Abstract
Breast cancer is a very common diagnosis in women; approximately 30% of patients with breast cancer will develop bone metastases. In addition, breast cancer patients experience adjuvant treatment related bone loss. Bisphosphonates (BPs) are the standard of care for maintaining bone health in patients with cancer. The purpose of this paper is to review the evidence on the use of zoledronic acid (ZA) in breast cancer. Therapy with ZA is generally safe and well tolerated. ZA has become established in the management of patients with breast metastatic bone disease (MBD), reducing the morbidity by decreasing the rate of skeletal-related events (SREs) and the time to develop first and subsequent SREs. Among different BPs, the strongest evidence in this setting is available for ZA. However, emerging data is currently addressing the benefit of anti-RANKL therapy versus ZA. Several trials in early breast cancer have also investigated ZA in other settings, such as for protection of bone density and in adjuvant therapy.
Keywords
Introduction
Breast cancer is the most common cancer in women, with more than 190,000 new cases estimated in 2009 in the United States. It is also a leading cause of cancer related deaths among women worldwide, with more than 1,3 million new cases and approximately 465,000 deaths every year. 1
The skeleton is one of the preferred sites of metastasis for breast cancer, with an incidence of metastatic bone disease (MBD) among the metastatic setting of 65%–75%. 2
Despite the progress that has been made in the treatment of breast cancer, most patients with advanced tumors and MBD are at high risk for developing skeletal-related events (SREs), with a 68% chance of experiencing at least one SRE in the first 24 months (pathologic fracture, need of radiation or surgical therapy, spinal cord compression and hypercalcemia of malignancy).3,4 SREs are associated with increased mortality; for example experience of a pathological fracture increases the risk of death in breast cancer patients by 32%. 5 SREs also cause considerable morbidity, dramatically reducing patients’ quality of life. Finally, MBD is a major cause of pain in breast cancer patients and bisphosphonates (BPs) are effective to palliate pain in this context. 6
MBD results from the interactions between cancer cells located in the bone marrow and normal bone cells, thereby providing the rationale for bone-targeted therapies.
Zoledronic acid (ZA) is a potent inhibitor of osteoclast-mediated bone resorption, and has became established in the management of patients with MBD, 7 with the aim of preserve patient's functional independence and quality of life, by preventing and delaying SREs, and palliating and controlling bone pain (reducing the need for analgesics and palliative radiotherapy).
In addition, patients with breast cancer face other challenges to skeletal health even before development of MBD. Adjuvant chemotherapy and endocrine therapy may have a negative impact on bone of women with early breast cancer. ZA, by inhibiting osteoclastic bone resorption, can also be used to prevent bone loss associated with breast cancer therapy.
Besides that, there is emerging data that suggests that ZA may also have anti-tumor effects and that may have a role in prevention of breast cancer recurrence. In fact, metastasis results from a multi-step process, including tumor cell growth, migration, adhesion to extracellular matrix, extravasation into distant tissues, angiogenesis, and avoidance of immune surveillance, 8 and both preclinical and clinical evidences suggest that ZA can inhibit many of these steps.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
BPs are stable analogues of pyrophosphate that bind to hydroxyapatite bone mineral surface and are selectively internalized by osteoclasts, where they affect osteoclast-mediated bone resorption in a variety of ways. 9
The first generation BPs, such as etidronate, tiludronate and clodronate are incorporated into adenosine triphosphate (ATP)-containing compounds, thus inhibiting cell function. The second generation of nitrogen-containing BPs (N-BPs), such as alendronate, risodronate and ZA, has a different mechanism of action and is more potent by several orders of magnitude. 9
Within the osteoclast, N-BPs target the mevalonate pathway, via inhibition of diphosphate synthase (FPPS).10–12 FPPS inhibition causes loss of pyrophosphates, required for prenylation (ie, post-translational lipid modification) of signaling GTPases, such as Ras, Rho and Rac, that are essential for cell function and survival. 13 As consequence, defective intracellular vesicle transport 14 and loss of prenylated proteins occur, leading to osteoclast apoptosis, via activation of the caspases's cascade. 15 Additionally, FPPS inhibition by N-BPs also causes an accumulation of isopentenyl diphosphate, which is metabolized to an intracellular ATP analogue, triphosphoric acid 1-adenosin-5‘-yl ester- [3-methylbut-3-enyl] ester (Apppi). 16 Mitochondrial ADP/ATP translocase is inhibited by Apppi, causing loss of mitochondrial membrane potential and direct apoptosis induction.16,17 Trough these mechanisms ZA is reported to be the most potent inhibitor of osteoclast-mediated bone resorption.
In addition to inhibition of osteoclast-mediated bone resorption, there is increasing evidence that BPs, including ZA, may directly affect tumor cells. BPs anti-tumor activity may include: inhibition of tumor cell adhesion and invasion;18,19 anti-angiogenic effects, 20 it has been shown that ZA induces significant and long-lasting modifications of circulating serum angiogenic factors in cancer patients;21,22 immunomodulating anti-tumor effects;23–25 inhibition of tumor cell growth and induction of tumor cell apoptosis;26,27 reduction on the release of bone derived cytokines and growth factors, essentially rendering bone “less attractive to tumor cells”.22,28–30
Furthermore, BPs enhance the ability of antineoplastic agents to inhibit breast cancer cell invasion,31,32 and synergistically induce breast cancer cell apoptosis when combined with clinically relevant concentrations of chemotherapy drugs such as paclitaxel and doxorubicin.26,33,34
In oncology clinical practice, ZA is administered intravenously (I.V.) (4 mg in 100 ml 0.9% saline over 15 minutes). In bone metastases setting, patients receive ZA 4 mg I.V. every three or four weeks. The I.V. administration of ZA results in a peak of concentration in plasma of 1–2 μM, which is cleared from the circulation within a few hours. 35 By 24 hours, ZA concentration in plasma falls to less than 1% of the concentration at the end of the infusion, although remaining detectable up to day 29 after administration. 36
Approximately half of the circulating BP is taken up by the skeleton, the exact amount depending on gender, age and rate of bone resorption at time of first administration. Nevertheless, the rate of uptake by bone is always very fast, and BPs are almost cleared from circulation upon the first passage through bone. The compound not retained in bone, is excreted in urine, with little accumulation of BPs in extraosseous tissues.37,38 BPs are then released from bone during osteoclast resorption, or through passive processes, 38 and once released can be excreted in urine or rebind to the bone, what explains the long retention times. 39 Retention of BPs in bone have been estimated to range from 1–10 years in humans. 37
Clinical Role of ZA in Breast Cancer–-Clinical Trials
ZA was studied in different settings; we will review the data from clinical trials that addressed its use in: metastatic breast cancer, breast cancer adjuvant therapy associated bone loss, and prevention of breast cancer recurrence. Key elements of the studies are summarized in Tables 1 and 2.
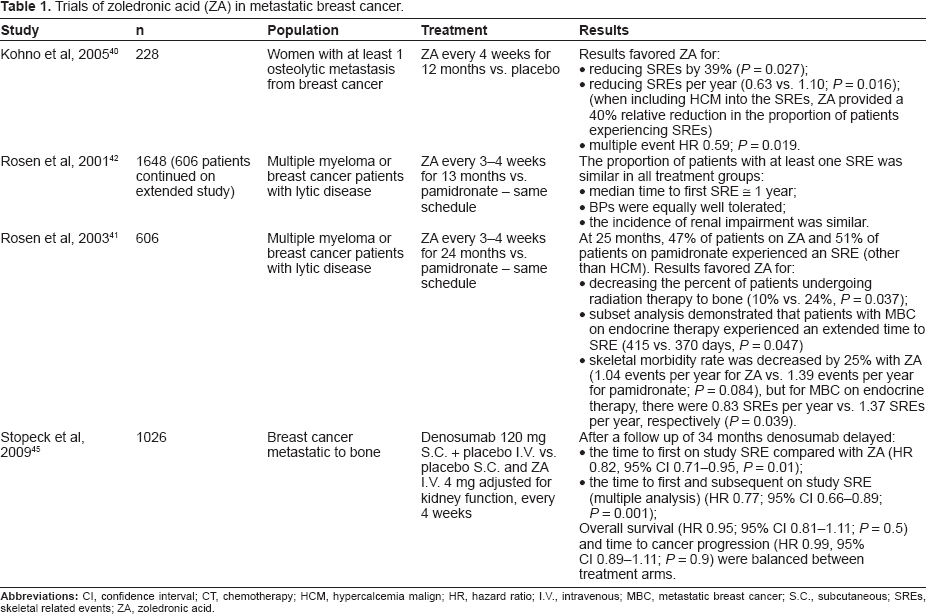
Trials of zoledronic acid (ZA) in metastatic breast cancer.
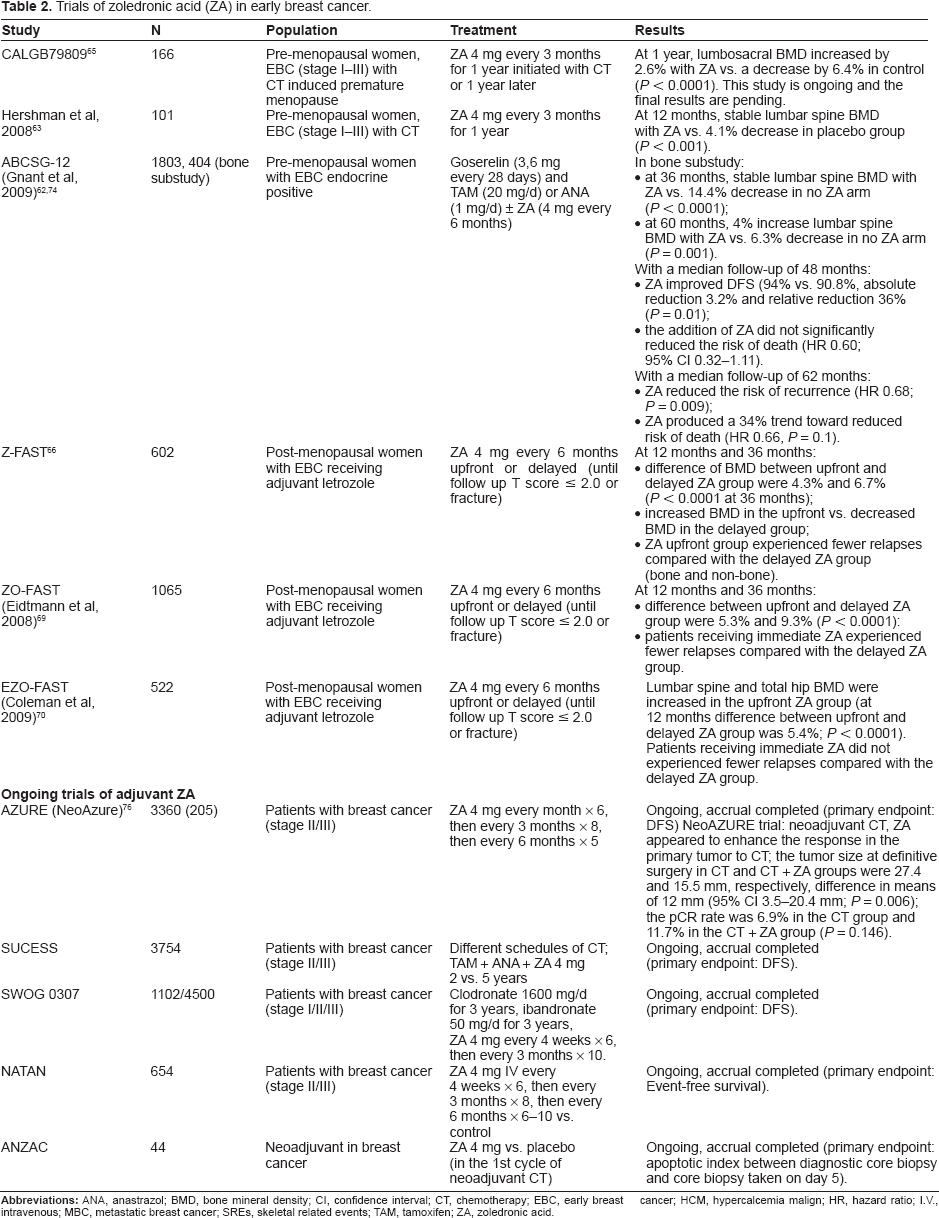
Trials of zoledronic acid (ZA) in early breast cancer.
Metastatic breast cancer
ZA effectively decreases the risk of SREs, which include fracture, need for surgery or radiation to bone, spinal cord compression, and hypercalcemia of malignancy, in women with breast cancer and bone metastases, having demonstrated statistically significant clinical benefit across multiple endpoints.40,41
In a trial designed to investigate the efficacy and safety of ZA for the treatment of bone metastases from breast cancer, 228 women with bone metastases were involved and ZA was compared to placebo, for one year. It was demonstrated that ZA reduced the rate ratio of SREs by 39% (P = 0.027); the number of SREs per year (0.63 vs. 1.1; P = 0.016); the risk of SREs (reduced by 41%; risk ratio = 0.59, P = 0.019); and the time to develop a first SRE (median not reached vs. 364 days on placebo; P = 0.007). 40
In a phase III study that compared pamidronate and ZA for two years, it was appropriately showed that the proportion of patients with at least one SRE was similar for pamidronate and ZA (46% vs. 50%). However, ZA reduced the overall risk of developing any skeletal complication of treatment by 20% when compared with pamidronate (relative risk (RR) = 0.799; P = 0.025). In the subset of patients with lytic bone lesions, it was demonstrated greater efficacy of ZA over pamidronate (RR = 0.80, 95% CI 0.66–0.97).41–43
There are some evidences that ZA can provide survival benefits in the metastatic setting. A retrospective analysis from patients with bone metastases enrolled in three phase III trials showed that in patients with highly aggressive or advanced disease, ZA had a positive effect on overall survival (OS) that was independent of SREs prevention. 44
A recent phase III study compared denosumab (anti-RANKL antibody) vs. ZA for the incidence of SREs in patients with breast cancer metastatic to bone. After a follow up of 34 months, denosumab significantly delayed the time to first SRE compared with ZA (HR 0.82, 95% CI 0.71–0.95, P = 0.01), and also significantly delayed the time to first and subsequent on study SREs (multiple analysis, HR 0.77; 95% CI 0.66–0.89; P = 0.001). OS (HR 0.95; 95% CI 0.81–1.11; P = 0.5) and time to cancer progression (HR 0.99, 95% CI 0.89–1.11; P = 0.9) were balanced between treatment arms. 45
Breast cancer adjuvant therapy associated bone loss
Adjuvant systemic therapies for breast cancer (chemotherapy or endocrine therapy) are associated with an increased risk for bone loss.46,47 Such risk is differentially defined in the post-menopausal or pre-menopausal setting.
Post-menopausal women have an increased risk of osteoporosis, independently of bone loss associated with breast cancer therapy. Besides that, several studies showed the impact of adjuvant breast cancer therapies in this scenario. A prospective observational study of post-menopausal women receiving adjuvant chemotherapy without endocrine therapy reported losses in lumbar spine bone mineral density (BMD) ranging from 0% to 10% within one year of chemotherapy. 48 Aromatase inhibitors, widely used in adjuvant endocrine therapy for breast cancer, are clearly associated with an increased risk of both loss of bone mass and fracture. In the Arimidex, Tamoxifen, Alone or in Combination (ATAC) trial, among anastrozole-treated patients, there was a decrease in median lumbar spine and total hip BMD from baseline to five years (-6.08% and -7.24%, respectively) compared with the tamoxifen group (lumbar spine BMD +2.77%; total hip BMD +0.74%).49–53
In the pre-menopausal setting, chemotherapy, associated with the development of amenorrhea, plus the anti-estrogen therapies (tamoxifen) and the ovarian ablation, have a negative effect on the bone. Previously, other BPs, such as clodronate or risedronate, have shown to halt bone mass loss in the context of adjuvant breast cancer.54–61 However, with the completing of adjuvant therapy, there can be recovery of bone mass, although it does not seem to return to baseline.59,61,62 For this subset of patients there are no established guidelines for screening of bone loss related with adjuvant treatment.
Several trials tested the efficacy of ZA in the prevention of bone loss associated with adjuvant treatments.
Pre-menopausal setting
Data from different trials support the use of ZA in the pre-menopausal scenario, for the management of chemotherapy related bone loss, where it decreases the rate of bone loss and improves bone mineral density.63–65
The CALGB 79809 trial assigned 166 women who were at least 40 years old, with stage I to III breast cancer, which developed chemotherapy-induced ovarian failure, to be randomized to ZA (4 mg every three months, initiated one year after chemotherapy or 12 months later) or placebo. In a preliminary report with one year of follow up, there was a significant increase in lumbosacral BMD in ZA arm (+2.6% vs. -6.4%). 65
In a trial with 101 patients with early breast cancer, ZA (4 mg) every three months for one year, lead at 12 months to stabilization in lumbosacral BMD in ZA arm, compared with 4.1% decrease in placebo group.63,64
Also, data from different trials support the use of ZA in the management of bone loss related with endocrine treatment. In the Austrian Breast and Colorectal Cancer Study Group (ABCSG)-12 trial, a subset of 401 pre-menopausal women receiving adjuvant GnRH therapy with goserelin plus either tamoxifen or anastrozole for three years, were randomly assigned to ZA (4 mg every six months) or placebo. After three years of treatment, there was a significant overall bone loss in patients who did endocrine treatment without ZA (-14.4%, mean T score reduction -1.4) compared with stabilization of BMD in ZA-treated patients. Two years after cessation of the endocrine intervention, although there was partial recovery of BMD, patients who received endocrine treatment without ZA still had bone loss (lumbar spine BMD -6.3%), and patients who received ZA showed a significantly increased lumbar spine BMD (+4%). 62
Post-menopausal setting
The adjuvant aromatase inhibitor studies, Z-FAST, ZO-FAST and EZO-FAST, enrolled nearly 2,200 post-menopausal women with early stage breast cancer, randomized to either upfront or delayed therapy with ZA 4 mg every six months (until follow up T score ≤ 2.0 or fracture). The primary endpoint was a change in lumbar spine BMD at 12 months. In each one of these studies, upfront ZA prevented bone loss, increased BMD above baseline, and decreased fracture incidence. However, the use of delayed ZA in the control arm decreased the statistical power for fracture prevention end-point.66–70
Prevention of breast cancer recurrence
As previously mentioned, preclinical and early clinical studies have suggested that BPs may possess inherent antitumor activity, including antiangiogenic, antiproliferative and proapoptotic properties, which may be synergistic with chemotherapy agents.26,31–34
Clinical trials investigating BPs as adjuvant therapy have been performed and additional studies are ongoing.
A secondary endpoint of Z-FAST, ZO-FAST and EZO-FAST trials was time to disease recurrence. In the 36-months update of the ZO-FAST trial, patients receiving upfront ZA experienced fewer relapses compared with the delayed ZA group (HR 0.57; P = 0.018),69,71 and fewer relapses outside bone were also observed. A reduction in disease free survival rates in favor of adjuvant ZA was observed in the smaller Z-FAST study, 66 but this is counter-balanced by a small excess of relapses in the upfront arm the EZO-FAST study. 72
In the study ABCSG-12, that included 1803 pre-menopausal patients, the primary end point was disease-free survival, with a median follow-up of 48 months. The addition of ZA (4 mg I.V. every 6 months for three years) to endocrine therapy, as compared with endocrine therapy without ZA, significantly improved disease-free survival (94% vs. 90.8%, with an absolute reduction of 3.2% and a relative reduction of 36%). Interestingly, as in Z-FAST and ZO-FAST studies, patients in the ZA arm also had fewer relapses outside bone. The addition of ZA did not significantly improved survival, besides having a trend toward reduced risk of death (HR = 0.60; P = 0.11), probably the number of events was insufficient for reliable statistical analysis (HR 0.60; 95% CI 0.32–1.11). 73
A 62 month follow-up of this study presented in the 10th CIBD Meeting in 2010, confirms a significant increase in disease-free survival in favor of the ZA arm. 74
A meta-analysis of trials which randomized patients to BPs or not in the adjuvant setting, did not find significant reductions in the overall number of deaths, overall disease recurrences, distant relapse, bone metastases, or any kind of relapse. 75 However, in the subgroup analysis, use of ZA was associated with a significant lower risk for disease recurrence (odds ratio (OR) 0.68; 95% CI 0.48–0.95), but not death (OR 0.62; 95% CI 0.39–1.06).
It is clear that the results of additional clinical trials are needed before BPs would be widely used in the adjuvant setting for improvement of breast cancer outcome. Important phase III studies are ongoing, like AZURE, SUCCESS, NATAN, SWOG 0307 and ANZAC that enrolled thousands of patients, and were designed to specifically investigate adjuvant ZA therapy on risk of breast cancer recurrence.
In the AZURE (Adjuvant Zoledronic Acid to Reduce Recurrence) trial, adjuvant ZA (4 mg every month x 6, then every three months x 8, then every six months x 5) with standard adjuvant treatment was compared to adjuvant treatment alone, in 3,360 patients with stage II/III breast cancer. Data from the subgroup analysis of 205 patients who received chemotherapy before surgery, suggest a possible anti-tumor effect of ZA in combination with neoadjuvant chemotherapy. The residual pathologically assessed tumor size at surgery was significantly smaller in patients receiving chemotherapy and ZA vs. chemotherapy alone (the mean residual tumor size was 27.4 mm for the chemotherapy alone group vs. 15.5 mm for the combination group; 95% confidence interval: 3.5–20.4 mm; P = 0.006). The complete pathological response rate was also higher in the group of chemotherapy and ZA (6.9% in the chemotherapy group and 11.7% in the combination group; P = 0.146). 76
Safety
Therapy with BPs is generally safe. The most common complications are acute phase reactions, renal insufficiency, hypocalcemia, and ONJ. Association of ZA with atrial fibrillation, stroke and atypical fractures of the femoral diaphysis remains uncertain, although it was reported.
The acute phase reaction may occur in approximately one third of patients receiving ZA for the first time. Usually it occurs within the first 48 hours of infusion and is self limited within 24 to 48 hours. 77 Typically, the symptoms of acute phase reaction can be managed with analgesics and do not constitute a reason for treatment discontinuation. 7
The mechanism of nephrotoxicity of ZA appears to be different from that responsible for the nephrotic syndrome in patients receiving pamidronate. In one report, biopsy-confirmed acute tubular necrosis was found. 78 Significant renal impairment attributable to ZA is uncommon; the nephrotoxicity of BPs is both dose- and infusion time-dependent (8 mg vs. 4 mg and with infusion durations less than 15 minutes). Therefore, renal toxicity may be reduced by respecting infusion duration, optimizing hydration prior to BP therapy and avoiding concurrent nephrotoxic medications.41–43,79–81
Relevant hypocalcemia related to ZA is not common due to the existence of compensatory mechanisms, the most important being the increased secretion of parathyroid hormone. However, in the case of blockade of these compensatory mechanisms (eg, prior parathyroidectomy, low vitamin D levels), hypocalcemia can occur. It is mandatory to screen calcium values before ZA administration.82,83
ONJ is considered by the American Association of Oral and Maxillofacial Surgeons as the persistence of exposed bone in the oral cavity, despite adequate treatment for 8 weeks, without local evidence of malignancy and no prior radiotherapy to the affected region. 84 ONJ it is increasingly recognized as a complication of chronic intravenous therapy using high potency BPs in patients with malignancy. ZA-related ONJ was first described in 2002. Nine months after ZA was approved in the United States for prevention of cancer-related SREs, FDA received reports of nine patients with MDB treated with ZA, who developed ONJ. 85
In breast cancer, the estimated incidence of ONJ varies between 1 and 4.6%.86,87 However the frequency of ONJ appears to be even lower in early stage cancer patients compared with the bone metastasis setting. In recent and ongoing trials of ZA, ONJ incidence varied between less than 0.1% with twice-yearly administration68,70,88 and 0.7% with dosing up to 8 times/year in the AZURE trial. 89 For unclear reasons, there appears to be a 10-fold greater risk of ONJ with ZA compared with pamidronate.88,90–93 This may be due to its greater potency, as demonstrated by larger reductions in collagen type-I degradation products (N-telopeptide). 42
The pathophysiology of ONJ is unknown. It is not clear whether ONJ is due to direct oral toxicity to the bone and/or soft tissues from BPs, posterior infected; or if it is primary due to osteomyelitis worsened by BPs. It is probably a multifactorial process, that includes and it is favored by BPs inhibition of bone resorption and bone remodeling, poor dental hygiene, and/or a dental manipulation of some sort (eg, dental extraction, advanced periodontal disease), and the simultaneous use of glucocorticoids or chemotherapeutic agents.92,94–102
The incidence of ONJ can be further reduced by implementing preventive dental care. In one study from the National Cancer Institute of Milan, preventive dentistry resulted in an approximately 70% reduction in ONJ incidence. 103
There are reports of the use teriparatide in the management of severe osteonecrosis of the jaw that was resistant to conventional therapy, however until data on safety are available it should not be used.104–106
The association of ZA with atypical femoral fractures was not identified in clinical trials. 107 However, several cases of atypical subtrochanteric hip fractures in patients taking long-term BPs have been reported.108–113
A secondary analysis of the results of three large, randomized BPs trials was performed, including the Fracture Intervention Trial (FIT), the FIT Long-Term Extension (FLEX) trial, and the Health Outcomes and Reduced Incidence with ZA Once Yearly (HORIZON) Pivotal Fracture Trial (PFT). 284 records for hip or femur fractures among 14,195 women were analyzed. A total of 12 fractures in 10 patients were classified as occurring in the subtrochanteric or diaphyseal femur, with a combined rate of 2.3 per 10,000 patients per year. In the HORIZON-PFT trial, ZA vs. placebo, had a RH = 1.50 (95% CI, 0.25–9.00) for ZA use. 114 Although increases in risk were not significant, confidence intervals were wide. In this study, the occurrence of fracture of the subtrochanteric or diaphyseal femur was very rare, even among women who had been treated with BPs for as long as ten years. Thereby, it seems that there was no significant increase in risk associated with ZA use. 114
At this point the association of ZA with atypical fractures of the femoral diaphysis remains uncertain, although additional research is needed.
The first study reporting a possible relation of cardiovascular events and ZA was the HORIZON trial, an international, multicenter, randomized, double-blind, placebo-controlled trial involving post-menopausal women with osteoporosis, randomly assigned to receive either ZA or placebo at baseline, 12 and 24 months. The number of patients who had arrhythmia in the ZA group was significantly higher than that in the placebo group (266 vs. 203 patients, 5.3% vs. 6.9%; P = 0.003). Serious atrial fibrillation was more common among ZA group (50 vs. 20 patients, 1.3% vs. 0.5%; P < 0.001). Among 559 patients who underwent electrocardiography, the prevalence of atrial fibrillation (2.1% in the ZA group and 2.8% in the placebo group) and other electrocardiographic abnormalities did not differed significantly between the study groups. No difference was observed in the occurrence of the serious adverse event of stroke (2.3% in both study groups), being the incidence of death due to stroke of 0.5% in the ZA group and 0.3% in the placebo group (P = 0.15). 115
In patients with recent hip fracture, randomized to ZA or placebo, the incidence of cardiovascular events was similar in the two groups. A total of 24 patients in the ZA group (2.3%) and 39 patients in the placebo group (3.7%) had a serious adverse event of arrhythmia. Atrial fibrillation occurred in 12 patients in the ZA group (1.1%) and in 14 patients in the placebo group (1.3%). 116
An FDA statement released in 2008, concluded that, across all studies, no clear association between BPs exposure and the rate of serious or no serious atrial fibrillation was observed, recommending that health care professionals “should not alter their prescribing patterns for bisphosphonates”. 117
However, in a recent study that included patients with cancer who were treated with I.V. BPs (using Surveillance, Epidemiology, and End Results (SEER)–-Medicare-linked data), BPs use was modestly associated with an increased risk for atrial fibrillation (HR = 1.30; 95% CI 1.18–1.43), all supraventricular tachycardia (HR = 1.28; 95% CI 1.19–1.38), and stroke (HR = 1.30; 95% CI 1.09–1.54). The risk for all supraventricular tachycardia increased 7% for each increase of five BP dose equivalents (HR = 1.07; 95% CI 1.02–1.12). 118
A clear association of atrial fibrillation, stroke, and BPs is unproven, but physicians must be aware of the possible cardiovascular adverse events associated with ZA.
Patient Preference
In general ZA administration is considered convenient. Most of patients with metastatic breast cancer and indication to BPs therapy agree with the I.V. administration, since it can help ensuring adherence and persistence and can be combined with chemotherapy in an outpatient clinical setting. The infusion times of ZA are the most convenient and preferred (15 min vs. up to 2 h). In a study evaluating patient preference for ZA vs. pamidronate, 92% preferred ZA because of the shorter infusion time, less disruptive to their daily schedule. 119
However, there are also oral BPs approved in metastatic breast cancer, that can be considered for those patients that prefer this route of administration and do not need to attend regular hospital care. Oral BPs imply regular intake, must be taken on an empty stomach and in an upright position, and there are reports of low persistency.7,120,121
Place in Therapy
Regarding the efficacy and safety data mentioned above, the current guidelines still recommend, as standard of care, ZA therapy for women with breast cancer and evidence of bone metastases. An appropriate option is 4 mg over 15 minutes every three to four weeks. ZA therapy should be continued, in the absence of toxicity, at least for two years. 7 There will be emerging data addressing the benefits of anti-RANKL therapy in this setting.
For pre-menopausal women receiving adjuvant endocrine/chemotherapy treatment with low BMD (T-score ≤ -2.5) or risk factors for osteoporotic fractures, can be rational to address the possibility of ZA treatment 4 mg every six months. The same principle can be used for post-menopausal women initiating aromatase inhibitors who have a BMD T-score ≤ -2.5, or for women with a BMD T-score between -1.0 and -2.5 who have risks for fracture, for which other than aromatase inhibitors can be recommended treatment with ZA (4 mg every six months is a reasonable dosage option).
Nevertheless, the preliminary results of the CALGB 79809 and Hershamn studies, that showed a benefit of ZA for bone health; the results of ABCSG-12 trial, with benefits for BMD and in disease free survival; and the results of the Z-FAST, ZO-FAST and EZO-FAST trials that showed benefits in prevention of associated bone loss, reduced risk of fractures with aromatase inhibitors therapy and probably increased disease free survival, can lead in the future to an overall use of upfront ZA in early breast cancer.
Conclusions
The use of ZA has an established place in the management of bone metastatic breast cancer, and in the prevention of bone loss due to endocrine therapy in women's at risk. Data of ongoing large phase III trials will further increase our knowledge about the adjuvant benefits of ZA. The use of ZA is generally safe, but physicians and nurses should have experience in the prevention of ONJ and provide proper surveillance of renal function and cardiovascular adverse events. This recommendation is more striking in the context of metastatic bone disease where the dose regimen should be 4 mg I.V. every three or four weeks.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and not under consideration by any other publication and has not been published elsewhere. Luis Costa has received honoraria from Novartis and Amgen, and speaker fees from Novartis and Amgen. The rest of the authors and peer reviewers report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
Footnotes
Acknowledgements
Authors want to acknowledge: S. Casimiro FCT grant BPD/34801/2007.
