undergoing bowel resection (68%) or hysterectomy (32%) two hours before surgery and b.i.d. thereafter. The hazard ratios for GI-2 and GI-3 recoveries (Fig. 3), together with hospital discharge endpoints favored alvimopan over placebo. Neither dose of alvimopan had a statistically significant effect on the GI-3 endpoint in the initial assessment (mean reductions of 7.5 and 9.9 hours, at 6 and 12 mg doses, respectively; Table 4). However, adjustment of the analysis for significant covariates (sex and surgery duration) resulted in a statistically significant reduction in GI-3 recovery time for both doses of alvimopan. GI-2 recovery was also significantly accelerated (by a mean of 16.4 or 13.7 hours at 6 or 12 mg, respectively). Reductions in time between surgery and hospital discharge, and for time to first bowel movement were observed with alvimopan relative to placebo at the 6 or 12 mg dose. However, neither dose of alvimopan had a significant effect on time to tolerance of solid food or first flatus. It was concluded that although the results for the individual endpoints were somewhat different from those for the 14CL302 study, accelerated GI recovery was evident with alvimopan. In this study, there was a larger reduction in the proportion of patients experiencing postoperative morbidity with alvimopan at 12 mg (Fig. 4).
Studies 14CL313 58 and GSK001 53 consisted primarily of small or large bowel resection with primary anastomosis (96% of treated patients in 14CL313 and 87% in GSK001). In study 14CL313, alvimopan at 6 and 12 mg b.i.d. resulted in a statistically significant shortening of GI-3 and GI-2 recovery times compared to placebo (by an average of 15 or 20 hours at 6 mg, and 22 or 28 hours at 12 mg, respectively; Table 4). Time to writing the hospital discharge was also shortened by alvimopan (reductions of 13 and 29 hours compared to placebo at the 6 and 12 mg doses, respectively). Study GSK001, performed primarily in Europe, had several distinct features compared to the other Phase 3 studies. The protocol did not prohibit the use of concomitant non-opioid analgesics, and the duration of the post-surgery, in-hospital observation period was longer (14 days compared to the typical 10-day evaluation). Furthermore, while all patients in the North American studies were scheduled to receive opioid analgesic by PCA, study GSK001 also permitted intravenous and intramuscular bolus dosing as determined by the pain management protocol in place at each participating hospital. The primary endpoint (GI-3 recovery) was not achieved in study GSK001 (a mean reduction of 4.8 hours with the 12 mg dose; Table 4). GI-2 recovery was reduced by 10.7 hours on average with alvimopan (12 mg) compared to placebo. The hazard ratios for GI-2 and GI-3 recoveries from both studies are shown in Figure 3. Interestingly, post hoc analysis indicated that alvimopan was more efficacious in patients using PCA for analgesia compared to those relying on bolus opioid analgesic dosing (a reduction in GI-3 recovery of 8.3 hours compared to an increase of 1.1 hours, respectively, relative to placebo). There was no effect of alvimopan on the hospital discharge-related endpoints in study GSK001. A larger reduction in the proportion of alvimopan treated patients who experienced postoperative morbidity occurred in study 14CL313 compared to GSK001 (Fig. 4).
Study 14CL314 55 differed from the other Phase 3 POI studies in several respects. Only patients undergoing laparotomy for segmental bowel resection with primary anastomosis were enrolled as it was concluded from earlier studies that these patients responded better to alvimopan than those undergoing hysterectomy. Additionally, GI-2, rather than GI-3, recovery was adopted as the primary endpoint as it was evident, based on prior data, that there was more variability with the latter endpoint, due to the flatus assessment. It is likely that time to first flatus is less objective as patients must be conscious and willing to report its occurrence. In study 14CL314 only the 12 mg dose of alvimopan was studied; its administration was allowed 30-90 minutes prior to surgery and, as in all other Phase 3 studies, twice daily thereafter. The hazard ratios for GI-2 and GI-3 recoveries (Fig. 3), and the hospital discharge endpoints, indicated that alvimopan was clearly favored over placebo. Alvimopan significantly reduced the mean time to GI-2 and GI-3 recoveries by 20 and 16 hours, respectively, compared to placebo (Table 4). The time to writing of the hospital discharge, and the actual discharge, were also shortened (by 18 and 17 hours, respectively). Patients who received alvimopan were less likely to receive nasogastric tube insertion relative to placebo, and in consequence there was a lower incidence of postoperative morbidity (Fig. 4).
Several large-scale pooled analyses of the Phase 3 data have been performed.55,60,61 Alvimopan (6 or 12 mg) significantly accelerated GI-3 recovery, time to writing of the hospital discharge order, and was associated with reduced post-operative morbidity relative to placebo together with reduced incidence of prolonger hospital stay or readmission. There was a lower incidence of postoperative nasogastric tube insertion. Pooled analysis indicated that although both 6 and 12 mg significantly accelerated GI recovery, the benefits appeared greater and more consistent with the higher dose. 59 Post hoc analysis of the Phase 3 studies in patients undergoing bowel resection by Senagore et al. 60 indicated that alvimopan accelerated GI recovery independent of age, gender, race and concomitant drug use (i.e. preoperative antibiotics, histamine antagonists or proton pump inhibitors).
Based on its receptor selectivity, the clinical activity of alvimopan is entirely consistent with antagonism of exogenous μ opioid receptor agonist action in the GI tract. A role for endogenous opioids in POI has been proposed,2,14 and so it is feasible that alvimopan may antagonize their influence on GI motility. Enkephalins and beta-endorphin are released in response to inflammation 62 and surgical manipulation, 23 and increased colonic transit has been observed with alvimopan in healthy subjects in the absence of an exogenous opioid agonist. 63 However, in another study, in patients with chronic idiopathic constipation, alvimopan had no effect on stool production. 64
In each clinical study, the ability of alvimopan to interfere with pain control was closely monitored (by assessing visual analogue scale pain scores, and measuring daily opioid analgesic consumption). Compared to placebo, alvimopan (6 or 12 mg) had no effect on pain intensity scores, and daily opioid analgesic consumption was similar in the preoperative, intraoperative and postoperative periods.55,57,60 The lack of effect on pain control confirms that following doses of 6 and 12 mg, alvimopan, (and its major circulating metabolite, ADL 08-0011) have an appropriate degree of peripheral selectivity in POI patients.
Safety Assessment of Alvimopan
Preclinical Studies
Data from preclinical safety studies have demonstrated that alvimopan has a large therapeutic index (Entereg® NDA 21-775 advisory panel briefing document). 34 Alvimopan is not genotoxic in the Ames test, the mouse lymphoma cell (L5178Y/TK+/-) forward mutation test, the Chinese hamster ovary cell chromosome aberration test or the mouse micronucleus test (Entereg® prescribing information). Alvimopan has no significant inhibitory activity in the hERG potassium channel and dog Purkinje fiber assays, nor has it any adverse effects on the cardiovascular systems of rats and dogs following oral or intravenous doses of up to 200 mg/kg or 2.5 mg/kg, respectively. In rats, intravenous doses of up to 10 mg/kg had no effect on neurobehavioral endpoints, while oral doses up to 200 mg/kg failed to affect renal function. Alvimopan, at single oral doses of 100 and 200 mg/kg reduced sensorimotor activity in mice. Chronic intravenous and oral dosing studies in rats and dogs did not reveal significant toxicological findings, and no reproductive issues were observed. 34 No adverse findings were observed in two long-term (2-year) carcinogenicity studies performed in rats and mice with alvimopan (maximum doses of 0.5 and 4 g/kg/day, respectively; Entereg® NDA 21-775 advisory panel briefing document). The alvimopan metabolite, ADL 08-0011, also had a low order of toxicity preclinically in multiple intravenous toxicity studies, and in a genotoxicity assay.
Clinical Studies
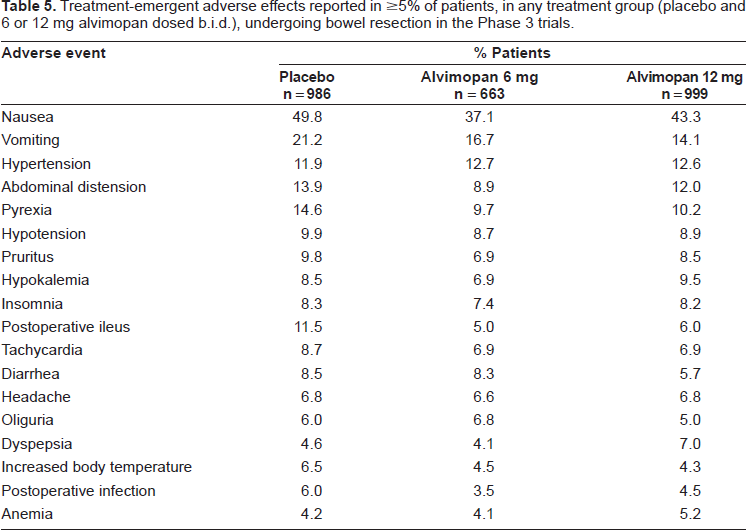
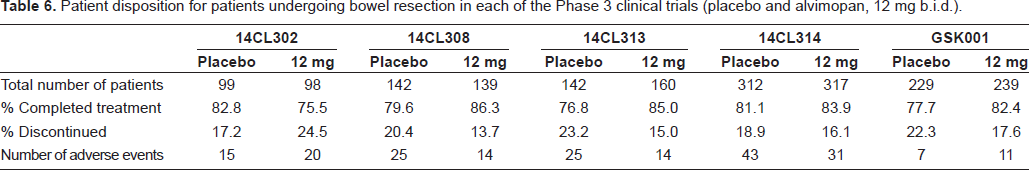
Tolerability data in Phase 1 and Phase 2 clinical trials indicate that alvimopan is generally well tolerated (Entereg® NDA 21-775 advisory panel briefing document). 54 Healthy subjects or POI patients tolerate alvimopan better than patients on chronic opioid analgesic therapy, who experience dose-limiting GI cramping, pain, diarrhea, nausea and vomiting, 13 likely as a result of a GI opioid withdrawal response. A thorough ECG Phase 1 study has been performed, and alvimopan (6 and 24 mg b.i.d. for 7 days) found to have no significant effect on the QT interval. The Phase 3 clinical studies in POI patients have provided a large safety database for alvimopan. Safety assessments included adverse event reporting, clinical laboratory tests, vital sign measurements and electrocardiograms. Adverse events were defined as treatment-emergent if they occurred after the first dose of study medication and up to 14 days after the last dose. Serious adverse events were defined as life-threatening if they required intervention to prevent permanent impairment, were associated with significant disability, or resulted in prolonged hospitalization or readmission. Adverse events in the Phase 3 trials were mainly GI-related (e.g. nausea, vomiting and abdominal distension), and consistent with those expected in patients undergoing bowel resection under general anesthesia with opioid analgesic therapy.54,55,57–60 The overall incidence of these adverse events was somewhat lower in alvimopan treated patients compared to those receiving placebo (Table 5). In study 14CL302, the lower incidence of vomiting in the alvimopan 12 mg group was statistically significant compared to that in the placebo group. 54 A smaller proportion of patients in the alvimopan (6 and 12 mg) groups had serious adverse events that required prolonged hospitalization or readmission in comparison to those receiving placebo. 58 Discontinuations as a result of adverse events occurred to similar extents for alvimopan and placebo in the Phase 3 trials (Table 6), and of the few deaths reported, none was considered drug-related.54,59 The incidence of anastomotic leak, a serious complication of bowel resection surgery, was low and similar for alvimopan and placebo treated POI patients. 60
Treatment-emergent adverse effects reported in ≥5% of patients, in any treatment group (placebo and 6 or 12 mg alvimopan dosed b.i.d.), undergoing bowel resection in the Phase 3 trials.
Patient disposition for patients undergoing bowel resection in each of the Phase 3 clinical trials (placebo and alvimopan, 12 mg b.i.d.).
The FDA's review of the alvimopan new drug application for POI included an interim (6-month) analysis of a Phase 3, 12-month safety study in patients with chronic OBD. Unexpectedly, this analysis revealed a non-statistically significant increase in the reported incidence of serious cardiovascular adverse events (i.e. seven subjects with myocardial infarctions, three with unstable angina and two with serious arrhythmia compared to none on placebo; Entereg® NDA 21-775 advisory panel briefing document). The reported events were in patients at high risk for cardiovascular disease, did not appear to be linked to the duration of dosing, and were consistent with epidemiological expectations for the subject population. Adolor and GlaxoSmithKline concluded that the cardiovascular adverse events represented a chance finding and were not causally related to alvimopan. It is noteworthy that in this long-term safety study, patients received a lower dose of alvimopan compared to that studied acutely in the POI setting (0.5 and 12 mg b.i.d., respectively). It would seem unlikely that the cardiovascular adverse events associated with alvimopan, if genuine, are related to μ opioid receptor antagonism. The opioid receptor antagonists, naltrexone and naloxone, have been used for many years in the treatment of addiction and opioid agonist overdosage, and while changes in blood pressure, tachycardia, fibrillation and cardiac arrest are listed as precautions for naloxone, these effects are generally only observed upon abrupt reversal of opioid depression post-operatively (Physicians Desk Reference). Naltrexone, approved at relatively high doses (≥50 mg orally) for addiction, has no cardiovascular liability. In rats, δ opioid receptor agonist activity has a cardioprotective effect against ischemic and hypoxic damage, 65 although clinical data are limited. Alvimopan and ADL 08-0011 are potent antagonists at human recombinant δ opioid receptors (pA2 values of 9.0 and 8.3, respectively); 32 on the basis of their systemic exposures at clinical doses of alvimopan, significant levels of δ receptor occupation are likely. It remains to be determined whether δ opioid receptor antagonism contributes to the apparent cardiovascular risk associated with chronic alvimopan administration. In the same long-term safety study, a numerical imbalance was noted in the incidence of bone fractures (3.7% with alvimopan vs. 1.1% with placebo), and benign (1.5% vs. 0.4% in favor of alvimopan) and malignant (0.9% vs. 0.4% in favor of alvimopan) neoplasms. The identification of these imbalances led to an interim analysis of safety data in a cancer pain study which revealed more deaths in the alvimopan treated patients compared to those on placebo. A causal relationship for these phenomena is uncertain.
While the potential cardiovascular and neoplasm findings have cast some doubt on the safety of alvimopan upon chronic use, for an acute POI indication there appears to be no such concern. Approximately 2,600 POI patients in the Phase 2 and Phase 3 trials received alvimopan, and the incidence of myocardial infarction, determined by both adverse event reporting and independent adjudication, was considered consistent with that expected in the patient population, and rates for the alvimopan and placebo groups were similar. However, based on the adverse effects noted in the 12-month safety study with alvimopan, the FDA required a Risk Evaluation and Mitigation Strategy (REMS) to be in place prior to approval. As part of the REMS, the Entereg Access Support and Education (EASE) program was established. Under the EASE program, alvimopan is made available only to hospitals that complete a registration process and meet all of its requirements. The EASE program is designed to maintain the benefits associated with short-term use in the bowel resection population and prevent long-term, outpatient use.
The recommended adult dose of alvimopan is a single 12 mg capsule administered orally 30 minutes to five hours prior to surgery, followed by a 12 mg capsule b.i.d. commencing the day after surgery for a maximum of 7 days. The alvimopan prescribing information includes a boxed warning stating that it is available only for short-term use (up to 15 doses) in hospitalized patients. Alvimopan is contraindicated in patients who have taken therapeutic doses of opioids for more than 7 consecutive days immediately prior to treatment to avoid intolerable GI cramping, pain, diarrhea, nausea and vomiting. As there are no studies of alvimopan tolerability in patients with severe hepatic or renal impairment undergoing bowel resection, its use in these populations is not recommended. While reproductive toxicity studies in animals failed to identify any evidence of impaired fertility or harm to the fetus, no clinical studies have been conducted with alvimopan in pregnant women. Alvimopan has a pregnancy category B status, and should be prescribed to pregnant women only if it is clearly needed. Caution is warranted in the treatment of lactating women as it is unknown whether alvimopan or its metabolite, ADL 08-0011, is excreted in human milk. The safety and effectiveness of alvimopan in pediatric patients have not been established.
Place in Therapy
The introduction of alvimopan is a significant advance for the treatment of POI in patients undergoing bowel resection. In the majority of clinical studies in bowel resection patients, the alvimopan-induced acceleration of GI recovery achieved statistical significance. Although the magnitude of response may appear relatively modest (e.g. hospital discharge up to one day earlier), the FDA advisory panel agreed with Adolor and GlaxoSmithKline that alvimopan provides a clinically meaningful benefit to patients. It seems reasonable to assume that alvimopan will be utilized in conjunction with less invasive surgical procedures, increased fluid intake, early feeding, and ambulation. Alvimopan therapy should reduce the use of nasogastric suction which is now acknowledged to provide little benefit, and may in fact be hazardous.7,8 Presumably in the event that the use of opioid agonists for postoperative analgesia diminishes significantly in the future, so may the clinical efficacy of, and need for, alvimopan.
In order to reduce the severity or duration of POI, administration of alvimopan should be initiated prior to surgery (i.e. when gastric emptying is functioning normally) to achieve sufficient systemic exposure. Therefore, this demands alvimopan be used routinely prior to bowel resection rather than restricted to patients who already have POI. A parenteral formulation, if feasible, would perhaps be more appropriate for POI therapy as administration could, in principle, be initiated at any time to stimulate GI recovery.
It remains to be determined whether the magnitude of the acceleration in GI recovery noted with alvimopan in POI patients is the maximum achievable via antagonism of peripheral μ opioid receptors, and whether other peripherally selective opioid receptor antagonists will be associated with improved efficacy. Methylnaltrexone, a quaternary derivative of the opioid receptor antagonist, naltrexone, was associated with a mean accelerated GI recovery, discharge eligibility and actual discharge of 27, 30 and 25 hours, respectively, in patients undergoing segmental colectomy following intravenous dosing (0.3 mg/kg infusions every 6 hours) in a Phase 2 trial, suggesting that increased efficacy relative to alvimopan may be possible. 65 However, in two large Phase 3 trials methylnaltrexone unexpectedly failed to demonstrate significant clinical efficacy (Progenics press releases March 12th and May 22nd 2008).
Concluding Remarks
Alvimopan, a potent μ opioid receptor antagonist, is the first approved therapy in the United States to reduce the duration and severity of POI in patients undergoing bowel resection. In the majority of the large clinical POI studies performed, alvimopan accelerated GI recovery, assessed with respect to time to toleration of solid food, first bowel movement or flatus, or readiness for hospital discharge and actual discharge time. Postoperative morbidity was improved by alvimopan. Due to its poor CNS penetration, postoperative analgesia, commonly provided by opioid analgesics, is unaffected by alvimopan. With respect to safety and tolerability, alvimopan appears to be well tolerated when dosed at 12 mg b.i.d. for up to 7 days. In one long-term safety study, however, an increased incidence of serious cardiovascular adverse events, and benign and malignant tumors was noted. It remains unclear whether or not these findings are attributable to alvimopan. It is hoped that the successful development of alvimopan for POI will stimulate further progress in identifying novel therapies to accelerate GI recovery and improve the quality of life of postsurgical patients.
Disclosures
The author's employer, Theravance, Inc., has a peripheral μ opioid receptor antagonist in development.
Footnotes
Acknowledgements
The author is grateful to Dr. Sharath Hegde, Dr. Jan Smith and Dr. Ross Vickery for their comments on the manuscript during its preparation.
