Abstract
Ciclesonide is a novel inhaled corticosteroid (ICSs) approved in most countries for the management of persistent asthma. Although inhaled corticosteroids are first-line therapy in the treatment of asthma, long term use and high-doses of these products may result in significant side effects. When developing a new ICSs, the goal is to identify a drug with comparable (or superior) efficacy to active comparators, and an improved safety profile. Ciclesonide is a prodrug which is administered through a hydrofluoroalkane-propellant metered dose inhaler (HFA-MDI). Once it reaches the lungs, the parent compound is metabolized by esterases to desisobutyryl ciclesonide (des-CIC), an active metabolite with a 100-fold greater affinity for the glucocorticoid receptor. Ciclesonide has a unique pharmacokinetic-pharmacodynamic profile which confers an improved therapeutic ratio. Several clinical trials have shown that its efficacy is superior to placebo and similar to several active comparators. However, its high pulmonary deposition and on-site activation minimizes the risk for local side effects. Also, its low oral bioavailability, high hepatic clearance, and extensive plasma protein binding, among other factors, decrease the risk for systemic side effects. Doses of ciclesonide as high as 1280 μg/day (ex-actuator) result in minimal hypothalamic-pituitary-adrenal (HPA) axis suppression, a measure commonly used to assess systemic bioavailability for an ICSs. This review will provide a summary of ciclesonide's role in the management of asthma, including a discussion of relevant clinical trials designed to evaluate its efficacy and safety.
Introduction
Asthma is a chronic respiratory condition which is characterized by bronchial hyperresponsiveness, airway inflammation, and airflow obstruction. 1 These processes result in recurring symptoms such as wheezing, coughing, chest tightness, and shortness of breath upon exertion. Based upon worldwide numbers, 300 million people suffer from asthma and it accounts for 1 out of every 250 deaths.2,3 The current treatment of asthma divides available medications into two classes, long-term controller medications and quick-relief medications. 1 Inhaled corticosteroids (ICSs) are long-term controller medications that improve the symptoms of asthma by reducing the inflammatory component of the disease. These drugs are widely accepted as firstline therapy in the management of asthma and have been shown to reduce hospitalizations due to asthma, 4 airway hyperresponsiveness, 5 and improve several measures of lung function.1,5,6 Although ICSs are the most effective anti-inflammatory drugs available for the treatment of asthma, they are associated with local (i.e. oropharyngeal) and systemic side effects. Thus, the development of newer drugs has focused on identifying molecules with similar (or superior) efficacy and an improved safety profile.
There are several ICSs currently available for use in the management of asthma. These include fluticasone propionate, budesonide, mometasone furoate, triamcinolone acetonide, beclomethasone dipropionate, flunisolide, and ciclesonide. Ciclesonide is a novel synthetic corticosteroid with distinct pharmacokinetic (PK) and pharmacodynamic (PD) properties, which is approved in many countries for the treatment of both asthma and allergic rhinitis. Ciclesonide is formulated as a solution and delivered via a hydrofluoroalkane (HFA)-propelled metered-dose inhaler (MDI), achieving a high pulmonary deposition. 7 Once it reaches the lungs, ciclesonide, a pro-drug, is converted to the active metabolite, des-isobutyryl ciclesonide (des-CIC). 7 Des-CIC has a 100-fold greater affinity for the glucocorticoid receptor (GR) than the parent compound and once it is bound to the receptor it can have a broad range of anti-inflammatory effects. 7 The following discussion will provide a concise review of the use of ciclesonide in the management of asthma, focusing not only on the efficacy and safety of the drug, but also on its unique PK-PD profile.
Mechanism of Action
ICSs improve asthma symptoms by increasing the expression of anti-inflammatory proteins and decreasing the expression pro-inflammatory proteins. 8 The latter may include inflammatory mediators involved in cell recruitment as well as multiple types of inflammatory cells (e.g. eosinophils). 8 In order to exert their beneficial effects, corticosteroids must first diffuse across the cell membrane, and bind to GRs located in the cytoplasm. These receptors are expressed in large quantities in airway epithelial cells and bronchial vascular cells. 9 Following receptor binding, there is a translocation into the nucleus and receptor dimerization. Inside the nucleus these receptors can bind to positive or negative glucocorticoid response elements (GRE), located in the promoter region of corticosteroid-responsive genes.8,10 More commonly though, GRs may indirectly affect transcription through an interaction with activated transcription factors or coactivator (and corepressor) proteins. 8 For example, GRs may bind to and inhibit co-activators with intrinsic histone acetyltransferase activity, thus decreasing the unwinding of DNA.8,10 Although binding to GRs in cells located in the lungs results in an improvement in asthma symptoms, binding to the same receptor outside the lungs results in serious side effects. Many of the systemic side effects associated with corticosteroids involve binding of the GRs to negative GREs. 11
Ciclesonide's anti-inflammatory effects have been described in several publications. However, it should be noted that these anti-inflammatory effects are not specific to ciclesonide, and all ICSs are believed to bind to the same GRs. The beneficial effects of ciclesonide, like with all other corticosteroids, is likely a result of a variety of different effects. Indeed, several studies have demonstrated the broad range of effects which may be seen following ciclesonide use. In a cell culture of human bronchial epithelial cells, ciclesonide inhibited the expression of intercellular adhesion molecule (ICAM)-1, granulocyte-macrophage colony stimulating factor (GM-CSF), and interleukin (IL)-8, when the cells were stimulated with IL-4 and tumor necrosis factor (TNF)-α. 12 Another study demonstrated that ciclesonide inhibits the growth factor-induced proliferation of fibroblasts, a key cell type involved in the airway remodeling process seen in asthmatic patients. 13 Furthermore, in a clinical study using asthmatic patients, low-dose ciclesonide was shown to decrease T-cell migration and cytokine production after allergen challenge. 14 Therefore, ciclesonide's beneficial effects are partly based on a change in expression of inflammatory cells as well as the mediators which recruit them.
Pharmacokinetic/Pharmacodynamic Properties
The fate of inhaled corticosteroids
Administration of a corticosteroid via inhalation allows for the local delivery of drug to the lungs, thus increasing the concentration at the site of action, and minimizing the quantity that reaches the systemic circulation. Following inhalation, about 10%-60% of the administered dose reaches the lungs, and the remaining amount (40%-90%) is deposited in the oropharynx (Fig. 1). 15 The fraction of the drug that is deposited in the oropharynx is then swallowed and absorbed from the gastrointestinal (GI) tract. Following GI absorption, the drug is exposed to pre-systemic clearance in the liver, where a small percentage may escape metabolism and then reach the systemic circulation. In this case, the amount of drug which reaches the systemic circulation is dependent on the extent of drug absorption as well as the efficiency of the metabolic processes in the gut and liver. 16 In contrast, for the drug which reaches the lungs, a large percentage of it will be directly absorbed into the systemic circulation (bypassing pre-systemic clearance). However, one cannot assume that the amount absorbed from the lungs is equal to the amount deposited (pulmonary deposition) because there are mucociliary processes which can remove the drug before it is absorbed into the systemic circulation.
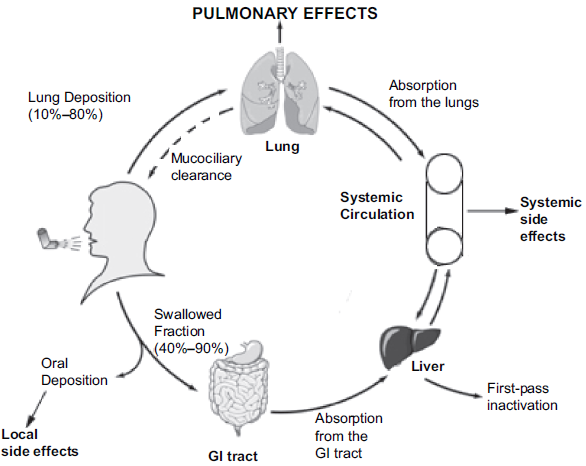
The fate of inhaled corticosteroids. (Figure adapted from 7 )
Bioavailability
The pulmonary bioavailability of a drug refers to the rate and extent to which the drug reaches the bloodstream from the lungs (for drug which was deposited in the lungs).15,16 On the other hand, the oral bioavailability (Foral) is the rate and extent to which it reaches the bloodstream after pre-systemic clearance in the gut and liver has occurred.15,16 Since drug absorbed from the GI tract does not add to the beneficial effects, ideally the oral bioavailability should be as low as possible. 16 However, as the pulmonary targeting of a drug is increased, the significance of having a very low oral bioavailability decreases (assuming Foral < 25%). 17 The absolute systemic bioavailability is a sum of the drug absorbed from the lungs and the GI tract.17,18 Thus, a higher pulmonary bioavailability will result in greater systemic concentrations, and possibly an increased risk for systemic side effects.
The oral bioavailability for ciclesonide and des-CIC is < 1%,16,19 which is comparable to that seen with fluticasone propionate (<1%) and mometasone furoate (<1%).16,20,21 On the other hand, the oral bioavailability of beclomethasone monopropionate (26%), 22 triamcinolone acetonide (23%), 23 budesonide (11%), 24 and flunisolide (7%), 25 are much higher. Ciclesonide's oral bioavailability is an important property, since it minimizes the amount of active drug which reaches the systemic circulation.
Pulmonary deposition
Pulmonary deposition refers to the quantity of drug which reaches the lungs. High pulmonary deposition not only guarantees that more drug reaches the site of action, but also it decreases the amount which is absorbed from the GI tract. 7 As stated above, one cannot assume that the pulmonary deposition and bioavailability are equivalent because mucociliary clearance may result in removal of drug from the lungs. The extent to which a drug reaches the lungs is largely dictated by the particle size, the drug's physical properties, application device, and the inhaler technique.15,16 Thus, the overall goal is to optimize these factors in an effort to increase pulmonary deposition of the drug.
The lung deposition of ciclesonide (after a single dose) administered via HFA-MDI was found to be approximately 52%, with a large fraction of the deposition occurring in the small airways and alveoli.26,27 This high deposition is explained by the small particle size delivered after inhalation of ciclesonide. The majority of the particles inhaled in the range of 1.1 to 2.1 urn. 7 This small particle size allows greater deposition in the small airways, which have an average diameter of 2 um.7,16 A major reason for the small particle size seen with ciclesonide is that it is formulated as a solution HFA-MDI. 7 When compared to solution formulated chlorofluorocarbon (CFC) MDIs, suspension MDIs, or dry powder inhalers (DPI), it has been shown that solution formulated HFA-MDIs deliver the largest amount of fine particles to the lungs.7,28,29 However, although the device and drug specific properties are important for improving lung deposition, an appropriate inhalation technique is a critical factor. 30 When the PK of des-CIC were compared after administration of a single dose of ciclesonide with or without the use of the AeroChamber Plus™ spacer, the systemic exposure was found to be similar. 31
Metabolism
A corticosteroid may be given as a pro-drug, which is activated in the target tissue, a strategy which may result in reduced local and systemic side effects. 16 Of the currently available ICSs, ciclesonide and beclomethasone dipropionate are inactive compounds, which are activated by esterases (in the lungs) to a metabolite with much greater affinity for the GR.32–34 Ciclesonide is metabolized to desisobutyryl-ciclesonide (des-CIC) in the lungs, followed by the formation of fatty acid conjugates. 32 The formation of these fatty acid conjugates allows for the drug to remain in the lungs for a longer period of time. Of the fatty acid conjugates which are formed, des-CIC oleate is the major metabolite. 32 Lipid conjugation will be discussed in greater detail later in this section.
Last, in the liver des-CIC is further metabolized by CYP isoenzymes (e.g. CYP3A4, CYP2D6, CYP2C8) to several polar, inactive metabolites. 32 When ciclesonide was co-administered with erythromycin, a CYP3A4 inhibitor, there were no changes in the PK of the parent drug or its active metabolite (des-CIC). 35 However, when it was given with ketoconazole (also a CYP3A4 inhibitor), there was a decrease in the elimination of des-CIC, while the conversion of ciclesonide to des-CIC remained unchanged. 36
Plasma protein binding
Corticosteroids can bind to plasma proteins (e.g. albumin, alpha-1-acid glycoprotein, transcortin) in the circulation, thus reducing the free, unbound drug concentration. Therefore, plasma protein binding would be a beneficial attribute of an ICSs since only the unbound drug can bind to GRs located outside of the lungs, and contribute to the systemic side effects. 7 In the case of ciclesonide, both the parent drug and des-CIC are highly protein bound (~99%). 37 Although all of the other ICSs have moderate to high protein binding, none are as highly bound as ciclesonide and its active metabolite (triamcinolone acetonide 71%, 23 flunisolide 80%, 33 beclomethasone dipriopionate 87%, 22 budesonide 88%, 34 mometasone furoate 98%, 38 and fluticasone propionate 90%).34,39 Ciclesonide's (and des-CIC) high degree of protein binding may be at least partially responsible for the minimal effects that the drug has on the hypothalamic-pituatary-adrenal (HPA) axis.40,41 However, one cannot assume that an increase in plasma protein binding minimizes the risk for side effects, while having no effect on efficacy. A study done in rats found that although unbound blood concentrations were lower with des-CIC when compared to budesonide, the higher protein binding also may result in a decrease in receptor occupancy at the site of action (lungs). 42
Clearance and half-life
Clearance refers to the volume of fluid which is cleared of drug per unit time. For drugs which have linear PK, this value is constant. 16 In the case of drugs which are cleared by the liver, hepatic clearance cannot exceed liver blood flow (~90 L/h). For corticosteroids, ideally the hepatic clearance should approach liver blood flow in order to have a low systemic bioavailability as well as systemic concentrations. 16 Indeed, most ICSs are high extraction drugs with very high hepatic clearance values. The clearance of ciclesonide is approximately 228 L/h, which indicates that extrahepatic clearance mechanisms may be involved in its elimination.15,16 The clearance values for other corticosteroids varies between 37 L/h (triamcinolone acetonide) and 84 L/h (budesonide),16,24 except for 17-beclometasone monopropionate (active metabolite of beclomethasone dipropionate), which has a clearance of 120 L/h.16,43–45 The high clearance of ciclesonide is remarkable since the drug is highly bound to plasma proteins. However, the metabolism is very rapid, so that the high binding does not limit its high-extraction clearance.
The half-life of elimination is the time it takes for drug concentrations in plasma to decrease by one-half, and is dependent on a drug's clearance and volume of distribution. The latter term is a hypothetical volume which helps to describe the extent of distribution of a drug. Most corticosteroids have a high volume of distribution due to their high lipophilicity. 16 If clearance is held constant, the larger the volume of distribution of a drug, the longer its half-life. The mean elimination half-life of des-CIC is approximately 3.5 hours.19,46
Lipid conjugation
As discussed in the metabolism section, des-CIC forms fatty acid conjugates in pulmonary cells.32,34,47 Aside from des-CIC, only budesonide and triamcinolone acetonide have been shown to form lipid conjugates.48,49 Since the conjugation process is reversible, it allows for the slow release of drug over time, prolonging its lung residence time. 16 This may be beneficial since the longer the drug is at the site of action, the more time it has to bind to the GR. Also, since ICSs are rapidly absorbed from the lungs, the formation of lipid conjugates may decrease the maximal systemic exposure to the drug.7,34 However, one should point out that there is no unambiguous evidence that lipid conjugation in the lungs results in a longer duration of action or an improved safety profile. Also, there is no good quantitative data about the pharmacokinetics of these corticosteroid-esters in the lung.
Receptor binding
In order for a corticosteroid to exert its beneficial effects it must reach the site of action and then bind to its receptor with high affinity. GRs are expressed in most cell types, and binding to these receptors is responsible not only for the drug's beneficial effects, but also for their adverse effects. 34 A corticosteroid's binding affinity is referred to as the relative receptor affinity (RRA).50–52 RRA are values used to compare the receptor affinities between the available steroids, and is measure of potency, not efficacy. 51 Thus, a lower RRA may be overcome by increasing the dose of the corticosteroid. 51 Dexamethasone's RRA is designated as 100, which is used as a reference for comparison with other corticosteroids. Ciclesonide's has a RRA of 12, which signifies that the parent drug is practically inactive. 52 On the other hand, des-CIC has a RRA of 1200, a 100-fold greater affinity than the parent drug. 48 When ranking des-CIC's RRA relative to other corticosteroids, mometasone fuorate has the highest affinity (2,200), followed by fluticasone propionate (1,800), 17-beclomethasone monopropionate (1,345), des-CIC (1,200), budesonide (935), triamcinolone acetonide (233), flunisolide (180), and beclomethasone dipropionate (53).16,34,50,51
Efficacy Studies
When developing a new inhaled corticosteroid, ideally the drug should be superior to placebo, at least equally efficacious to active comparators, and have an improved safety profile. As discussed in the previous section, ciclesonide displays most of the PK and PD properties which would maximize its efficacy and reduce the risk for both local and systemic side effects. Several clinical trials have been conducted comparing the efficacy of ciclesonide to that of both placebo and active comparators. Although not comprehensive, relevant clinical trials published between 2006 to 2009 are discussed below. All doses stated in this section correspond to the ex-actuator dose, unless stated otherwise.
Comparison with placebo
When compared to placebo, ciclesonide was shown to improve several measures of pulmonary function, including peak expiratory flow (PEF), airway hyperresponsiveness to adenosine monophosphate (i.e. PC20FEV1), and exhaled nitric oxide (NO) levels in patients with mild-to-moderate asthma.53,54 The improvement in morning (and evening) PEF as well as a reduction in the use of rescue medication was evident with a dose as low as 100 μg/day. 53 The time course with which these improvements occur has also been evaluated in patients with mild asthma. 54 The beneficial effects of ciclesonide on airway hyperresponsiveness, assessed by measuring the provocative concentration of adenosine monophosphate causing a 20% reduction in FEV1 (PC20FEV1), were evident within 2.5 hours after administration of the first dose (320-640 μg). Also, a significant reduction in median exhaled NO levels were seen after 3 days of treatment. Neither of these effects was dose-dependent.
Ciclesonide has a high pulmonary deposition (52%), with a large portion of the deposition likely occurring in the small airways (airways with an internal diameter <2 mm). 55 One study sought to evaluate ciclesonide's effects on small airway function, though the comparison was made with placebo and not an active comparator. 55 A total of 16 subjects with mild-to-moderate asthma were randomized to 5-6 weeks of treatment with once-daily ciclesonide (320 μg) or placebo. Then several measures of small airway function were compared between the groups. The only significant improvements which were noted were in exhaled NO and computed tomography measurements of expiratory lung volume. Ciclesonide treated patients had median alveolar eNO values which were significantly lower than those in patients given placebo (8.5 vs. 16.5 ppb). Also, measurements taken by computed tomography scan following methacholine challenge were lower in the ciclesonide group. The results of this study do not allow one to make any claims regarding ciclesonide's effects on small airway inflammation because there was no active comparator. In order to reach such conclusions, one would need to compare the effects seen with ciclesonide to those of another active comparator with a larger particle size.
A key measure of efficacy for an ICSs is a reduction in asthma exacerbations and oral steroid use. After 12 weeks of treatment with ciclesonide (640 μg/day and 1,280 μg/day) or placebo, drug treatment resulted in a significant decrease in oral prednisone dose (47% for 640 μg/day, 63% for 1,280 μg/day, and 4% increase for placebo). 56 In addition, there was a significantly greater number of patients in the ciclesonide group who were able to discontinue the use of prednisone altogether (30% vs. 11%). Therefore, it is clear that ciclesonide is superior to placebo based on improvements in lung function, number of asthma exacerbations, and systemic steroid use.
Comparison with active comparators
Most published studies comparing ciclesonide to active comparators have focused on budesonide or fluticasone propionate. Fluticasone propionate is a widely used ICSs, which explains why a plethora of clinical trials have been performed to compare it with ciclesonide. When ciclesonide given at a dose of 80 or 160 μg/day was compared with fluticasone propionate (88 μg twice daily) in patients with persistent asthma, similar improvements were seen in pulmonary function tests and asthma symptoms after 12 weeks of treatment.57,58 When higher doses of both ciclesonide (320 μg once daily) and fluticasone propionate (200 μg twice daily) were compared in patients with moderate persistent asthma, again both groups had similar improvements in lung function. 59 However, in the intention-to-treat population, ciclesonide resulted in a significantly greater improvement in health related quality of life. Another study demonstrated non-inferiority of ciclesonide (320 μg twice daily) to fluticasone propionate (330 μg twice daily) in patients with moderate-to-severe asthma. 60 Similar results were obtained when ciclesonide (80 μg twice daily) and fluticasone propionate (88 μg twice daily) were compared in children with asthma. 61 Furthermore, it has been shown that patients well controlled on twice-daily fluticasone propionate (250 μg/dose), may be “stepped down” to once-daily ciclesonide (160 μg), and still maintain similar asthma control. 62 However, it should be mentioned that in some countries (e.g. United States), ciclesonide is currently only approved for twice-daily use.
In contrast, when ciclesonide (320 μg once daily) was compared to budesonide (400 μg once daily), a significantly greater increase in forced expiratory volume in 1 second (FEV1), peak expiratory flow (PEF), and forced vital capacity (FVC) was seen in the group receiving ciclesonide. 63 The difference between the two groups was even more apparent in patients with a history of smoking. Although a significant improvement in asthma symptom scores and decreased use of rescue medication was seen with both drugs, there were no statistically significant differences between the two groups. When the onset of treatment effect was compared, ciclesonide resulted in a significant improvement in morning PEF by day 2 of treatment (versus baseline). Budesonide's effects were not as rapid, with a significant effect seen by day 7 of treatment (versus basline).
On the other hand, several other studies have found ciclesonide and budesonide to be equally efficacious. In a previous study, ciclesonide (80 μg or 320 μg once daily) was found to be non-inferior to budesonide (200 μg twice daily) when evaluating changes in FEV1, FVC, and asthma symptom scores. 64 In children with asthma, ciclesonide (160 μg once daily) and budesonide (400 μg once daily) showed similar improvements in pulmonary function, asthma symptoms, and quality of life. 65 Again, similar results were seen when higher doses of each drug (ciclesonide 320 μg once daily and budesonide 800 μg once daily) were evaluated in adolescents. 66
Ciclesonide has also been compared with beclomethasone dipropionate in adults with moderate-to-severe asthma. 67 In this study there were three treatment groups, ciclesonide 320 μg once daily, ciclesonide 320 μg twice daily, and beclomethasone dipropionate 400 μg twice daily (ex-valve). 67 Ciclesonide 320 μg once daily was shown to be non-inferior to beclomethasone dipropionate 400 μg twice daily (ex-valve). However, a significantly greater increase in morning (and evening) PEF was seen with ciclesonide 320 μg twice daily compared to beclomethasone dipriopionate. The use of rescue medication also decreased significantly in the group receiving high-dose ciclesonide, but not with lower-dose ciclesonide or beclomethasone dipropionate.
Efficacy in exercise induced bronchospasm
The effect of ciclesonide on exercise induced bronchospasm (EIB) has also been investigated. One study sought to evaluate the relationship between sputum eosinophil levels and response to ciclesonide in asthmatic patients with EIB. 68 Twenty-six steroid-naïve patients were randomized to one of two groups, with each group comparing two dose levels. One group compared ciclesonide 40 vs. 160 μg given once daily, whereas the second group compared 80 vs. 320 μg once daily. Low-doses provided a significant improvement in EIB at week 1, but no further improvement was noted thereafter, regardless of baseline eosinophil count. The authors reported that sputum eosinophil percentage was significantly correlated with EIB severity and only high doses of ciclesonide resulted in significant decreases in this percentage. Also, patients with sputum eosinophilia (≥5%) benefited more from high-dose therapy when compared to patients with eosinophil counts <5%.
Safety
Commercially available ICSs differ in their propensity to cause local and systemic side effects. Thus, careful consideration of a drug's safety profile is warranted because significant adverse effects may worsen a patient's quality of life, discourage adherence to drug treatment, and diminish the beneficial effects of a drug.69,70 Side effects which may be seen with ICSs use can be divided into local (oropharyngeal) or systemic side effects. Although local side effects are usually not regarded as serious or life-threatening, they are clinically relevant because they may impact compliance to drug treatment. 69 Dysphonia (or hoarseness) and oropharyngeal candidiasis are examples of local side effects which may result from ICSs use, particularly in patients with poor oral hygiene. 69 These side effects may be minimized by rinsing the oral cavity with water following ICSs use and through the use of a spacer device.69,71 Also, as stated earlier, all ICSs do not cause local side effects to the same extent. The propensity of an ICSs to cause local side effects is likely multifactorial and dependent on factors such as the steroid and the inhaler device. 69
Related to the inhaler device, a high oropharyngeal deposition would increase the risk for experiencing local side effects. When the oropharyngeal deposition of ciclesonide (Total 800 μg, ex-valve) and fluticasone propionate (Total 1000 μg, ex-valve) were compared using oropharyngeal washes, 3-fold higher Cmax values were observed with fluticasone propionate (Fig. 2). 71 The calculated mean AUC0-60 min for ciclesonide and des-CIC were significantly lower than that of fluticasone propionate (ciclesonide 250.4 nmol*h/L, des-CIC 37.8 nmol*h/L, fluticasone propionate 636.2 nmol*h/L). Thus, the low oropharyngeal deposition seen with ciclesonide use along with minimal conversion to des-CIC, likely contributes to a low incidence of local side effects. Indeed, several clinical trials comparing active comparators have reported significantly fewer oropharyngeal side effects with ciclesonide treated patients compared with those treated with fluticasone propionate.58,60 However, several studies have also reported no differences between ciclesonide and active comparators.56,62,66 This conflicting information is likely a result of differences in the measures used to assess (or diagnose) local side effects as well as differences in the study design. 69
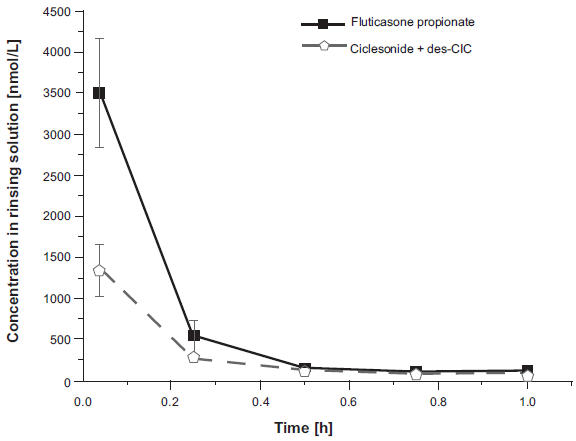
Concentration-time curve for fluticasone propionate and the sum of ciclesonide and desisobutyryl-ciclesonide. The samples were collected from 18 asthmatic patients following inhalation of ciclesonide 800 μg (ex-valve) and fluticasone propionate 1000 μg (ex-valve). (Reference 71; permission to reuse this figure was obtained).
Systemic side effects are dose-dependent and are directly related to unbound drug concentrations in the circulation (drug exposure).69,72 Therefore, their incidence increases with high doses and long-term use of an ICSs. 73 Systemic side effects include HPA-axis suppression, growth suppression, osteoporosis, cataracts, and skin thinning/bruising.72,73 HPA-axis suppression is used as a measure of systemic bioavailability of ICSs. This phenomena can occur because ICSs bind to GRs in the hypothalamus and anterior pituitary, exerting a negative feedback which results in a decreased production of corticotropin-releasing hormone and adrenocorticotropic hormone.72,74,75 This in turn results in a decrease in endogenous cortisol production in the adrenal cortex.
A randomized, placebo-controlled study demonstrated that healthy subjects given ciclesonide 800 μg/day (ex-valve) for 7 days did not experience significant HPA-axis suppression, as assessed by a 24-h profile of serum cortisol. 76 Also, the time of administration did not affect the results. Several trials have compared the effects of fluticasone propionate and ciclesonide on HPA-axis suppression. One study randomized subjects with mild-to-moderate asthma to receive placebo, ciclesonide 320 μg once daily, ciclesonide 320 μg twice daily, or fluticasone propionate 440 μg twice daily, for a period of 12 weeks. 75 Neither of the ciclesonide groups caused significant changes in 24-hour urinary free cortisol levels or peak serum cortisol levels following a sequential low- and high-dose cosyntropin stimulation. However, the group receiving fluticasone propionate displayed significant decreases in 24-hour urinary free cortisol levels and serum cortisol levels following highdose cosyntropin stimulation. In the intention-to-treat population, this included a 60.8% decrease in the mean level of 24-hour urinary free cortisol compared with a 19.8% decrease seen with placebo. A similar study conducted in asthmatic patients showed that a ciclesonide dose as high as 640 μg twice daily did not result in a significant decrease in the AUC0-24 for plasma cortisol following 9 days of treatment (Fig. 3). 77 Urinary excretion of cortisol over a 24 hour period also remained unaffected.
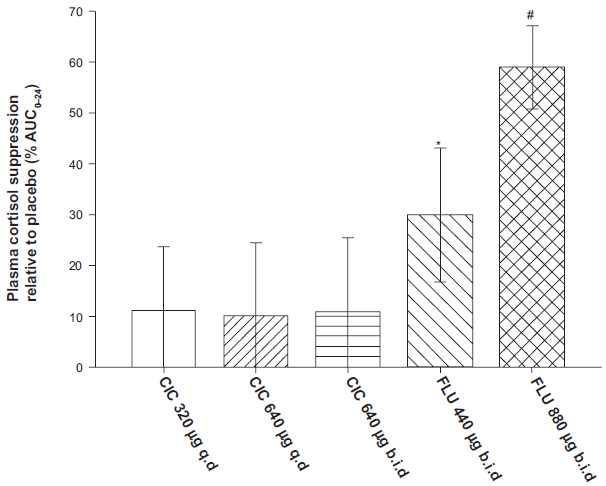
Mean plasma cortisol suppression after 9 days of treatment with ciclesonide 320 μg once daily, ciclesonide 640 μg once daily, ciclesonide 640 μg twice daily, fluticasone propionate 440 μg twice daily, and fluticasone propionate 880 μg twice daily. The degree of suppression is measured as AUC0-24 and expressed as a percentage fall from placebo for each drug treatment. (Figure adapted from 77 )
Treatment with fluticasone propionate 440 μg twice daily and 880 μg twice daily resulted in a 29% and 59% decrease, respectively, in plasma cortisol AUC0-24. Similar to plasma cortisol, a significant and dose-dependent decrease in urinary cortisol excretion was seen for both regimens of fluticasone propionate. Several other randomized clinical trials which have evaluated high doses of both drugs, report minimal to no HPA-axis suppression with ciclesonide, whereas significant suppression was seen with high-doses of fluticasone propionate.40,78
ICSs use may also cause growth suppression, a side-effect that requires special consideration in children that are prescribed an ICSs. A randomized, double-blind, placebo-controlled clinical trial sought to evaluate the effects of ciclesonide (40 or 160 μg/day) on growth in a large sample of children with mild, persistent asthma (Fig. 4). 79 After 1 year of treatment, both ciclesonide treatment regimens were noninferior to placebo in terms of growth velocity and mean change in stadiometer height (cm) from baseline. The low incidence of systemic side effects seen with ciclesonide use is a result of its favourable PK-PD profile. In particular, its high pulmonary deposition, low oral bioavailability, high plasma protein binding, and high hepatic clearance, minimize the amount of free, unbound drug in the circulation that is able to bind to GRs. This then may result in its improved therapeutic ratio.
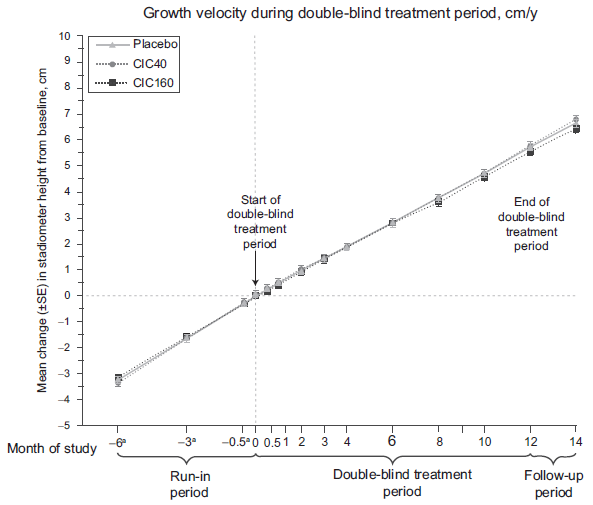
Mean change in stadiometer height (cm) from baseline for children receiving placebo, ciclesonide 40 μg, or ciclesonide 160 μg (ex-actuator) once daily. (Reference 79; permission to reuse this figure was obtained).
Place in Therapy
The role of ciclesonide in the management of asthma still requires further study since no clinical trials have compared ciclesonide with mometasone furoate, flunisolide, or triamcinolone acetonide. 80 Clinical trials have documented ciclesonide's positive effects on pulmonary function, and have concluded that it is at least equally efficacious to fluticasone propionate and budesonide. Based on available evidence the main characteristic which distinguishes ciclesonide from active comparators is its improved safety profile. The low incidence of local side effects and lack of HPA-axis suppression result in an improved therapeutic ratio. As a result of this, in patients with severe asthma who require high-doses of an ICSs, ciclesonide may again be a favourable choice due to its lack of HPA-axis suppression.
Conclusions
ICSs are considered first-line therapy in the treatment of asthma. Ciclesonide is a novel ICSs which is approved in most countries for the management of allergic rhinitis and asthma. Its unique PK-PD profile results in an improved safety profile, with minimal to no effects on HPA-axis function. Its favourable PK and PD properties include a high pulmonary deposition, prolonged pulmonary residence time, on-site activation, high clearance rate, extensive plasma protein binding, and an active metabolite with a high RRA. Efficacy studies have shown that ciclesonide is at least as effective as fluticasone propionate and budesonide. Few to no studies have been conducted with beclomethasone dipropionate, flunisolide, and triamcinolone acetonide. More studies are needed before ciclesonide's place in therapy can be further described. However, based on ciclesonide's low incidence of systemic side effects, it is clear that several groups of patients could benefit from this drug. Some examples would be children who require use of an ICSs, and patients who use high-doses of an ICSs in order to control their asthma.
Disclosure
Dr. Derendorf has been a consultant for Sanofi-Aventis US and ALTANA Pharma US, Inc-a NYCOMED Company.
