Abstract
High bone turnover disorders are characterized by excessive osteoclastic activity resulting in loss of bone mass and quality, fractures, deformities, debilitating pain, loss of mobility and hypercalcemia. These complications have a major impact in quality of life, and control of osteoclast activity is critical to reduce or prevent these skeletal events. Over the last 2 decades bisphosphonates became an integral part of the management of these disorders. Bisphosphonates, analogues of pyrophosphate, bind to hydroxyapatite crystals and have a very high affinity for bone mineral. Their retention in bone and rapid clearance from the circulation make them safe and effective agents for inhibition of osteoclast activity. Pamidronate is one such bisphosphonate that has been effective in treating Paget's disease, myeloma bone disease and osteolytic bone metastases from breast cancer. This review will focus on the effects of pamidronate in these disorders.
Keywords
Introduction
The first bisphosphonate synthesized in the end of the 19th century was used in the industry as an antiscaling agent to prevent calcium deposition. 1 By the mid 20th century, studies on bisphosphonates revealed their ability to bind avidly to bone and showed that their major effect in bone metabolism was inhibition of osteoclast activity.2–4 For this reason, bisphosphonates have been used successfully to treat conditions in which bone resorption is a major component of the disease process such as Paget's disease of bone, osteoporosis, hypercalcemia of malignancy and metastatic bone disease.3,4
Bisphosphonates are composed of two phosphate groups linked by a central carbon atom and two covalently bonded side chains referred to as R1 and R2 (Fig. 1). There are two classes of bisphosphonates: the non-nitrogen compounds that include clodronate and etidronate and the nitrogen-containing bisphosphonates that include alendronate, risedronate, ibandronate, zoledronic acid and pamidronate (Fig. 1).2–4
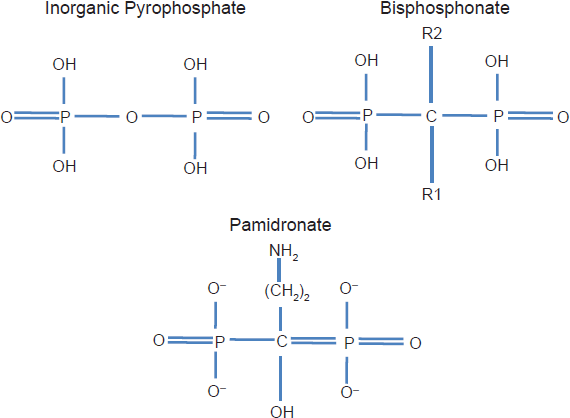
The structure of pyrophosphate, geminal bisphosphonate and of pamidronate.
The nitrogen-containing bisphosphonates have a stronger affinity for bone and greater potency; they inhibit the enzyme farnesyl diphosphate synthase of the mevalonate pathway resulting in inhibition of prenylation of small GTPases that are critical for osteoclast activity. Direct effects of bisphosphonates on osteoclasts include an impairment of the ruffled border, altered cytoskeleton, reduction in acid production and enzyme activity, effects that result in inhibition of osteoclast function.4,5
For the past 2 decades bisphosphonates have been the mainstay therapy for bone disorders associated with increased bone resorption. In 1992, the only available bisphosphonate in the United States was etidronate. However, the development of second and third generation bisphosphonates with greater potency and fewer side effects rapidly took place. Pamidronate is one such bisphosphonate that has been effective in treating Paget's disease, myeloma bone disease and osteolytic bone metastases from breast cancer. This review will focus on the effects of pamidronate in these disorders.
Osteolytic Bone Metastases in Breast Cancer
Bone metastases are common in patients with advanced breast, prostate, and lung cancers, multiple myeloma and to a lesser extent other solid tumors. The skeletal complications result in major impact on quality of life.
Early detection of localized breast cancer is associated with a 5-yr survival rate of 98%. However, the survival rate drops to approximately 30% for those patients diagnosed with distant metastases. Bone metastases occur in approximately 70%-80% of these patients; most have osteolytic lesions characterized by increased osteoclastic activity. 6 Despite higher mortality risk, the continuous development of more efficacious anti-cancer drugs has resulted in better survival which requires supportive therapies that are not only effective but also safe for prolonged use.
Pathogenesis
The skeleton is a preferred site for breast cancer metastasis. After detaching from the primary tumor, breast cancer cells are attracted to bone sites through interactions between tumor cells and bone cells.7–9 This process is facilitated by several factors that includes vascular endothelial growth factor (VEGF) and connective tissue growth factor (CTGF) that promotes angiogenesis, chemokine receptor 4 (CXCR4) promoting migration and chemotaxis, and matrix metalloproteinase (MMP) promoting detachment. After the colonization of the bone by the tumor cells, crosstalk between these cells and the bone microenvironment takes place resulting in a vicious cycle that promotes bone destruction and tumor growth (Fig. 2). 9 Bone destruction or osteolysis is stimulated by the release of factors produced by the tumor cell including parathyroid hormone-related protein (PTH-rP), transforming growth factor-beta (TGF-β) and interleukin-11 (IL-11).10–12 These factors induce osteoclast activation and bone resorption which leads to the release of growth factors such as TGF-β, insulin-like growth factor-1 (IGF-1), insulin-like growth factor 2 (IGF-2), fibroblast growth factor (FGF), bone morphogenetic proteins and platelet-derived growth factor (PDGF); these factors stimulate not only bone cell but also tumor cell proliferation (Fig. 2).7,8,13 The net effect is increased bone destruction and a continuous stimulus for tumor growth that results in pain, fractures, spinal cord compression and hypercalcemia.
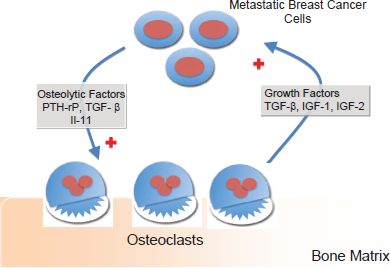
The vicious cycle of breast cancer metastases. The crosstalk between breast cancer cells and the bone microenvironment promotes release of osteolytic factors such as PTH-rP, TGF-β and 1 that results in osteolysis. Upon osteolysis, growth factors are released from bone promoting further tumor growth.
Treatment
The treatment of bone metastases involves a multidisciplinary approach including not only the systemic therapy for tumor control but also bisphosphonate treatment to reduce excessive bone resorption as well as local treatments needed for pain control and/or maintenance of bone integrity such as radiotherapy, surgery and radiofrequency ablation.
Over the last 2 decades, bisphosphonates became an integral part of the management of bone metastases. The initial studies involving clodronate suggested that the use of a bisphosphonate in association with anticancer therapy reduced the incidence of skeletal complications. Subsequently, trials with intravenous pamidronate in breast cancer patients clearly demonstrated the efficacy of this drug in reducing the so-called “skeletal-related events”. Skeletal-related events (SRE) are used as end points in clinical trials investigating the benefit of bisphosphonates; these events include pathologic fracture, spinal cord compression, radiation or surgery to bone and hypercalcemia of malignancy.
Pamidronate was the first bisphosphonate to be approved for the treatment of bone metastases. Table 1 summarizes the pivotal studies that have lead to its approval and clinical use.14–17
In 1996, Hortobagyi and colleagues reported the results of a randomized, placebo-controlled study of 12 months of treatment with pamidronate in breast cancer patients with osteolytic bone metastases treated with systemic chemotherapy. The addition of pamidronate (monthly infusions of 90 mg) resulted in significantly less SREs and in a significant delay to the development of the first skeletal complication. 14 Subsequently, Theriault et al reported that pamidronate was also effective in reducing SREs in women with breast cancer metastatic to bone treated with hormonal therapy. 15 Follow-up results from these 2 prospective, multicenter, placebo controlled trials confirmed the efficacy of pamidronate in reducing skeletal complications in women with breast cancer and bone metastases. In these studies, the most common complications were pathologic fractures and the need for radiation to bone.16,17 The number of skeletal complications over time was reduced by 35% in the pamidronate group, and 20% fewer pamidronate-treated patients experienced any of the skeletal complications. 18 In addition, the time to a first SRE was significantly longer in the pamidronate-treated group compared to the placebo-treated group (Table 1). Bone resorption markers remained significantly decreased from baseline during the 2 years of treatment demonstrating the long-term inhibition of osteoclastic activity. 19 Pamidronate became then a standard treatment for breast cancer induced bone metastases.
Subsequent clinical trials with other nitrogen-containing bisphosphonate such as zoledronic acid demonstrated its efficacy in the management of metastatic breast cancer and myeloma bone disease, and extended the benefit of bisphosphonate therapy to other tumor types and to patients with osteoblastic metastases.20–22 Other approved bisphosphonates for the treatment of breast cancer bone metastases include clodronate and ibandronate. 23 The focus of this review is to discuss the effects of pamidronate in the management of these bone disorders; discussion about other bisphosphonates can be found in excellent reviews.5,23,31
Most of the bisphosphonate clinical trials showed clinical efficacy and safety up to 2 years of treatment. However, because of their good safety profile and apparent clinical benefit, patients with breast cancer and lytic bone disease have continued treatment indefinitely.18,19,24–26 The knowledge that treatment with bisphosphonates has the potential to cause osteonecrosis of the jaw, a complication that has been associated with prolonged, greater accumulative doses of bisphosphonate and with the potency of the bisphosphonate has raised questions regarding the most appropriate dose and duration of bisphosphonate therapy.27–31 The retrospective review of 3994 patients treated with intravenous bisphosphonates for osteolytic bone disease at The University of Texas MD Anderson Cancer Center revealed that development of osteonecrosis of the jaw was associated with longer median duration of bone metastases which relates to longer bisphosphonate treatment duration, and greater cumulative doses of pamidronate and/or zoledronic acid. The multivariate Cox proportional hazards regression analysis of the breast cancer cohort identified treatment with zoledronic acid [hazards ratio (HR) 15.01 (95% CI: 2.41-93.48),P = 0.0037], treatment with pamidronate followed by zoledronic acid [HR 4.00 (95% CI: 0.86-18.70),P = 0.078] and dental extractions [HR 53.19 (95% CI: 18.20-155.46),P < 0.0001] as significant risks for osteonecrosis of the jaw. 30 Similar findings were observed in other studies.28,32,33 The knowledge that long-term use of bisphosphonates is not free of complications has raised several questions–-how much suppression of bone resorption is necessary to control osteolytic bone metastases? For how long should these patients be treated with bisphosphonates? How frequent should bisphosphonates be administered? Should we consider effective but less potent bisphosphonates for patients with more indolent disease known to require treatment for several years? A few ongoing trials have been designed to answer some of these questions; hopefully they will provide enough information to guide us towards the best way to treat these patients.
Myeloma Bone Disease (MBD)
Multiple myeloma (MM) is a clonal disease with the highest incidence of skeleton involvement among malignancies. Up to 90% of the patients will develop bone lesions during the course of disease and 60% of patients will present with bone pain at diagnosis. Progressive bone destruction is the most severe cause of morbidity in MM. 34 Myeloma bone disease can be catastrophic resulting in complications such as bone pain, pathological fractures requiring surgery and/or radiation to bones, spinal cord compression and hypercalcemia; a great impact in quality of life. Saad et al 35 retrospectively analyzed the effect of pathologic fractures on survival of patients with malignant disease. Patients with MM have the highest fracture incidence (43%), compared with patients with breast, lung and prostate cancer. These are not only associated with pain and reduced mobility but have also been associated with increased risk of death.
Pathogenesis
The pathogenesis of myeloma bone disease (MBD) is not fully understood; hyperactivity of osteoclasts is the predominant event. The basic principle is uncoupling of normal bone remodeling with enhanced bone resorption and decreased bone formation. Myeloma patients treated with glucocorticoid-containing regimens lose bone more rapidly than age-matched controls and greater than 30% of these patients develop fractures or require radiation therapy. 36 These skeletal complications are thought to be secondary to the highdose glucocorticoid therapy combined to the MM itself.
A major histological finding in MBD is osteoclast accumulation with enhanced bone resorption adjacent to the myeloma cells; these findings are not observed in areas not invaded by myeloma. In the invaded areas, no major histomorphometric difference was found between patients receiving chemotherapy and untreated patients, demonstrating that if usual chemotherapies reduce the tumor mass, they do not improve histological bone lesions in areas still invaded by plasma cells. 37 These findings have led to the hypothesis that local cytokines produced or induced by myeloma cells are responsible for the increased bone resorptive activity in MM. The interaction between MM cells and the bone microenvironment 38 results in the release of several cytokines that are critical for osteoclast recruitment and activation. These cytokines include: receptor activator of nuclear factor-kB ligand (RANKL), osteoprotegerin (OPG), the chemokines macrophage inflammatory protein (MIP) 1α, (MIP)1β, IL-6, IL-3 and stromal derived factor-1α(SDF-1α).
The RANK/RANKL signaling pathway, composed of RANK, RANKL and OPG, is a critical component of the bone remodeling process. RANK is a transmembrane signaling receptor, which is a member of the tumor necrosis receptor superfamily. 39 RANKL is expressed as a membrane-bound protein on marrow stromal cells and osteoblasts and secreted by the activated lymphocytes. Its expression is induced by cytokines that stimulate bone resorption. 40 RANKL induces differentiation, formation and survival of preosteoclasts 41 and it has a direct stimulatory effect on mature osteoclasts. OPG is a decoy receptor antagonist for RANKL 42 secreted by osteoblastic and stromal cells. A balance between RANKL/OPG is essential for normal bone turnover. Studies in bone marrow biopsies of MM patients demonstrate that expressions of RANKL and OPG are upregulated and downregulated, respectively. 43 In another study, circulating levels of OPG and RANKL correlated both with clinical activity and severity of MBD. 44
MIP-1α and MIP-1 β are also important for MBD. MIP-1α is a chemokine produced by MM cells in 70% of MM patients and is a potent inducer of osteoclast formation. MIP-1α can act independently of RANKL pontentiating the effects of both RANKL and IL-6 in osteoclast formation. 45 A recent study of gene expression profiling in MM showed that MIP-1α is the gene most highly correlated with bone destruction. 46 Other cytokines also involved in increased osteoclast function in MM include IL-3 and IL-6.47,48
The importance of osteoblast function in the pathogenesis of MBD has been recently demonstrated. In addition to increased osteoclast activity, osteoblast activity is suppressed in MM; the uncoupling between bone resorption and bone formation results in a greater degree of bone loss and destruction. 49 This may explain the fact that MM patients have low levels of bone formation markers such as alkaline phosphatase and osteocalcin. 50 Wingless-type (wnt) pathway plays an important role in bone formation as it promotes proliferation and survival of osteoblasts. Wnts are soluble glycoproteins that engage receptor complexes composed of low density lipoprotein receptor-related Lrp5/6 and Frizzled proteins. A subgroup of Wnts induces a cascade of intracellular events that stabilize β-catenin, facilitating its transport to nuclei where it binds Lef1/Tcf transcription factors and alters gene expression to promote osteoblast expansion and function. 51 Wnt antagonists, Dickkopfs (DKK) and secreted frizzled-related proteins (sFRP), impair osteoblast function and inhibit bone formation. Tian et al 52 demonstrated that elevated DKK1 levels in bone marrow and peripheral blood from patients with MM correlated with the gene-expression patterns of DKK1 and were associated with the presence of focal bone lesions. Moreover, bone marrow serum containing an elevated level of DKK1 inhibited the differentiation of osteoblast precursor cells in vitro. In a recent study the authors demonstrated that serum DKK-1 levels decrease in MM patients responding to treatment, irrespective of the regimen chosen. These findings suggest that myeloma cells are the main source of circulating DKK-1 protein. 53
Treatment of myeloma bone disease
Bisphosphonates are the gold standard treatment of MBD. The evidence supporting the use of bisphosphonates to prevent MBD comes from 4 randomized phase 3 clinical trials. Two of these trials investigated the use of clodronate; these will not be discussed in this review. The first trial that evaluated the use of intravenous pamidronate in patients with mm54 was a randomized, placebo controlled trial involving 392 patients with stage III MM and at least 1 lytic lesion. Patients were randomized to placebo or pamidronate, 90 mg, given as a 4-hour infusion every 4 weeks for 21 cycles. The primary end points were the reduction of SREs and evaluation of safety and survival. A total of 377 patients were assessable; at 9 months, the number of SREs per year was 2.1 in the placebo group and 1.1 in the pamidronate group (P = 0.0006), findings that persisted at 21 months. The median time to the first SRE was 10 months in the placebo group and 21 months in the pamidronate group (P < 0.001). Another benefit of pamidronate treatment was a significant decrease in pain and reduced use of analgesics. This prospective study subsequently led to the Food and Drug Administration approval of the use of this drug for MM.
Two other randomized trials evaluated the efficacy of the more potent bisphosphonate, zoledronic acid, compared to pamidronate. They showed that zoledronic acid had similar efficacy and safety compared to 90 mg of pamidronate. The first, a randomized phase 2 study, investigated the dose of zoledronic acid required to reduce the need for radiation to less than 30% of treated patients and compared it to pamidronate. 55 Radiation treatment was required in a similar proportion of patients receiving pamidronate and zoledronic acid at 2 mg or 4 mg (18% to 21%). Subsequently, a larger phase 3 randomized trial compared 4- or 8-mg doses of zoledronic acid to 90 mg of pamidronate every 3 to 4 weeks in patients with MM or breast cancer who had lytic disease. 56 The higher dose of zoledronic acid was associated with reduced renal function which led to the reduction of the dose to 4 mg. The study enrolled 1648 patients in an intent-to-treat analysis; 510 had MM, and the remainder had metastatic breast cancer. The portion of patients with any SRE after 13 months did not differ among the 3 treatments and did not differ between patients with breast cancer and those with MM.
Upon the results of these trials, an American Society of Clinical Oncology (ASCO) expert panel advocated the use of either pamidronate or zoledronic acid monthly for patients with MM and lytic bone disease. 57 It was recommended the use of pamidronate at 90 mg delivered intravenously for at least 2 hours or zoledronic acid at 4 mg intravenously for 15 minutes every 3 to 4 weeks for patients with MM and lytic bone disease. 58 These guidelines were followed by several other expert meetings and consensus summarized in Table 2.57–62
Placebo-controlled trials with pamidronate in breast cancer bone metastases.
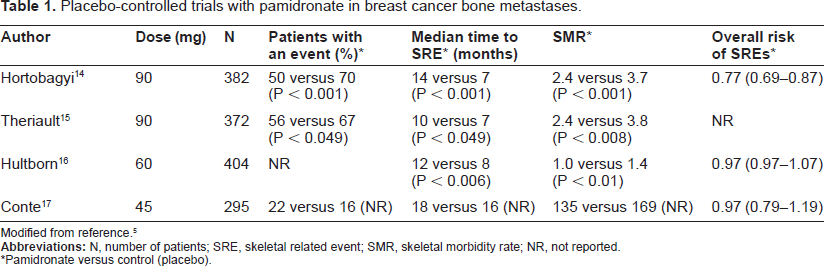
Modified from reference. 5
*Pamidronate versus control (placebo).
Summary of recently published guidelines in multiple myeloma.
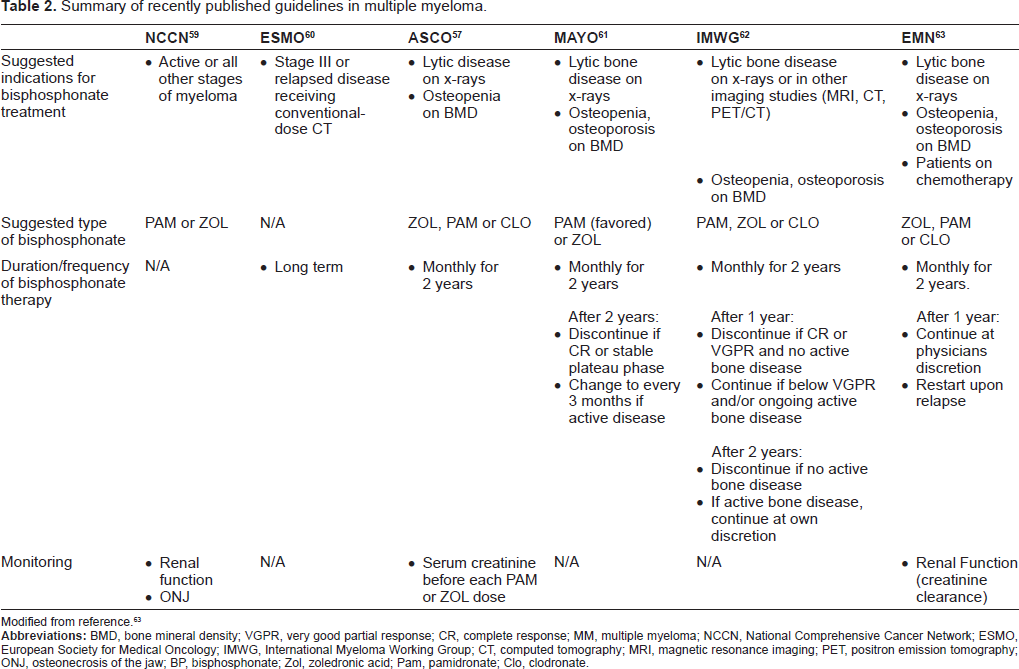
Modified from reference. 63
Paget's Disease of Bone
Paget's disease of bone is a disorder of excessive and abnormal bone remodeling involving 1 or more areas of the skeleton. In addition to the significant increase in bone resorption there is also excessive bone formation; however, the bone formed is irregular and chaotic with formation of both woven and lamellar bone. The primary and driving defect that causes Paget's disease rests in the osteoclast, and therefore the inhibition of bone resorption by bisphosphonates generally leads to normalization of bone turnover markers and to a marked improvement in clinical manifestations. 64
The majority of patients with Paget's disease of bone are asymptomatic and the disease is incidentally identified by x-rays performed for another reason, or by the identification of an abnormal alkaline phosphatase level. In symptomatic patients, the most common complain is bone pain. The pain is usually local over the bone lesion and may be due to the expansion of the lesion with pressure to the periosteum or due to the increased vascularity of the area. In a few cases, the pain is related to nerve root compression or nerve entrapment due to the expanding lesion. The disordered bone remodeling can result in new bone with altered architecture; this is the cause of the bone deformities observed in patients with Paget's disease. Common deformities include an increase in skull size, lateral bowing of long bones and dorsal kyphosis. Abnormal bone formation and quality and increased vascularity can result in fracture, nerve entrapment, deafness, high-output cardiac failure and hypercalcemia. Very rarely, sarcomatous transformation of the pagetic lesion occurs. 64
Pathogenesis
The pathogenesis of Paget's disease has not been clearly defined. It is known that genetic predisposition is important; in some families Paget's is transmitted as an autosomal dominant pattern. Mutations in two genes have been reported, in the RANK and SQSTM1 genes. Mutations of the SQSTM1 gene are more frequent and have been found in approximately 30% of familial cases.65,66 Chronic paramyxovirus infection has also been implicated as having a pathogenetic role in Paget's disease. Ultrastructural studies of pagetic osteoclasts have shown nuclear and cytoplasmic inclusions similar to nucleocapsids from paramyxoviruses.67,68 While several studies have shown the presence of measles or canine distemper virus (CDV) in pagetic osteoclasts others were unable to confirm this presence, therefore this possibility remains controversial. 64
Treatment
The goal of treatment in Paget's disease is to inhibit osteoclastic activity to reduce pain, improve mobility and to prevent future complications. Bone resorption inhibitors that have been used for Paget's disease include calcitonin, plicamycin, gallium nitrate and bisphosphonates. The higher potency in suppressing increased bone resorption has made the bisphosphonates the drug of choice for this disorder. 69
Several bisphosphonates have been used in the treatment of Paget's disease including etidronate, tiludronate, alendronate, risedronate, zoledronic acid and pamidronate. 70 Pamidronate is usually given at a dose of 60-90 mg per infusion. Patients with mild disease and slight elevation of serum alkaline phosphatase and other bone turnover markers tend to require only one infusion; patients with more severe disease may require several infusions (generally 3 to 4) that can be given once a week. Therefore, the dose of pamidronate should be individualized according to rate of bone turnover and clinical manifestations. 64 Normalization of alkaline phosphatase is observed in approximately 50% of patients and the nadir of the alkaline phosphatase level occurs 1-3 months after the course of treatment.64,71 Merlotti and colleagues compared different intravenous bisphosphonates in Paget's disease and reported remissions rate above 90% with zoledronic acid and neridronate compared to 45% in patients receiving pamidronate. Non-responders to pamidronate were crossed-over to zoledronic acid or to neridronate and more than 90% of these patients then achieved a therapeutic response. 72 Therefore, patients with more severe disease may benefit from more potent bisphosphonates such as zoledronic acid. Patients initially treated with pamidronate that do not achieve biochemical response should be switched to zoledronic acid.
All patients treated with bisphosphonates should receive calcium and vitamin D replacement to avoid hypocalcemia and secondary hyperparathyroidism.64,69 Side effects are more common with the oral bisphosphonates that are associated with upper gastrointestinal symptoms. A few cases of osteo necrosis of the jaw have been reported in patients with Paget's disease treated with intravenous bisphosphonates, most of these patients were treated for prolonged period of time with zoledronic acid or pamidronate. 73 Despite being rare, it is important to inform any patient considered for bisphosphonate treatment about the risks of osteonecrosis of the jaw and to ensure good oral health before starting treatment.
Conclusion
Osteolytic bone metastases, myeloma bone disease and Paget's disease of bone are disorders associated with excessive osteoclastic activity. When not controlled they cause significant complications such as fractures, bone pain, bone deformities, cord compression and hypercalcemia. Since its introduction, bisphosphonates such as pamidronate have prevented and reduced these complications resulting in a significant impact in the outcome and quality of life of these patients.
Disclosure
The authors report no conflicts of interest.
