Abstract
Allergic rhinitis affects more than 20% and is responsible for very high direct and indirect costs. Nasal corticosteroids are the principle pharmaceutical option for the treatment of allergic rhinitis. They are more effective than all other therapeutic options and in addition to improving symptoms of allergic rhinitis they also improve quality of life in patients suffering from allergic rhinitis. There are many intranasal corticosteroids approved for the treatment of allergic rhinitis in pediatric and adult populations. Ciclesonide has shown so far a very good safety and pharmacokinetic profile and is effective for treatment of symptoms in both seasonal and perennial allergic rhinitis. This review summarizes the studies describing these effects of intranasal ciclesonide.
Allergic Rhinitis
Allergic rhinitis is one of the most common chronic conditions and is estimated to affect at least 20% of the adult and close to 40% of the pediatric population in the United States. 1 A cross-sectional, random-digit-dialling telephone survey of Canadian households showed that 45% of Canadian adults report allergyrelated nasal symptoms and a little less than half of them have been diagnosed with allergic rhinitis by a physician. 2 A study in 10 European countries showed a prevalence of respiratory allergic conditions between 11.7% and 36.6%. 3 The direct cost for allergic rhinoconjunctivitis in the US in 1996 was estimated to be between $1.9 and $3 billion,4,5 while an additional $4 billion was spent on co-morbidities. 5 The cost in 2004 was estimated to be between $2 and $5 billion. 6 In addition to the direct cost for the disease productivity losses due to allergic rhinoconjunctivitis are estimated to be between $2.4 and $4.6 billion. 7 In a German study the average annual cost for patients with seasonal allergic rhinitis was estimated to be €1,089 for children and adolescents and €1,543 for adults. 8
Allergic rhinitis may not be life threatening or lead to significant morbidity from other organ systems, but it greatly affects quality of life in both adults and children. 9 It is now recognized that the main goal of treatment should be improvement of quality of life for suffering individuals. Many attempts have been made to measure quality of life in patients with allergic rhinitis. There are indications that a generic instrument such as the Medical Outcomes Study Short-Form Health Survey (SF-36) may be useful for accessing quality of life in patients with allergic rhinitis. 10 This study showed that perennial allergic rhinitis impairs quality of life to a degree similar to what is seen in patients with asthma. There also exists a more specific questionnaire for allergic rhinitis, the Rhinoconjunctivitis Quality of Life Questionnaire (RQLQ), 11 which has also been adapted for adolescent and pediatric populations. There is however poor correlation between the number and severity of symptoms reported by patients and the impairment of their quality of life. Therefore, outcomes that focus only on symptom improvement may not be adequate to describe the presence or absence of positive effects of medical treatment on a patient's well being.
Efficacy of intranasal corticosteroids in allergic rhinitis
Nasal corticosteroids are the principal pharmaceutical option for the treatment of allergic rhinitis. The 2008 ARIA report considers them the most effective drugs for the treatment of allergic rhinitis (ARIA report). Meta-analyses have shown that nasal corticosteroids are more effective than oral H1-antagonists, 12 topical H1-antagonists 13 and leukotriene-receptorantagonists 14 for the treatment of allergic rhinitis. Intranasal corticosteroids have also been shown to improve quality of life15,16 in patients with allergic rhinitis.
Intranasal corticosteroids are the first line treatment for nasal congestion, since they are more effective for congestion than any other treatment. Intranasal corticosteroids have also been shown to be effective in decreasing ocular symptoms in subjects with allergic rhinoconjunctivitis. 17 Meta-analyses have shown that intranasal corticosteroids are not inferior to oral 12 or topical antihistamines 13 for the treatment of eye symptoms in these subjects.
Nasal corticosteroids have minimal side effects and can be combined with other treatments with only minor interactions if any.
Comparisons between corticosteroids
The high cost of allergic rhinitis to the patient, the health care system, and the society in general, increases the significance of finding effective therapeutic options to decrease disease burden, decrease direct and indirect costs of the disease and improve patient quality of life. Many new therapeutic options are currently under study for allergic diseases including allergic rhinitis. However, we already have a very effective treatment in nasal corticosteroids and we should strive to improve appropriate diagnosis and patients’ compliance with this therapeutic option if appropriate. Corticosteroids with improved tolerance, efficacy and lower side-effect characteristics are also a valuable goal.
Few studies have focused on the relative potency or relative side effect profile of the different nasal corticosteroids. There are only a few direct head-to-head comparisons.
Improving efficacy of corticosteroids, while at the same time minimizing the side effect profile of these drugs, has been the holy grail of research in this area. Systemic side effects are less of a problem in allergic rhinitis as compared to asthma, because the doses of corticosteroids used in allergic rhinitis are much lower. However, any improvement in the effectiveness to side effect ratio is always an important advancement in therapeutics.
The local side effect profile is also important for a medication that has to be used daily and may affect olfaction and the nasal cavity. Many patients complain of an unpleasant smell when using nasal corticosteroids, while nasal dryness and bleeding are also common occurrences.
A recent review has concluded that all inhaled corticosteroids provide similar efficacy and safety when used at low to medium doses. 18 Differences in side effects between topical corticosteroids may appear when they are used at high doses, since there is some evidence that their effect on HPA axis suppression depends on absorption and bioavailability, which may differ between molecules. However, there is not expected to be much difference between them at the small topical doses used for the treatment of allergic rhinitis. The Updated Practice Parameters for Diagnosis and Management of Rhinitis, published in 2008, state the following in summary statement 77: “when comparing the available intranasal corticosteroids, the overall clinical response does not appear to vary significantly between products irrespective of the differences in topical potency, lipid solubility, and binding affinity.” 19
Intranasal Ciclesonide
Active compound and relative potency
Endogenous glucocorticoids and all the synthetic corticosteroids used clinically mediate their effects through activation of the corticosteroid receptor (GR), which is a member of the steroid-hormone receptor family of proteins. Corticosteroids bind to the receptor with high affinity and promote the dissociation of molecular chaperones, including heat–-shock proteins. The corticosteroid–-glucocorticoid receptor complexes move to the nucleus, homo-dimerize and bind to DNA sequences called glucocorticoid-responsive elements (GRE). Co-activator or co-repressor proteins are then recruited and the assembly of transcription machinery is either promoted or inhibited. These complexes can also interact directly with other transcription factors and alter their activation status. The therapeutic effect of all of these corticosteroids comes from the inhibition of a variety of pro-inflammatory pathways. 20 Human glucocorticoid receptor mRNA has alternative splice variants that lead to the production of two isoforms of the GR protein, GRα and GRβ. There is evidence that the presence of the GRβ isoform leads to corticosteroid resistance in asthma 21 and in nasal polyposis. 22
Ciclesonide is a non-halogenated corticosteroid that is administered as an inactive parent ester compound. The parent compound is hydrolyzed to desisobutyryl-ciclesonide (des-CIC) by esterases in the lung tissue. 23 Airway epithelial cells 24 and lung fibroblasts 25 have the potential to metabolize CIC to des-CIC. In addition, nasal epithelium from multiple species activates ciclesonide into des-CIC. 26 Human nasal epithelial cells also activate ciclesonide and fatty acid conjugates of des-CIC can be retained by these cells for more than 24 h. 27 Long retention of these fatty acid conjugates is important for ciclesonide efficacy 28 and allows once daily dosing of the medication for allergic rhinitis. An in vivo study in rabbits has shown that ciclesonide activation and retention by nasal epithelium is more efficient when ciclesonide is administered in a hypotonic solution, 29 which is the current formulation of the medication for intranasal administration.
Ciclesonide undergoes extensive first-pass metabolism in the liver predominantly by the cytochrome P450 (CYP) isoenzyme 3A4. 30 Since the drug has low bioavailability and low potential for systemic side effects, dose adjustment is not considered necessary. Systemic absorption of the drug is very low, which further decreases the potential for systemic side effects. Elimination is predominantly via feces; complete elimination requires 80–120 h. 31 Although maximum serum concentration of des-CIC increased during co-administration with the strong CYP3A4 inhibitor ketoconazole, there was no difference in the rate of metabolism of ciclesonide to the active compound. 32 Most of these studies have been performed after single or multiple administrations of inhaled medication, but the characteristics of intranasal administration are expected to be the same. The pharmacokinetic and pharmacodynamic profile of inhaled ciclesonide has been reviewed elsewhere. 33
The relative binding activity of ciclesonide to corticosteroid receptors in the rat lung was approximately 8-fold lower than that of dexamethasone, but the relative binding activity of des-ciclesonide was 12-fold higher than that of dexamethasone and 100fold higher than that of ciclesonide, suggesting that des-ciclesonide is the active metabolite of ciclesonide. 34 Des-ciclesonide had a number of in vitro and in vivo anti-inflammatory effects, including the inhibition of eosinophil accumulation in the airways of ovalbumin sensitized and challenged Brown-Norway rats and bradykinin-induced protein leakage in rat airways. 34 Another study showed that des-ciclesonide and fluticasone had comparable binding to corticosteroid receptors and comparable efficacy in the Brown-Norway rat model of allergic airway inflammation. 35 In addition, that study showed that ciclesonide-induced adrenal involution was much less pronounced than that induced by fluticasone. In addition, ciclesonide inhibited airway remodeling in a similar model. 36
Onset of action
A double-blind, randomized, parallel-group, placebo-controlled, single-center study was conducted in an environmental exposure chamber in order to identify onset of action of ciclesonide nasal spray. It was shown that 200 μg of ciclesonide significantly decreased total nasal symptom scores 6 to 12 h after administration in patients with seasonal allergic rhinitis (SAR). 37 Another similar study showed this onset of action to be much earlier, at 1 h after administration of a single dose of 200 μg of ciclesonide. 38 The reason for this difference is not clear, but there are suggestions that the onset of action identified in different studies depends also on the study design. 39 The published data on onset of action of different intranasal corticosteroids vary a lot. 39 For example, studies have shown onset of action at 8 h for fluticasone furoate, 40 12 h for budesonide 41 and 4 h for mometasone furoate. 42 The data on ciclesonide onset of action presented above show that its onset of action is at least as quick as many of the other nasal corticosteroids.
Studies on efficacy of intranasal corticosteroids discussed in the next section have shown in general that ciclesonide is more effective than placebo in relieving chronic nasal symptoms after 2 to 5 days of treatment.
Efficacy
The published data on the efficacy of intranasal ciclesonide in allergic rhinitis are limited. There are 3 published studies on its efficacy in SAR and 2 in perennial allergic rhinitis (PAR). We have much more information on the efficacy of ciclesonide in asthma. Information on the effects of inhaled ciclesonide in patients with asthma has been reviewed recently. 43 The results from the published studies on the efficacy of intranasal ciclesonide are summarized in Table 1 and presented in more details below.
Summary of ciclesonide clinical trials in allergic rhinitis.
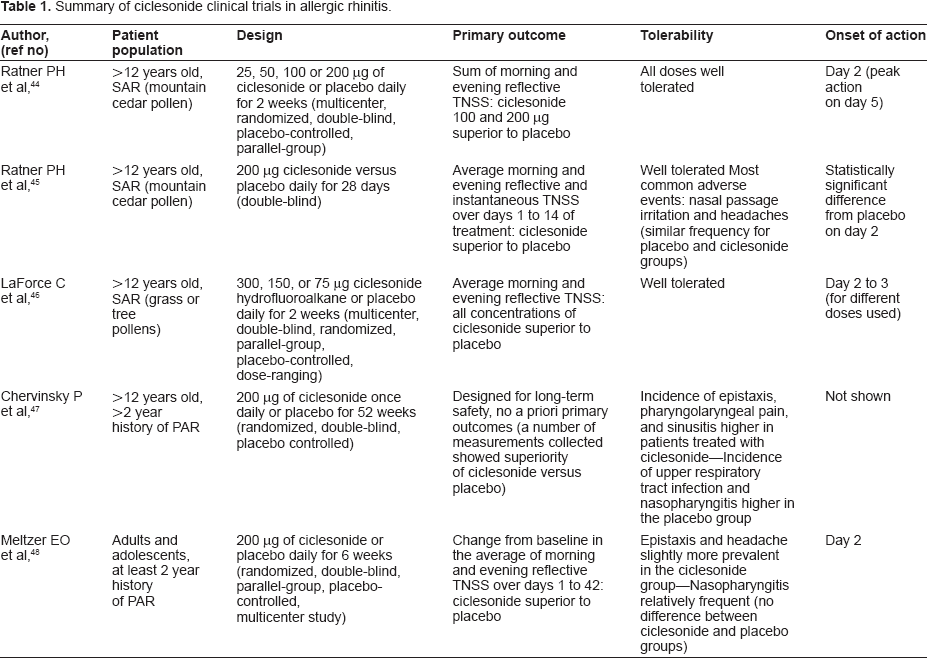
A multicenter, randomized, double-blind, placebo-controlled, parallel-group study conducted during the 2002 to 2003 mountain cedar pollen season evaluated the change in the sum of morning and evening reflective total nasal symptom scores (TNSS) over 2 weeks of 25, 50, 100 or 200 μg of ciclesonide versus placebo and showed that the two higher doses of ciclesonide led to improvement of these outcomes. 44 Onset of action of ciclesonide was rapid, with a statistically significant difference between the placebo and the 200 μg/day ciclesonide groups already present by day 2 and with a peak difference between the two groups on day 5. Among the secondary outcomes the study also showed that the sum of morning and evening instantaneous TNSS decreased with the two higher doses of ciclesonide while there was no difference in rescue medication use between the placebo and ciclesonide groups.
A second double-blind study from the same group with a similar patient population, evaluated 200 μg of intranasal ciclesonide versus placebo given daily for 28 days in patients over 12 years of age with seasonal allergic rhinitis to mountain cedar pollen. The study showed improvement of average morning and evening reflective and instantaneous TNSS with ciclesonide versus placebo over days 1 to 14 of treatment. 45 This effect of ciclesonide was maintained throughout the duration of the study; significant improvement of average morning and evening reflective TNSS was also seen over days 1 to 28 and 14 to 28 of treatment. Onset of action of ciclesonide was again quite rapid, with a difference from placebo appearing by day 2 of treatment. In addition, ciclesonide showed a trend towards improvement of reflective non-nasal symptoms. Ciclesonide, however, did not improve physician-assessed overall nasal signs and symptom severity or the results from the Rhinoconjunctivitis Quality of Life Questionnaire.
More recently a study similar in design was performed to evaluate the efficacy of a ciclesonide hydrofluoroalkane nasal aerosol in patients with seasonal allergic rhinitis. This aerosol showed improvement in average morning and evening reflective TNSS in doses higher than 75 μg. 46 There has been no attempt to compare this new ciclesonide formulation to the previous one in this or any other studies so far.
There are two studies on the effectiveness of ciclesonide in perennial allergic rhinitis. A randomized, double-blind, placebo controlled study of patients over 12 years of age with a more than 2 year history of perennial allergic rhinitis 47 studied the efficacy of 200 μg of ciclesonide once daily for 52 weeks. 663 patients were randomized to placebo or ciclesonide in a 1:2 ratio and a little over 70% from each group completed the study. The study was primarily designed to study the long-term safety of the preparation so there were no primary efficacy outcomes defined a priori, but a number of efficacy measurements were collected. Patient-evaluated morning 24-hour reflective TNSS, patient-evaluated individual nasal symptom scores (sneezing, runny nose, nasal itching, and nasal congestion), and physician evaluation of overall nasal signs and symptom severity (PANS) were collected throughout the treatment period. Patients recorded daily 24-hour reflective TNSS in diaries each morning before the administration of study medication. At 24 and 48 weeks, the authors also collected combined adult and adolescent Rhinoconjunctivitis Quality of Life Questionnaires (RQLQ) data. The 24-hour reflective TNSS decreased from baseline in both placebo and ciclesonide groups, but ciclesonide achieved a significantly greater reduction. Differences in morning 24-hour reflective TNSS were greater for subjects on ciclesonide treatment starting from the second week. Ciclesonide produced a significantly greater improvement in nasal stuffiness and congestion, nasal itching, sneezing, and runny nose compared to placebo, although again these symptom scores improved in both groups. Ciclesonide induced a greater improvement in RQLQ compared to placebo but the difference did not reach the point that would be considered clinically significant. There were, however, no appreciable differences between the ciclesonide and placebo treatment groups in overall or individual signs or symptoms of PANS at the end point.
A second study also evaluated the efficacy of daily ciclesonide nasal spray in PAR. 48 Adults and adolescents with at least a 2 year history of PAR were treated with 200 μg of ciclesonide or placebo daily for 6 weeks in a randomized, double-blind, parallel-group, placebo-controlled, multicenter study. The primary efficacy measure was the change from baseline in the average of morning and evening patient-evaluated reflective TNSS over days 1 to 42, where baseline was the average of responses obtained up to 7 days before randomization. A number of secondary outcomes, similar to the other studies discussed, were also evaluated. Intention to treat analysis was used for all efficacy measures. The study showed that ciclesonide decreased the average morning and evening reflective TNSS significantly more than placebo over the 42 days of the trial. In addition, ciclesonide improved average morning and evening reflective TNSS significantly over days 1–14, 15–28 and 29–42. The magnitude of treatment difference between placebo and ciclesonide increased from the first evaluation (days 1–14) to the last (days 29–42). Ciclesonide showed a trend towards improvement of PANS scores compared to placebo at the end of the study, but improved PANS scores on day 22 of the study.
The last study showed a large improvement of symptoms with placebo treatment. This effect was also evident in the previous studies we discussed as well as in most other studies with nasal corticosteroids or other nasal topical medications. This potent effect of placebo in allergic rhinitis may have many explanations. Saline solution may keep the nasal cavity moist, or it may decrease nasal irritation. Finally saline solution may enhance mucociliary clearance and drainage of the nasal cavity, although the mucociliary clearance is thought to be affected primarily by hypertonic solutions during nasal irrigation rather than just the administration of a nasal spray.
Eye symptoms have not been evaluated in most studies with intranasal ciclesonide. In many cases Rhinoconjunctivitis Quality of Life Questionnaire scores are evaluated and improve with the use of ciclesonide, but there is no mention of specific improvement in ocular symptoms. One study assessed reflective non-nasal symptoms, which included primarily eye symptoms, but also itching of ears and palate, using patient diaries, and demonstrated a numerically larger response in the ciclesonide group versus the placebo group. 45 Further study of the potency of ciclesonide for the treatment of eye symptoms in subjects with allergic rhinoconjunctivitis should be performed to elucidate this issue.
Tolerability/safety
Tolerability of the drug has been studied extensively in healthy and asthmatic individuals. The drug was very well tolerated and induced only mild to moderate adverse reactions, many of them deemed to be unrelated to the drug. These studies have been reviewed recently. 43 Here we will focus exclusively on the tolerability of intranasal ciclesonide.
The studies discussed above regarding efficacy of intranasal ciclesonide also studied the tolerability of the preparation in patients with SAR and/or PAR.
Ratner et al 44 demonstrated that all doses of ciclesonide were remarkable well tolerated over 2 weeks. Headaches and pharyngitis were the two most common adverse events reported in the ciclesonide groups. The number of patients with increased intraocular pressure was small and equally distributed between placebo and ciclesonide groups. Discontinuation of the study because of side effects was also rare and occurred both in subjects receiving placebo and subjects receiving ciclesonide. A similar safety profile was also seen in study where patients with seasonal allergic rhinitis were treated for 28 days with 200 μg of ciclesonide daily. 45 The most common adverse events in patients receiving ciclesonide were nasal passage irritation and headaches, but the frequency of these adverse events was not statistically different from subjects receiving placebo. There was only one serious adverse event in subjects receiving ciclesonide, which was considered to be unrelated to the study medication. When ciclesonide hydrofluoroalkane nasal aerosol was studied in seasonal allergic rhinitis there was no difference between the active compound and the placebo in the number, severity, and type of adverse events seen. 46 One of the main reasons for having a new formulation of intranasal ciclesonide would be increased tolerability. However, this study did not compare this new formulation to the one that is currently available in North America. Therefore, we could not evaluate whether the new formulation is superior to the one currently used.
In a 6 week study treating patients with PAR using 200 μg of ciclesonide daily, the adverse events were again mild to moderate. 48 Epistaxis and headache was slightly more prevalent in the ciclesonide versus the placebo group. Nasopharyngitis was another relatively frequent ciclesonide side effect, but its frequency was not different than that seen in subjects receiving placebo. The number of subjects that discontinued the study medication was similar between the ciclesonide and placebo groups. A longer duration study treated patients with PAR for 52 weeks with 200 μg of ciclesonide or placebo daily. 47 Treatment-emergent adverse events were reported by ¾ of the patients in each group, while severe adverse events were reported by 13.4% of patients in the ciclesonide group and 11.7% of patients in the placebo group. Upper respiratory tract infection, nasopharyngitis, epistaxis, sinusitis, and pharyngolaryngeal pain were the most frequent adverse events regardless of treatment group. The incidence of epistaxis, pharyngolaryngeal pain, and sinusitis was higher in patients treated with ciclesonide, whereas the incidence of upper respiratory tract infection and nasopharyngitis was higher in the placebo group. No clinically relevant differences were observed between the ciclesonide and placebo groups in 24-hour urinary free or morning plasma cortisol levels. Similarly, no clinically relevant differences were found between treatment groups in intraocular pressure, visual acuity, or lens opacification.
Finally, another study evaluated the safety of adding intranasal ciclesonide in patients already being treated for asthma with inhaled beclomethasone dipropionate. 49 The investigators showed that the addition of ciclesonide did not induce any decrease in mean plasma cortisol levels or urinary free cortisol compared to placebo. Adverse events in general were also not different between he two groups.
In general these studies show that daily intranasal ciclesonide is very well tolerated for up to 52 weeks, with a small number of side effects, which are generally mild to moderate in severity. Tolerability of ciclesonide also compares favourable with the tolerability of other intranasal corticosteroids, such as fluticasone furoate 50 and budesonide. 51
Formulation
There are a number of formulation characteristics that may render one nasal corticosteroid preferable to another. 52 Osmotic pressure can modulate absorption and retention of the solution on the nasal mucosa and influence clinical efficacy. Other characteristics may affect sensory attributes of the formulation, such as unpleasant taste or smell and drying of the nasal mucosa. Finally, the choice of device used to deliver the medication may also influence patient preferences.
Ciclesonide is a white to yellow-white powder that is almost insoluble in water but freely soluble in ethanol and acetone. Ciclesonide is marketed as a metered-dose, manual pump spray formulation of a hypotonic aqueous suspension, under the trade name Omnaris® Nasal Spray. The formulation also contains microcrystalline cellulose, carboxymethylcellulose sodium, hypromellose, potassium sorbate and edetate sodium. The pH is adjusted to 4.5 using hydrochloric acid. Omnaris Nasal Spray must be gently shaken and then the pump must be primed by actuating eight times prior to initial use. Once primed, each actuation of the pump delivers 50 μg ciclesonide in a volume of 70 μl (Omnaris Prescribing Information, http://www.omnaris.com/OMNARIS-Prescribing-Information.pdf).
Indications
Ciclesonide nasal spray has been approved as a treatment for allergic rhinitis in many countries including Canada, the US and countries of the European Union. In the US and Canada it is approved for adults and children older than 6 years with seasonal allergic rhinitis and for adults and children over 12 years old with perennial allergic rhinitis. The recommended total dose in all cases is 200 μg/day administered as two sprays in each nostril (50 μg/spray) once a day (Omnaris Prescribing Information, http://www.omnaris.com/OMNARIS-Prescribing-Information.pdf).
Conclusions
Ciclesonide is a well-tolerated nasal corticosteroid that is effective for prevention and treatment of nasal symptoms in seasonal and perennial allergic rhinitis. Although there are no direct data comparing the efficacy of ciclesonide to other nasal corticosteroids, ciclesonide has a number of advantages that would indicate that it should be at least as good if not better. These include the fact that it is administered as a prodrug and activated only in the area of its effectiveness, and the fact that it has high binding to proteins in the serum and rapid clearance, which decreases its bioavailability for development of side effects. Ciclesonide has also a favorable side effect profile and is well tolerated. Ciclesonide, or other nasal corticosteroids, should be considered as a first line treatment for every patient suffering from seasonal or perennial allergic rhinitis.
Disclosure
This manuscript has been read and approved by the author. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The author reports no conflicts of interest.
