Abstract
The development of new, abnormal and leaky blood vessels, termed choroidal neovascularization, causes loss of vision and quality of life as a result of many ocular diseases. A current therapy for a leading cause of irreversible blindness in older people in the United States, age-related macular degeneration, is intravitreal injection of ranibizumab. However, ranibizumab has also been used off-label to successfully treat a number of other ocular diseases causing significant ocular morbidity, including retinal vascular occlusion and diabetic macular edema. Despite its efficacy, the association of detectable serum levels of ranibizumab and arterial thromboembolic events in major follow-up studies since its approval by the United States Food and Drug Administration as well as its high cost relative to the possible existence of cheaper, equally efficacious alternatives has underscored the importance of understanding the mode of action and clinical utility of this novel pharmacotherapy.
Keywords
Introduction
The development of new blood vessels in the retina and choroid, with subsequent bleeding, exudation and edema, results in loss of vision and quality of life in many ocular diseases including diabetic retinopathy and age-related macular degeneration (AMD). The pathogenesis of retinal and choroidal neovascularization (CNV) has not been fully elucidated, but extensive evidence implicates that abnormal production of vascular endothelial growth factor (VEGF) plays a critical role.1–6 Intravitreal anti-VEGF agents have recently become commonplace for treating ocular neovascularization and in 2006, the United States Food and Drug Administration approved the use of ranibizumab (Lucentis, Genentech, USA) for the treatment of neovascular (also known as “wet” or “exudative”) AMD. Since then, this agent has become a first line treatment for neovascular AMD. It has also been used off-label to successfully to treat a number of other ocular diseases. Despite the efficacy of ranibizumab, the high cost associated with its use and the possible existence of cheaper, equally efficacious alternatives have underscored the importance of understanding its mechanism of action and clinical trials behind this novel therapy.
Pharmacology and Early Studies
Ranibizumab is an affinity-matured, recombinant humanized immunoglobulin monoclonal antibody fragment with a molecular weight of 48 kD that binds to, and inhibits, the biologic activity of all isoforms of human VEGF by preventing interaction with its receptors. 7 The fragment is one third the size of a full-length antibody and readily penetrates all layers of the retina after intravitreal injection, consequently decreasing cell proliferation, vascular permeability and formation of CNV. 8 Early preclinical investigation of ranibizumab conducted in primate monkey models indicated a reduction in the likelihood of CNV leakage in ranibizumab-treated groups compared with controls and decreased occurrence and size of CNV lesions in one group that was crossed over from control to treatment. 9 Based on these findings it was concluded that intravitreal injections of ranibizumab in animals were well tolerated, preventing the formation of significant CNV, and causing regression of existing CNV. Early human clinical trials determined that ranibizumab had predictable pharmacokinetic properties, with no unexpected accumulation of drug following multiple dosing with serum ranibizumab concentrations predicted to be 90,000-fold lower than vitreous concentrations.10–11
Treatment of Age-related Macular Degeneration
Age-related macular degeneration (AMD) is the leading cause of irreversible vision loss in people 50 years of age or older in the United States.12,13 Currently, the most common therapies for neovascular age-related macular degeneration are intravitreal ranibizumab and bevacizumab (Avastin, Genentech, USA). See Table 1 for a list of major treatment studies using ranibizumab.
Major ocular treatment studies using ranibizumab.
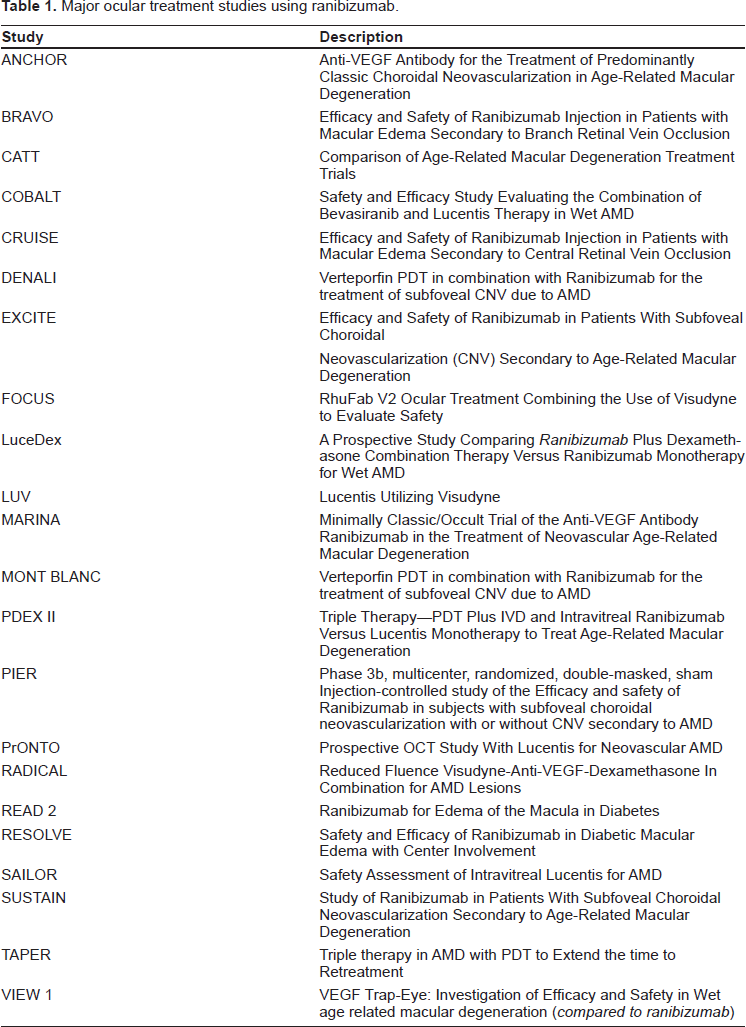
In 2006, the MARINA (Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular Age-Related Macular Degeneration) trial showed that of 240 patients enrolled, 90.0% of patients with neovascular age-related macular degeneration treated with 0.5 mg of ranibizumab lost fewer than 15 letters (either doubling of the visual angle or three lines of visual loss on a standardized visual acuity chart) at a 2-year follow-up, compared with 52.9% of control patients. In addition, 33.3% of treated patients had visual acuity improvement by 15 letters or more, as compared with only 3.8% of controls.
Concurrently with MARINA, the ANCHOR (Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in Age-Related Macular Degeneration) trial compared intravitreal ranibizumab to the previous standard of care for wet AMD, photodynamic therapy (PDT) with verteporfin. The results showed that of the 423 patients enrolled, 94.3% of those given 0.3 mg of ranibizumab and 96.4% of those given 0.5 mg lost fewer than 15 letters, as compared with 64.3% of those in the verteporfin group. Visual acuity improved by 15 letters or more in 35.7% of the 0.3-mg group and 40.3% of the 0.5-mg group, as compared with 5.6% of the verteporfin group. Mean visual acuity increased by 8.5 letters in the 0.3-mg group and 11.3 letters in the 0.5-mg group, as compared with a decrease of 9.5 letters in the verteporfin group. Among 140 patients treated with 0.5 mg of ranibizumab, there were 2 cases of presumed endophthalmitis (1.4%) and 1 case of serious uveitis (0.7%). 17 This rate differs from a previous review of controlled clinical trials by Jager et al. which determined an endophthalmitis rate of 1 to 2 cases per 1000 injections. 18 Because of the serious and potentially vision-threatening nature of endophthalmitis, more extensive use of prophylactic antibiotics and povidone—iodine, as well as greater emphasis on aseptic technique (including use of a lid speculum), was designed into the protocols of subsequent trials and clinical use of ranibizumab in an effort to decrease the likelihood of infection following intravitreal injection. Two more recent, large case series reported an incidence of endophthalmitis after intravitreal injection with either ranibizumab or bevacizumab to be between 0.02% to 0.029%; much lower than was previously reported. Fintak et al. looked at almost 27,000 injections performed in 4 large clinical centers and found a rate of endophthalmitis of approximately 1 in 4500. 19 Pilli et al. looked at over 10,000 injections in community office settings and found the rate of endopthalmitis to be closer to 1 in 3,300. 20
The FOCUS (RhuFab V2 Ocular Treatment Combining the Use of Visudyne to Evaluate Safety) trial was designed to compare the safety, tolerability, and efficacy of ranibizumab treatment in conjunction with PDT versus PDT alone in patients with subfoveal CNV secondary to AMD. At 2 years, 88% of ranibizumab/PDT patients had lost less than 15 letters from baseline visual acuity, compared to 75% for PDT alone, 25% had gained greater than 15 letters compared to 7% for PDT alone, and the two treatment arms differed by 12.4 letters in mean visual acuity change. The visual acuity benefit of adding ranibizumab to PDT in year one persisted through year two. Endophthalmitis and serious intraocular inflammation occurred in 2.9% and 12.4% of ranibizumab/PDT patients respectively and 0% of PDT-alone patients. Incidences of serious nonocular adverse events were similar in the two treatment groups. 21 When comparing the ANCHOR results with the FOCUS results, it appears that the combination of ranibizumab with PDT does not necessarily result in better visual acuity outcomes, and the use of PDT may even reduce the visual acuity benefits achieved with ranibizumab alone. It seems unlikely that combination therapy provides any significant advantage over ranibizumab alone unless the combination of PDT and ranibizumab can decrease the need for frequent retreatment. 15
Other, larger randomized clinical trials examining combination therapies for AMD are currently underway in the United States and Europe, including combining PDT and VEGF inhibitors (LUV, DENALI, MONT BLANC trials), PDT with VEGF inhibitors and corticosteroids (RADICAL and TAPER trials), PDT with corticosteroids (VERITAS trial) as well as comparison of newer agents such as bevasiranib and VEGF-Trap to ranibizumab (VIEW-1 and COBALT trials.)
An alternative dosing regimen decreasing the frequency of injections was hypothesized based on evidence from studies indicating that the pharmacodynamic activity of intravitreal ranibizumab administered monthly for three doses may last 90 days.10,22 The PIER trial was designed to determine whether a less frequent ranibizumab dosing schedule (monthly for three months and then once every three months) would also prevent loss of visual acuity in patients with AMD-related subfoveal CNV and to provide additional safety information. Although quarterly ranibizumab therapy during months 3-2 maintained baseline visual acuity, this regimen was associated on average with a 4.5-letter decline in the maximal visual acuity benefit achieved with monthly induction therapy during months one to three. This loss was accompanied with an increase in vascular leakage on fluorescein angiography and increases in mean retinal thickness once the quarterly dosing regimen began. This overall increase in mean retinal thickness and increase in leakage suggests that at least some of the subjects needed ranibizumab injections more frequently to control the neovascular leakage and that decreased frequency of intravitreal ranibizumab therapy from monthly to quarterly injections may attenuate the improvement in visual acuity that was observed with monthly injections. 23
The PrONTO trial was a two-year study in which 40 patients with subfoveal CNV and a central retinal thickness of at least 300 μm as measured by a noninvasive office-based test called optical coherence tomography (OCT) were enrolled to receive three consecutive monthly intravitreal injections of 0.5 mg ranibizumab. Subsequent retreatment was performed if one of the following changes was observed between visits: a loss of five letters in conjunction with fluid in the macula as detected by OCT, an increase in OCT central retinal thickness of at least 100 μm, new-onset CNV, new macular hemorrhage or persistent macular fluid detected by OCT at least one month after the previous injection. At 1 year, the mean visual acuity improved by 9.3 letters and the mean OCT central retinal thickness decreased by 178 μm. Visual acuity improved 15 or more letters in 35% of patients. These outcomes were achieved with an average of 5.6 injections over 12 months. After a fluid-free macula was achieved, the mean injection-free interval was 4.5 months before another reinjection was necessary. This OCT-guided, variable-dosing regimen with ranibizumab resulted in visual acuity outcomes similar to the MARINA and ANCHOR studies and required fewer intravitreal injections. 24
Ocular and Systemic Complications
Intravitreal ranibizumab injection may lead to detectable levels in the serum. 25 In early phase clinical trials involving patients with AMD, mean serum concentration of ranibizumab one hour after 0.3 mg intravitreal administration was 1.01 ± 1.35 ng/ml. After 28 days serum concentrations were <0.300 ng/ml in 96% of subjects.25,26 Such levels of serum ranibizumab are below the ~10 ng/ml threshold estimated to affect VEGF-A-related activity in humans. There is a single case report of visual and anatomic effect on the contralateral eye following intravitreal ranibizumab injection in a patient with macular edema of different etiologies in each eye, which the authors hypothesized was a result of systemic absorption, 27 however they were unable to substantiate this theory with serum studies.
Early phase III clinical data from the SAILOR trial (Safety Assessment of Intravitreal Lucentis for AMD) reported that serious ocular adverse events were rare but included endophthalmitis and uveitis. 14 A subsequent report determined the incidence of endophthalmitis following ranibizumab treatment to be ~1 in 4,500 injections. 28 Other reported ocular complications include retinal pigmented epithelial tears, 29 persistent ocular hypertension. 30 However RPE tears are rare and may result from a predisposition rather than an effect of treatment, 29 and in a study of 213 consecutive anti-VEGF injections to 120 eyes, intraocular pressure was reduced to less than 30 mmHg in 100% of eyes by 30 minutes. 31
Systemic adverse events, including arterial thromboembolic events and hypertension, were also rare and of similar incidence in the treated and control groups. 14 Subsequent reports from SAILOR have examined the differences in rates of non-ocular significant adverse events in patients receiving intravitreal injections of ranibizumab either 0.5 mg or 0.3 mg. A planned interim analysis found that patients receiving the larger dose had a 1.2% incidence of stroke, compared with 0.3% of patients receiving ranibizumab 0.3 mg. The risk appeared to be higher in patients with previous stroke, 15 however at one year the incidence of non-ocular significant adverse events did not differ between dose groups. 16 In spite of this, a recent meta-analysis of the MARINA, ANCHOR, and FOCUS studies identified 19 (2.2%) of ranibizumab-treated subjects compared to 3 (0.7%) of sham-treated subjects as having experienced a cerebral vascular accident (CVA), whereas 16 (1.9%) of ranibizumab-treated compared to 13 (3.0%) of sham-treated were found with myocardial infarction (MI) during the 2-year observation period. This demonstrates an apparent association of intravitreal ranibizumab with CVA (p = 0.045; OR, 3.24; 95% CI = 0.96-10.95), though not MI (p = 0.193). 32
Treatment of other Ocular Diseases with Ranibizumab
Ranibizumab has also demonstrated efficacy in resolving neovascularization secondary to retinal angiomatous proliferation, as well as several other retinal diseases associated with secondary choroidal neovascularization in a number of small studies (see Table 2).33–46 Spontaneous resolution of vitreomacular traction following a ranibizumab injection has also been reported. 47 However a significant number of larger studies examining ranibizumab therapy for non-AMD ocular conditions have focused on the treatment of central retinal vein occlusion (CRVO),48–51 branch retinal vein occlusion (BRVO), 49 and diabetic macular edema (DME).52–56
Selection of small reports utilizing ranibizumab.
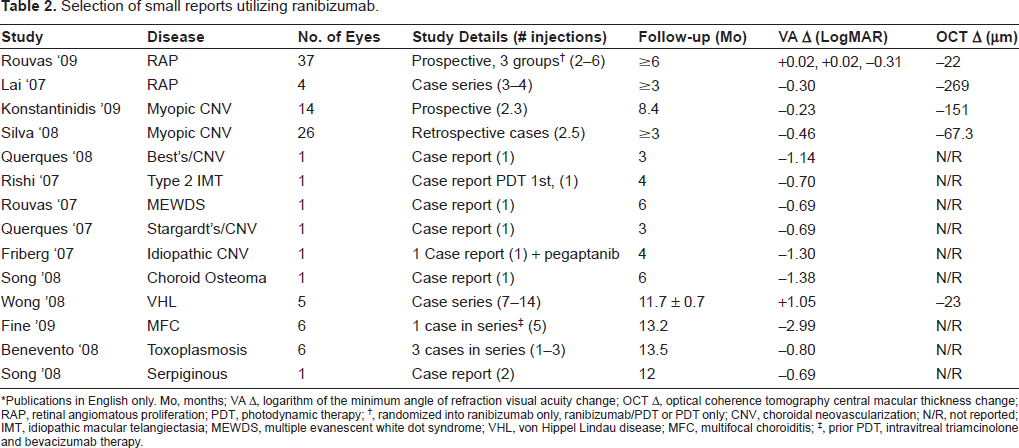
*Publications in English only. Mo, months; VA Δ, logarithm of the minimum angle of refraction visual acuity change; OCT Δ, optical coherence tomography central macular thickness change; RAP, retinal angiomatous proliferation; PDT, photodynamic therapy;
, randomized into ranibizumab only, ranibizumab/PDT or PDT only; CNV, choroidal neovascularization; N/R, not reported; IMT, idiopathic macular telangiectasia; MEWDS, multiple evanescent white dot syndrome; VHL, von Hippel Lindau disease; MFC, multifocal choroiditis;
, prior PDT, intravitreal triamcinolone and bevacizumab therapy.
A recent prospective study examined 20 patients with CRVO and demonstrated that at 12 months of follow-up, the mean visual acuity improved by 18.5 letters and the central macular thickness decreased from 574 to 186 μm using a mean of 8.5 injections. 48 The CRUISE trial (Efficacy and Safety of Ranibizumab Injection in Patients with Macular Edema Secondary to Central Retinal Vein Occlusion) is designed to evaluate the safety and efficacy of either 0.3 or 0.5 mg of ranibizumab versus sham injection in the treatment of macular edema from CRVO. This multicenter trial will study 390 patients and is currently ongoing.
The BRAVO trial (Efficacy and Safety of Ranibizumab Injection in Patients with Macular Edema Secondary to Branch Retinal Vein Occlusion) is also a Phase III, multicenter trial, like CRUISE. However, it is designed to evaluate the safety and efficacy of macular edema from BRVO. This ongoing study will also look at 390 patients.
The READ 2 (Ranibizumab for Edema of the mAcula in Diabetes) trial examined 115 patients randomly assigned to either 0.5 mg of ranibizumab, focal laser photocoagulation (the current gold standard of treatment for DME) or a combination of the two. In the ranibizumab-only group, patients received two monthly injections, then every other month to match up with the combination group that had an injection, then 1 month later had laser photocoagulation. In the ranibizumab-alone group the central fovea thickness decreased by nearly 50% and there was a mean improvement in visual acuity of approximately eight letters, whereas both the laser and combination groups still had substantial residual retinal thickness and only a 3.8 letter improvement. 55 The ongoing RESOLVE (Safety and Efficacy of Ranibizumab in Diabetic Macular Edema With Center Involvement) trial is also evaluating the safety and efficacy of ranibizumab on retinal edema and visual acuity in patients with diabetic macular edema with center involvement in patients who have had focal laser photocoagulation withheld for at least three months prior to enrollment. Six month results have shown a visual acuity improvement of 9.3 letters in the ranibizumab group compared with a loss of 3.5 letters in the control group. 56
Economic Considerations and Alternate Therapy
A vial of ranibizumab containing a single dose of 0.5 mg/ 0.05 mL costs ~$1950 at treatment centers in the United States, Ranibizumab is administered once monthly and initial studies for the management of AMD had lasted 2 years suggesting that costs for a course of treatment may be $46,800. In the United States, treatment for AMD is partially covered through Medicare Part B, with patients responsible for a 20% copayment for each injection. 57 Currently Medicare does not cover any portion of off-label use of the drug. One study carried out at a Veteran's Administration showed that one vial of ranibizumab actually contains enough drug for 4 injections, which can theoretically decrease the cost by 75%. 58 Despite this high cost, Fletcher et al. developed a computerized model of cost-utility analysis for different treatments for AMD and determined that ranibizumab has significant value above observation with supportive care, photodynamic therapy and pegaptanib, and is the most cost effective treatment in most cases. 59 However, this analysis did not include bevacizumab because only short-term data were available for visual acuity and adverse event outcomes. Bevacizumab has a much lower unit cost (~$50 for 1.25 mg/ 0.5 mL) and may be equally therapeutically effective.60–62 The ongoing CATT (Comparison of Age-Related Macular Degeneration Treatments Trials) trial is a large, prospective, double-blinded clinical trial which is comparing ranibizumab to bevacizumab in standard and variable dosing regimens. The results of this study will play a pivotal role in determining the future therapeutic applications of ranibizumab for the treatment of retinal diseases.
Conclusion
Intravitreal anti-VEGF agents have sparked a revolution for the practice of ophthalmology and the prevention of blindness due to conditions involving abnormal vascular development including retinopathy of prematurity, diabetic retinopathy and age-related macular degeneration. Ranibizumab has been approved as a first line treatment for neovascular AMD and other, off-label uses of the drug have shown varying degrees of efficacy. Its application for a wide variety of ocular disorders will likely continue to expand in the future. However, despite its efficacy, high costs and the potential for serious ocular and systemic side effects may limit its ubiquitous use. The existence of cheaper alternatives has emphasized the need for a better understanding of ranibizumab's mechanism of action and expanding clinical spectrum as well as the need for structured clinical trials studying the cost, efficacy and continued development of other intravitreal anti-VEGF agents.
Conflict of Interest
The authors have no financial interest in any of the products mentioned herein.
