Abstract
Chronic Obstructive Pulmonary Disease is a prevalent disease throughout the world. Recently updated treatment guidelines were published and still endorse a general step-wise approach to therapy based on severity of disease. The heterogeneity of the disease makes it difficult to determine appropriate therapy based on the data available. Currently, combination therapy, specifically triple therapy with a long acting beta2 agonist, a long acting anticholinergic agent, and an inhaled corticosteroid appear to achieve the best patient outcomes. In addition, there is a desperate need for new therapy with a different mechanism of action than those already on the market. The recommendations concerning stable COPD treatment will be reviewed, as well as, efficacy data regarding combination therapy. In addition, a few new and prospective therapies will be evaluated.
Background
Chronic obstructive pulmonary disease (COPD) is a condition of airflow limitation that is not fully reversible and is usually progressive. 1 It is the fourth most common cause of death worldwide. Based on Canadian data, about 28% of the population will be diagnosed with COPD during their lifetime. 2 This is more than those diagnosed with heart failure, myocardial infarction, and some common cancers such as breast and prostate. In addition, it is a leading cause of disability, hospitalization, and health care cost.1,2
The most common cause of COPD is smoking; however, even in the United States up to 19% of COPD in smokers and 31% of COPD in nonsmokers may be attributable to indoor and outdoor air pollutants including occupational dust and fume exposure.1,3,4 This number increases in countries where air pollution is prevalent and in which solid fuels are used for heating and cooking. 1 Chronic exposure to smoke and other noxious particles cause inflammation in the lungs resulting in tissue destruction and small airway narrowing. Lung elastic recoil is eventually decreased and the airways have a difficult time remaining open during expiration. Inflammation is present in all stages of COPD and affects tissue throughout the lungs, including the proximal and peripheral airways, lung parenchyma, and pulmonary vasculature.
The inflammatory response in COPD patients is amplified compared to those without the disease. 1 Evidence shows that only a subset of smokers or people chronically exposed to noxious particles develop COPD suggesting that other predisposing factors cause amplification of inflammatory response making them more likely to develop the disease.3–5 These predisposing factors include genetics, epigenetic changes, and altered reaction to oxidative stress. 1
Smoking and irritants activate both innate and adaptive immunity in the lungs.6,7 Macrophages in the respiratory tract release a large number of inflammatory mediators that damage tissue and cause progression of disease including leukotrienes, cytokines, chemokines, transforming growth factor (TGF), and proteases. Chemotactic factors are released and attract neutrophils, monocytes, and CD8+ T lymphocytes in lung tissue. In addition, there is an imbalance between proteases that break down connective tissues and antiproteases that protect tissues in the lungs of COPD patients. Proteases including neutrophil elastase and metalloproteinases are released breaking down connective tissue including elastin in the lung parenchyma leading to emphysema and stimulating mucus hypersecretion leading to chronic bronchitis. TGF-β and connective tissue growth factor (CTGF) are also present and mediate small airway fibrosis. In addition, oxidants are present damaging lipids and proteins leading to cell death. Chronic inflammation leads to changes within the airway presenting with a combination of signs and symptoms including chronic bronchitis, emphysema, and/or pulmonary hypertension.
In addition to the inflammation, the lung undergoes tissue destruction and changes including air trapping on expiration and hyperinflation.1,6,7 Hyperinflation has been shown to be a major contributor to exertional dypsnea by effecting muscles and causing mechanical changes in the lung decreasing respiratory capacity. It is also a predictor of mortality. 8 In addition, some patients have destruction and loss of alveoli walls and enlargement of airways resulting in loss of gas exchange surface area.1,6,7 Small airways collapse and patients become hypoxemic. Smooth muscle and connective tissue becomes thickened, scarred and fibrotic. Eventually, pulmonary vessels become thickened and may lead to pulmonary hypertension and cor pulmonale. The degree to which forced expiratory volume in 1 second (FEV1) and FEV1 to forced vital capacity (FVC) ratio are affected determine the stage and severity of the disease (Table 1). 1
Severity and treatment of COPD as outlined by GOLD.
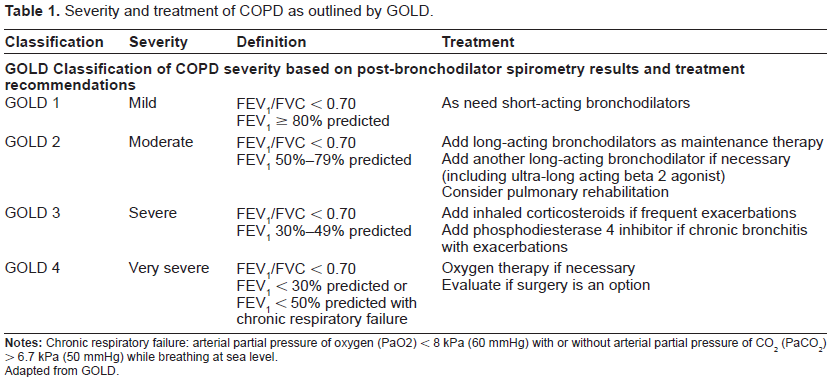
Adapted from GOLD.
This article will review the current COPD treatment guidelines and recommendations concerning stable COPD treatment will be reviewed.1,9 Recent efficacy data regarding combination therapy with currently marketed agents will be presented and evaluated. In addition, a few new and prospective therapies will be discussed.
A MEDLINE search of English-language literature was performed from 1950 to December 2011 using the search terms chronic obstructive pulmonary disease, chronic bronchitis, emphysema, and treatment guidelines. Separate searches were performed using the search terms COPD and beta2 agonists, anticholinergics, inhaled corticosteroids, phosphodiesterase-4 inhibitors, and combination and triple therapy. A thorough review of data regarding roflumilast is included. Data on dual therapy are summarized and all trials evaluating triple therapy were included in the review.
Current Guidelines
Current COPD treatment goals include prevention and treatment of symptoms and exacerbations and improving exercise tolerance and quality of life.1,9 Although two long-term goals are to modify the long-term decline in lung function and decrease mortality, there is a paucity of data showing pharmacotherapy has a positive impact on these two outcomes; however, cessation of smoking, supplemental oxygen and lung reduction surgery have affected these outcomes.1,10–12
The importance of prevention of COPD exacerbations should not be minimized. Exacerbations contribute to accelerated decline in lung function and mortality is up to 40% one year after an exacerbation.1,13–19 Exacerbations require a long recovery period before patients’ lung function return to baseline. About 50% of hospitalized patients will be readmitted at least once in six months following an acute exacerbation and hospital mortality in patients with exacerbations is 8%-11%.1,13–19 Therefore, preventing exacerbations will not only decrease hospitalizations and costs, it should prevent morality, maintain quality of life, and potentially help maintain lung function.
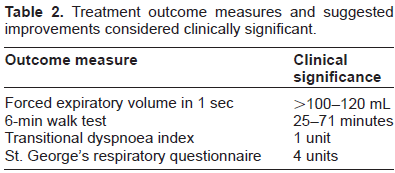
Consensus guidelines recommend a step-wise approach to chronic treatment of COPD (Table 1).1,9 Pharmacotherapy regimens need to be individualized because of the heterogeneity of the disease presentation. After spirometry results and clinical symptoms determine the severity of disease, therapy should be added and response to medication monitored. (Table 1) Patients will require at least several weeks to months on medications to see effects and improvement in outcome measures. It is unclear what the best outcome measures are to determine efficacy. Most COPD patients have minimally reversible airflow disease based on spirometry improvements after bronchodilators are used. 1 Bronchodilators do not change or minimally improve FEV1 in these patients. In addition, the improvements in FEV1 are often not considered clinically significant; however, these same therapies show positive effects on quality of life, dypsnea scores, exercise tolerance, exacerbation rates, and hospitalizations (Table 2). 20 Therefore, improvement in FEV1 should not be the only and/or best determinant of efficacy. This needs to be considered when evaluating not only trial results, but also evaluating therapy in individual patients. Ultimately the number of medications will be cumulative as the disease progresses and daily maintenance therapy will eventually be required.
Treatment outcome measures and suggested improvements considered clinically significant.
Bronchodilators
Bronchodilators which includes beta2-agonists (B2 agonists), anticholinergic agents, and methylxanthines are considered the foundation of stable COPD treatment.1,9 Short acting inhaled B2 agonists and anticholinergics are recommended first line as needed for symptom relief early in disease progression. As the disease progresses from mild to moderate severity, patients will begin to have poor symptom control on short acting agents. At this point, long-acting bronchodilators should be prescribed. The inhaled route is preferred to oral or intravenous route because efficacy is usually the same when proper administration techniques are used and it limits systemic adverse effects. Bronchodilators relax bronchial smooth muscle, improve lung emptying, reduce thoracic hyperinflation, and improve exercise tolerance. 20 As stated previously, these benefits are seen even without improvements in FEV1 considered clinically significant. Although, they have shown some efficacy in COPD patients, methylxanthines are not considered first line agents because of their narrow therapeutic window and toxicities. 1
Beta2-agonists
B2 agonists work by stimulating B2 adrenergic receptors. As a result of stimulation of the B2 receptor, cytoplasmic G proteins are activated which then activate adenylyl cyclase to produce cyclic adenosine monophosphate (cAMP). 21 cAMP relaxes bronchial smooth muscle which leads to bronchodilation. Although it may have greater impact in asthma compared to COPD, B2 agonists may suppress release of leukotrienes and histamine from mast cells in the lung, enhance mucociliary function, decrease microvascular permeability, and inhibit phospholipase A2.
Preferred short acting B2 agonists include albuterol, levalbuterol, and pirbuterol.1,9 Their short onset of action of 5 minutes makes them ideal for as needed use in stage I disease. Their duration of action is 4-6 hours. These agents have been shown to improve lung function, dyspnea, and exercise endurance. 22 Adverse effects are dose related and a result of excess activation of B2 receptors. More common adverse effects include tachycardia, tremors, and hypokalemia.
Albuterol is a racemic mixture of (R)-albuterol which produces a bronchodilator effect and (S)-albuterol which has no therapeutic effect, but may worsen airway inflammation and increase intracellular calcium levels promoting smooth muscle contraction and bronchial hyperresponsiveness.23–26 It may also recruit and activate eosinophils in the airway. Levalbuterol is the single isomer of (R)-albuterol and some believe this gives it an advantage over albuterol. There is controversy whether this is truly the case clinically and whether any benefits outweighs the cost of the medication.27–31
Maintenance therapy with long-acting beta2 agonists (LABAs) salmeterol, formoterol, and arformoterol may be added in moderate disease.1,9 All are dosed twice daily. Formoterol and arformoterol have a quick onset of action compared to salmeterol, 5 minutes versus 15 minutes, respectively. 21 Arformoterol is the (R, R)-enantiomer of formoterol and accounts for formoterol's activity. Studies have shown arformoterol to be as efficacious salmeterol and formoterol without clinical advantage over the other agents.32,33 LABAs have been shown to significantly improve lung function, quality of life, exercise endurance, and reduce symptoms and exacerbations.34–37 Regardless, the guidelines currently do not recommend one LABA over another.1,9
Chronic administration of B2 agonists causes downregulation and decreased binding affinity to the B2 receptors. 26 As a result, patients develop tolerance to the effects of the drugs; however the lungs seem less affected than other tissues because of the large amount of B2 receptors found there. Tolerance usually develops the first week after initiation of chronic use and does not seem to worsen after this. In addition, patients will experience less systemic side effects with chronic use. Tolerance often has more effect on duration of action rather than peak effect. If necessary, B2 agonist dose and/or frequency may be increased. It may be particularly clinically relevant in a patient on a chronic LABA using as needed doses of the short acting B2 agonists for acute dyspnea.
Anticholinergics
Short acting anticholinergics, ipratropium and oxitropium, are also available to use as needed in Stage I disease.1,9 These agents interact with muscarinic receptor subtypes, M1, M2, and M3 located on medium and large bronchial smooth muscle.20,38 When activated, M1 and M3 receptors cause airway smooth muscle contraction and increased mucus secretion. Therefore, an anticholinergic agent blocks these effects and bronchodilation occurs. M2 receptors located on postganglionic parasympathetic nerves inhibit acetylcholine release which is beneficial. These agents also decrease tracheobronchial secretions increasing the risk of mucous plugging; however, this has not been a problem clinically. 38
Ipratropium's and oxitropium's onset of action is 15-20 minutes which is slower than the short acting B2 agonists and duration of effect is 6 hours. 1 Patients should be made aware of a delayed onset if these agents are being used as needed. They have been shown to improve lung function similar to short acting B2 agonists with less systemic adverse effects. 1 They increase exercise performance, decrease symptoms, and improve quality of life.39–43 Ipratropium also improves arterial oxygen saturation during sleep and improves sleep quality. 44 All of the inhaled anticholinergic agents are well tolerated. 1 Dry mouth is the most common complaint.
Tiotropium is a long-acting anticholinergic agent with an onset of 30 minutes, peak effect in 3 hours, and duration of 24 hours.1,45,46 One advantage of tiotropium over ipratropium is that it dissociates slowly from Ml and M3 receptors allowing it to be dosed daily. At the same time, it dissociates quickly from M2 receptors. Tiotropium improves lung function, dyspnea, and quality of life and decreases the frequency of exacerbations when compared to placebo and ipratropium. 46 When compared to salmeterol, tiotropium improved FEV1, dyspnea, and quality of life significantly. 46 Tiotropium has also been shown to reduce hyperinflation during exercise and at rest increasing endurance time. It also increases oxygen saturation during sleep. 46
The UPLIFT trial studied the effects of tiotropium compared to placebo over 4 years in 5,993 patients. 11 Patients were allowed to continue on concomitant COPD therapy except short acting anticholinergics. Mean improvements in both pre- and post-bronchodilator FEV1 were significant as compared to placebo. Mean St. George's Respiratory Questionnaire (SGRQ), measuring quality of life, significantly improved in the tiotropium group as compared to placebo; however the improvement was not considered clinically significant. Tiotropium reduced exacerbations, hospitalizations, and respiratory failure. It did not reduce the rate of decline of FEV1. Although mortality in the intention to treat analysis was not statistically different between tiotropium versus placebo groups (Hazard Ratio (HR) 0.89; 95% confidence interval (CI) 0.79-1.02), those patients receiving tiotropium and following protocol during 4 years, showed significantly reduced mortality as compared to placebo (14.4% versus 16.3%; HR 0.87, 95% CI 0.76-0.99). For this reason, in addition to all its other benefits tiotropium is the anticholinergic of choice for maintenance therapy.
Inhaled corticosteroids
In patients with severe to very severe disease who have a history of repeated exacerbations (≥ 2 in 1 year), guidelines recommend adding inhaled corticosteroids (ICS) to a patient's regimen which should already include a long acting bronchodilator.1,9 Oral corticosteroids should only be used during acute exacerbations. ICS reduce capillary permeability decreasing mucus, inhibiting release of proteolytic enzymes from leukocytes, and inhibiting prostaglandins. 47 They decrease the frequency of exacerbations and improve health status.48–51 Withdrawal from them has been associated with an increased recurrence of exacerbations and deterioration of health.52,53 Hoarseness, sore throat, oral candidiasis, and skin bruising are the most common adverse effects. 1 Higher dose therapy increases the risk of systemic side effects including dose-based risk of fractures. Some data shows ICS may increase the risk of pneumonia.53–56
Combination therapy
Guidelines recommend combination therapy in COPD patients with moderate to severe disease when one long acting bronchodilator is no longer controlling patient symptoms.1,9 Rationally, combination therapy with agents with different mechanisms of action makes sense and there is data to show benefit.
Beta2 agonists + anticholinergic agents
The combination of B2 agonists and anticholinergic agents offer bronchodilation through two different mechanisms of action. Additionally, there is some evidence that sympathetic activity is more prominent during the day and parasympathetic activity is more prominent at night. 57 Therefore, the combination of drug classes would provide maximum effect over the entire 24 hours. The combination of short acting anticholinergics and LABAs show improvements in trough FEV1 and decrease rates of exacerbations and symptoms as compared to either agent alone.58–64
Tiotropium has been studied in combination with formoterol, arformoterol, and salmeterol. Overall, data shows combinations of the LABAs with tiotro-pium significantly improves dyspnea scores, decreases need for rescue medication, and improves quality of life.65–73 Based on this data, when one long acting bronchodilator is not adequately controlling patient symptoms, addition of another agent with a different mechanism of action is warranted.
Beta2 agonists + inhaled corticosteroids
Combination therapy with salmeterol/fluticasone and formoterol/budesonide have shown significant improvements in lung function including FEV1, health status, and symptoms. There is also a reduction in rescue inhaler use and the frequency of exacerbations as compared to either placebo or either agent alone.74,75 The TORCH study is one of the largest trials showing benefit of using the combination of LABA and inhaled corticosteroids. 54 This landmark trial enrolled over 6,000 patients to placebo, salmeterol 50 mcg twice daily, fluticasone 500 mcg twice daily or the combination of salmeterol/fluticasone. Combination therapy showed an absolute risk reduction for death of 2.6% when compared to placebo (HR 0.825; 95% CI 0.681-1.002, P = 0.052). This is a 17% relative reduction in mortality over three years as compared with placebo (P = 0.052). The combination therapy also reduced the annual rate of exacerbations by 25% (P < 0.001 versus placebo). Hospitalizations were 17% lower in the combination group than placebo (P ≤ 0.03). SGRQ total scores also increased in all three treatment groups as compared to placebo; however the improvement was not considered clinically significant. Over three years, the mean baseline FEV1 in the combination group increased 29 mL and the placebo group decreased 62 mL. This difference of 92 mL was statistically significant when comparing the combination group to placebo, salmeterol alone or fluticasone alone. Pneumonia was more likely to occur with fluticasone therapy–-the probability was 19.6% in the combination group, 12.3% in placebo group, and 18.3% in fluticasone group (P < 0.001 for both combination versus placebo and fluticasone versus placebo). Although, mortality difference was not statistically significant, the reduction was notable.
Triple therapy
Although triple therapy with a LABA, an inhaled corticosteroid, and a long acting anticholinergic has been used in clinical practice for many years, there has not been data showing benefits until recently. Current data suggests triple therapy may be the most effective way to treat moderate to very severe COPD.
Aaron et al conducted a multicenter, randomized, double-blind, placebo-controlled trial comparing tiotropium 18 mcg daily plus placebo (n = 156), tiotropium 18 mcg daily plus salmeterol 50 mcg twice daily (n = 148), and tiotropium 18 mcg daily plus fluticasone-salmeterol (FSC) 500/50 mcg twice daily (n = 145) over one year. 76 Over the course of the study significantly less patients withdrew from the triple therapy group (26%) as compared to tiotropium (47%) and tiotropium plus salmeterol groups (43%, P < 0.001 for all). No difference was seen in the proportion of patients who experienced an exacerbation within 52 weeks and the rate of exacerbations per patient year. The time to first exacerbation was 130 days in the tiotropium group, 128 days in the tiotropium plus salmeterol group, and 217 days in the triple therapy group. The results were not statistically significant; however some may consider a difference of 3 months clinically significant. Triple therapy showed lower rates of severe exacerbations requiring hospitalization (incidence rate ratio for exacerbations was 0.53; 95% CI, 0.33-0.86, P < 0.01) and all cause hospitalization (P = 0.04) than those treated with tiotropium alone. Health related quality as measured by SGRQ improved in the tiotropium plus salmeterol and the triple therapy groups as compared to tiotropium alone. SGRQ scores decreased by 4.5 points in the tiotropium alone, 6.3 points in the tiotropium plus salmeterol group (P = 0.02 versus tiotropium alone), and 8.6 points in the triple therapy group (P = 0.01 versus tiotropium alone). These improvements were considered clinically significant. Significant improvements in prebronchodilator FEV1 was seen in the triple therapy group (86 mL) versus the tiotropium plus placebo group (27 mL) (P = 0.049). No difference was seen in dyspnea scores and adverse events including death.
Singh et al conducted a double-blind, placebo-controlled crossover study evaluating the effects of SFC plus tiotropium (n = 31) to SFC alone (n = 37) and tiotropium alone (n = 31) in patients with moderate to severe COPD. 77 Treatment lasted 14 days with a 2 week washout period between treatments. Doses were the same as in the previous trial by Aaron et al. Day 14 AUC0-4 hr sGaw was significantly higher with triple therapy compared to tiotropium (22%) or SFC alone (27%) (both P < 0.001). On day 14, mean FEV1 at trough was significantly improved with triple therapy compared to tiotropium alone (212 mL, P < 0.001), 2 hours (198 mL, P < 0.001), and 4 hours (251 mL, P < 0.001). Mean FEV1 at trough with triple therapy was significantly improved over SFC alone (110 ml, P = 0.017) and 4 hours after dose (97 mL, P = 0.023). There was a significant difference in dyspnea score as measured by the Transitional Dyspnea Index (TDI) at day 14 between triple therapy and tiotropium (2.2 units, P < 0.001) and this was considered clinically significant. Less rescue medication was used in the triple therapy group versus tiotropium alone group (1 less occasion over 24 hours, P < 0.001) and SFC alone (0.6 less occasion over 24 hours, P = 0.013).
Welte and colleagues conducted a triple therapy study that differed from the other trials by enrolling patients with severe to very severe disease and using budesonide/formoterol instead of FSC. 78 In this randomized, placebo-controlled, double-blind, multicenter trial, patients received tiotropium 18 mcg daily plus placebo (n = 331) or tiotropium 18 mcg daily plus budesonide/formoterol 320/9 mcg twice daily (n = 329) for 12 weeks. Predose FEV1 was 65 mL higher in the triple therapy group compared to tiotropium alone (P < 0.001). Postdose FEV1 was 123 mL higher at 5 minutes and 131 ml higher at 60 minutes in the triple therapy group compared to tiotropium alone (P < 0.001 for both). This improvement was maintained over the 12 weeks of treatment. SGRQ total score was increased in the triple therapy group by 3.8 units versus 1.5 units in the tiotropium group (P = 0.023). Clinically significant improvements (>4 units) occurred in 49.5% in the triple therapy group compared to 40% in the tiotropium group alone (P = 0.016). Triple therapy also significantly decreased the inhalations of rescue medication needed per day, the rate of severe exacerbations, number of hospital/emergency room visits, time to first severe exacerbation, and time to first hospital/emergency room visit compared to tiotropium alone (P < 0.05 for all). All symptom scores (breathlessness, nighttime awakening, chest tightness and cough) improved significantly in the triple therapy group as compared to tiotropium alone (P < 0.001 for all). Adverse effects were similar between groups.
The most recent data published regarding triple therapy showed mortality benefit with triple therapy. 12 Short and colleague conducted a retrospective cohort using a database of COPD patients in Scotland. 12 Their trial was a retrospective cohort using a database of COPD patients in Scotland. COPD patients were separated into those receiving triple therapy (n = 1857) and those receiving dual therapy with inhaled corti-costeroids + LABAs (n = 996). The mean FEV1 was 50.8% in the triple therapy group and 62.7% in the dual therapy group. Mean follow-up was 4.65 years. The hazard ratio was adjusted for confounders including history of respiratory and cardiovascular disease, diabetes mellitus, and smoking pack years, age at diagnosis, sex, and deprivation index. Authors discovered a reduction in all cause mortality in the triple therapy group as compared to the dual therapy group. The adjusted hazard ratio was 0.65 (95% CI 0.57-0.75); a 35% reduction in the triple therapy group. Triple therapy also reduced death from respiratory (HR 0.70; 95% CI, 0.57-0.84) and cardiovascular (HR 0.49; 95% CI, 0.33-0.73) causes. COPD exacerbations were identified through prescription data looking for short courses of oral corticosteroids. Triple therapy decreased the rate of exacerbations based on this data (HR 0.71; 95% CI, 0.63-0.80). In addition, hospital admissions due to respiratory disease were also decreased (HR 0.85; 95% CI 0.73-0.99). Clearly this mortality data has limitations based on study design, but it does suggest if triple therapy was evaluated in a prospective, randomized trial for a longer period of time, mortality benefit may be seen.
Based on the data available regarding triple therapy, it improves lung function, decreases symptoms including dyspnea, reduces rescue medication, improves quality of life, decreases exacerbations and hospitalization, and may decrease mortality compared to single and dual therapy.12,76–78 In addition, it does not appear to increase adverse effects over using single or dual therapy. Data supports the common practice of prescribing triple therapy and it should be considered in those patients not controlled with dual therapy alone. Future triple therapy studies need to be longer duration to verify mortality benefit and the question remains whether triple therapy should be started during earlier stages of disease.
New and prospective therapy
Ultra-long acting beta2 agonists
There are several ultra-long acting B2 agonists with a duration of action > 24 hours including carmoterol, olodaterol, vilanterol, and the recently approved indacaterol. 79 In addition, to easier dosing and increased patient adherence, recent data shows that the duration of bronchodilation effects clinical efficacy and outcomes. One theory is that the longer duration of action allows peripheral airways to stay open because of more consistent drug levels. 80 Area under the curve (AUC) over 24 hours for FEV1 and FVC are maximized because of decrease in the number of trough concentrations per day. The longer airways stay open, trapped air is emptied reducing hyperinflation and helping breathing mechanics.
Indacaterol is a partial B2 agonist, a weak B1 agonist and a full B3 receptor agonist. It has a similar binding affinity to the B2 receptor as formoterol and higher intrinsic activity than salmeterol. 81 Within 5 minutes, the drug takes effect and this is attributed to its faster membrane permeation compared to agents with a slower onset such as salmeterol. Indacaterol has been compared to placebo, salmeterol, formoterol, and tiotropium.
In the INLIGHT 1 and INDORSE studies, indacaterol was compared to placebo.81,82 Results showed indacaterol increased trough FEV1 and decreased need for rescue inhalers and the rate of exacerbations. Efficacy was not lost over 52 weeks. There was a higher incidence of cough in the indacterol group within 5 minutes of administration. The cough was not associated with bronchospasm and did not lead to study withdrawal.
Indacaterol has also been compared to salmeterol and formoterol. INSIST and INLIGHT-2 compared inacaterol 150 mcg daily to salmeterol 50 mcg twice daily in patients with moderate to severe COPD.83,84 INSIST, a 12 week multicenter, randomized, controlled trial, examined the efficacy and safety of indacaterol (n = 560) versus salmeterol (n = 563). 83 A trough FEV1 and FEV1 AUC difference of 50 mL between treatment groups was considered clinically significant. Indacaterol increased FEV1 AUC(5 minutes to 11 hours and 45 minutes) by 57 mL over salmeterol (P < 0.001). Based on the authors’ definition, this difference was clinically significant. Trough FEV1 was higher in the indacaterol group at week 12 with a mean difference of 60 mL (P < 0.001). Indacaterol also significantly improved TDI score as compared to salmeterol with a statistical but not clinically significant mean difference of 0.63 points (95% CI 0.30-0.97). More patients did have clinically significant improvements in the indacaterol group compared to the salmeterol group (69.4% versus 62.7%) with an odds ratio of 1.41 (95% CI 1.07-1.85, P < 0.05). Patients in the indacaterol group also used fewer puffs per day of rescue medication (mean difference of -0.18, P < 0.05) and experienced a greater percentage of days with no rescue medication use (mean difference 4.4, P < 0.05). Adverse effects were similar between groups.
The INLIGHT-2 trial was similarly designed to the INSIST trial; however this trial lasted 6 months and had a placebo arm. 84 A difference in trough FEV1 of 120 mL between indacaterol and placebo was considered clinically significant. Trough FEV1 on day 2, week 12, and week 26 was significantly increased (P < 0.001) in both the indacaterol (n = 333) and salmeterol (n = 334) groups versus placebo (n = 335). Trough FEV1 at week 12 increased from baseline in the indacaterol group by 150 mL and salmeterol group by 90 mL. The placebo group decreased trough FEV1 by 30 mL over the same period of time. Trough FEV1 was significantly greater at weeks 12 (60 mL) and 26 (70 mL) in the indacaterol group compared to salmeterol (both P < 0.001). The SGRQ total score was significantly lower in the indacaterol and salmeterol groups at weeks 4, 8, 12, and 26 as compared to placebo (all P < 0.01). Indacaterol improved SGRQ score by a clinically significant amount at weeks 8, 12 and 26. Salmeterol reached clinical significance by weeks 12 and 26. The only time point indacaterol was statistically better than salmeterol in this measurement was at week 12 (P < 0.05). Over 26 weeks, both indacaterol and salmeterol decreased puffs per day of rescue inhaler, days without rescue inhaler use, change in morning and evening peak expiratory flow, nights without awakenings, and days with no daytime symptoms compared to placebo (all P < 0.05). In addition, indacaterol increased the days able to perform usual activities (P < 0.05) as compared to placebo while salmeterol did not. Indacaterol decreased the days without rescue therapy, change in morning and evening peak expiratory flow, and days able to perform usual activities significantly (P < 0.05) as compared to salmeterol. More patients in the indacaterol group experienced cough following inhalation of (17.6%) compared to salmeterol (0.9%) and placebo (2.5%). This was not associated with bronchospasm or discontinuation rates.
The INTEGRAL trial was a multicenter, placebo-controlled, crossover study evaluating indacaterol 300 mcg daily (n = 61) to salmeterol 50 mcg twice daily (n = 59), and placebo (n = 61) in moderate to severe COPD over 14 days. 85 At 14 days, indacaterol increased trough FEV1 by 200 mL over placebo (P < 0.001) and 90 mL over salmeterol (P = 0.011). Salmeterol also increased trough FEV1 significantly more than placebo (P = 0.001). In addition, serial measurements of FEV1 over 24 hours on day 1 and day 14 were taken. Indacaterol and salmeterol increased FEV1 greater than placebo all time points (P < 0.05 for all). Indacaterol increased FEV1 statistically over salmeterol for the first 12 hours post dose and again at 23 hours (P < 0.05). Adverse events were similar between groups.
INVOLVE was a randomized placebo-controlled trial comparing the efficacy of indacaterol 300 mcg daily (n = 437), 600 mcg daily (n = 425), formoterol 12 mcg twice daily (n = 434) and placebo (n = 432) in moderate to severe COPD patients over one year. 86 At 12 weeks, trough FEV1 for both doses of indacaterol was 170 mL higher than placebo (P < 0.001) and 100 mL higher than formoterol (P < 0.001). This difference was maintained between indacaterol and formoterol and indacaterol and placebo until week 52. Formoterol showed significant increases in trough FEV1 when compared to placebo on both day one and week 52 (P < 0.05). Both indacaterol doses and formoterol were statistically superior to placebo when comparing change in TDI scores at week 12 and 52, percent days of poor COPD control, decrease in mean daily inhalations of rescue medication, days without rescue medication, percent nights with no awakenings, and change from morning and evening peak expiratory flow rates (P < 0.05 for all). Both doses of indacaterol decreased the mean daily inhalations of rescue medications and days without rescue medications as compared to formoterol (P < 0.05). SGRQ scores improved in all treatment groups as compared to placebo at weeks 12 and 52. Indacaterol 300 mcg and formoterol showed clinical significant change at week 52 and indacaterol 600 mcg show clinical significant change at both 12 and 52 weeks. Both indacaterol and formoterol increased the time to first exacerbation and reduced the rate as compared to placebo. The BODE index which includes a dyspnea scale and a 6 minute walk test changed significantly in favor of both doses of indacaterol and formoterol as compared to placebo at weeks 12 and 52 (P < 0.05 at all points). Adverse events were similar between groups.
Indacaterol has been compared to tiotropium in three trials. INTIME was a randomized, double-blind, placebo controlled, crossover study comparing indacaterol 150 mcg daily, indacaterol 300 mcg daily, tiotropium 18 mcg daily, and placebo in moderate to severe COPD. 87 Patients (n = 169) were randomized to receive three of the four treatments over 14 days with a 14 day washout between crossover. A non-inferiority comparison was done between indacaterol and tiotropium. Non-inferiority margin was considered a difference between treatments of <55 mL in trough FEV1. Clinical importance was considered a change of at least 120 mL in trough FEV1 when compared to placebo. At 14 days, trough FEV1 statistically and clinically improved for both doses of indacaterol as compared to placebo. Indacaterol 150 mcg increased trough FEV1 from baseline by 170 mL and indacaterol 300 mcg increased by 150 mL. Indacaterol was also found to meet the criteria for noninferiority with tiotropium. Indacaterol 150 mcg increased trough FEV1 by 40 mL higher than tiotropium and indacaterol 300 mcg increased trough FEV1 by 30 mL over tiotropium. Both doses of indacaterol had a faster onset of action on day 1 as compared to tiotropium. FEV1 at 5 minutes post indacaterol 150 mcg dose increased FEV1 by 120 mL, indacaterol 300 mcg by 130 mL, and tiotropium by 50 mL (P < 0.001 for each indacaterol dose as compared to tiotropium). Adverse effects were similar between groups.
INHANCE compared indacaterol 150 mcg daily (n = 416) and indacaterol 300 mcg daily (n = 416) to tiotropium 18 mcg daily (n = 415) and placebo (n = 418) over 26 weeks in patients with moderate to severe COPD. 88 The indacaterol and placebo arms were blinded, but tiotropium was open-label. At week 12, the difference in trough FEV1 was significantly improved in both indacaterol groups (180 mL for both groups) and tiotropium groups (140 mL) over placebo (P < 0.001 for all versus placebo). In addition, both doses of indacaterol were considered non-inferior to tiotropium (P < 0.001). The TDI score significantly increased at weeks 4, 8, 12, and 26 in both indacaterol groups as compared to placebo (P < 0.001 at all points). TDI score increased significantly in the tiotropium group at weeks 4, 12, and 26 versus placebo (P < 0.05 at all three times). TDI scores were significantly better in the indacaterol 300 mcg group at week 4, 8, and 12 when compared to tiotropium (P < 0.05 for all). In addition, the proportion of patients experiencing clinically significant improvement in TDI score (≥ 1 point) was greater in the indacaterol 300 mcg group as compared to tiotropium (P < 0.01) group. SGRQ scores improved significantly in both indacaterol groups as compared to placebo (P < 0.001 for both). Patients needed less rescue medication with all treatment groups as compared to placebo (P < 0.05) and was lower in the indacaterol group as compared to tiotropium (P ≤ 0.001 for both indacaterol doses). Similar to other trials, more patients in the indacaterol groups experienced cough after inhalation (16.6-1.3%) as compared to tiotropium (0.8%) and placebo (2.4%).
The third trial comparing indacaterol to tiotro-pium was a multicenter, double-blind, placebo-controlled noninferiority study completed by Buhl et al. 89 Patients with moderate to severe COPD were randomized to indacaterol 150 mcg daily (n = 794) or tiotropium 18 mcg daily (799) for 12 weeks. The trough FEV1s at week 12 were essentially the same in each group (1.44 L indacaterol versus 1.43 L tiotropium). Indacaterol was considered non-inferior to tiotropium. Trough FEV1 increased from baseline by 130 mL in the indacaterol group compared to 120 mL in the tiotropium group. TDI total scores were statistically and clinically improved in the indacaterol group as compared to tiotropium (2.01 versus 1.42, P < 0.001). Patients in the indacaterol group were more likely to achieve clinically significant improvements (≥4 points) in SGRQ scores compared with tiotropium (odds ratio 1.43, P < 0.001). Adverse effects were similar between groups.
Overall, indacaterol is more effective at improving lung function as compared to the shorter acting beta-2 agonists, salmeterol and formoterol and is at least as effective as tiotropium in increasing trough FEV1.81–90 Indacaterol did not consistently effect exacerbations, but most trials had shorter durations than would be necessary to see significant differences. When data from four trials were compiled, indacaterol 150 mcg and 300 mcg improved dyspnea both statistically and clinically over salmeterol, formoterol, and open-label tiotropium. 91 Indacaterol statistically improved SGRQ scores over placebo but not over salmeterol, formoterol, or open-label tiotropium. Coughing after inhalation was the main adverse effect, but this was of short duration and was not associated with bronchospasm or withdrawal. Based on the data available and the advantages of once daily dosing and ability to dose titrate with quick onset of action and its beneficial effects on hyperinflation, it appears that when a patient needs a maintenance B2 agonist in moderate to severe COPD, indacaterol is a good option. More studies need to be done comparing indacaterol long term to tiotropium. In addition, the role of indacaterol needs to be evaluated in combination with other agents.
Phosphodiesterase-4 (PDE4) inhibitors
Phosphodiesterases are enzymes that inactivate cAMP and cGMP.92,93 There are eleven phosphodiesterase isoenzymes identified and they are widely distributed in different tissues and inflammatory cells. Theophylline is a nonspecific PDE inhibitor. This nonspecificity is a major cause of the its associated adverse effects. The hope is that by focusing therapy on inhibition of specific PDE enzymes, particularly PDE4 efficacy can be maximized and adverse effects are limited. 94 All immune and inflammatory cells, except platelets express PDE4 and it is the main enzyme in airway smooth muscle and pulmonary nerves. PDE4 inhibitors increase intracellular levels of cAMP by preventing inactivation by PDE enzymes. As a result, there is inhibition of numerous pro-inflammatory mediators (Table 3). By impacting these mediators, PDE4 inhibitors should improve airway narrowing and limit damage from inflammation. In addition to reducing inflammatory mediator release, they improve pulmonary circulation and inhibit lung vasculature remodeling. 95
Consequences of PDE4 inhibition on cells and mediators involved in COPD.
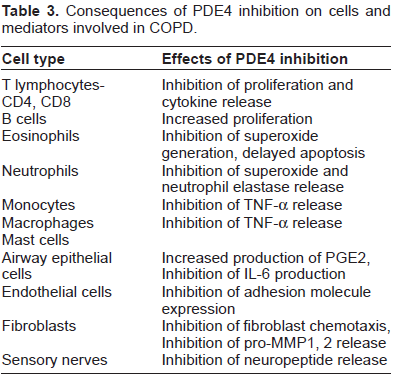
Rolipram, cilomilast, and roflumilast are selective PDE-4 inhibitors. 94 Rolipram has a high-affinity for rolipram binding sites (HARBS) and exhibits more adverse effects such as nausea and emesis compared to those agents with a low-affinity for rolipram binding sites (LARBS), such as cilomilast and roflumilast. LARBS are thought to have more anti-inflammatory effects and less adverse effects.
There are many PDE4 variants encoded by four genes PDE4 A, B, C, and D. Cilomilast selectively inhibits PDE4D. 93 Trials evaluating its efficacy have shown some improvements in FEV1 and FVC, health status, and exacerbations; however, there are safety concerns related to the medication.96,97 The most common adverse effects were nausea, diarrhea, abdominal pain, and dyspepsia. These usually occur within the first few weeks of therapy. The main concern with cilomilast was arteritis occurring in rats. There is a relatively narrow margin for safety when dosing the drug since arteritis is related to dose. Based on this animal data, the dose used clinically in humans is close to that shown to cause toxicity. The dose-effect relationship is steep and small increase in doses increase the risk of arteritis greatly. In 2003, the FDA reviewed the drug for approval in the United States and based on safety concerns and the marginal efficacy data, the FDA asked for more studies to be completed prior to final approval.
Roflumilast is an oral agent recently approved in Europe, Canada, and the United States. In the US, it is indicated to reduce the risk of COPD exacerbations in patients with severe disease associated with chronic bronchitis and a history of exacerbations at the recommended dose of 500 mcg daily. 98 Roflumilast and its active metabolite are highly selective for PDE4 and inhibit all PDE4 variants, unlike cilomilast which selectively inhibits PDE4D. 92 Roflumilast reaches peak concentrations in 1 hour and has a mean elimination half-life of 17 hours. It is 80% bioavailable and widely distributed in tissues and highly protein bound (99%). The active metabolite accounts for 90% of the medication's action. The metabolite's Cmax is one to two times higher than roflumilast and has both a longer tmax between 4-12 hours and an elimination half-life of 27 hours. In addition, the AUC of the metabolite is 10 times higher than the parent drug. Based on the pharmacokinetics of the metabolite, roflumilast needs to be dosed once daily.
There are four published trials comparing roflumilast to placebo. All have shown roflumilast to improve FEV1 significantly and two have shown a significant decrease in exacerbations per year. Rabe et al conducted a multicenter, double-blind, randomized, placebo-controlled trial comparing roflumilast 250 mcg (n = 576), roflumilast 500 mcg (n = 555), or placebo (n = 280) give once daily for 24 weeks to patients with moderate to severe COPD. 99 Both doses of roflumilast significantly increased postbronchodilator FEV1 from baseline and as compared to placebo at all visits during the 24 weeks (P < 0.05 for all). At 24 weeks, FEV1 improved by 74 mL in the roflumilast 250 mcg group and 97 mL in the 500 mcg group as compared to placebo. Prebronchodilator FEV1 also improved over baseline for both doses of roflumilast.
The 250 mcg dose increased FEV1 by 64 ml and the 500 mcg dose increased it by 88 ml. SGRQ score improved significantly from baseline in all three treatment groups, although the change in scores were not clinically significant. Exacerbations were lower in the treatment arms compared to placebo (P = 0.0029, one sided for both). The mean number of exacerbations was 1.13, 1.03, and 0.75 per patient/year in the placebo, 250, and 500 mcg groups respectively. There was a 34% lower rate of exacerbations in the 500 mcg group compared to placebo. More withdrawals occurred in the treatment arms of this trial and was highest in the roflumilast 500 mcg group. As expected, the most common adverse effect of roflumilast was diarrhea which was most common in first four weeks and mild to moderate.
Calverley and colleagues conducted a similar double-blind, randomized, placebo controlled trial, but this trial enrolled severe to very severe COPD patients. 100 Patients were randomized to roflumilast 500 mcg orally daily or placebo for 1 year. Inhaled corticosteroids and short-acting anticholinergics were allowed at a constant dose if the patients were using them prior to the study. Roflumilast improved postbronchodilator FEV1 from baseline and was maintained from week 4 to week 52. At 52 weeks, postbronchodilator FEV1 was 39 mL higher in the roflumilast group as compared to placebo (P = 0.001). Authors believe that continued use of corticosteroids and severity of COPD may have caused the lower increase in FEV1 with roflumilast compared to the Rabe trial. The overall rate of exacerbations was not different between groups; however a post hoc analysis showed patients with very severe disease in the roflumilast group experienced less exacerbations a year compared to those in the placebo group, 1.01 versus 1.59 exacerbations/patient/year respectively (P = 0.024). By week 12, SGRQ total score improved significantly in both groups compared to baseline. However, there was no difference in the change between groups and the changes were not clinically significant. Withdrawals for adverse events occurred more frequently in the treatment group (OR 1.5, P = 0.002). Most common adverse effects related to roflumilast were diarrhea, nausea, and headache.
Caverley also reported the findings of two additional studies comparing roflumilast to placebo. 101 The studies were identical and results were published in one paper. Patients with severe to very severe COPD were randomized to either roflumilast 500 mcg daily or placebo for 52 weeks. These patients must have had cough and sputum scores greater than 14 in the week before randomization. Patients could continue LABAs or short acting anticholinergics. No corticosteroids were allowed in these trials. Pooled data showed the mean prebronchodilator FEV1 increased by 48 mL more in the treatment arm as compared to placebo (P < 0.0001). Pooled data also showed an increase in postbronchodilator FEV1 by 55 mL more in the treatment group compared to placebo. The rate of moderate or severe exacerbations decreased significantly in the treatment groups. Pooled data showed an exacerbation rate of 1.14 exacerbations per patient per year compared to 1.37 in the placebo group (RR 0.83; 95% CI 0.75-0.92, P = 0.0003). This benefit is caused by a significant decrease in moderate exacerbations in the treatment group (RR 0.83; 95% CI 0.75-0.92, P = 0.0007). No difference was seen in severe exacerbations. The median time to first exacerbation was only significantly different with pooled data (HR 0.89; 95% CI 0.80-0.98, P = 0.0185). Median time to second exacerbation was significantly better in the treatment group (HR 0.79; 95% CI 0.69-0.91, P = 0.0014). TDI score was also significantly improved in the treatment group compared to placebo; however the difference was not clinically significant.
There are two other published studies examining the effects of roflumilast used in combination with bronchodilators. 102 These results were also published in one paper. Both trials were double-blinded, placebo-controlled, multicenter trials enrolling patients with moderate to severe COPD for 24 weeks. The first trial, M2-127, randomized patients to roflumilast plus salmeterol (n = 466) or salmeterol plus placebo (n = 467). No inhaled corticosteroids, short acting anticholinergic drugs, other long acting bronchodilator drugs, theophylline, or other respiratory drugs were allowed. At 24 weeks, the roflumilast group had greater improvements in prebronchodilator (49 mL, P < 0.0001) and postbronchodilator FEV1 (60 mL, P < 0.0001) as compared to the placebo group. Pre-and postbronchodilator FVC also improved more than in the placebo group by 47 mL (P = 0.0128) and 58 mL (P = 0.0028), respectively. The rate of mild, moderate, or severe exacerbations was not different between groups. The median time to first moderate or severe exacerbation was significantly longer in the roflumilast group (HR 0.6, P = 0.0067). The proportion of patients with a moderate or severe exacerbation or a mild, moderate or severe exacerbation was significantly less in the roflumilast group (P < 0.05 for both). There was no difference seen in the change in puffs per day of rescue medication or dyspnea scores between groups. Diarrhea, nausea and weight loss occurred significantly more in the roflumilast group. Mean weight loss in the roflumilast group was 2 kg over 24 weeks. There were significantly more withdrawals in the roflumilast group (77/466 versus 45/467, P = 0.0019) due to adverse events.
In the second trial, M2-128, patients were randomized to roflumilast plus tiotropium (n = 371) or tiotropium plus placebo (n = 372). 102 Participants met the same inclusion criteria as in M2-127, but they also had to have chronic cough and sputum production, and frequent use of as needed short acting B2 agonists (at least 28 puffs per week) while they were being treated with tiotropium for at least 3 months before enrollment. This trial showed that tiotropium plus roflumilast increased prebronchodilator FEV1 by 80 mL and post-bronchodilator FEV1 by 81 mL over tiotropium alone (both P < 0.0001). Pre- and postbronchodilator FVCs also increased significantly between groups. Patients in the roflumilast group increased prebronchodilator FVC 95 mL (P = 0.0001) and postbronchodilator FVC by 101 mL (P = 0.0004) more than tiotropium alone. The only effect roflumilast appeared to have on exacerbations in this trial was a significant increase in the median time to any exacerbation (HR 0.7, P = 0.0264). As opposed to the M2-127 trial, roflumilast plus tiotropium significantly improved TDI scores over tiotropium alone, but the difference was not clinically significant. A significant difference was also seen in the SGRQ score between the groups (P = 0.0051). The roflumilast group also used less puffs per day of rescue medications compared to the tiotropium alone group (-1.56 versus -1.05, P = 0.0004). Diarrhea, nausea, and weight loss occurred more in the roflumilast group. Mean weight loss in the roflumilast group was 1.8 kg over 24 weeks. More patients withdrew from the trial in the roflumilast group, but the difference was not statistically significant.
Recently, ameta-analysis was published that included twenty-three randomized controlled trials examining the efficacy of roflumilast (nine trials, n = 9211) and cilomilast (14 trials, n = 6457) in patients for up to one year. 103 The trials included in the meta-analysis enrolled patients with mild to very-severe severe disease. Primary outcomes included change in lung function from baseline including FEV1, FVC, or PEF and quality of life as measured by SGRQ. Secondary outcomes included incidence of COPD exacerbations, symptoms including dyspnea, exercise tolerance, and adverse effects. The changes in FEV1 from baseline were statistically significant for both the subgroup patients with GOLD stage I or stage II disease, as well as a subgroup including patients with stage III or IV disease. Regardless of COPD severity and concomitant COPD therapy, FEV1 improved significantly when compared to placebo (45.6 mL; 95% CI 39.2-22.0). FVC increased significantly from baseline (82.7 mL; 95% CI 66.1-99.2, P < 0.00001) and PEF increased by 6.1 L/min (95% CI 3.45-8.75) as compared to controls. The SGRQ total score improved significantly compared to controls by -1.04 points (95%CI -1.66 to -0.41); however this change is not considered clinically significant. PDE4 inhibitors decreased the numbers of patients experiencing exacerbations (OR 0.75; 95% CI 0.72-0.85, P < 0.00001). No significant benefit was seen in symptoms or exercise tolerance. Diarrhea, nausea, headache, vomiting, dyspepsia, abdominal pain, and weight loss occurred significantly more often with PDE4 inhibitor therapy as compared to control.
Roflumilast is effective in increasing FEV1 when used alone or in combination with salmeterol or tiotropium.99–103 It also appears to impact exacerbation rates positively and decrease rescue medication use. To date it remains unclear the role it has in overall therapy. Indications state it should be used to decrease exacerbations and in the United States indication is limited to chronic bronchitis patients. Where it fits into current therapy is uncertain. Current guidelines recommend using for its approved indication; however, additional studies will need to determine its place in combination therapy. Specifically, should it be added to patients on triple therapy or should it be used to spare corticosteroid use in patients with frequent exacerbations. In addition, the gastrointestinal side effects occur frequently and patient acceptability is an issue. It should be remembered cilomilast caused mesenteric arteritis in animals. Although this does not appear to be as big a concern with roflumilast, prescribers should still be vigilant as the drug becomes more widely used. It should be noted that package labeling includes a psychiatric warning regarding the risk of insomnia, anxiety, depression, and suicidal thoughts that needs to be taken into consideration. 98 There are other PDE4 inhibitors in development including those administered via inhalation.
Long-acting muscarinic antagonists
Aclininium bromide and glycopyrronium bromide are two long acting muscarinic antagonists (LAMAs) currently being studied in Phase III clinical trials for use in COPD. Aclininium, an inhaled agent, is a M2 and M3 receptor antagonist. 104 It is epuipotent to tiotropium and 8-16 times more potent than ipratropium. It has a half-life of 29 hours and therefore can be dosed daily. Aclininium's duration of effect on the M2 receptor is 3 times shorter than tiotropium and this is an advantage. Drug that reaches systemic circulation is rapidly hydrolyzed in the plasma by the enzyme butyryl-cholinesterase into an alcohol and a carboxylic acid metabolite. Both are inactive. Aclidinium significantly improves FEV1 and decreases hyperinflation.105,106 It increases endurance time, improves TDI and SGRQ total scores as compared to placebo.107–109 Early studies have shown aclidinium achieves spirometic results similar to tiotropium. 110 Head ache and dry mouth are the most common adverse effects.
Glycopyrronium bromide is an inhaled LAMA with an onset of action of 5 minutes and duration of 24 hours.111,112 Studies have shown improved FEV1, TDI and SGRQ total scores, and exercise endurance.113–117 It prolonged the time to first exacerbation and decreased rescue inhaler use and hospitalizations. Improvements and FEV1 are similar to tiotropium. 118 Adverse effects were similar to placebo.
LAMAs could potentially replace tiotropium in the future; however, data shows significant effects of tiotropium alone and combination.11,12,45,46,65–72,76–78 This includes data out to 4 years and a potential to decrease the decline in FEV1 and mortality in COPD patients.
Future therapies
There are a number of other therapies currently being studied to treat COPD including other new LABAs and PDE4 inhibitors. Expect to see a number of new combination products such as, LABAs with ICS and LABAs with long-acting anticholinergic or LAMAs. Elastase inhibitors are in phase II and III trials. Other medications in early stages of development include an IL1β antagonist, a CXCR2 and CXCR1/CSCR2 antagonist, and a p38 MAP kinase inhibitor. There are multitude of products being developed for COPD such as inflammatory mediator antagonists and anti-proteases as well as medication aimed at fibrosis and mucous hypersecretion prevention and alveolar repair (Table 4). Medications need to be developed that will decrease the decline of lung function and slow progression of disease, reverse damage, and ideally impact mortality.
Prospective agents in the treatment of COPD.

In the future, therapy will be studied based on disease phenotypes where patients are classified into subgroups based on clinical presentation, physiologic and radiologic changes, frequency of exacerbations, and systemic inflammation; however these phenotypes still need to be defined. 119 It is believed that certain therapies or combinations of medications may be more effective in some phenotypes as opposed to others. In addition, biomarkers must be identified to potentially detect modifiable risks and potentially serve a role in monitoring therapy. 120 Personalized medicine will also play a role in the future treatment of COPD whether it be to identify risk factors, take preventive measures, or identify medications and doses specific to the patients based on pharmacogenomics.
Conclusion
COPD is so complex and patients present in so many different ways, it is difficult to define best therapy and follow a guideline for similar treatment of all COPD patients. In addition, differences in study protocols make it difficult to compare data between trials. Regimens will need to be individualized until studies determine best therapy based on disease phenotype and it should be remembered that therapy does not need to greatly impact FEV1 to be considered efficacious.
COPD guidelines recommend a step-wise approach to therapy depending on severity of disease. Data clearly shows that combinations of products result in improvements in spirometry parameters more than monotherapy. In addition, symptom improvement, quality of life, exacerbations, and hospitalizations are all impacted with combination therapy. Data also is beginning to show that combination therapy may decrease the rate of decline in FEV1 and decrease mortality.
Based on the data presented, once as needed B2 agonists are no longer sufficient, tiotropium with an ultra-long acting B2 agonist is the combination of choice. Guidelines recommend adding inhaled corticosteroids when patients experience frequent exacerbations; however, triple therapy appears to be beneficial in moderate disease and appears to give the best patient outcomes. Therefore, once a patient presents with moderate disease triple therapy should be implemented. It is felt the benefits of this regimen greatly outweigh the risks.
What role roflumilast has is also unclear. Labeling suggests it should be used in patients with chronic bronchitis with exacerbations when there is a desire to avoid ICS; keeping in mind outcomes may not be as good as with triple therapy with a LABA, long acting anticholinergic and ICS. Roflumilast has been used in studies in combination with inhaled corticosteroids and data shows addition of roflumilast would be warranted in a patient who progresses to severe disease while on triple therapy and/or has moderate disease with frequent exacerbations. Until new agents are proved effective in a clearly defined clinical phenotype, newly approved agents should not replace triple therapy as the backbone of COPD treatment.
Author Contributions
Conceived and designed the experiments: TS. Analysed the data: TS. Wrote the first draft of the manuscript: TS. Contributed to the writing of the manuscript: TS. Agree with manuscript results and conclusions: TS. Jointly developed the structure and arguments for the paper: TS. Made critical revisions and approved final version: TS. All authors reviewed and approved of the final manuscript. Provenance: the authors were invited to submit this paper.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
