Abstract
Cefazolin sodium is a first-generation cephalosporin antibiotic and has been used worldwide since the early 1970s. It is used for the treatment of bacterial infections in various organs, such as the respiratory tract, skin and skin structure, genital tract, urinary tract, biliary tract, and bone and joint infections. It has also been used for septicemia due to susceptible gram-positive cocci (except Enterococcus), some gram-negative bacilli including E. coli, Proteus, and Klebsiella may be susceptible, and for perioperative prophylaxis. After the introduction of penicillins and other cephalosporins, occasional outbreaks of methicillin-resistant Staphylococcus aureus were noted. As a result, vancomycin use was increased; however, very recently and most alarmingly, vancomycin-resistant strains have been described. In this setting, to avoid the risk of the development of vancomycin-resistant strains further, vancomycin use should be curtailed. In consideration of this historical background, the appropriate use of antibiotics, such as dosage, dosage intervals, and the duration of administration is required not only for the protection of patients’ health but also for the prevention of the development of drug resistance. Cefazolin has been used in clinical practice for about 40 years, and a large body of evidence has been accumulated, and its efficacy and safety are well established compared with other antibiotics. Therefore, cefazolin has been chosen as a first-line anti-microbial for prophylaxis after various surgical procedures, including cardiovascular surgery, hysterectomy, arthroplasty and so on. Based on these facts, especially for the prophylaxis of surgical site infections, the first-generation cephalosporin, cefazolin, is now being “re-visited”.
Introduction
Cephalosporin is a member of beta-lactam antibodies and are commonly grouped into four generations based on the spectrum of activity against aerobic and gram-negative bacilli. Cefazolin sodium (Fig. 1, CAS number 25939-19-9) is a first-generation cephalosporin antibiotic, a derivative of 7-amino-cephalosporanic acid, and is used worldwide (AnceP and Kefzol® in the US and Canada, Kefzol® in the UK, Gramaxin® and Elzogran® in Germany, Cefamezin® in Holland and Japan, and Cefacidal® in France) since the early 1970s. 1 It is used for the treatment of bacterial infections in various organs, such as the respiratory tract, skin and skin structure, genital tract, urinary tract, biliary tract, and bone and joint infections. It has also been used for septicemia due to susceptible gram-positive cocci (except Enterococcus) and against some gram-negative bacilli, including E. coli, Proteus, and Klebsiella, which may be susceptible.2,3
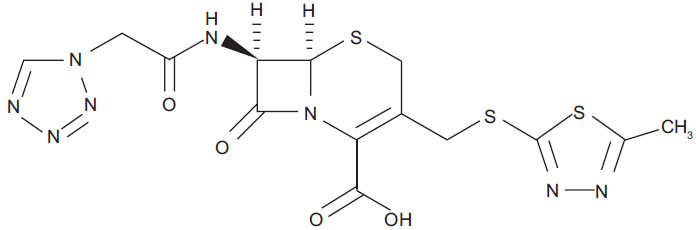
Chemical structure of cefazolin, (C14H13O4N8S3Na; molecular weight, 476.3).
After the introduction of penicillins and other cephalosporins, β-lactamase-producing staphylococci became more prevalent, and occasional outbreaks of methicillin-resistant Staphylococcus aureus (MRSA) were noted. As a result, vancomycin use increased; however, very recently and most alarmingly, vancomycin-resistant strains, suchasvancomycin-resistant Enterococcus (VRE) and the vancomycin-resistant MRSA, which had acquired the enterococcal vanA gene, have been described in the US. 4 In addition, several findings suggested that MRSA might be emerging as a community-acquired pathogen, perhaps following the historic precedent of penicillin-resistant S. aureus. 5 In this setting, to avoid the risk of the development of further vancomycin-resistant strains, vancomycin use should be curtailed and cefazolin has again become a focus as a first-line antibiotic for prophylaxis after surgical operations. In fact, several clinical studies proved that there are no superiority in vancomycin administration as surgical prophylaxis compared with cefazolin use.6–9 In this review, the pharmacokinetics, pharmacodynamics, and clinical use of cefazolin, especially for prophylaxis following surgical operations, are summarized.
Review of the Mode of Action, Pharmacokinetics and Pharmacodynamics of Cefazolin
Mode of action
Cefazolin inhibits bacterial cell wall synthesis by binding to one or more of the penicillin-binding proteins, which in turn inhibits the final transpeptidation step of peptidoglycan synthesis in bacterial cell walls, thus inhibiting cell-wall biosynthesis. Bacteria eventually lyze due to the ongoing activity of cell-wall autolytic enzymes (autolysins and murein hydrolases) while cell-wall assembly is arrested.10,11
Antibacterial spectrum
Cefazolin is active against a wide range of bacteria, including the gram-positive cocci (except Enterococcus) and some gram-negative bacilli, such as E. coli, Proteus, and Klebsiella.1,3 However, there is a high prevalence of β-lactam resistance among gram-positive cocci. A representative strain, MRSA, was first reported in the UK in 1961 and spread worldwide in the early 1960s.12,13 Furthermore, soon after the emergence of MRSA, it was realized that the issue of methicillin resistance involved not only S. aureus but also coagulase-negative staphylococci (CNS). 14 Although there are marked geographic variations, there are high prevalence rates of S aureus or CNS that are methicillin resistant. 15 Thus, because cefazolin can no longer act against all of the gram-positive cocci sufficiently, physicians must choose appropriate antibiotics in consideration of the proportion of methicillin-resistant organism in each institution.
Concerning the other first cephalosporins such as Cefradine, Cefalotin, Cefapirin, which are now available for intravenous administration, there is no apparent difference in antibacterial spectrum between Cefazolin and these. With regard to the other generation of cephalosporins, they are defined according to their spectrum against gram-negative cocci. Each newer generation of cephalosporins has significantly greater gram-negative antimicrobial properties than preceding generation, in most cases with decreased activity against gram-positive bacteria. 16 However, fourth-generation cephalosporins are extended-spectrum drugs with similar activity against the gram-positive organisms as first generation of cephalosporins.
Pharmacokinetics
When a single dose of 250 or 500 mg of cefazolin is given intramuscularily, a peak serum concentration of 29.8 or 44.6 μg/ml, respectively, is attained at 1 hour after administration.1,3 The serum half-life of cefazolin was reported to be approximately 2 hours for the intramuscular route.1,17,18 For the intravenous route, it was slightly shorter, approximately 1.5 hours, compared with the intramuscular route (Fig. 2). 1 These are slightly longer than that for other cephalosporins. 19
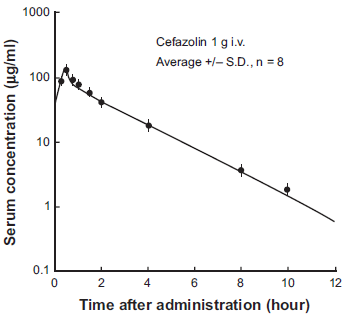
Serum concentration of cefazolin after 1 g intravenous administration in patients with normal renal function.
The distribution volume for cefazolin was 10 to 12 L,17,20,21 approximately, which was smaller than for other cephalosporins.19,22 Cefazolin is widely distributed into most body tissues and fluids including the gallbladder, liver, kidneys, bone, sputum, bile, pleural, and synovial fluid; however, CSF penetration is poor. A total of 74%-86% of cefazolin is bound to serum proteins.3,23 Approximately 90% of cefazolin sodium is excreted in the urine as unchanged drug and is minimally metabolized in the liver.3,24
Recommended dosage and dosage intervals based on pharmacokinetics and pharmacodynamics (PK-PD) concepts
Recently, animal PK-PD infection models have served as a method for antibacterial and dose and dosing interval selection. 25 From the view of this PK-PD concept, the anti-bacterial activity of cefazolin in vitro is dependent on the duration of time that a drug concentration remains above the MIC. 25 Based on this fact, it is important that cefazolin should be administered at appropriate intervals so as not to allow the drug concentration below the MIC. Recommended dose and dosing intervals for patients with normal renal function is 0.5-1 g by intravenous administration at 6 to 8 hours intervals for moderate to severe infections. Furthermore, for severe, life-threatening infections (e.g. endocarditis, septicemia) 1-1.5 g by intravenous administration every 6 hours is recommended. 1
Cefazolin dose and dosing intervals adjustments for the patients under specific conditions
Renal impairment
Because approximately 90% of cefazolin sodium is excreted in the urine, 1 its elimination half-life is prolonged in patients with renal impairment. 18 As shown in Figure 3, the relationship between creatinine clearance and serum half-life is established 18 and Craig et al demonstrated that, when creatinine clearance falls below 20 ml/min, the serum half-life rises dramatically and in severely uremic patients may be extended to as long as 35 hours.18,26 In addition, seizures associated with cefazolin were reported in severe renal insufficiency patients (see adverse effects section). 27 Based on these facts, modification of the administration interval and dosage schedule is required. Prince et al proposed a recommendation of dosing intervals of 8 to 12 hours for mild impairment, 12 to 16 hours for moderate impairment, and 24 to 48 hours for severe impairment. 24 Leroy et al proposed that as long as creatinine clearance remains above 50 ml/min, dose modification is not required. When creatinine clearance is reduced to 10-20 ml/min, intramuscular 500 mg administration at a 24-hour interval is adequate. 18 More precisely, Bergan proposed a dose adjustment as intramuscular 500 mg administration with 8-hour intervals when creatinine clearance is above 60 ml/min, with 12-hour intervals for creatinine clearance of 30-60 ml/min, with 24-hour intervals for creatinine clearance of 15-30 ml/min, and at 36-hour intervals for creatinine clearance of less than 15 ml/min. 17
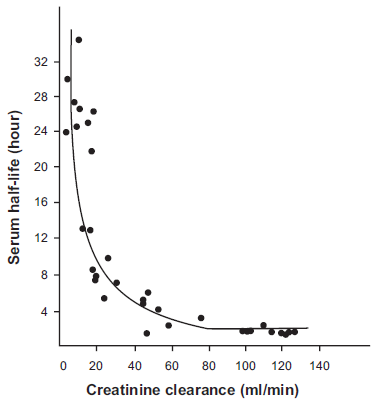
Relationship between renal function (creatinine clearance) and the half-life of cefazolin in serum after a single intramuscular dose.
In hemodialysis patients, Marx et al proposed that approximately 20 mg/kg of cefazolin administered post-hemodialysis appears to be a safe and effective empiric therapy. 28 Sowinski et al proposed that 15 or 20 mg/kg after hemodialysis maintained appropriate serum concentration of cefazolin throughout a 2-3-day interdialytic period. 29
In the recent topic concerning the treatment of dialysis catheter related bacteremia, the guideline proposed by the Infectious Diseases Society of America was published in 2009. 30 Based on the fact that a substantial proportion of staphylococcal infections in dialysis patients are methicillinresistant species, Vancomycin was recommended as an empirical antibiotic therapy in that guideline. 30 However, Cefazolin is still a reasonable choice as a first line therapy in dialysis units with low prelvalence of methicillin-resistant species, 31 because dialysis patients continued on Vancomycin therapy have a 3-fold greater risk of treatment failure than those switched to Cefazolin therapy for the methicillinsusceptible staphylococci. 32
In continuous ambulatory peritoneal dialysis patients, cefazolin is usually administered into the peritoneal cavity by mixing with dialysate. Cefazolin is absorbed well from the peritoneal cavity. 33 When 1 g of cefazolin was left in the peritoneal cavity, serum levels rose to a mean of 63 μg/ml, with most of the rise occurring within 30 min. 33 In addition, cefazolin was very poorly dialyzed out by the intraperitoneal route. After 17 to 24 hours of dialysis with no cefazolin in the fluid, the serum concentration was still 43%-66% of the starting serum concentration.33–35
In addition, concerning the treatment of PD-related peritonitis, the first-line agents recommended by the International Society of Peritoneal Dialysis are the combined intraperitoneal administration of firstgeneration cephalosporins and a ceftazidime in patients with cloudy effluent, without fever and/or severe abdominal pain, and no risk factors for severe infection.36,37 In these recommendations, the dose of cefazolin was 15 mg/kg, which should be administered intraperitoneally once daily, 37 based on the previous pharmacokinetic study by Manley et al. 38
Pediatric dosage
In pediatric patients, a total daily dosage of 25-50 mg per kg of body weight, divided into 3 or 4 equal doses, is effective for most mild to moderately severe infections.1,16,39 However, safety for use in premature infants and in neonates has not been established. 1
Pregnancy
As there are no adequate and well-controlled studies in pregnant women, Cefazolin should be used during pregnancy only if clearly needed. Moreover, its pharmacokinetics is different from nonpregnent adult. Protein binding capacity of Cefazolin becomes lower and the free Cefazolin concentration becomes higher during the pregnancy due to the lower albumin concentration. 40 Furthermore, in addition to the increased plasma volume, the Cefazolin distribution volume is increased (12.04 L) compared with nonpregnant adults (6.9 L). 41 As a result, Cefazolin clearance during the pregnancy (7.44 L) becomes approximately twice that of nonpregnet adults. 41 On the basis of these considerations, although the efficacy has not been evaluated enough yet, Allegaert et al recently proposed that the administered dose should be modified as 2 g of Cefazolin at 6 hours intervals. 41
General Recommendations for Prophylaxis of Surgical Site Infections
Surgical site infection is a major cause of nosocomial infections, and prophylaxis using anti-microbials is important to prevent these infections. Cost-effectiveness, safety, and savings in nursing are required when choosing anti-microbials for prophylaxis. The goal of antimicrobial prophylaxis is to achieve serum and tissue drug levels that exceed, for the duration of the operation, the MICs for the organisms likely to be encountered during the operation. 39 On the basis of these considerations, infusion of the first microbial dose should begin within 60 min before incision.16,39 Concerning the duration of antimicrobial prophylaxis, the majority of evidence showed that prophylaxis after wound close is unnecessary, and there were no beneficial effects to multiple-dose prophylaxis compared with single dose.42–48 There are numerous advantages of the single-dose regimen, including pharmacy costs and reducing the opportunity for errors. In this setting, the American Society of Health-System Pharmacists (ASHP) recommended that prophylaxis within 24 hour might be appropriate.39,44 Concerning anti-microbial dosing, there are limited data on appropriate dosing for prophylaxis. One study of obese patients undergoing gastroplasty demonstrated that blood and tissue levels of cefazolin were consistently below the MICs for prophylaxis against gram-positive and gram-negative organisms in patients who received a 1 g dose preoperatively. 49 This study also demonstrated that 2 g of cefazolin reduced surgical site infection compared with 1 g. 49 Thus, the suggested initial dose of cefazolin for surgical prophylaxis is 1-2 g intravenously (20-30 mg/kg, approximately). In addition, for operations of long duration, anti-microbials should be re-administered at intervals of 1-2 times the half-life of the drug and the recommended a re-dosing interval of cefazolin is 2-5 hours. 39 One study by Scher et al proved that when a colorectal surgery operation lasted more than 3 hours, the infection rate with a single dose of cefazolin was 6.1%, which was significantly higher than that observed with two doses of cefazolin (1.3%). 50 From this fact, the authors recommended that cefazolin should be re-administered at intervals of 3 hours.
Cardiovascular surgery
Deep sternal infection and mediastinitis are reported to occur in 0.25%-2% of patients undergoing cardiac surgery 51 and appropriate prophylaxis has been shown to reduce the incidence. 52 S. aureus and S. epidermidis are the most common culprits in postcardiothoracic surgical site infection, accounting for 34%-54% and 12%-44%, respectively.53–58 Furthermore, another analysis demonstrated that 41.4% of S. aureus and 64% of S. epidermidis isolates were resistant to methicillin. 51 Despite the high prevalence of β-lactam resistance among these organisms, several studies demonstrated that there were no superiorities for the prevention of surgical site infection by glycopeptide administration compared with that by cefazolin.6–9
Concerning less invasive cardiac surgery, such as pacemaker insertion, a meta-analysis suggested that systemic antibiotic prophylaxis significantly reduces the incidence of short-term pocket infection, skin erosion, and septicemia. 59 In particular, one study showed that there was no incidence of infection in a patient given 2 g of intravenous cefazolin before the operation, whereas infection occurred in 12% of untreated control patients. 60 From these findings, cefazolin administration should still be chosen as a standard prophylaxis for cardiac surgery.
Gastric and colonic surgery
For percutaneous endoscopic gastrostomy, several studies revealed that antibiotic prophylaxis significantly reduced the risk for peristomal wound infection associated with this procedure.61,62 The rate of incidence is reported to be between 4.3% and 16%, and another 3%-8% of patients may develop local cellulitis or abscess formation. 63 The superiority of cefazolin for preventing these complications was proved in two studies. Jain et al suggested that a preoperative 1 g dose of cefazolin reduced the incidence of local infection (7.4% for cefazolin versus 32.1% for placebo). 62 In another study, Sturgis et al suggested that a preoperative 1 g dose of cefazolin reduced the incidence of local infection (13.3% for cefazolin versus 19.3% for placebo). 64
In colorectal surgery, it is now generally accepted that antimicrobial prophylaxis is beneficial for the prevention of surgical site infection. 65 However, the choice of anti-microbials is still controversial. There is no convincing evidence to suggest that new-generation cephalosporins are more effective than first-generation cephalosporins. 65 In contrast, De Lalla proposed that cefazolin monotherapy is not recommended for colorectal surgery because of the requirement for anti-anaerobic coverage.66,67 However, combination therapy of intravenous cefazolin and oral metronidazole could be alternative regimen for prophylaxis in elective and clean-contaminated colorectal surgery. 16 On the basis of these considerations, for the surgical prophylaxis of upper gastro-intestinal tract operations, cefazolin monotherapy should be chosen. However, in lower gastro-intestinal tract operations, oral metronidazole administration should be added to cefazolin for anaerobic coverage.
Gynecological surgery
For cesarean operation, the routine use of a single dose of cefazolin is safe and effective in emergency but not elective cesarean sections.16,68 Another study by Sullivan et al showed that administration of prophylactic cefazolin prior to skin incision resulted in a decrease in both endomyometritis and total postcesarean infectious morbidity, compared with administration at the time of cord clamping. 69
In sepsis occurring after hysterectomy, the source of bacteria recovered from infected sites is most commonly from the patient, including cervicovaginal flora, the skin, and the respiratory or gastrointestinal tract. 70 Of the gram-positive organisms found in wound infections, S. aureus is most frequently associated with surgical sepsis. Others organisms include β-hemolytic streptococci and occasionally E. coli, enterococci, and anaerobes. 70 These observations indicate that a regimen of first- or second-generation cephalosporins is efficacious in prophylaxis. With regard to the duration of prophylaxis, long- and shortterm regimens were found to have equal efficacy. In particular, Lett et al proposed that single-dose cefazolin administration was as effective as multiple-doses for prophylaxis in vaginal hysterectomy. 71
Orthopedic surgery
Tang et al analyzed the incidence of wound infection of primary total hip and knee arthroplasties and revealed that approximately 1% of patients developed wound infections. 72 Staphylococci account for approximately 70% of postoperative infections in orthopedic prosthetic surgery, with the leading organism being S. epidermidis. Therefore, the antibiotics most widely used for prophylaxis are cefazolin, cefamandole, and cefuroxime. 73 In addition, Williams et al analyzed bone antibiotic concentrations and revealed that cefazolin and ceforanide presented the highest bone concentrations. 74 Therefore, the antibiotics most widely used for prophylaxis are cefazolin, cefamandole, and cefuroxime, by virtue of their excellent activity against the indicated pathogens. On the basis of these findings for the prophylaxis of surgical site infections in the hip and knee arthroplasty, cefazolin or cefroxine are recommended.16,42–44 However, methicillin-resistant coagulase-positive and -negative staphylococci are increasingly being reported as the causative agents of postoperative infection in clean prosthetic surgery, which has prompted the use of glycopeptide antibiotics (vancomycin and teicoplanin) in prophylaxis for total joint replacement, particularly in hospitals in which there is high level of methicillin-resistance among pathogens. Periti et al demonstrated that a single preoperative dose of teicoplanin ensured adequate surgical antisepsis, with results similar to a standard multiple-dose regimen of cefazolin. 75 In this setting, although cefazolin is still first-line prophylaxis in arthroplasty, physicians must choose glycopeptides in consideration of the proportion of methicillin-resistant organisms in each institution.
Safety and Tolerability
Side-effects from cefazolin are not common. The major and notable adverse effects of cefazolin were summarized as follows.
Allergic reaction
Despite their low prevalence, anaphylaxis, eosinophilia, itching, drug fever, skin rash, and Stevens-Johnson syndrome due to cefazolin administration have been reported. To avoid these fatal reactions, careful review should be made as to whether the patient has had the previous hypersensitivity to not only cefazolin but also other cephalosporins or penicillins. In particular, in penicillin hypersensitive patients, cross-hypersensitivity among β-lactams has been clearly documented and may occur in up to 10% of patients with a history of penicillin hypersensitivity. 1
Pseudomembranous colitis
Pseudomembranous colitis is antibiotic-associated colitis caused by Clostridium difficile, which colonizes the human intestinal tract after the normal gut flora have been altered by antibiotic therapy.76,77 Patients with more than mild clinical manifestations of C. difficile (e.g. diarrhea, abdominal pain or nausea and vomiting) and a positive diagnostic assay should receive antibiotics for treatment for C. difficile. 78 The most commonly administered agents whose use preceded the onset of this disease were cefazolin, trimethoprim-sulfamethoxazole, ampicillin, ticarcillin/clavulanate, and gentamicin. 77 Several reports on cefazolin associated pseudomembranous colitis, even for single-dose administration, were published previously.79–81 Furthermore, C. difficile was detected in 14.3% of patients who received a single 2 g intravenous prophylactic dose of cefazolin, though the patient does not experience diarrhea. 82 The most important step in the treatment of diarrhea induced by C. difficile is cessation of the inciting antibiotic as soon as possible. Standard therapy for C. difficile associated disease consists of oral metronidazole or oral vancomycin. 78
Central nervous system effects
Although less common, cefazolin has been reported to cause encephalopathy and seizures in patients with severe renal insufficiency.27,83–85 In these case reports, cefazolin was administered 3 to 8 g/day for several days despite of severe renal impairment (at least, more than 7.2 mg/dL in serum creatinine level). In two cases, cefazolin concentration measurement revealed 455 and 280 μg/ml, which are about 10-20 times the treatment level. Schwankhaus et al emphasized the possible mechanism that high-dose cefazolin may cause neurotoxicity by competitive inhibition of the active transport of toxic organic compounds from cerebrospinal fluid, which was also observed on an occasion of toxic encephalopathy secondary to high-dose intravenous penicillin administration.83,86 In addition, the protein binding of many drugs may change in renal failure, either by decreases in serum albumin concentrations or displacement by azotemic products. 87 Approximately 86% of cefazolin bound to protein in healthy volunteers, 23 whereas this decreased to 65%-70% in uremic patients,26,88 which resulted in a remarkable increase in the free concentration of cefazolin. Subsequently, highly concentrated free cefazolin facilitated penetration into cerebrospinal fluid. 27 Based on these findings, excessive cefazolin concentration, mainly caused by overdoses compared with renal function might induce seizures, and appropriate dose adjustment is required.
Conclusion
The development of multiple drug resistance microorganisms and the safety of drugs are now major concerns in worldwide. The appropriate use of antibiotics, including dosage, dosage intervals, and the duration of administration, is required not only for the protection of patients’ health but also for the prevention of the development the drug resistance. Because cefazolin has been used for about 40 years in clinical practice, a large body of evidence is available and its efficacy and safety are well established compared with other antibiotics. Most recently, appropriate antibiotic use has been decreasing the incidence of MRSA central line-associated bloodstream infection. 89 Based on these facts, especially for the prophylaxis of surgical site infection, the first-generation cephalosporin, cefazolin, is now being “re-visited”.
Conflict of Interest
The author reports no conflict of interest.
