Abstract
Clopidogrel is an antiplatelet drug that is used in patients who have had previous cerebrovascular events, acute coronary syndromes, or who underwent percutaneous coronary interventions (PCI) with bare metal or drug-eluting stents. About 5% of patients who undergo PCI have to undergo non-cardiac surgery within 1 year of coronary stent implantation. Patients who receive clopidogrel may be at increased risk of bleeding complications during surgery. The risk of coronary thrombosis after non-cardiac surgery increases, especially when surgery is performed early after stenting, and particularly when antiplatelet agents are withdrawn before surgery. The decision to continue or withhold clopidogrel should reflect a balance of the consequences of perioperative hemorrhage versus the risk of perioperative vascular complications. Close communication among surgeons, anesthesiologists, and cardiologist is necessary to minimize both adverse cardiac risk and surgical risk in those patients.
Keywords
Introduction
The thienopyridine derivative clopidogrel is an antiplatelet drug that is used in patients who have had previous cerebrovascular events, recent acute coronary syndromes (ACS), or who underwent recent percutaneous coronary interventions (PCIs) with bare metal or drug-eluting stents. Around 2 million patients undergo coronary dilatation each year in Western countries, and more than 90% of these PCIs involve the placement of intracoronary stents. Several large-scale clinical studies showed that long-term therapy (9–12 months) with aspirin and clopidogrel reduces ischemic complications compared with aspirin therapy alone among patients with ACS or after percutaneous coronary revascularization.1–3 Clopidogrel monotherapy is effective for secondary prevention of ischemic stroke. 4 Currently, clopidogrel is one of the most widely prescribed medications with worldwide sales in 2008 of $8.6 billioAbout 5% of patients who undergo PCI need to undergo non-cardiac surgery within 1 year of coronary stent implantation.5,6 Increasing number of patients who receive antiplatelet treatment for cardiovascular diseases undergo both cardiac and non-cardiac surgical procedures.7,8 Perioperative management of antiplatelet therapy in these situations is often left to the individual experience and discretion of the physician. The traditional approach of stopping the medication 7–10 days before surgery seems to pose considerable danger and requires revision.9,10 This review will focus on one of the most widely employed antiplatelet agents, clopidogrel, its safety in the perioperative setting, and the efficacy in reducing postoperative cardiovascular complications.
Mechanism of Action, Metabolism, and Pharmacokinetic Profile
Many drugs are used specifically to inhibit platelet function. Thienopyridines affect the adenosine diphosphate (ADP) pathway by irreversibly blocking the ADP receptor P2Y12, thereby inhibiting the activation of the GP IIb/IIIa complex and platelet aggregation. 11 The antiplatelet benefits of thienopyridines are additive to those of aspirin that irreversibly inhibits cyclooxygenase 1. Clopidogrel and ticlopidine are equally effective in antiplatelet therapy, but clopidogrel appears to have a slightly more favorable toxicity profile with less frequent liver injury, neutropenia, and thrombocytopenia.12,13 Therefore, clopidogrel has largely replaced ticlopidine. The usual dose is 75 mg per day with or without an initial loading dose of 300 mg. Clopidogrel is a prodrug, and only 10%–15% of absorbed clopidogrel is available for hepatic conversion to an active metabolite in a two-step process. Metabolic conversion occurs rapidly and neither native clopidogrel nor active metabolites are detectable in the plasma beyond 2 h following oral clopidogrel administration. Its pharmacological activity depends on the cytochrome P-450 (CYP)-dependent oxidation to the active antiplatelet agent. The CYP isoenzymes CYP2C19, CYP1A2, and CYP2B6 are responsible for the first step of metabolic conversion, whereas CYP2C19, CYP2C9, CYP2B6, and CYP3A4 are responsible for the second step. CYP2C19 contributes prominently to both steps and CYP3A4 substantially to the second step. Variants in genes encoding CYP enzymes that less efficiently metabolize clopidogrel have been associated with poorer cardiovascular outcomes in patients with post-myocardial infarction. 14 CYP2C19 is also involved in the metabolism of proton pump inhibitors (PPIs), which are commonly co-prescribed with clopidogrel to avoid gastrointestinal (GI) bleeding. 15 Platelet studies have shown that PPIs reduce the antiplatelet effects of clopidogrel. 16 A meta-analysis showed that there was an increased risk (odds ratio [OR], 1.43; 95% confidence interval (CI), 1.15–1.77) for adverse outcomes in patients co-prescribed clopidogrel and a PPI. 17 On the contrary, secondary analyses of clinical trial data have shown no association existed between PPI use and adverse cardiovascular outcomes. 18 Currently, the evidence for a clinically significant interaction between PPI therapy and clopidogrel is highly controversial; more studies are needed.
While the half-life of the active metabolite of clopidogrel is 7–8 h, it seems to be 2–3 times longer in elderly patients. Recovery from the drug requires 7 days because of irreversible platelet inhibition. 19 Clopidogrel seems to have its effect on the platelet precursor, ie, the megakaryocyte, rather than the mature platelet itself. Hence, its platelet inhibition is corrected only by platelet transfusions or through production of new platelets.
Efficacy of Clopidogrel in Ischemic Heart Disease and Stroke
Antiplatelet therapy after coronary stenting
Platelets play a central role in the development of ACS, and antiplatelet agents have been shown to improve clinical outcomes. The beneficial effect of dual antiplatelet therapy over aspirin alone in patients with ACS without ST-segment elevation was established in the Clopidogrel in Unstable Angina to Prevent Recurrent Events (CURE) trial. 1 Analysis of a subset of patients in the CURE trial also showed the efficacy of dual antiplatelet therapy in PCI. 2 There are 2 major types of coronary stents: bare metal stents (BMSs) and drug-eluting stents (DESs). DESs were designed in response to the high incidence of stent re-stenosis due to neointimal hyperplasia with the BMS. Several drugs such as biolimus, zotarolimus, sirolimus, everolimus and paclitaxecel have demonstrated safety and efficacy in this application. Polymer-based sirolimus-eluting stents (Cypher; Cordis Corp., Miami, FL) and paclitaxel-eluting stents (Taxus; Boston Scientific Corp., Natick, MA) are the 2 major DESs for human use. Everolimuseluting stents (Xienence V; Abbott Lab., Abbott Park, IL, Promus; Boston Scientific Corp., Natick, MA) are now increasingly being used. While DESs have significantly reduced the risk for in-stent restenosis, the incidence of late cardiac death or nonfatal myocardial infarction (MI) after discontinuation of clopidogrel is greater in DES-compared with BMS-treated patients. 20 Most of this difference was attributed to an increased rate of thrombosis-related events. The DES thrombosis-related events had a mortality rate of nearly 20%. From an autopsy study, BMSs showed significantly greater endothelialization than DESs regardless of implant duration. Even beyond 40 months, DESs are not completely endothelialized, whereas BMSs are completely covered within 6–7 months. These partially endothelialized, fibrin-rich sites seem to remain a potent thrombogenic stimulus. In high-risk clinical situations such as cessation of antiplatelet therapy or perioperative period, stent thrombosis may develop at these sites. 21 Although the risk is relatively low, stent thrombosis is associated with a high rate of MI and is sometimes fatal. 22 Notably, the collateral function 6 months after coronary stenting is impaired with DESs when compared with BMSs. In the event of stent thrombosis, impaired collateral function in patients with DES could render the thrombosis more dangerous. 23 This raises a difficult question about the replacement of BMSs by DESs: Are we trading a short-term benefit on a relatively benign disorder, namely, re-stenosis with BMS, for a long-term mortality disadvantage with DES?24,25 Accordingly, the joint guidelines of the American College of Cardiology (ACC), the American Heart Association (AHA), and the Society for Cardiovascular Angiography and Interventions (SCAI) recommended that for all patients with ST elevation MI-ACS, aspirin therapy (75–162 mg/day) should be continued life-long, and clopidogrel 75 mg daily (or prasugrel 10 mg daily) should be given for at least 12 months in patients receiving a stent (BMS or drug-eluting stent [DES]) during PCI for ACS,26,27 (Table 1).
Recommended duration of dual antiplatelet therapy after PCI.

Antiplatelet therapy for secondary prevention of stroke
Antiplatelet therapy is used for both the management of acute ischemic stroke and for the prevention of stroke. Aspirin is the most commonly used agent. Long-term administration of clopidogrel to patients with ischemic stroke is also beneficial with a slightly lower frequency of GI bleeding compared with aspirin. 28 The combination of aspirin and extended-release dipyridamole (ER-DP) had greater benefits for secondary stroke risk reduction than aspirin alone. 29 The results of the Prevention Regimen for Effectively Avoiding Second Strokes (PRoFESS) trial showed that clopidogrel monotherapy and aspirin plus ER-DP are equivalent in terms of effectiveness and safety for secondary prevention of ischemic stroke. 4 Adding aspirin to clopidogrel in high-risk patients with recent ischemic stroke or transient ischemic attack did not reduce the risk of major vascular events compared with clopidogrel monotherapy. The risk of life-threatening or major bleeding is increased by the addition of aspirin. 30 Thus, only selected patients with a coronary or arterial stent placement should be treated with clopidogrel plus aspirin. Compared with coronary artery disease, most acute events are caused by unstable plaque rupture, and intense platelet inhibition does not seem to be efficacious for secondary stroke protection. Stopping antiplatelet therapy in high-risk patients, particularly those with coronary heart disease, increases the risk of ischemic stroke. 31
Safety of Perioperative Use of Clopidogrel
Preoperative revascularization
The risk of perioperative cardiac complications can be assessed by clinical predictors, cardiac functional status, and the underlying risk of the surgical procedure, 32 (Table 2). Many patients with coronary disease who require non-cardiac surgery do not benefit from preoperative revascularization. In the Coronary Artery Revascularization Prophylaxis (CARP) trial, which enrolled 510 patients, coronary artery revascularization [coronary artery bypass graft (CABG) or PCI] before elective vascular surgery did not significantly alter the long-term outcome. 33 The trial also showed no reduction in the number of postoperative MIs, deaths, or days in the hospital. The results of the CARP trial suggest that coronary revascularization may be necessary for only selected patients with unstable coronary syndrome or other very high-risk diseases.
Cardiac risk stratification for noncardiac surgical procedures.
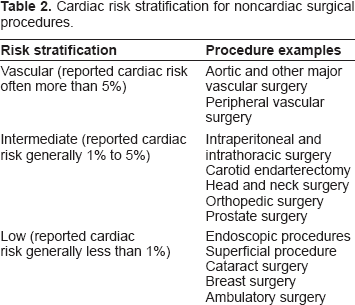
Several reports have shown that in those patients who require PCI and need subsequent non-cardiac surgery within 6 weeks after PCI, the rate of MI and mortality (average 30% and 20%–40%, respectively) was 5–10 times higher than that in matched patients undergoing the same operation under maximal medical therapy or after appropriate delay.34,35 These events are often due to stent thrombosis, which may be caused by premature discontinuation or reduction of antiplatelet therapy. Although stents are currently used in the vast majority of PCIs, coronary revascularization in such patients seems to be more safely performed with balloon angioplasty. Surgery should be delayed for at least 1 week after balloon angioplasty to facilitate healing of the vessel injury at the balloon treatment site. Sometimes, stenting cannot be avoided during PCI due to the complexity of the lesion. Thus, ACCF/AHA guidelines 36 state that if the bleeding risk of surgery is low, stenting may be performed and dual antiplatelet therapy may be continued. If the bleeding risk of surgery is high, balloon angioplasty without stenting is suggested for surgery needed in 14–29 days, bare metal stenting for surgery needed in 30–365 days, and drug-eluting stenting for surgery needed after 12 months. Administration of aspirin should be continued perioperatively.
Patients with prior PCI
Unscheduled non-cardiac surgery in patients who have undergone prior PCI presents special challenges, particularly with regard to the management of dual antiplatelet agents required in those patients who have received coronary stents. 37 Most ischemic episodes tend to start at the end of surgery and during emergence from anesthesia. In the postoperative setting, the risk of ACS is further aggravated by the augmented release of endogenous catecholamines, increased platelet adhesiveness, and decreased fibrinolysis, which are characteristic of the acute phase reaction.38,39 A recent meta-analysis revealed that aspirin withdrawal preceded up to 10.2% of acute cardiovascular syndromes in patients after PCI. 40 In patients undergoing early surgery, discontinuation of antiplatelet therapy during the perioperative period may be a major cause of the increase in major cardiac events. The earlier the surgery is performed after stenting, the higher is the risk for stent thrombosis. 41 Although the risk associated with non-cardiac surgery performed after 6 weeks was lower than that when the surgery was performed before 6 weeks, the mortality was still 4-fold higher for surgeries performed between 6 weeks and 1 year compared with those performed beyond 1 year. 5 The excessive risk of early surgery after PCI might be due to the high-risk of in-stent thrombosis during the perioperative period. In addition to premature discontinuation of antiplatelet therapy, other key predictors of stent thrombosis are renal failure, bifurcation lesions, diabetes mellitus, and low ejection fraction. 42 For at least 2 years after PCI, cardiac outcomes after non-cardiac surgery are similar for both DESs and BMSs. 5 Some considerable risk endures over at least 2–3 years. 43 In fact, perioperative stent thrombosis has been reported even after 5.5 years of DES implantation. 44 The interruption of aspirin therapy alone is also risky and may lead to stent thrombosis even more than 1 year after DES implantation.
Surgical hemorrhage with perioperative antiplatelet therapy
Patients receiving antiplatelet therapy may be at increased risk of bleeding complications. The most common site of spontaneous bleeding in patients treated with clopidogrel is the gastrointestinal tract. Hematuria and epistaxis are also common. Intracranial or intraoccular is rare complication. 45
A meta-analysis of 474 studies comparing surgical bleeding in patients with or without aspirin treatment revealed that aspirin therapy increased the rate of bleeding complications by a factor of 1.5. It did not lead to increased severity of bleeding complications, except for intracranial surgery and transurethral prostatectomy. In intracranial neurosurgery, aspirin is a major contributing factor to a fatal outcome, increasing the risk of postoperative intracerebral hematoma. 46
Clopidogrel has become a double-edged sword, particularly when surgical intervention is required. This issue was first raised in cardiac care. 47 Pre-operative clopidogrel exposure increased the risk of hemostatic reoperation and the requirements for blood and blood product transfusion during and after CABG surgery on average by 30%–50%, 40 but most of the studies have been performed in cardiac surgery with full intraoperative heparinization for cardiopulmonary bypass. In off-pump elective CABG surgery with prophylactic-dose heparin, clopidogrel exposure even within 2 days of surgery evoked only a minor bleeding risk. 48 Few studies are available on the effect of clopidogrel in non-cardiac surgery. There is no indication to interrupt dual antiplatelet therapy for dental procedures and oral surgery. In urological surgery, prostate biopsy, flexible nephroscopy, and transurethral resection of the prostate (TURP) can be performed without causing major complications in patients receiving antiplatelet agents. 49 Photoselective laser vaporization is associated with a lower bleeding risk in endoscopic prostatectomy. 50 In orthopedic surgery, hip fractures, which are the most common non-elective surgery in elderly patients, can be operated on 3 days after stopping administration of clopidogrel without increased bleeding risk and mortality.51,52 In ophthalmic surgery, clopidogrel use before cataract surgery increased minor complications of sharp needle and subtenon's cannula local anesthesia but did not increase sight-threatening local anesthetic or operative hemorrhagic complications. 53 Posterior chamber procedures can be performed in patients on aspirin therapy, but these procedures require clopidogrel withdrawal. In GI endoscopy, antiplatelet agents do not need to be discontinued for low-risk procedures such as endoscopic biopsy. Stopping clopidogrel therapy should be considered in patients undergoing high-risk procedures such as endoscopic sphincterotomy, polypectomy, and endoscopic submucosal resection. In a study of 147 patients undergoing colonoscopic polypectomy, the rate of delayed post-polypectomy bleeding was higher in patients receiving clopidogrel compared with those who did not (3.5% vs. 1.0%), but those were not serious. 54 In transbronchial lung biopsy, despite the fact that clopidogrel use greatly increases the risk of bleeding, no patient required transfusion, and each bleeding episode was controlled by the endoscopic route. 55 In patients undergoing pacemaker or cardioverter-defibrillator implantation, dual antiplatelet therapy increased the risk of bleeding complication defined as need for pocket exploration or blood transfusion. 56
Antiplatelet therapy also presents problems in regional anesthesia. Spinal-epidural hematoma is a rare but potentially devastating complication of neuraxial blockade in patients treated with clopidogrel. Epidural hematomas have been reported after both spinal and epidural anesthesia; in both cases, administration of clopidogrel was stopped 7 days before the regional technique.57,58 Current recommendations by the American Society of Regional Anesthesia and Pain Medicine (ASRA) produced in 2003 suggest that without prior platelet transfusion, clopidogrel therapy should be stopped 7 days before neuraxial blockade. 59 On the other hand, from the chart review of 306 vascular surgical patients who received epidural analgesia without perioperatively stopping clopidogrel therapy, no postoperative neurological complications resulting from the use of epidural analgesia were found in any of these patients. 60 Stopping clopidogrel therapy or platelet transfusion before surgery may be necessary if regional neuraxial blockade is thought to be essential.
Dual antiplatelet therapy with aspirin and clopidogrel carries a 0.4%–1.0% increased absolute risk of major bleeding compared with aspirin alone, and the available reports indicate that it does not appear to cause an increase in surgical complications or mortality in most of the cases. For elective procedures, it is safer to postpone surgery until clopidogrel can be withdrawn with lower risk.
Recommendations
There are no alternative therapies to oral antiplatelet therapy. Although most surgeons are familiar with warfarin as an anticoagulant and perioperative management in terms of elective heparin bridging or reversal, there is no evidence to support heparin bridging in preventing intraoperative stent thrombosis, probably because antithrombins have no antiplatelet activity. In contrast, the cessation of heparin in a patient who does not receive antiplatelet agents has been shown to cause platelet activation and a rebound phenomenon that may increase the risk of perioperative stent thrombosis. Furthermore, preoperative use of enoxaparin has been shown to increase postoperative bleeding and re-exploration in cardiac surgery compared to clopidogrel use. 61 A bridge using an anti-GP IIb/IIIa agent such as eptifibatide or tirofiban (half-life: 2 h) is a possible substitution for clopidogrel. The substance can be given safely until 6 hours before surgery. 62
In clopidogrel monotherapy for primary prevention, clopidogrel can be withheld 5 days before surgery as needed. For secondary prevention of ischemic stroke, clopidogrel should be continued, except in high-risk surgeries in a closed space, eg, the skull, spinal canal, or posterior chamber of the eye. Dual antiplatelet therapy for preventing coronary stent thrombosis after PCI raises an important question regarding the safety of interrupting clopidogrel therapy. The perioperative cardiac risk should be assessed before surgery. Anesthesiologists and surgeons should determine the type and date of implantation. High-risk cardiac situations include surgery within 6 weeks after PCI and BMS, within 3 months after sirolimus-eluting stent implantation, and within 6 months after paclitaxel-eluting stent placement. There have been few reports on perioperative management of patients with second generation DESs. They should be treated following to the first generation DESs. Patients with high-risk stents include those with multiple stents, long stents, stents at proximal location (left main), those with bifurcation lesions, and those with previous stent thrombosis. Aspirin should be continued perioperatively when prescribed for secondary prevention after ACS, MI, stroke, vascular surgery, or PCI with any type of stent. Surgery should be performed in centers with 24-h interventional cardiology coverage. Emergency PCI for revascularization of a thrombosed coronary vessel during the early postoperative period is associated with a greater risk than red blood cell transfusions and surgical hemostasis during the operation (Fig. 1).
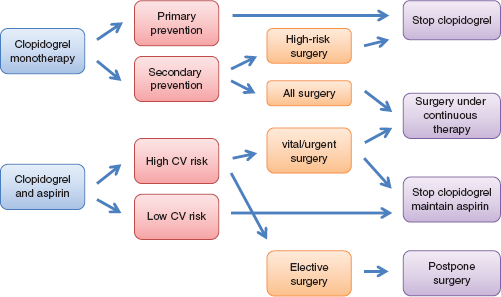
Algorithm for perioperative management of patients receiving clopidogrel.
If the bleeding risk is low, dual antiplatelet therapy should be continued during and after surgery. This includes thoracic, abdominal, vascular, orthopedic, and superficial general surgery, endoscopies, and biopsies. If the bleeding risk is intermediate, administration of clopidogrel should be stopped 5 days before surgery. This is reflected in the ACC/AHA recommendation suggesting that clopidogrel should be withheld for at least 5–7 days in patients scheduled for elective CABG surgery. If the bleeding risk is high, elective surgery should be postponed. This strategy alone will move most patients to the low-risk group requiring continued single agent antiplatelet therapy (aspirin or clopidogrel). If an urgent surgery is needed, administration of clopidogrel should be discontinued 5 days before surgery. Some experts recommend stopping treatment 10 days before surgery. For urgent and emergent surgery, a delay of the surgery until the platelet function has recovered is usually not a feasible option. Under these circumstances, some experts recommend platelet transfusions for the treatment of hemorrhage that continues despite application of the usual hemostatic techniques. Immediately after postoperative bleeding has ceased, clopidogrel therapy should be restarted with a loading dose of 300 mg, because of concerns with regard to stent thrombosis during the postoperative phase of hypercoagulability. Some experts recommend 600 mg clopidogrel as the loading dose, because the complete antiplatelet effect can be observed after 2 h, compared with after 6 h with 300 mg as the loading dose.
Conclusions
The risk of coronary thrombosis after non-cardiac surgery appears to increase, especially when surgery is performed early after stenting, and particularly when antiplatelet agents are withdrawn before surgery. The hemorrhagic risk is usually modest when antiplatelet therapy is continued throughout surgery. The decision to continue or withhold clopidogrel should reflect a balance of the consequences of perioperative hemorrhage versus the risk of perioperative vascular complications. There should be a close liaison among surgeons, anesthesiologists, and cardiologist to minimize both the adverse cardiac and surgical risk in those patients.
Disclosure
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author and peer reviewers of this paper report no conflicts of interest. The author confirms that they have permission to reproduce any copyrighted material.
