Abstract
This review considers treatments of proved efficacy in secondary stroke prevention, with an emphasis on antiplatelet therapy. Most strokes could be prevented, if readily available lifestyle and risk factor modifications could be applied to everyone. In secondary stroke prevention, the same lifestyle and risk factor modifications are also important, along with anticoagulation for patients with cardiac sources of embolus, carotid procedures for patients with significant internal carotid artery stenosis, and antiplatelet therapy. For patients with noncardioembolic ischemic strokes, FDA-approved antiplatelet agents are recommended and preferred over anticoagulants. ASA, clopidogrel, and ASA + ER-DP are recognized as accepted first-line options for secondary prevention of noncardioembolic ischemic stroke. Combined antiplatelet therapy with ASA + clopidogrel has not been shown to carry benefit greater than risk in stroke or TIA patients. Aspirin and extended release dipyridamole appeared to carry a greater benefit over aspirin alone in individual studies, leading to a recommendation of this agent in the AHA guidelines, but the recently completed PRoFESS trial showed no difference in efficacy between clopidogrel and aspirin with extended release dipyridamole, and clopidogrel had better tolerability and reduced bleeding risk.
Introduction to Stroke Prevention
Stroke is the third leading cause of death in the U.S, and the leading cause of neurological disability in adults. Every year, approximately 780,000 persons in the United States suffer strokes, of which about 25% are recurrent strokes. 1 After a stroke or transient ischemic attack (TIA), patients are most likely to have a stroke as the next clinical event, more than myocardial infarction or any other vascular complication.2–5 The first responsibility of the physician managing a patient with a stroke or TIA, therefore, is the prevention of another stroke, or secondary stroke prevention. Over a longer time horizon, however, patients with stroke and TIA are also at risk of myocardial infarctions,6,7 and the most common ultimate cause of death in stroke patients is heart disease.7–9 For this reason, attention must also be directed to cardiac prevention. For secondary stroke prevention, the recommended treatments include risk factor modification, such as management of blood pressure, lipids, and glucose; smoking cessation; anticoagulation for patients with atrial fibrillation and related cardiac sources of embolic stroke; carotid endarterectomy or stenting for patients with carotid stenosis; and antiplatelet therapy. 10
Based on evidence from clinical trials, most strokes could be prevented by the control of five common risk factors: hypertension, hyperlipidemia, smoking, atrial fibrillation, and excessive alcohol consumption. 11 The problem we have in the United States with stroke prevention is not a lack of scientific knowledge, as in the quest for a cure for cancer, but rather a failure to apply readily available knowledge to the people at risk. It is a defect in our health care delivery and health information systems, not a failure to achieve a scientific breakthrough.
The vascular disorders underlying stroke are related to atherosclerotic disease in the coronary and peripheral vascular circulations, but there are some major differences in pathophysiology among the different vascular disease locations. Myocardial infarction virtually always results from an atherosclerotic plaque rupture in a coronary artery, but stroke can result from a variety of mechanisms (see below). Differences are also seen in the risk factors for each vascular disease, even though the same major risk factors, listed above, are all related to stroke, MI, and peripheral arterial disease. For example, the largest single risk factor for stroke is hypertension, whereas dyslipidemia is much more closely associated with coronary artery disease. In peripheral arterial disease, diabetes and smoking are the most prevalent risk factors. Stroke patients are thus not identical to patients with other vascular diseases, and there are some differences in preventive treatments. 12 Another difference between patients with stroke and those with other vascular disorders is that stroke patients appear more susceptible than those with myocardial infarction or peripheral arterial disease to bleeding caused by antithrombotic agents. 12 Stroke patients tend to be older than those with coronary events. In the recent TRITON-TIMI-38 trial for acute coronary syndrome, the bleeding risk of prasugrel over clopidogrel did not outweigh the benefit in most patients, but major bleeding occurred in more patients with a history of stroke or TIA. 13
A variety of pathophysiological mechanisms can lead to stroke, as compared to the homogeneous cause of coronary events, large artery disease with plaque rupture. The principal stroke subtypes are large vessel disease (LVD), small vessel or “lacunar” disease (SVD), cardioembolic, other known cause (e.g. hypercoagulable state, arterial dissection), and cryptogenic (unknown cause).10,14 When strokes recur, approximately 60%-70% are of the same subtype as the initial stroke.15–17 Ischemic stroke subtypes differ somewhat in risk factor associations. Stronger risk factors for SVD include hypertension, smoking, and diabetes,18–20 though the differences may not be as great as previously suspected. 21 Risk factors for LVD include smoking,19,20 abdominal obesity, 22 and dyslipidemia. 23 Stroke subtypes are also associated with varying risks of recurrence, as well as different degrees of severity and impairment. Lacunar stroke has been associated with lower 30-day risk of recurrence, lower 5-year mortality, and better functional outcomes than the other subgroups. 16 LVD stroke has demonstrated the highest 30-day recurrence of ischemic stroke subtypes, and cardioembolic stroke the highest 5-year mortality, >80%. 16
This article will review treatment strategies for secondary stroke prevention. All stroke patients can benefit from lifestyle and risk factor modifications such as low fat and low salt diet, exercise, smoking cessation, and control of blood pressure, glucose, and lipids. Patients with LVD strokes related to severe extracranial internal carotid artery stenosis can benefit from endarterectomy or endovascular procedures such as angioplasty and stenting. Those with large vessel disease involving the extracranial vertebral arteries or intracranial arteries may also benefit from angioplasty and stenting, though these procedures are still considered investigational. Antiplatelet therapy is also typically recommended. In cardioembolic stroke patients with a high-risk source of embolism such as atrial fibrillation or prosthetic heart valves, anticoagulant agents such as warfarin are indicated. 10
Guidelines for Secondary Stroke Prevention
In this article, the principal guidelines cited are The American Heart Association and American Stroke Association (AHA/ASA) evidence-based guidelines for prevention of stroke in patients with history of ischemic stroke or TIA, 10 along with the recent 2008 update. 24 The levels of evidence in these guidelines 10 are included as Table 1.
Definition of classes and levels of evidence used in AHA recommendations.

Recommendations regarding modifiable risk factors
Smoking
All stroke/TIA patients should be counseled not to smoke and to avoid environmental smoke. In addition to counseling, direct medical assistance to help patients quit smoking is recommended, including nicotine products and medications such as bupropion or varenicline (ChantixR).
Alcohol
Small quantities of alcohol (≤2 alcoholic drinks/day for men and 1 drink/day for women) may protect against ischemic stroke, but any alcohol intake increases the risk of hemorrhagic stroke. Excessive alcohol intake increases both ischemic and hemorrhagic stroke risk. Note that the AHA guideline recommendation for avoiding heavy alcohol consumption is Class I, Level of Evidence A, whereas that for the protective effect of small quantities of alcohol is weaker, Class IIb, Level of Evidence C.
Obesity
Obesity appears to be an indirect risk factor for stroke. Recommendations for treatment include weight management through caloric limitation, physical activity, and behavioral counseling. The goals of treatment are BMI 18.5-24.9 kg/m2 and waist circumference <35 inches for women and <40 inches for men.
Exercise
Moderate exercise for ≥30 minutes on most days is encouraged for ischemic stroke/TIA patients who are able to engage in physical activity. For those whose disability precludes independent exercise, a supervised therapeutic regimen is recommended.
Diabetes
In diabetics, strict control of lipid levels and blood pressure is recommended, with angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs) preferred as first-line antihypertensives. Strict glycemic control should target near-normal glucose levels and hemoglobin A1c ≥ 7%, though excessively tight glucose control may result in hypoglycemia and increased mortality. 25
Hypertension
Control of blood pressure is one of the most powerful and effective treatments for secondary stroke prevention. In the PROGRESS trial, treatment with the ACE inhibitor perindopril, usually in combination with a diuretic, indapamide, produced a 28% relative risk reduction in recurrent stroke over a four year period. 26 Two trials involving ACE receptor blockers, or ARBs, ACCESS 27 and MOSES, 28 also found that these drugs were efficacious in secondary stroke prevention. Recently, the ARB telmisartan was shown to be equal in efficacy in primary stroke prevention to the ACE inhibitor ramipril in the ONTARGET trial; 29 ramipril has had a unique approval for primary stroke prevention, based on the HOPE trial. 30 The combination of telmisartan and ramipril, however, had no better efficacy in stroke prevention and excessive side effects, as compared to either agent alone. 29
The AHA guidelines recommend antihypertensive treatment for all ischemic stroke/TIA patients, though there has been controversy about how early after the stroke to begin therapy. Recent studies suggest that initiation of therapy before discharge from the hospital improves adherence to antihypertensive therapy. 31 Clinicians should follow the JNC7 (The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure) guidelines 32 in choosing antihypertensive drugs, but the clinical trial evidence in stroke patients supports the use of diuretics with or without ACE inhibitors or ACE receptor blockers. Calcium channel blockers have also been effective (VALUE), 33 but in the LIFE trial, 34 the beta blocker atenolol was less effective in stroke prevention than the ARB losartan. The only caveat to the ACE inhibitor recommendation is the lesser efficacy of these agents in African American patients in the ALLHAT trial.35,36 For this group, diuretics and calcium channel blockers may be the most effective drugs, though combinations of these agents with ACE inhibitors or ARB's can be considered.
Lipids
Lipid lowering therapy has also been found effective in secondary stroke prevention, but this evidence has accumulated only over the past few years. The National Cholesterol Education Panel (NCEP) III guidelines for lifestyle modification, diet, and medications explicitly apply only to those ischemic stroke/TIA patients with elevated cholesterol, coronary artery disease, or evidence of large vessel atherosclerotic disease such as internal carotid artery stenosis. 37 Lipid-lowering therapy, especially with HMG CoA-reductase inhibitors, or “statins,” is recommended, with a goal of LDL-C < 100 mg/dL, <70 mg/dL for very high-risk patients.10,37 Patients with low HDL-C may be treated with niacin or gemfibrozil. 37
Three randomized, placebo-controlled trials of patients with acute myocardial infarction have shown that HMG CoA reductase inhibitors (“statins”) prevent strokes as well as recurrent myocardial infarctions.38–41 The Heart Protection Study 42 also demonstrated a lessened risk of stroke in high risk patients, age 40-80, treated with simvastatin 40 mg daily. Simvastatin was approved for secondary stroke prevention based on this trial, but a subsequent analysis of the stroke subgroup of HPS did not confirm significant secondary stroke prevention; the benefit was primarily in preventing MI or death. 43 Most recently, the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) trial has shown that statin therapy with high dose atorvastatin reduced the risk of recurrent stroke in patients with a history of stroke or TIA. 44 Based on these findings, the AHA/ASA guidelines have been updated in 2008 to recommend statin therapy for all ischemic stroke or TIA patients with LDL > 100. 24
Recommendations for surgical and endovascular treatment in LVD patients
Extracranial carotid disease
Carotid endarterectomy (CEA) is recommended for patients with recent TIA or ischemic stroke and severe (≥70%) ipsilateral internal carotid artery stenosis.10,45–47 CEA is also effective in patients with symptomatic ipsilateral moderate (50%-69%) stenosis of the internal carotid artery, but the benefit is less.10,48 Based on one clinical trial, balloon angioplasty and stenting of the internal carotid artery may be considered for “high risk” patients with symptomatic, severe stenosis of the internal carotid artery. The high risk group included those with inaccessible stenosis, contralateral internal carotid occlusion, radiation-induced carotid disease, restenosis after prior CEA, severe comorbid conditions such as unstable coronary or pulmonary disease, or age over 80 years. 49 Many clinicians have adopted this indication for endovascular rather than surgical therapy. Since patients over 80 years were not included in the studies of carotid endarterectomy for symptomatic carotid artery disease, angioplasty and stenting are often preferred for patients in this age group. A three-year follow-up of the “high risk” study showed no differences between endarterectomy and angioplasty/stenting. 50 Two studies of angioplasty with or without stenting, however, have shown either equivalent 51 or inferior results 52 of stenting as compared to surgical treatment. For non-high risk patients, endarterectomy is still the procedure most supported by the evidence, until further trials, especially the long-awaited CREST trial, 53 are completed.
Extracranial vertebrobasilar disease
Endovascular therapy (angioplasty, stenting) is also carried out on patients with stroke or TIA symptoms and evidence of stenosis in the extracranial vertebral arteries. In the absence of a clinical trial, many clinicians reserve such treatment for patients who fail medical therapy. Vertebral artery angioplasty/stenting is still considered to be an investigational treatment.
Intracranial atherosclerosis
The WASID study 54 showed that patients with TIA or stroke symptoms related to greater than 50% stenosis of the intracranial internal carotid, middle cerebral, distal vertebral, or basilar arteries had approximately a 20% risk of stroke over a two year period. Despite this high risk of recurrent stroke, the study found no significant difference in poor outcomes between high-dose aspirin and warfarin therapy. Warfarin therapy was associated with higher risk of hemorrhage or death. Endovascular therapy such as balloon angioplasty or stenting is employed in such patients, especially those who fail medical therapy, but this treatment, too, is considered investigational. A clinical trial comparing angioplasty and stenting with the Wingspan stent is in progress.
Recommendations for antithrombotic therapy in cardioembolism
In patients with cardioembolic stroke, the principal secondary stroke prevention measure is anticoagulation, usually with warfarin. 10 In the absence of a clear contraindication, patients with atrial fibrillation (AF) and a recent stroke or TIA should receive oral anticoagulants, not antiplatelet therapy. The recommended range of anticoagulation with warfarin is an International Normalized Ratio (INR) of 2.0-3.0.
Oral anticoagulants are also recommended in stroke patients with acute MI and left ventricular thrombus, rheumatic mitral valve disease, and prosthetic heart valves (Table 2). Some clinicians use higher levels of anticoagulation in patients with prosthetic heart valves. Other possible sources of embolus such as reduced cardiac ejection fraction, patent foramen ovale, and atrial septal aneurysm, carry less definite evidence of benefit with anticoagulation, and antiplatelet therapy is a good alternative. Current clinical research is continuing on agents which impair coagulation in a more predictable, dose-dependent manner, such as Factor Xa inhibitors and direct thrombin inhibitors. These agents promise efficacy at least equal to warfarin, with less bleeding risk. It is hoped that these agents will become available soon.
Summary of AHA/ASA guidelines for antithrombotic therapy for prevention of stroke in patients with cardioembolism.10

Recommendations for antithrombotic therapy in TIA or noncardioembolic stroke (atherosclerotic, lacunar, or cryptogenic infarcts)
In patients with noncardioembolic ischemic stroke or TIA, the AHA/ASA Guidelines recommend antiplatelet agents rather than oral anticoagulation to reduce the risk of recurrent strokes (Table 3).10,24 Oral anticoagulants are generally not recommended for patients with noncardioembolic stroke, because of both the lack of evidence of greater efficacy and the increased risk of bleeding complications.
Summary of AHA/ASA Guidelines for Antithrombotic Therapy for Prevention of Stroke in Patients with Noncardioembolic Stroke or TIA (Specifically Atherosclerosis, Lacunar, or Cryptogenic Infarcts).10,24
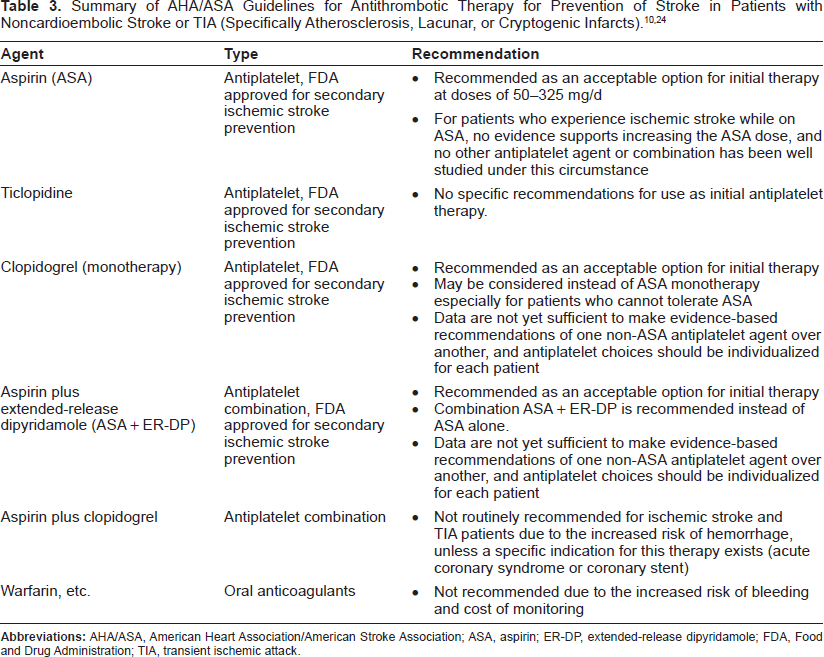
Four antiplatelet regimens have been approved by the FDA for secondary ischemic stroke prevention: 1) aspirin (ASA), 2) ticlopidine, 3) clopidogrel, and 4) a combination of ASA plus extended release dipyridamole (ASA+ER-DP). Ticlopidine is virtually no longer used, because of bleeding complications, thrombotic thrombocytopenic purpura, and other side effects. ASA, clopidogrel and ASA + ER-DP are currently the recommended antiplatelet agents for secondary stroke prevention.10,24
Aspirin
Aspirin inhibits platelet function by binding irreversibly to the cyclooxygenase enzyme in the platelet, thereby reducing the generation of prostaglandins such as thromboxane-A2, a stimulator of vasoconstriction and platelet aggregation. The platelet is inhibited for its lifespan of approximately 10 days; the platelet does not have a nucleus and cannot produce more cyclooxygenase enzyme. Prostaglandin systems in the vessel wall, which produce prostacyclin, a vasodilator and platelet inhibitor, can recover after low dose aspirin. ASA has been proved effective in many studies, with a recurrent-event risk reduction of approximately 13%-22%.55–57 These numbers, however, point to the limited efficacy of aspirin. Most patients with TIA and stroke will fail aspirin therapy, and this should be expected. Other problems with aspirin include the interference with its function by nonsteroidal anti-inflammatory drugs such as ibuprofen, gastrointestinal bleeding risk, and occasional aspirin allergy.
Another controversial issue concerning the efficacy of aspirin in stroke prevention is the lack of proof of an optimal dose. Two trials comparing ASA dosing regimens demonstrated no additional benefit and a greater risk of nonfatal major gastrointestinal hemorrhage with higher as compared to lower doses.58,59 In the United Kingdom Transient Ischaemic Attack trial (UK-TIA), patients with minor ischemic stroke or TIA (n = 2435) were randomized to 600 mg ASA twice daily, 300 mg ASA once daily, or placebo. The risk of major stroke, MI, or vascular death was 15% less with ASA than placebo, but the two aspirin doses were equal in efficacy. Gastrointestinal hemorrhage was more common with the 1200 mg dose than the 300 mg dose. 58 In the Dutch TIA trial of patients with TIA or nondisabling stroke (n = 3131), 30 mg ASA was compared with 283 mg in preventing vascular death, nonfatal stroke, or nonfatal MI. Again, there was no difference in stroke prevention between the two doses of aspirin, but the 30 mg dose group had fewer bleeding complications. 59
For patients who have a stroke while receiving ASA, no clinical trial evidence supports an increase in the aspirin dose. “Aspirin resistance” has also been a controversial topic, but there is poor agreement on the best measurement of such resistance, since the methods of assaying platelet inhibition do not always agree, and there has also been little control for lack of adherence to the aspirin regimen. Likewise, switching to alternative antiplatelet agents has not been studied for “aspirin failures.” In the WARSS trial, 60 a clinical trial of aspirin versus warfarin in noncardioembolic stroke patients, the two agents had no significant difference in efficacy. Moreover, patients who had already failed aspirin when they had their index strokes had a higher risk of recurrent stroke in the course of the trial, but there was still no superior efficacy of warfarin over aspirin.
Clopidogrel
Clopidogrel inhibits platelet aggregation by binding to the adenosine diphosphate (ADP) site on the platelet. This is a different mechanism of action from that of ASA, but the same as ticlopidine and the newer agent, prasugrel.
13
The principal clinical trial supporting the use of clopidogrel in stroke patients is the CAPRIE study, a large trial of 19,185 patients with recent stroke, recent myocardial infarction, or peripheral vascular disease. Patients were randomized to receive aspirin 325 mg daily or clopidogrel 75 mg daily.
4
In the combined groups, clopidogrel monotherapy was more effective than ASA (8.7% overall RRR, p = 0.043) in reducing the risk of stroke, myocardial infarction, or vascular death. The difference did not reach statistical significance in the subgroup of stroke patients. The greatest benefit, RRR 23.8% (
Dual therapy with clopidogrel and ASA for up to 12 months has been shown to be more effective than ASA monotherapy in patients with acute coronary syndrome (ACS), 61 acute ST elevation MI,62,63 and in patients following coronary stents. 64 Even in acute coronary syndrome, however, combined ASA plus clopidogrel carried a higher risk of major bleeding, especially when the aspirin dose was 325 mg (bleeding risk 4.9%). 61
These studies of combination antiplatelet therapy in patients with coronary artery disease created the expectation that combined aspirin plus clopidogrel therapy would also be effective in stroke patients. Stroke patients, however, appear to differ from acute coronary syndrome patients in their response to antiplatelet agents. In comparison with clopidogrel or ASA monotherapy, the dual antiplatelet regimen has not been shown to have any better efficacy in the prevention of recurrent stroke or TIA than single antiplatelet drug therapy.65,66 Furthermore, clopidogrel plus ASA increased bleeding risk in this patient population.65,66 Two trials have compared the effect of combination clopidogrel plus ASA with monotherapy for prevention of vascular events in stroke/TIA patients (Table 3). The MATCH trial
65
compared clopidogrel alone versus aspirin plus clopidogrel, whereas the CHARISMA trial compared aspirin alone versus aspirin plus clopidogrel.
66
In the MATCH trial, the combination clopidogrel + ASA did not demonstrate significantly greater efficacy in prevention of the primary composite end point (ischemic stroke, MI, vascular death, or rehospitalization secondary to ischemic event) than clopidogrel alone, but major bleeding was almost twice as common (2.6% versus 1.3%).
66
The CHARISMA trial compared clopidogrel (75 mg/day) plus ASA (75-162 mg/day) with ASA alone in 15,603 patients with cardiovascular disease or multiple risk factors, including about 3,000 without an index vascular event. Overall, clopidogrel plus ASA was not significantly more effective than ASA in reducing incidence of the primary composite endpoint of ischemic stroke, MI, or cardiovascular death. In a prespecified subgroup analysis of 12,153 patients with documented coronary disease, PAD, or ischemic stroke/TIA within the previous 5 years, the combination was slightly more effective than ASA alone in reducing the primary endpoint (6.9% vs. 7.9%, RR 0.88,
ASA + extended-release dipyridamole
Dipyridamole (DP), a phosphodiesterase inhibitor, has both antiplatelet and endothelial effects, for a complex mechanism of action. Whereas ASA inhibits thromboxane-A2 formation, DP raises intracellular levels of cyclic adenosine monophosphate and cyclic guanosine monophosphate (cAMP and cGMP), producing a weak antiplatelet effect, but the drug also increases cGMP, augmenting downstream signaling pathways of nitric oxide. This may, in effect, produce effects on the vascular endothelium, including vasodilation. 5
Extended-release dipyridamole (ER-DP) has advantages over immediate release dipyridamole. Immediate release DP has a half-life of 40 minutes, which would result in rapidly declining plasma concentrations. The ASA plus ER-DP capsule also contains tartaric acid, which results in better gastrointestinal absorption. 5
The ESPS-2 trial showed that ASA + ER-DP was significantly more effective than ASA alone in secondary stroke prevention, with similarly low risk of severe bleeding.
5
The ESPS-2 randomized 6602 patients with recent ischemic stroke or TIA to ASA 25 mg twice daily, ER-DP 200 mg twice daily, ASA + ER-DP, or placebo, and followed them for 2 years. Primary endpoints were stroke, death, and stroke or death together (combined endpoint). Compared with placebo, risk of stroke was reduced 18% with ASA monotherapy (
A recent open-label study confirmed the findings of ESPS-2. The ESPRIT trial randomized 2739 patients with recent TIA or minor ischemic stroke to ASA or ASA + ER-DP (separately or as fixed-dose combination). The ASA dosage, determined by the treating physician, varied from 30 to 325 mg (median 75 mg/d) daily, and the ER-DP dosage was 200 mg twice daily. The mean follow-up was 3.5 years. On intention-to-treat analysis, the incidence of composite primary outcome (nonfatal MI, nonfatal stroke, vascular death, or major bleeding complication) was significantly lower with ASA + ER-DP than with ASA alone (13% versus 16%, HR 0.80, 95% CI 0.66-0.98). There were 35 major bleeding complications with ASA + DP versus 53 with ASA alone (HR 0.67, 95% CI 0.44-1.02). More patients discontinued the combination therapy than ASA alone, mainly because of adverse effects, primarily headache. 70
A revised meta-analysis including these data and all 6 studies comparing ASA with ASA + ER-DP or ASA + immediate release DP demonstrated an overall risk ratio for composite stroke, MI, or vascular death of 0.82 (95% CI 0.74-ss0.91) with the combination versus ASA alone, an RRR of 18%. 70
The most recent, long awaiting head-to-head, antiplatelet stroke prevention trial was the PRoFESS study. 71 This randomized, double-blind study (n = 20,332) compared efficacy of ASA + ER-DP versus clopidogrel for prevention of recurrent stroke. Recurrent stroke rates were similar with ER-DP+ASA and clopidogrel therapy (9.0% vs. 8.8%; HR 1.01, 95% CI 0.92-1.11); no significant differences in the incidence of the composite event (stroke, MI or vascular death) were reported (13.1% vs. 13.1%; HR 0.99, 95% CI 0.92-1.07). Ischemic stroke occurred less often with ER-DP + ASA (7.7% vs. 7.9%), but hemorrhagic strokes occurred more often (0.8% vs. 0.4%). More major hemorrhages occurred in patients receiving ER-DP + ASA than in those receiving clopidogrel (4.1% vs. 3.6%, HR 1.15, 95% CI 1.00-1.32), but no significant difference was found in the benefit-to-risk ratio expressed as combined recurrent stroke and major hemorrhage (11.7% vs. 11.4%, HR 1.03, 95% CI 0.95-1.11). 71 Dropouts were more common with aspirin and ER-dipyridamole than with clopidogrel, mainly because of headache. In sum, the evidence does not support any major difference in efficacy between clopidogrel and aspirin plus ER-dipyridamole in patients with noncardioembolic strokes, and clopidogrel was better tolerated.
The current AHA/ASA guidelines recommend aspirin, aspirin plus ER dipyridamole, or clopidogrel in secondary stroke prevention of noncardioembolic strokes.10,24 Clopidogrel monotherapy is comparable in efficacy and safety to ASA in secondary stroke prevention and is recommended over ticlopidine, because of fewer gastrointestinal symptoms and hemorrhages. In addition, the combination of ASA + extended release dipyridamole is recommended instead of ASA alone; evidence from the ESPRIT trial and meta-analysis of previous data motivated the AHA/ASA, in the 2008 update to the Guidelines, 24 to upgrade this recommendation to Class I supported by grade B evidence. New guidelines are expected to be published in 2009, likely reflecting the PRoFESS trial, with its overall lack of support for any difference in efficacy between aspirin plus ER-dipyridamole versus clopidogrel. For the present, the available evidence on antiplatelet therapies are not yet sufficient to make evidence-based recommendations preferring one agent over another, and choices should be individualized for each patient, considering allergies and side effects, costs, comorbidities, and adherence.10,24
Conclusions
In ischemic stroke and TIA patients, prevention of recurrent cerebrovascular events is the primary treatment goal, though prevention of other long-term complications such as MI and cardiac death is also important. Most strokes could be prevented, if readily available lifestyle and risk factor modifications could be applied to all stroke patients, along with anticoagulation for patients with cardiac sources of embolus, carotid procedures for patients with significant internal carotid artery stenosis, and antiplatelet therapy. For patients with noncardioembolic ischemic strokes, FDA-approved antiplatelet agents are recommended and preferred over anticoagulants. ASA, clopidogrel, and ASA + ER-DP are recognized as accepted first-line options for secondary prevention of noncardioembolic ischemic stroke. For patients who cannot tolerate regimens containing ASA, clopidogrel is recommended as a reasonable alternative. Although dual antiplatelet therapy with ASA + clopidogrel has been shown to be beneficial for use in ACS, 61 it is not recommended for secondary prevention in stroke or TIA patients because of the increased risk of hemorrhage, without definite added benefit. Aspirin and extended release dipyridamole appeared to carry a greater benefit over aspirin alone in the ESPS2 and ESPRIT studies than did clopidogrel over aspirin in CAPRIE, leading to a recommendation of this agent in the AHA guidelines, 24 but the PRoFESS trial did not confirm a clear advantage of this agent over clopidogrel. 71
Disclosure
The authors report no conflicts of interest.
