Abstract
Thrombocytopenia is the most common hematological abnormality in patients with chronic, advanced liver disease. In these patients, the presence of severe thrombocytopenia is an obstacle to the performance of invasive diagnostic and therapeutic procedures, and the current standard treatment for these patients is platelet transfusions, a remedy whose characteristics are far from being ideal. Furthermore, thrombocytopenia in patients with chronic hepatitis C virus infection may render the patients ineligible to antiviral treatment or may limit its efficacy because of premature discontinuation. Although the cause of thrombocytopenia in patients with chronic liver disease is likely multi-factorial, decreased thrombopoietin production by the liver undoubtedly plays a significant role. In this regard, eltrombopag, a non-peptide, orally bioavailable thrombopoietin receptor agonist has been shown to safely increase platelet count in a dose-dependent fashion in both healthy subjects and thrombocytopenic patients with chronic hepatitis C. Furthermore, in this latter group of patients, it has been shown to be superior to placebo in counteracting the myelosuppressive effect of short-term pegylated interferon treatment.
Introduction
The prevalence of thrombocytopenia in patients affected by chronic liver disease is highly variable, and depends upon both the threshold used to define this hematologic abnormality and the severity of the underlying liver disease. 1 Indeed, any thrombocytopenia (i.e. platelet count below 150 × 10 9 /L) can be observed in 5.5% of non-cirrhotic patients with chronic liver disease, and in up to 64% of patients with liver cirrhosis. 2 However, in these patients, this degree of thrombocytopenia is usually associated with no major bleeding, both spontaneous or during invasive procedures. On the contrary, in patients with advanced liver disease, platelet count tends to decrease in parallel with a decrease in liver function and worsening of portal hypertension, 3 and in these patients there is a higher prevalence of severe thrombocytopenia (i.e. platelet count below 50 × 10 9 /L). 4 In these patients, thrombocytopenia and the presence of liver-related coagulopathy may increase the risk of bleeding during both minor and major invasive procedures, 5 and platelet transfusion may be required so as to safely carry out the required diagnostic or therapeutic interventions. Nevertheless, platelet transfusions have several drawbacks, as they may expose the patients to the risks of reactions, alloimmunization and consequent refractoriness to further transfusions, and infections. 6 In patients with advanced liver disease, repeated transfusions may also lead to volume expansion with a detrimental consequence on portal pressure, and in the liver transplant setting platelet transfusions have been associated with adverse outcomes.7,8 Thus, in this clinical situation there is an unmet need for a drug able to safely increase platelet count so as to avoid platelet transfusions.
The description of thrombopoietin (TPO) and of its biological functions put the bases for the development of growth factors with thrombopoietic activity.9,10 Although the development of antibodies that cross-reacted with endogenous TPO prematurely halted the progress of studies with recombinant human TPO and pegylated recombinant human megakaryocyte growth and development factor, the encouraging results in terms of modality of action and thrombopoietic activity of these compounds opened an avenue for the development of “second generation” thrombopoietic growth factors. 11 These are either peptide or non-peptide molecules that have the capacity of binding the TPO receptor and activate megakaryocytopoiesis yet without eliciting the formation of antibodies against the native molecule. 12
Eltrombopag (Promacta®/Revolade™, GlaxoSmithKline) is the first orally bioavailable, small molecule, non-peptide TPO receptor agonist that has been evaluated for the treatment of thrombocytopenia in patients with various pathologic conditions.11,12 A phase I study has shown that eltrombopag administration determines a dose-related increase in platelet count in healthy subjects, 13 and both phase II and III studies in patients with chronic Idiopathic Thrombocytopenic Purpura (ITP) have demonstrated that eltrombopag significantly increases and maintains elevated platelet counts as compared to placebo with a favourable safety profile.14,15 Lastly, in thrombocytopenic patients with chronic liver disease due to hepatitis C virus (HCV) infection, eltrombopag administration proved to be able to safely increase platelet count and enable patients to initiate pegylated interferon and ribavirin treatment. 16
Rationale for the Use of Thrombopoietic Growth Factors in Patients with Chronic Liver Disease
Thrombocytopenia may characterize the clinical picture of patients with various degrees of severity of chronic liver disease, and is the most common hematological abnormality encountered in patients with liver cirrhosis. 17 The pathophysiological mechanisms leading to decreased platelet count in patients with liver disease are several and complex, and more than one mechanism at a time may be responsible for thrombocytopenia in a given patient. 18 Splenic sequestration due to portal hypertension, myelosuppression–- either disease-related or treatment-induced–-and immune-mediated phenomena have all been suggested as putative etiological factors for decreased platelet count in patients with liver disease. 1 The description of TPO and of the alteration in its production in patients with liver disease added a new possible culprit for thrombocytopenia in these patients.19,20 Briefly, TPO is almost exclusively produced–-at a constant rate–-by the adult liver, and is cleared from the circulation upon binding to its receptor (c-Mpl) on both megakaryocytes and platelets. 19 Thus, circulating TPO levels depend upon its hepatic synthesis and peripheral uptake, 20 and it is evident that impairment of the liver functioning mass may therefore cause a decrease in TPO production. In this regard, a series of recent studies showed that in patients with liver disease TPO serum levels may be normal, high, or low although they are nevertheless too low for the degree of thrombocytopenia observed while other studies elegantly showed that liver transplantation restores the correct platelet count-TPO feed-back.21–28
On this basis, it is apparent that molecules able to promote megakaryocytopoiesis through stimulation of the TPO receptor would circumvent the inadequate TPO production inherent to patients with chronic liver disease. However, before entering the clinical arena, these molecules should be proven to be safe, effective across various disease states, have a dose-related effect, and offer cost-effectiveness. Currently, eltrombopag is the only thrombopoietic drug with safety and efficacy data in patients with liver disease which have been publicly presented and published in full. 16 In this review we will detail the pharmacology and mode of action of eltrombopag, evaluating the preclinical and phase I-II study results, and the results of the study which demonstrated the thrombopoietic activity and safety of eltrombopag in chronic liver disease patients.
Pharmacology, Mode of Action and Pharmacokinetics of Eltrombopag
Eltrombopag is a member of the biarylhydrazone class of compounds, with an empirical formula of C25H22N4O4 and a molecular weight of approximately 442D (Fig.1). It is an orally bioavailable, non-peptide, species specific, small molecule that has been identified by screening chemical libraries with the use of BAF-3/TPO-R cell lines. 29 The identification of compounds on the basis of biological activity rather than mere receptor binding affinity has the advantage that the receptor is in its native conformation–-in this case a preformed, unliganded dimer–-and retains the capability of binding the native molecule, thus leading to an additive effect on thrombopoiesis.
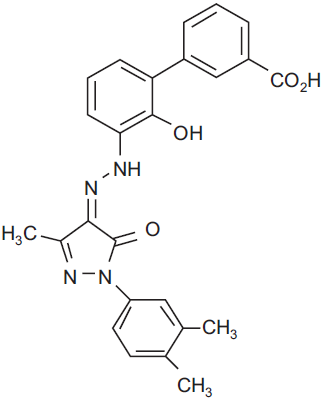
Structural formula of eltrombopag.
In-vitro results have shown that eltrombopag promotes the growth of TPO-dependent cell lines, stimulating differentiation of bone marrow progenitors (CD34 cells) into cells of megakaryocyte lineage, and thus promotes production of platelets. 30 It has been hypothesized that eltrombopag selectively interacts with the transmembrane domain of the TPO receptor, which is expressed on the surface of megakaryocytic precursors and megakaryocytes. 31 Binding of eltrombopag either induces dimerization of the TPO receptor or directly activates the signal transduction mechanism. Likely, eltrombopag binds the TPO receptor at a distance from the binding site for TPO thus initiating signal transduction by a mechanism different from the native molecule, although both the exact site of binding and modality of signal transduction have not been characterised completely (Fig. 2). 11 Stimulation of the TPO receptor results in activation of various signal transduction pathways such as the Janus kinase type 2 signal transducers and activators of transcription signaling pathway, and mitogen-activated protein kinase/extracellular signal-regulated kinase eventually leading to changes in gene expression promoting progression of stem cells along the megakaryocytic pathway, megakaryocyte maturation, and release of normally functioning platelets into the peripheral circulation. 32 Although native TPO has also the potential for platelet activation and degranulation, a recent study has demonstrated that eltrombopag stimulates platelet signal transduction with little or no effect on overall platelet function. 33
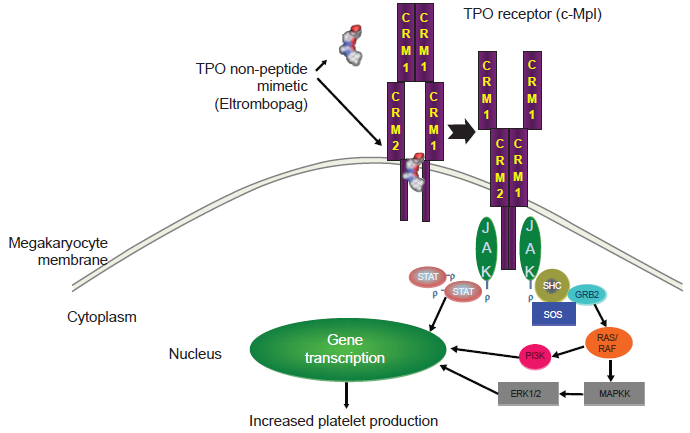
Postulated mechanism of action of eltrombopag. Upon binding to the transmembrane region of the thrombopoietin receptor (c-Mpl), eltrombopag activates the intracellular pathways leading to megakaryocyte differentiation and platelet production.
The pharmacokinetics of eltrombopag is dose-dependent and linear. In healthy humans administered 75 mg eltrombopag for 10 days, the mean Cmax was 7.3 μ/mL (15% coefficient of variability) at approximately 2.5 hours to 5 hours after dosing, with a mean area under the curve of 79.0 μγ.hour/mL (23% coefficient of variability). 34 The T1/2 averages more than 12 hours on day 10 across eltrombopag doses of 10, 20, 30, 50, and 75 mg. There is no accumulation of eltrombopag after once daily dosing at 5 mg and 10 mg, but there is approximately 40% to 50% accumulation at the 20 mg, 30 mg, 50 mg, and 75 mg dose. 34 Concomitant administration of eltrombopag with high-calcium food or an antacid containing aluminium and magnesium is associated with significantly reduced systemic exposure, whereas low-calcium meals is not. 35
Phase I and II Studies
In a phase I study 73 healthy male subjects were randomised to receive either eltrombopag (5, 10, 20, 30, 75 mg daily) or placebo for 10 days. 34 In this study a single oral dose of eltrombopag was not able to increase platelet count; however, repeated dosing led to a dose-dependent rise in platelet count. In particular, none of the subjects in the 5 and 10 mg eltrombopag dose groups, 11% of the subjects in the 20 mg dose group, 67% of the subjects in the 30 mg dose group, and 100% of the subjects in the 50 and 75 mg dose groups recorded an increase in platelet count greater than 20% of the baseline value. The mean increase in platelet count for the 30, 50, and 75 mg eltrombopag dose groups were 24%, 43%, and 50%, respectively (Fig. 3). An evident increase in platelet count was observed after 8 days of repeated dosing, with a the time-to-peak of platelet count after the start of drug administration of 16 days and a return to baseline values by day 22. Noteworthy, there was no rebound thrombocytopenia, this finding being consistent with the additive effect of eltrombopag and native TPO. Both platelet function and aggregation were reportedly unaffected by eltrombopag administration. From the safety standpoint, both incidence and type of adverse events were not different between the groups receiving placebo and eltrombopag and across the treatment groups, and there were no serious adverse events. 34
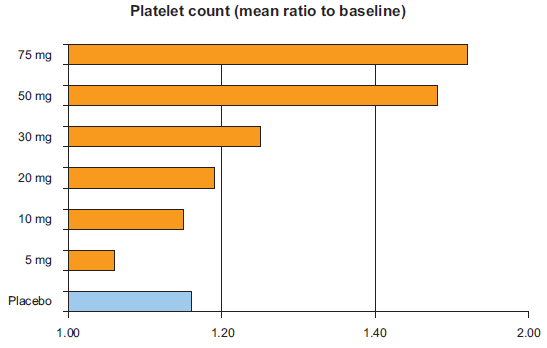
Mean platelet increase as compared to baseline in healthy subjects receiving eltrombopag or placebo daily for 10 days.
This study clearly showed that oral administration of eltrombopag to healthy subjects was able to increase platelet count in a dose-related fashion and without major safety issues, thus supporting further studies in patients with thrombocytopenia.
The initial phase II study of eltombopag in patients with thrombocytopenia was carried out in patients with ITP. Chronic ITP is a severe disease, with 11%-35% of affected patients refractory to conventional treatment, and therefore at risk of bleeding and death. 36 Although ITP has often been regarded as a disease where antibody-mediated destruction of platelets in the peripheral circulation is the main mechanisms responsible for the clinical manifestations, recent clinical studies have demonstrated the presence of co-existent ineffective thrombopoiesis. 37 In a randomized, double-blind, placebo-controlled, phase II study, patients were given placebo or oral eltrombopag at 30, 50 or 75 mg daily for 6 weeks. 38 Platelet counts increased to levels greater than 50 × 10 9 /L in 27%, 65% and 86% in the 30, 50 and 75 mg eltrombopag dosing groups, respectively, as compared to 12% in the placebo group. The platelet counts were maintained for the 6-week period of treatment and returned to baseline on treatment withdrawal. There was a trend toward fewer bleeding events on therapy in patients receiving the 50 mg and 75 mg doses. No treatment-related adverse events were reported from eltrombopag therapy. 38 The efficacy, safety, and tolerability of eltrombopag in ITP patients were further confirmed by the results of a phase III study showing a superiority of eltrombopag 50 mg (increased to 75 mg in the case of non-response) as compared to placebo in increasing platelet count above 50 × 10 9 /mL (59% versus 16%) and decreasing bleeding episodes (odds ratio: 0.49; 0.26-0.89, 95% confidence interval), with a similar safety profile. 39
Phase II Study with the Use of Eltrombopag in Patients with Chronic Liver Disease
The use of eltrombopag in patients with chronic liver disease was recently evaluated in a phase II study. 16 This study evaluated the ability of eltrombopag to initiate and maintain pegylated interferon and ribavirin therapy in patients with chronic hepatitis C virus infection and thrombocytopenia. This is an important clinical setting as patients with a platelet count below 75 × 10 9 /L are usually excluded from antiviral treatment with pegylated interferon and ribavirin. Furthermore, development of thrombocytopenia during antiviral treatment–-a phenomenon related to the myelosuppressive activity of interferon–- dictates a decrease in the dosage of the antiviral drug and even treatment withdrawal, thus representing a major obstacle to successful treatment in a sub-group of patients who are more in the need of cure. In this phase II study, 74 patients with chronic hepatitis C and thrombocytopenia (platelet count below 70 × 10 9 /L; median platelet count 55 × 10 9 /L) were randomized to receive eltrombopag at three different dosages (30 mg, 50 mg, and 75 mg daily) or placebo. Noteworthy, approximately 35% of the study population was in the very-low platelet count sub-group (i.e. below 50 × 10 9 /L). The primary end-point of the study was obtaining a platelet count above 100 × 10 9 /L after 4 weeks of treatment, and it was reached by 75%, 79%, and 95% of the patients treated with 30, 50, and 75 mg of eltrombopag, respectively, and none of the patients treated with placebo (Fig. 4). The increase in platelet count was confirmed to be dose-dependent in patients with liver disease, with a significant increase as compared to baseline in all the groups of patients treated with eltrombopag, and with each dose as compared to placebo (Fig. 4). Forty-eight of the 49 patients who subsequently proceeded to antiviral therapy had a platelet count above the threshold for starting antiviral treatment (i.e. platelet count above 90 × 10 9 /L for pegylated interferon alfa-2a and above 100 × 10 9 /L for pegylated interferon alfa-2b). In an intention-to-treat analysis, the 12-week course of pegylated interferon and ribavirin was completed by 36%, 53%, and 65% of patients receiving 30 mg, 50 mg, and 75 mg of eltrombopag, respectively, and by 6% of the patients who received placebo. Noteworthy, although platelet counts tended to decrease during interferon treatment they remained above the recommended threshold for pegylated interferon dose reduction (i.e. 50 × 10 9 /L) in all the active treatment groups and none of the patients treated with placebo. During the initial 4 weeks of eltrombopag administration side effects were mild in degree (headache, dry mouth, pruritus) and were not differently prevalent across the eltrombopag and placebo groups, while the side effects during the antiviral part of the study were mainly related to pegylated interferon and ribavirin treatment. During the whole study, eltrombopag was discontinued in three patients alone (one due to upper abdominal pain and ascites, one due to neutropenia, one due to retinal exudates), although the study did not allow to evaluate whether these side effects were related to the investigational drug or pegylated interferon and ribavirin therapy.
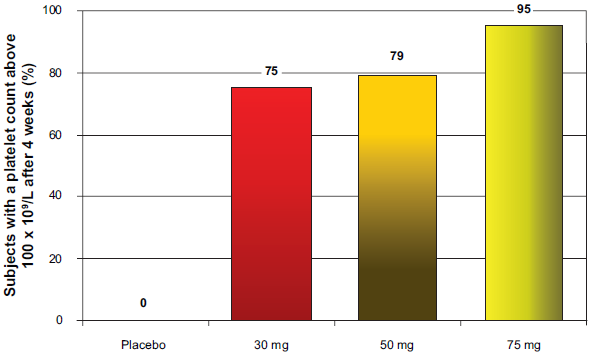
Conclusions
There is an unmet need for drugs able to safely increase platelet count in patients with advanced liver disease. Indeed, in these patients, thrombocytopenia is a limit to both antiviral therapy with pegylated interferon and performance of invasive procedures. As of today, platelet transfusion is the only therapeutic option available to treat this condition. However, platelet transfusions are associated with several shortcomings such as the possibility of transfusion reactions, infectious disease transmission, alloimmunization with subsequent refractoriness to further transfusions, and their effect is only temporary. In patients with advanced liver disease undergoing liver transplantation–-a clinical setting with a high prevalence of severe thrombocytopenia–-platelet transfusions have been associated with negative outcomes, mainly due to transfusion-related acute lung injury.7,8 Thus, the availability of an orally available, effective, and safe drug able to increase platelet counts in patients with advanced liver disease would be welcome. The only study published so far with the use of a thrombopoietic drug in thrombocytopenic patients with liver disease has shown that eltrombopag is able to safely increase platelet count in these patients and enable them to initiate pegylated interferon antiviral therapy. Long-term studies are undoubtedly necessary in order to evaluate the possible occurrence of late side effects of a drug interacting with proliferation and maturation of hematopoetic progenitors. Until this time, a cautious use of the drug should be recommended despite the doubtless importance of such new drugs for critically ill patients in the absence of alternative drugs. Finally, it remains to be established whether the use of eltrombopag will enable more thrombocytopenic patients with hepatitis C virus infection to achieve a sustained virological response to antiviral treatment, and whether its use will decrease the need for platelet transfusions when patients with advanced liver disease have to undergo invasive procedures.
Conflict of Interest
Edoardo G. Giannini reports receiving lecture and consulting fees from GSK.
