Abstract
Background
Sitagliptin was launched into the UK Market in 2007, as the first member of a new class of oral glucose lowering medications, the Dipetidyl Peptidase 4 inhibitors.
Aim
To review the efficacy and safety of sitagliptin and discuss its place in therapy.
Method
Expert review using published reviews and papers published on sitagliptin, information on guideline recommendations for sitagliptin and DPP4 inhibitors, and discussion of the author's use of the agent.
Results
Evidence from a Cochrane review and meta-analysis of 14 trials or study arms suggests that sitagliptin lowers HBA1c by 0.7% in sitagliptin versus placebo trials. Evidence from a pooled safety database of 3415 people taking sitagliptin, and the Cochrane review show that the drug is well tolerated, causes no hypoglycaemia and is weight neutral. No specific signals of concern for the safety of sitagliptin have so far arisen in the pooled database. Guidelines recommend its use in triple therapy with metformin and sulphonylurea in dual therapy with metformin or sulphonylurea or thiazolidinedione in certain circumstances.
Conclusion
Sitagliptin from this initial data appears to be a safe, weight neutral and effective anti-diabetic agent.
Introduction to Sitagliptin
Sitagliptin was approved for use in the USA. in October 2006 and launched into the UK Market in May 2007, as the first member of a new class of oral glucose lowering medications, the Dipeptidyl Peptidase 4 inhibitors, for use in Type 2 diabetes. It is licensed in Europe to be used second line in combination with metformin, thiazolidinedione, or sulphonylurea and third line in combination with metformin plus sulphonylurea.
This article will review its pharmacology, mode of action and its pharmacokinetics. It will discuss the published efficacy studies, and data about its safety and tolerability. It will conclude with a discussion of where sitagliptin has been recommended for use in national and international guidelines, and its emerging place in therapy.
Pharmacological Background–-The Incretin Story
Over 30 years ago it was observed that glucose given orally produced a threefold greater stimulation of insulin release than when an equivalent glucose level was achieved by intravenous infusion. This was called the “incretin” effect. 1 It took a number of years of further research before this was explained through the discovery of the hormones Glucagon like peptide 1 (GLP-1) and Glucose-dependent insulinotropic polypeptide (GIP). These are called “incretin” hormones. These hormones are released by cells in the small bowel in response to food and have a variety of actions including the stimulation of insulin release from pancreatic beta cells…
The incretin effect, which is responsible for up to 50% of the normal release of insulin following glucose ingestion is significantly reduced in Type 2 diabetes. 2 This is believed to be a consequence of impaired GLP-1 secretion, combined with reduced responsiveness of pancreatic islet cells to GIP. 3
The original finding that an intravenous infusion of GLP-1 increased insulin release and reduced fasting glucose and glucagon in people with Type 2 diabetes 4 has resulted in the development of GLP-1 agonists. GLP-1 itself has to be given by continuous infusion as it is rapidly degraded in the circulation by the enzyme dipeptidyl peptidase 4 (DPP-4) which is present in the small bowel and liver, as well as in the kidneys and bone marrow. 7
The development of incretin based therapies has been based upon the development of agents with GLP-1 like actions which are resistant to breakdown by DPP4 or by inhibiting the action of the DPP4 enzyme, using small molecular weight compounds that can be taken orally. This new class of drugs are called DPP4 inhibitors. Sitagliptin is the first of these DPP4 inhibitors to be launched in the UK, Vildagliptin is the second and there are several other DPP4 inhibitors in development.
DPP4 inhibitors prevent GLP-1 degradation and improve circulation time of the active forms of a lot of peptides including GLP-1 and GIP thereby increasing the biological activity of the incretin hormones.
DPP4 Inhibition
DPP4 is the founding member of a family of proteins that exhibit DPP4 activity and/or are structurally similar. 5 They have enzymatic activity characterized by their common post proline cleaving serine dipeptidyl peptidase mechanism. 6 In addition to DPP4, family members include quiescent cell protein peptidase (QPP or DPP7), DPP8, DPP9, fibroblast activation protein and attractin. The action of DPP4 on GLP-1 is clear but the functions of these other enzymes are unknown. 5 DPP4 is widely expressed in many tissues such as liver, lung, kidney, intestine, lymphocytes, capillary endothelium and T-cells, B-cells and natural killer cells. 7
Given the unknown consequences of inhibiting one or more of these DPP4 family members, a potentially important consideration in the selection of DPP4 inhibitors for clinical development is the degree of selectivity for DPP4 over other members of the DASH family of proteins.
Substances that preferentially inhibit other members of the DASH family, particularly those that inhibit DPP8 and DPP9 have been shown to be profoundly toxic in dogs and rats. 5 It is suggested that inhibition of DPP8/9 is also likely to be responsible for at least some of the effects on immune function that have been previously attributed to DPP4 inhibition. 5
The biological importance of DPP4 has been examined in mice with targeted genetic inactivation of DPP4. The mice are healthy, fertile, and have improved metabolic function. They have increased levels of GLP-1 and GIP, they are resistant to diet induced obesity, and are more insulin sensitive. They fail to develop hyperinsulinaemia, hepatic steatosis, or islet hyperplasia after high-fat feeding. 8
Pharmacology of Sitagliptin
Sitagliptin is a potent, competitive, reversible inhibitor of the DPP4 enzyme. It is highly selective for DPP4, and does not inhibit QPP, or DPP 8 or 9. This is very important as the enzymes DPP8 and 9 are very similar to DPP4, but inhibition of them has produced serious toxicity in animal models.
The selectivity and specificity of sitagliptin for DPP4 alone is indicated in Table 1.
Selectivity of Sitagliptin.5

There are two studies that evaluate the effect of sitagliptin on plasma activity of DPP4 in humans. One trial in 11 healthy men given sitagliptin revealed inhibition of plasma DPP4 of 80% or higher on day 10 versus placebo. In addition it was found that sitagliptin at doses of 25 mg/day or higher resulted in a twofold or greater increase in GLP-1 concentrations compared with placebo. 9
A follow up study of 32 middle-aged, normoglycemic obese people compared sitagliptin 200 mgs twice daily with placebo for 28 days. In this study sitagliptin caused 90% inhibition of DPP4 versus placebo and increased active GLP-1 levels 2.7 fold. 10
The dosing of sitagliptin at 100 mgs once daily is recommended as
Pharmacokinetics of Sitagliptin
A double blind, randomized, placebo controlled incremental dose, parallel group study was performed in 11 healthy volunteers. Steady state plasma concentrations were reached on day 3. 9 Sitagliptin is eliminated renally, with a terminal half live of 11.8-14.4 hours. 11 There does not seem to be a clinically meaningful effect of age, gender or obesity on the pharmacokinetics of sitagliptin. 12
A study of sitagliptin has been performed in people with renal insufficiency. To get plasma levels similar to those obtained by taking 100 mgs of sitagliptin daily in subjects with normal renal function a dose of 50 mgs daily was used in people with moderate renal insufficiency and 25 mgs in people with severe renal insufficiency. 13
Published Efficacy Studies–-Cochrane Review
A Cochrane review on dipeptidyl peptidase 4 (DPP4) inhibitors for Type 2 diabetes was published in 2008. 14 It included 14 trials or study arms on sitagliptin.
There were 6 Trials or study arms which compared sitagliptin monotherapy with placebo.
Aschner: 15 sitagliptin 100 mg or 200 mg daily versus placebo.
Goldstein: 16 sitagliptin 100 mg daily versus placebo.
Hanefeld: 17 sitagliptin 25 mg or 50 mg or 100 mg daily versus placebo.
Nonaka: 18 sitagliptin 100 mg daily versus placebo.
Raz: 19 sitagliptin 100 mg or 200 mg versus placebo.
Scott: 20 sitagliptin 10 mg or 25 mg or 50 mg or 100 mg daily versus placebo.
Two trials or study arms compared sitagliptin monotherapy with another oral agent monotherapy.
Goldstein: 16 sitagliptin 100 mg daily versus metformin 1000 mg or 2000 mg daily.
Scotts: 20 sitagliptin 100 mg daily versus glipizide 5 mg to 20 mg daily.
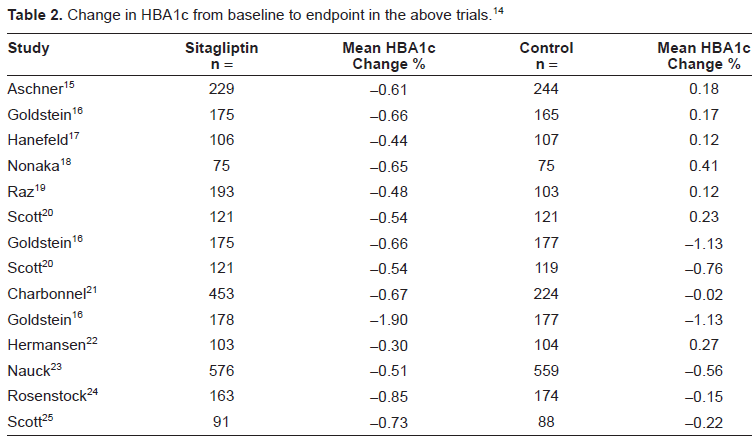
Change in HBA1c from baseline to endpoint in the above trials.14
Six trials or study arms compared sitagliptin combination therapies with other combination therapies of hypoglycemic agents.
Charbonnel: 21 sitagliptin 100 mg daily add on to metformin therapy versus placebo add on to metformin therapy.
Goldstein: 16 sitagliptin 50 mg daily plus metformin 1000 mg or 2000 mg daily versus metformin 1000 mg or 2000 mg daily.
Hermansen: 22 sitagliptin 100 mg daily add on to on going stable doses of glimepiride alone or in combination with metformin versus placebo add on to ongoing stable doses of glimepiride alone or in combination with metformin.
Nauck: 23 sitagliptin 100 mg daily add on to metformin therapy versus placebo add on to metformin therapy.
Rosenstock: 24 sitagliptin 100 mgs daily add on to pioglitazone versus placebo add on to pioglitazone therapy.
Scott: 25 sitagliptin 100 mg daily add on to metformin therapy versus rosiglitazone 8 mg add on to metformin therapy versus placebo add on to metformin therapy.
Trial subjects
The subjects reported in these trials were mostly white, obese, around 55 years of age with a duration of diabetes of between 3 and 5 years.
Sitagliptin Efficacy Results from Cochrane Review 14
Hba1c reduction versus placebo
In the sitagliptin versus placebo trials the weighted mean HBA1c difference between intervention groups was -0.7% (95% confidence interval -0.8% to -0.6%).
In the two studies that investigated sitagliptin monotherapy versus other agent monotherapy a reliable pooled estimate cannot be reported.
In the studies of sitagliptin combined treatments versus another combination of anti-diabetic agents no meaningful pooled estimate could be calculated but generally there was a trend for DPP4 therapy to decrease HBA1c more compared with control.
Weight change
In the placebo versus sitagliptin trials the pooled estimate was a weighted mean difference of 0.7 Kg in favour of placebo. The weight change in the comparator studies suggested less weight gain on sitagliptin than occurred with sulphonylurea.
Sitagliptin Non Inferiority to other Oral Agents–-Data from Papers in Cochrane Review
Non inferior to sulphonylurea–-glipizide
In the Nauck 23 trial the addition of sitagliptin 100 mg compared to glipizide (5 mgs daily up titrated to 20 mgs/daily) provided similar HBa1c lowering over 52 weeks (HBA1c lowering of 0.67%) in patients on stable ongoing metformin therapy.
Non inferior to rosiglitazone
In the Scott 25 study in people inadequately controlled on metformin therapy sitagliptin was non inferior to rosiglitazone 8 mgs when either was added to metformin. The HBA1c reduction in the rosiglitazone group at 18 weeks was -0.79% and 0.73% with sitagliptin.
Sitagliptin Efficacy in Triple Combination
In the Hermansen 22 study people with Type 2 diabetes inadequately controlled on glimepiride and metformin were randomized to sitagliptin 100 mgs daily or placebo. The sitagliptin treatment lowered HBA1c by 0.89% compared to placebo.
Sitagliptin Efficacy Data from Another Systematic Review
Another systematic review and meta-analysis which included DPP4 inhibitor therapy was published in 2007. 26 In this review 8 sitagliptin studies and 12 on vildagliptin (all of which were included in the Cochrane review) were subject to meta-analysis. The conclusion was that DPP4 inhibitors lowered HBA1c by 0.74% (95% confidence interval 0.85%-0.62%) compared with placebo and that they were weight neutral.
Efficacy Data from other Trials Published since the Cochrane Review
In a trial of sitagliptin as monotherapy in 530 Chinese, Indian and Korean subjects with Type 2 diabetes inadequately controlled on diet and exercise, sitagliptin 100 mgs daily reduced HBA1c by 1.0% versus placebo. 27
In a trial of 91 subjects with Type 2 diabetes and moderate to severe renal insufficiency, 65 were allocated to receive sitagliptin. Doses were 50 mgs in moderate renal insufficiency or 25 mgs daily in severe renal insufficiency. These doses were designed to achieve plasma concentrations of sitagliptin observed in people with normal renal function taking 100 mgs daily. Initially the control group of 26 received placebo for 12 weeks. The mean change from baseline was -0.6% in the sitagliptin group and -0.2% in the placebo group, The placebo group then received glipizide 2.5 mgs or 5 mgs daily, up titrated to a maximum of 20 mgs daily. At 54 weeks patients treated with sitagliptin had a mean change from baseline of HBA1c of -0.7%. 13
Safety Data for Sitagliptin
Results from the cochrane review
The review concludes that sitagliptin was well tolerated. It states that discontinuation due to adverse events did not differ significantly between sitagliptin intervention and control arms. The risk ratios of serious adverse events did not show statistically significant differences between groups.
Severe hypoglycemia was not reported in patients receiving sitagliptin. There were no statistically significant differences between sitagliptin and comparator groups.
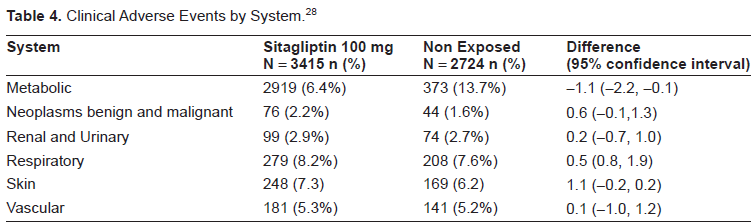
All cause infections (for example nasopharyingitis, upper respiratory tract infection, urinary tract infection) showed a statistically significant increase after sitagliptin treatment (RR 1.15 95% confidence interval 1.02-1.31).
Results from earlier systematic review 26
Studies with DPP4 inhibitors reported no risk of adverse gastrointestinal events (nausea, vomiting, diarrhea and abdominal pain) compared with placebo. DPP4 inhibitors were overall very well tolerated with low absolute rates of adverse events. There was no difference in reported mild to moderate hypoglycemia between DPP4 inhibitors and comparator group. There was an increased risk of nasopharyingitis (6.4% for DPP4 versus 6.1% for comparator) which was more evident for sitagliptin than vildagliptin and an increased risk of urinary tract infection (3.2% with DPP4 versus 2.4% for comparator). Headache was also reported more commonly with DPP4 inhibitors (5.1%) than with comparator (3.9%).
Results from a pooled analysis
Pooled safety and tolerability data from 12 phase 2b and phase 3 studies was published in 2008. 28 The analysis included 6139 patients with Type 2 diabetes receiving either sitagliptin 100 mgs daily (n = 3415) or a comparator agent (placebo or an active comparator, the non exposed group (n = 2724). Patients had been treated with sitagliptin for at least 18 weeks up to 2 years.
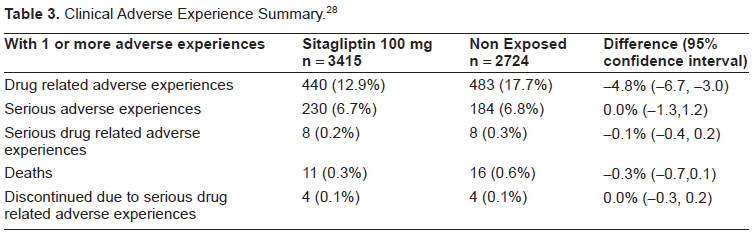
For clinical adverse experiences, the incidence rates of adverse experiences overall, serious adverse experiences, and discontinuations due to adverse experiences were similar in the sitagliptin and non-exposed groups. The incidence rates of specific adverse experiences were also generally similar in the two groups, with the exception of an increased incidence rate of hypoglycemia observed in the non-exposed group. The incidence rates of drug-related adverse experiences overall and discontinuations due to drug-related adverse experiences were higher in the non-exposed group, primarily due to the increased incidence rate of hypoglycemia in this group. For cardiac and ischaemia-related adverse experiences (including serious events), there were no meaningful between-group differences. No meaningful differences between groups in laboratory adverse experiences, either summary measures or specific adverse experiences, were observed.
Inhibition of DPP4 with a highly selective compound like sitagliptin has been shown not to alter measures of immune function in animals in vivo and in human immune cells in vitro. 5 Consistent with these observations, in the pooled analysis there were no meaningful differences observed between treatment groups in the incidence rate, severity and Type of infections. Amori 26 suggested an increased risk for certain infections (nasopharyingitis and urinary tract infection) with DPP-4 inhibitors (including sitagliptin and vildagliptin) In the pooled analysis, only a small numeric increase in the incidence rate of nasopharyingitis in the sitagliptin group was observed, while the incidence rates of urinary tract infections were similar in the two groups. Differences between the pooled database findings and those of Amori et al. may be due to several factors, including the larger cohort of patients followed for a longer duration in the pooled analysis of clinical trials of sitagliptin. Further, the present analysis included all available clinical trial safety data for the specified populations while the Amori meta-analysis could have only included selected adverse experiences meeting different criteria for inclusion in the source manuscripts referenced.
The paper concludes by saying that in patients with Type 2 diabetes, sitagliptin 100 mg/day was well tolerated in clinical trials up to 2 years in duration.
A summary of the adverse clinical events found in the pooled analysis are listed in Tables 3 and 4.
Clinical Adverse Experience Summary. 28
Clinical Adverse Events by System. 28
Safety in patients with renal failure
Sitagliptin is renally excreted. One study, Chan, 13 has looked at the safety of sitagliptin in patients with moderate renal insufficiency, when a dose of 50 mgs daily was used, and in severe renal insufficiency when a dose of 25 mgs daily was used. It was only a small study of 91 patients treated for 54 weeks but in this study sitagliptin was well tolerated and provided effective glycemic control even in patients with end stage renal disease on dialysis.
Summary of safety data
The safety data published from the systematic reviews and from the pooled safety data show that the drug is well tolerated, causes no hypoglycemia and is weight neutral. No specific signals of concern for the safety of sitagliptin have so far arisen. However most studies that have been published so far are short term ones. Long term data on safety and cardiovascular outcomes is therefore needed.
Sitagliptin long term trial
A large, simple trial designed to assess cardiovascular outcome of long-term treatment with sitagliptin used as part of usual care compared to usual care without sitagliptin in patients with Type 2 Diabetes Mellitus and a history of cardiovascular disease with an HbA1c of 6.5%-8.0% on single agent or dual combination antihyperglycemic therapy, has been announced. It is expected to begin recruiting in 2009 with an estimated completion date in 2014/5. The trial number is NCT00790205. 29
Sitagliptin and Effects on Beta Cell Function
A progressive reduction in beta cell mass contributes significantly to the gradual loss of glycemic control seen in individuals with Type 2 diabetes. A major goal of diabetes research is to restore beta cell mass typically lost during the natural progression of Type 2 diabetes. Currently used therapies including metformin, sulphonylurea and insulin do not reduce beta cell loss, and sulphonylurea therapy has been shown to induce beta cell apoptosis in cultured human islets. 30
DPP4 inhibitors in contrast exhibit favourable actions on islet and beta cell mass, morphology and survival in rat and mice animal studies. 8 At present there is no direct way of measuring beta cell mass and morphology in living human beings.
Beta cell function can be estimated using the homeostasis model assessment beta (HOMA-beta) by measuring fasting insulin and fasting glucose. HOMA measurements were reported in some trials included in the Cochrane review and the results are tabulated in that review. The authors state that until more studies are available they will refrain from a meta-analysis pooling of this data. They however report that inspection of the sitagliptin HOMA data seems to indicate that sitagliptin compared to placebo results in increased values of beta cell function measurements. The effect in comparison with other hypoglycemic agents does not seem to be clear cut. 14
Licence Indications for Sitagliptin
In Europe sitagliptin has a license to be used to treat people with Type 2 diabetes
To improve glycaemic control in combination with metformin when diet and exercise plus metformin alone do not provide adequate glycaemic control
To improve glycaemic control in combination with a sulphonylurea when diet and exercise plus maximal tolerated dose of a sulphonylurea alone does not provide adequate glycaemic control and when metformin is inappropriate due to contraindication or intolerance
To improve glycaemic control in combination with a sulphonylurea and metformin when diet and exercise plus dual therapy with these agents do not provide adequate glycaemic control
For patients with type 2 diabetes in whom the use of a thiazolidinedione is appropriate, sitagliptin is indicated
In combination with the thiazolidinedione when diet and exercise plus the thiazolidinedione agonist alone do not provide adequate glycaemic control.
The Place of Sitagliptin in Current Diabetes Treatment Guidelines and Recommendations
Scottish medicines consortium 31
The Scottish Medicines Consortium (SMC) accepted the following new drug for use within NHS Scotland:
sitagliptin (Januvia) for patients with Type 2 diabetes mellitus.
Sitagliptin (Januvia)
Sitagliptin (Januvia) is accepted for use for patients with Type 2 diabetes mellitus to improve glycemic control in combination with a sulphonylurea when diet and exercise plus maximal tolerated dose of a sulphonylurea alone do not provide adequate glycemic control, and when metformin is inappropriate due to contraindications or intolerance; or in combination with a sulphonylurea and metformin when diet and exercise plus dual therapy with these agents do not provide adequate glycemic control.
When added to a sulphonylurea with or without metformin, sitagliptin had a modest beneficial effect on glycated hemoglobin (HbA1c) levels.
Sitagliptin is also licensed for use in combination with thiazolidinedione drugs. The manufacturer's submission related only to the use of sitagliptin in combination with sulphonylureas with or without metformin. SMC cannot recommend the use of sitagliptin in combination with thiazolidinediones at this time.
The manufacturer estimated a net drug budget impact of £3k in 2009 rising to £5k in 2012 based on 50 patients uncontrolled on a sulphonylurea alone in 2009 and 99 in 2012 when adding in sitagliptin rather than a thiazolidinedione. The net drug budget impact of adding sitagliptin instead of a thiazolidinedione in patients uncontrolled on metformin and a sulphonylurea was £11k in 2009 and £21k by 2012 based on 197 patients in 2009 and 392 in 2012. Patient numbers seem low given the Scottish prevalence of Type 2 diabetes so the actual budget impact may be greater than these estimates.
NICE guideline on Type 2 diabetes 32
The NICE guideline on Type 2 diabetes published in May 2008 did not consider DPP4 inhibitors. A rapid update of the glycemic lowering section of this guideline is being undertaken and is due to be published in March 2009. The draft of this rapid update was published on the NICE website for stakeholder comment in October 2007 (www.nice.org.uk).
It gave the following recommendations Consider adding a DPP4 inhibitor as second line therapy instead of a sulphonylurea when blood glucose control remains or becomes inadequate (HBA1c at or above 6.5%) with metformin if
The person is at significant risk of hypoglycaemia and its consequences. This may include older people and those in certain occupations (for example those working at heights or with heavy machinery) or those in certain social circumstances (for example living alone)
The person does not tolerate a sulphonylurea (or it is contraindicated)
Consider adding a DPP4 inhibitor as second line therapy to sulphonylurea monotherapy when blood glucose control remains or becomes inadequate (HBa1c at or above 6.5%) if
The person does not tolerate metformin (or it is contraindicated)
If a trial of metformin in combination with a sulphonylurea does not adequately control blood glucose (HBA1c at or above 7.5%) and human insulin is unacceptable or inappropriate, consider adding sitagliptin
Continue DPP4 inhibitor therapy only if a beneficial metabolic response (at least 0.7% HBA1c reduction in 6 months) occurs and is maintained
Discuss the potential benefits and harms of treatment with a DPP4 inhibitor with the person so that an informed decision can be made
A DPP4 inhibitor may be preferable to a thiazolidinedione for people
In whom further weight gain would cause or exacerbate psychological or medical problems associated with high body weight
In whom a thiazolidinedione is contraindicated
Who have previously had a poor response to or were intolerant of thiazolidinedione therapy
ADA/EASD consensus statement 33
A consensus algorithm for the initiation and adjustment of therapy was published in January 2009 by 7 authors representing the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD) It lists DPP4 inhibitors in Table 1–-Summary of glucose-lowering interventions in the document. It does not however list them in the treatment algorithm under either tier 1 or tier 2 therapies. The text states that DPP4 inhibitors (along with 3 other groups of agents) are not included in the 2 tiers of preferred agents in this algorithm, owing to their lower or equivalent glucose lowering effectiveness compared with the first and second tier agents and/or to their limited clinical data or relative expense. However they may be appropriate choices in selected patients.
Sitagliptin use in my clinical practice
My practice is an urban teaching general practice with 15208 registered patients, 604 of whom are registered as having diabetes (data from 19th September 2008). This gives a prevalence rate of 4.1%. We began to prescribe sitagliptin from around the middle of 2007. We audited the results from prescribing this new treatment in September 2008.
14 people had been prescribed sitagliptin. One was in dual therapy with metformin in someone in whom sulphonylurea was contraindicated, and one was in someone on sulphonylurea in whom metformin was not tolerated. The remaining 12 patients were inadequately controlled on metformin and sulphonylurea and received sitagliptin as a third agent.
Two of the 14 patients did not respond to sitagliptin and it was stopped after 6 months. One went onto basal insulin, the other to exenatide.
Of the 12 who responded to sitagliptin the mean HBA1c reduction after 3 months therapy was -1.08% and the mean weight change was -0.9 Kg. No patient reported any side effects or problems with taking sitagliptin.
The use of sitagliptin in our practice is in line with the draft NICE guidelines, and indicate the major use of sitagliptin is third oral agent addition when metformin and sulphonylurea together do not give adequate glycaemic control.
Summary
Sitagliptin is a potent, competitive, reversible inhibitor of the DPP4 enzyme, and is the first agent in this class to be launched onto the world market. Evidence from a Cochrane review and meta-analysis of 14 trials or study arms suggests that it lowers HBA1c by 0.7% in sitagliptin versus placebo trials.
Evidence from a pooled safety database of 3415 people taking sitagliptin, and the Cochrane review show that the drug is well tolerated, causes no hypoglycemia and is weight neutral. No specific signals of concern for the safety of sitagliptin have so far arisen in the pooled database. However most studies that have been published so far are short term ones. Long term data on safety and cardiovascular outcomes is therefore needed. A long term trial has been designed and registered to provide this data and should begin recruiting patients in 2009.
In Europe sitagliptin has a license to be used to treat people with Type 2 diabetes who are inadequately controlled on monotherapy with metformin, thiazolidinedione or sulphonylurea or who are not adequately controlled on metformin plus sulphonylurea.
A draft of the rapid update of the glycemic lowering section of the Type 2 Diabetes NICE guideline (due for publication in March 2009) suggests that this guideline will recommend its use in line with its license in defined situations.
Disclosure
The author reports no conflicts of interest.
