Abstract
Bone loss and its associated risk of fracture is a serious long-term health issue for breast cancer and prostate cancer survivors. Hormone ablation therapy, in particular aromatase inhibitors (AIs) for breast cancer and androgen deprivation therapy (ADT) for prostate cancer, causes marked reduction in circulating estrogen or testosterone levels, resulting in increased bone resorption, decreased bone mineral density (BMD), and an increased risk of fragility fracture. In several clinical trials with small sample sizes and short follow-up periods, oral and intravenous bisphosphonates have been shown to improve BMD, but not actual fracture rates, in cancer patients on hormone ablation therapy. A number of professional organizations and expert panels recommend the use of bisphosphonates for selected patients at risk. Although bisphosphonates are generally well tolerated, physicians should be aware of safety concerns, including the risk of osteonecrosis of the jaw. With the growing number of older breast and prostate cancer survivors, additional research is needed to characterize patients who would benefit from pharmacotherapy and optimize strategies to prevent cancer treatment-induced bone loss.
Keywords
Introduction
The most commonly diagnosed non-skin cancers among U.S. women and men are breast and prostate, respectively. 1 Many patients of both cancers are over the age of 65 and may have low bone mineral density (BMD), the risk of falls, or any of a multitude of medical problems that increase the risk of fracture. Several therapies commonly used in the treatment of both cancers accentuate the age-related bone loss (cancer treatment-induced bone loss, CTIBL) (Fig. 1). 2 Hormone ablation therapy, in particular aromatase inhibitors (AIs) and androgen deprivation therapy (ADT), improves recurrence-free survival in patients with breast and prostate cancer, respectively. The American Society for Clinical Oncology (ASCO) recommends an AI as adjuvant therapy in postmenopausal women with hormone receptor-positive early breast cancer, either as initial monotherapy or after tamoxifen. 3 Currently, more than 50% of postmenopausal women with hormone-receptor positive breast cancer in the U.S. are being treated with an AI. 3 The role of androgen deprivation, either by orchiectomy or gonadotropin-releasing hormone (GnRH) agonists with or without an antiandrogen, has expanded from the treatment of metastatic prostate cancer to adjuvant therapy for localized disease and for rising prostate-specific antigen (PSA) after local therapy for prostate cancer. Currently, about one-third of the estimated 2 million prostate cancer survivors in the U.S. receive treatment with a GnRH agonist. 4 Although the consequences of fracture attributable to CTIBL have not formally evaluated, inferences for mortality, morbidity, and quality of life can be made from outcome studies in the general population.5,6 For example, about one out of four hip fracture patients dies within a year of injury. One out of five of these who were ambulatory before their hip fracture requires long-term care afterward. It is estimated that each hip fracture costs $33,000 in the first year and $8,000 per year thereafter. Vertebral fracture may be asymptomatic but often cause back pain, height loss, and kyphosis. Since most patients are likely to be long-term survivors after breast and prostate cancer diagnosis, it is of vital importance to manage accelerated bone loss from CTIBL and reduce the fracture risk in their remaining lifetime. The objective of this narrative review is to outline the problem of CTIBL in women and men receiving hormone ablation therapy for breast and prostate cancer and to describe recent findings from clinical trials of the pharmacotherapy, mainly bisphosphonates, for CTIBL.
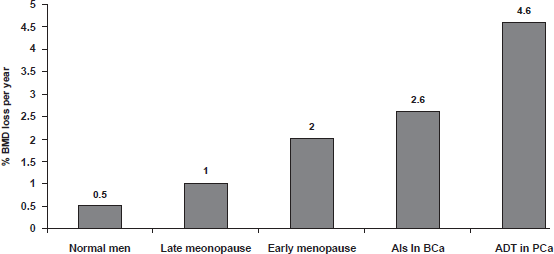
A comparison of cancer treatment-induced bone loss versus normal bone loss in men and women.
Methods
We searched MEDLINE from inception to August 2009 using “Medical Subject Heading” keywords of breast neoplasm or prostate neoplasm, aromatase inhibitor, osteoporosis, and diphosphonates and an original phrase of androgen deprivation therapy. We also identified references cited in relevant articles. Emphasis was placed on methodologically sound articles, particularly reports of randomized, controlled trials and well-designed observational studies. We limited the search to English-language articles.
Cancer Treatment-Induced Bone Loss
Pathophysiology
By potently inhibiting the aromatase enzyme, which converts androgen to estrogen, third generation non-steroidal (anastrozole and letrozole) and steroidal (exemestane) AIs suppress circulating estrogen levels to almost undetectable levels. ADT (i.e. GnRH agonists) decreases serum concentration of testosterone by more than 95% and estrogen by approximately 80%.7,8
In adults, healthy bone is continually being remodeled in the sequence of bone resorption followed by bone formation. Estrogen plays a dominant role in skeletal homeostasis, tightly regulating the bone remodeling process by two cell lineages; osteoblasts (bone forming cells) and osteoclasts (bone resorption cells). 9 Receptor activator of nuclear factor kappa B (RANK) ligand is an essential mediator for osteoclast formation, function, and survival in bone.10–12 RANK ligand is expressed from the osteoblast lineage cells. RANK ligand subsequently binds to its receptor, RANK, on osteoclasts, which leads to maturation of immature osteoclasts to activated osteoclasts. The human body also naturally produces a protein called osteoprotegerin to neutralize the effect of RANK ligand and defend against bone loss. When RANK ligand is bound and neutralized by osteoprotegerin, osteoclasts cannot form, function, and survive. Estrogen inhibits bone resorption by directly suppressing early osteoblastic and osteoclastic precursors through the inhibition of RANK ligand production by osteoblastic precursors.13,14 There is also evidence that estrogen stimulates the production of osteoprotegerin.15–17 Thus, estrogen deficiency shifts the balance of bone remodeling toward increased bone resorption through the upregulation of RANKL and downregulation of osteoprotegerin.18–25 Androgen deficiency increases bone resorption, probably because of decreased aromatization of androgen to estrogen at the tissue level. 26 Androgen levels are positively associated with osteoprotegerin concentrations in a cross-sectional study in healthy men. 27 Androgen replacement appeared to lower osteoprotegerin levels in men made acutely hypogonadal by GnRH agonist and who also received aromatase inhibitor blockade of estrogen production. 28 GnRH agonists also increase parathyroid-mediated osteoclast activation, suggesting that changes in skeletal sensitivity to parathyroid hormone play an additional role in the pathogenesis of ADT-induced bone loss. 29
Epidemiology
Al-induced bone loss in women with breast cancer
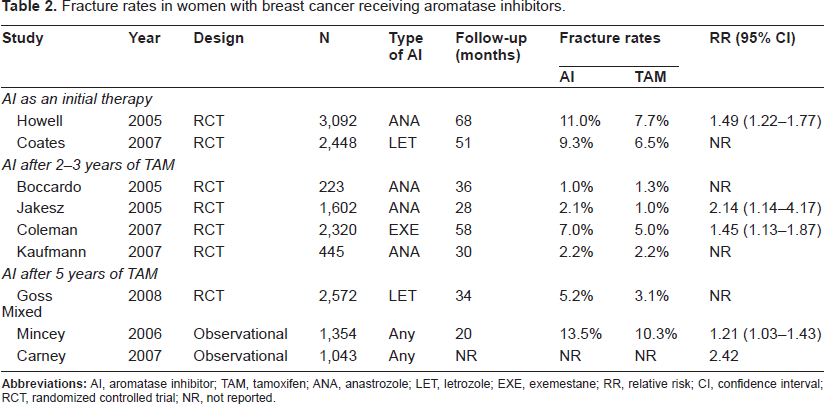
Table 1 outlines the longitudinal studies performed in postmenopausal women with early breast cancer who were treated with AIs.30–36 Compared with the annual 1% bone loss observed in otherwise healthy postmenopausal women, patients receiving AIs for breast cancer experience an average of 2% bone loss per year. Bone loss may also be observed in patients switching between tamoxifen and AIs during adjuvant therapy. Among AIs, the steroidal AI (i.e. exemestane) is structurally related to androstenedione, a compound that has been shown to protect against bone loss. Thus, exemestane was hypothesized to have protective effects on bone metabolism that are distinct from the nonsteroidal AIs. However, comparative studies of AIs showed conflicting results. In a randomized trial of healthy volunteers, no significant differences were observed among the three AIs in their effects on bone metabolism. 37 In contrast, an observational study showed that exemestane was associated with an increase in bone formation markers exceeding the expected increase based on the observed bone resorption increase. 38 The prospective longitudinal substudy of the Arimidex, Tamoxifen, Alone or in Combination trial showed that BMD was reduced at the lumbar spine (6.1%) and total hip (7.2%) at 5 years of therapy. 39 Data beyond 5 years are currently limited. Patients treated with AIs will continue to lose bone at a higher rate or will return to the lower rate noted in healthy postmenopausal women once AIs have been suspended; for example, 3-6 months after withdrawal in patients who received 2 years of adjuvant exemestane, lumbar spine BMD loss tended to return to normal rates and hip BMD stabilized. 40 Whether this BMD loss translates into an increased rate of fragility fractures in patients with breast cancer has not been prospectively evaluated. Nevertheless, the results of a number of clinical trials and retrospective observational studies consistently demonstrated an increased fracture risk in this patient population. Table 2 summarizes the fracture data in patients receiving adjuvant AIs for breast cancer. Data show that the increases in annual fracture rate are similar for all three AIs.41–48 Following 2-3 years of tamoxifen, fracture incidence was significantly higher in patients receiving AIs compared with those who continued to receive tamoxifen. However, these trial results must be placed in the context of their protocol design. Most trials have compared AIs with tamoxifen, which exerts partial estrogen-agonist activity in bone. Therefore, it is impossible to know whether difference in fracture rates observed between AIs and tamoxifen were due to estrogen suppression by AIs, the bone-sparing effect of tamoxifen, or the combination of both. In these trial protocols, the use of screening tests and the administration of antiresorptive agents were at the discretion of treating physicians. These trials may have underestimated the true incidence of fracture because they relied on self-report.
Change in bone mineral density in women with breast cancer receiving aromatase inhibitors.
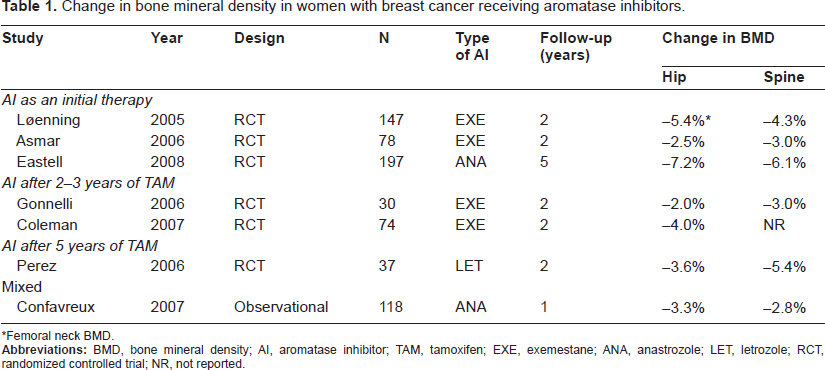
Femoral neck BMD.
Fracture rates in women with breast cancer receiving aromatase inhibitors.
ADT-induced bone loss in men with prostate cancer
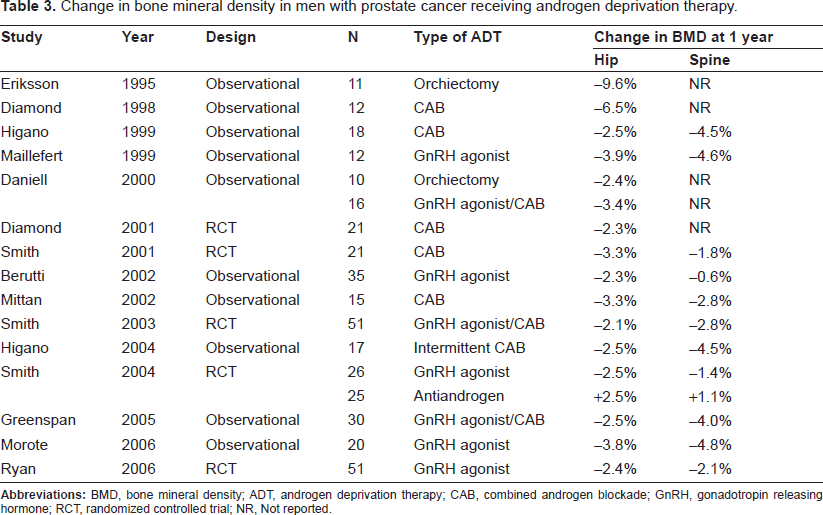
Table 3 outlines the longitudinal studies performed in men with localized prostate cancer or rising PSA who were treated with ADT.49–61 Compared with patients not receiving ADT or healthy controls, men with localized prostate cancer receiving ADT have a 2- to 4-fold higher annual bone loss rate. The rates are similar in men receiving GnRH agonists with or without an antiestrogen. Notably, the accelerated bone loss was observed despite concurrent administration of supplemental calcium and vitamin D, and with careful exclusion of secondary causes of osteoporosis. The duration of ADT may impact BMD significantly. Although longitudinal data beyond 1 year are limited, cross-sectional data demonstrated that spinal and femoral BMD were 1.7 and 5.2% less after 2 years and 14 and 28% less after 10 years of ADT, respectively, compared with age matched controls. 62 The prevalence of osteoporosis also increases with the duration of GnRH agonists administered. 63 Several factors unrelated to BMD may also account for the high fracture rates, including an increased risk of falling due to the physical effects of treatment-related frailty.64,65 Although the relative contribution of each of these factors have not been formally evaluated, treatment-related changes in BMD are sufficient to explain the increase in fracture risk for men with prostate cancer. Table 4 summarizes retrospective cohort studies that examined fracture rates in patients receiving ADT for prostate cancer.54,66–80 The fracture risk increases with the number of doses of GnRH analogues received. 75 Fractures significantly correlates with shorter survival in men with prostate cancer. The medical survival was shorter in men who sustained a fracture than in men who did not (121 versus 140 months, p = 0.04). 81
Change in bone mineral density in men with prostate cancer receiving androgen deprivation therapy.
Fracture rates in men with prostate cancer receiving androgen deprivation therapy.
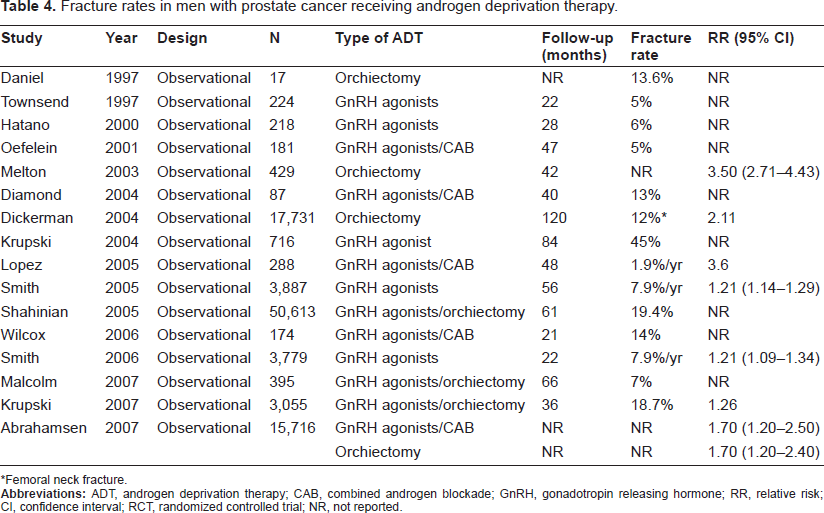
Femoral neck fracture.
Bisphosphonates for Cancer Treatment-Induced Bone Loss
Oral bisphosphonates are widely used in the treatment of postmenopausal osteoporosis. Alendronate, risedronate, and ibandronate have been shown to increase BMD and decrease fracture occurrence in women with established postmenopausal osteoporosis.82–87 Zoledronic acid was recently approved by the U.S. Food and Drug Administration (FDA) in once-yearly or biennial intravenous treatment for osteoporosis in postmenopausal women.88,89 To date, only oral alendronate and risedronate have been shown to reduce actual fracture rates in otherwise healthy men with osteoporosis.90–92 Data on the intermediate BMD endpoint are generally available in patients with breast cancer or prostate cancer. Although loss of BMD is a primary predictor of fracture, alone it provides an incomplete measure of fracture risk. Most studies evaluated the effects of bisphosphonates for one year and was not designed or powered to detect a difference in fracture rates. Whether the positive effects of bisphosphonates on BMD would be maintained for multiple years or correlates with a decreased fracture incidence with time has yet to be determined. Currently, the FDA does not approve bisphosphonates specifically for the treatment for CTIBL.
Mechanism of action, metabolism, and pharmacokinetic profile
Bisphosphonates are synthetic analogs of the endogenous pyrophosphate molecule. At least two types of bisphosphonates exist: non-nitrogen-containing bisphosphonates (clodronate, etidronate) and the more potent nitrogen-containing bisphosphonates (alendronate, ibandronate, pamidronate, risedronate, and zoledronic acid). These drugs are available in oral and intravenous formulations. Bisphosphonates selectively absorb to mineral surfaces of bone that are surrounded by osteoclasts. The drugs are then released from the bone surface, where they are internalized by and inhibit osteoclast activities and reduce bone resorption and turnover, leading to increased bone mass. Bisphosphonates are not metabolized in the liver. About 40% of intravenous dose of zoledronic acid is eliminated unchanged in urine. The remaining 60% binds to bone and is released slowly into the systemic circulation, producing low plasma concentrations. Serum concentration of ibandronate are 2-fold higher in patients with creatinine clearance <30 mL/min. Bisphosphonates stay in bone until remodeling occurs, sometimes for years, and may accumulate with retreatment.
Efficacy research
AI-induced bone loss in women with breast cancer
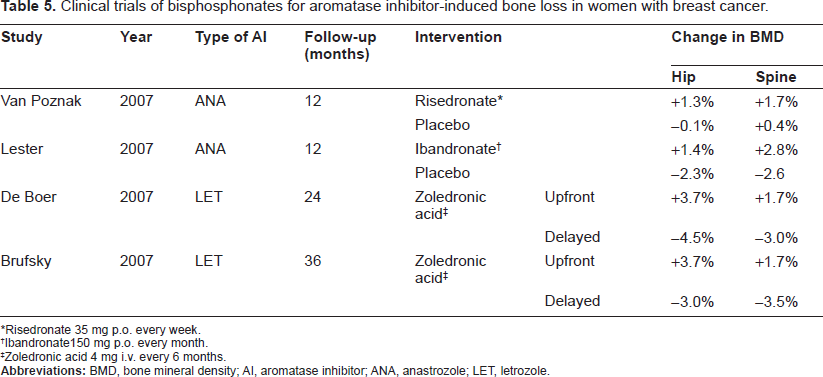
Table 5 summarizes clinical trials of bisphosphonates for the treatment of AI-induced bone loss in postmenopausal women with early hormone receptor-positive breast cancer. There are several ongoing trials with a number of oral bisphosphonates, such as anastrozole and risedronate in the Study of Anastozole with the Bisphosphonates RisedronatE (SABRE), anastrozole and ibandronate in the Arimidex-Bondronat (ARIBON) study.93,94 One-year preliminary results of the double-blinded placebo-controlled part of the SABRE indicated that oral 35 mg risedronate once weekly prevented bone loss in patients with the baseline T-score between -2.0 and -1.0 who are receiving adjuvant anastrozole. In the open-label, non-comparative part of the trial, risedronate also preserved or improved BMD in a cohort of women with the baseline T-score > -1.0 or < -2.0. A one-year analysis of the ARIBON study found that oral 150 mg ibandronate once monthly prevented bone loss in patients with the baseline T-score ≤ -1.0 who are receiving adjuvant anastrozole. These two randomized, double-blinded, placebo-controlled trials suggest that bisphosphonates at the dose and schedule used in postmenopausal osteoporosis are effective in the setting of AI-induced bone loss. The results of several intervention studies with intravenous bisphosphonates have been published recently. The largest of these trials, the Zometa-Femara Adjuvant Synergy Trials (Z-FAST [U.S.] and ZO-FAST [Europe]), are open-label, randomized, multicenter trials to evaluate intravenous 4 mg zoledronic acid every 6 months in patients with the baseline T-score ≥ -2.0 who are receiving adjuvant letrozole.95,96 Patients were randomized to either “upfront” or “delayed” zoledronic acid. The “upfront” group received zoledronic acid regardless of their baseline BMD. The “delayed” group received zoledronic acid when their T-score decreased to -2.0 or in the event of fragility fracture. Both at the end of 36 months in the Z-FAST, and at the end of 24 months in the ZO-FAST, the primary endpoint of changes in lumbar spine and total hip BMD were in favor of the “upfront” strategy. These trials give an insight into the optimal timing of starting bisphosphonates in preventing bone loss and fracture in women receiving AIs for early breast cancer.
Clinical trials of bisphosphonates for aromatase inhibitor-induced bone loss in women with breast cancer.
Risedronate 35 mg p.o. every week.
Ibandronate150 mg p.o. every month.
Zoledronic acid 4 mg i.v. every 6 months.
ADT-induced bone loss in men with prostate cancer
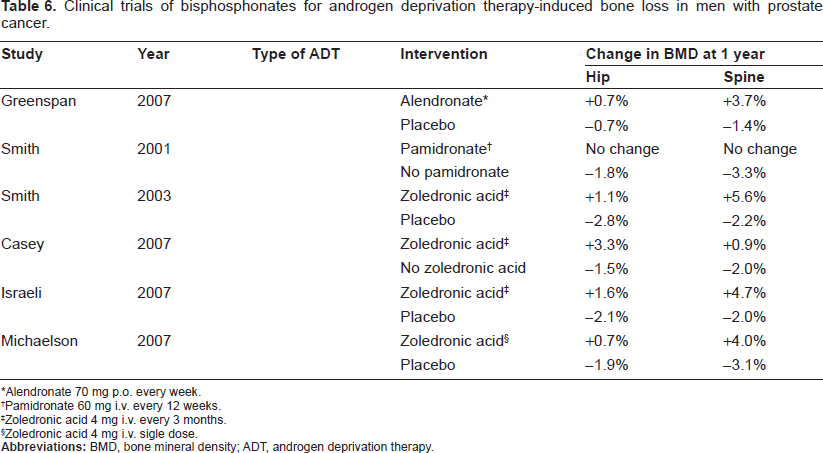
Table 6 summarizes clinical trials of bisphosphonates for the treatment of ADT-induced bone loss in men receiving ADT for localized prostate cancer or the rising PSA. The efficacy of oral 70 mg alendronate once weekly in patients with localized disease recently initiating ADT or receiving ADT for 6 months or greater was evaluated in a randomized, double-blinded, placebo-controlled, partial crossover trial.97,98 After 1 year of therapy alendronate increased lumbar spine and total hip BMD, and patients receiving placebo had a decrease in BMD. In an open label, randomized trial, men with localized disease or rising PSA newly initiating ADT were randomly assigned to ADT alone or ADT in combination with intravenous 60 mg pamidronate every 12 weeks. 55 Men receiving ADT plus pamidronate experienced no changes in lumbar spine and total hip BMD at 1 year, whereas those receiving only ADT experienced a decrease in BMD from baseline. Several randomized, double-blinded, placebo-controlled trials and a open-label, randomized trials have evaluated the intravenous 4 mg zoledronic acid every 3 weeks for ADT-induced bone loss in patients with localized prostate cancer or rising PSA.58,60,99,100 When initiated concurrently or during the first year of ADT, zoledronic acid not only maintained but increased lumbar spine and total hip BMD beyond baseline at 1 year. A recent randomized, double-blinded, placebo-controlled trial of a single, one-time administration of 4 mg zoledronic acid reported 1-year increase in lumbar spine BMD similar to those observed with every 3-momth administration. 101 Of note, all of these trials excluded men with low T-score at baseline.
Clinical trials of bisphosphonates for androgen deprivation therapy-induced bone loss in men with prostate cancer.
Alendronate 70 mg p.o. every week.
Pamidronate 60 mg i.v. every 12 weeks.
Zoledronic acid 4 mg i.v. every 3 months.
Zoledronic acid 4 mg i.v. sigle dose.
Effectiveness research
All of clinical trials reviewed so far concern about the efficacy of bisphosphonates for CITBL. While efficacy is a measure of the ability of a treatment to improve health under ideal (i.e. randomized controlled) settings, effectiveness is a measure of the overall risks and benefits of a treatment in the real world. Estimates of treatment effectiveness are almost always lower than estimates of treatment efficacy. For example, poor tolerability and compliance with oral bisphosphonates has the potential to offset the clinical benefit of these agents. A large study of women receiving oral bisphosphonates for postmenopausal women found that 57% of patients were noncompliant with therapy during the 2-year study period. Noncompliance with oral bisphosphonates resulted in a 37% increased risk of hip and vertebral fractures compared with compliant patients. 102 In one study evaluating persistence on oral bisphosphonates in cancer patients (47% of them had breast cancer), only 35% of patients remained on therapy at month 6. 103 On the other hand, bisphosphonates may have a clinical benefit unrelated to their effect on BMD. Several studies have been initiated to assess whether bisphosphonates reduce cancer recurrence. The Austrian Breast and Colorectal Cancer Study Group trial 12 showed that the addition of zoledronic acid to adjuvant endocrine therapy improved disease-free survival in premenopausal women with breast cancer. 104 The combined analysis of Z/ZO-FAST reported that patients who received the upfront zoledronic acid experienced fewer number of disease recurrence compared with those who received the delayed zoledronic acid. 105 Few studies have evaluated the impact of bisphosphonates on the natural history of prostate cancer. Clodronate, which is not available in the U.S. because of concerns about leukemia, did not improve the bone metastasis-free survival and overall survival in men with locally advanced prostate cancer. 106
Safety considerations
Gastrointestinal disturbances, including heartburn, esophageal irritation, esophagitis, abdominal pain, or diarrhea, have occurred with oral bisphosphonates. Trials reported perforations, ulcerations, and bleeding events from all bisphosphonates except zoledronic acid. The tablets must not be chewed or allowed to dissolve in the mouth, and must be taken with 8 ounces of plain water while in an upright position. For 30 minutes after taking the drug, the patient must not eat or drink anything other than water and must avoid lying down. The incidence of these side effects is very low if proper administration instructions are followed. Despite the controversy surrounding a link between esophageal cancer and oral bisphosphonates, the FDA recommends that bisphosphonates not be used in patients with Barrett's esophagus. 107 In the clinical trial weekly alendronate for men receiving ADT for localized prostate cancer, esophageal symptoms were less frequent than in the placebo group (2% versus 4%, respectively). Severe bone, joint, and muscle pain have been reported with oral alendronate and risedronate. The pain started an average of 91 days (range: 1 day to 52 months) after beginning therapy and remitted in 66% of patients when the drug was discontinued. 108 Pain may become diffuse and disabling in some patients, requiring opioids or other analgesics. In some patients, pain returned after rechallenge. Ocular inflammation with abnormal or blurred vision, ocular pain, conjunctivitis, uveitis, and scleritis has been reported with oral bisphosphonates. In one report, scleritis resolved only after stopping the drug. 109
Intravenous bisphosphonates may be associated with redness and swelling at the injection site. A transient acute-phase reaction including fever, flulike symptoms, headache, arthralgia, and/or myalgia has been reported in approximately 20% of patients receiving intravenous bisphosphonates, most frequently with the first infusion. 110
Renal toxicity has been more common with an 8-mg dose of zoledronic acid and less common with 15-minute infusion times. The combined analysis of Z/ZO-FAST reported only one patient out of the total 827 patients on the “upfront” arm experienced grade 2 renal impairment believed to be related to zoledronic acid; severe renal dysfunction was not reported. 96 Of the more than 217 patients, included in the 5 studies evaluating intravenous pamidronate or zoledronic acid for men receiving ADT for localized prostate cancer, an increase in serum creatinine was reported in only 2 patients receiving zoledronic acid. Nevertheless, ASCO guidelines recommend that creatinine be monitored prior to each dose of pamidronate or zoledronic acid. 111 Unexplained azotemia (an increase of ≥0.5 mg/dL in serum creatinine, or an absolute level of >1.4 mg/dL among patients with normal baseline values) should prompt temporary discontinuation of the bisphosphonate. Hypophosphatemia and asymptomatic hypocalcemia have occurred, requiring regular assessment of serum calcium, phosphate, and magnesium. Patients receiving bisphosphonates should routinely receive calcium and vitamin D supplementation.
Bisphosphonate-associated osteonecrosis of the jaws (BAONJ) is a relatively recently described entity, and has gained attention through the case reporting of both community care givers and academicians.112–114 The condition is characterized by necrotic exposed bone in the maxillofacial region. Symptoms may range from mild to significantly problematic and include pain, drainage, and paresthesia. Recent studies suggest that the rate of BAONJ in non-cancer patients receiving bisphosphonates for bone loss is considerably low, possibly as low as 1 in 100,000 patient-years, compared with those with metastatic cancer receiving high doses of intravenous bisphosphonates. However, available data are based on anecdotal reports, single institution's case series, spontaneous reports to the FDA, or manufacture's data, using inconsistent definitions of the condition. No confirmed cases of BAONJ were reported in 1667 patients included in the combined analysis of Z/ZO-FAST. 107 Of the more than 217 patients included in the 5 studies evaluating intravenous pamidronate or zoledronic acid, no patient had BAONJ. Also, no cases of BAONJ were reported in the alendronate study. The true incidence of BAONJ has yet to be proven by a population-based registry that collects systematic information on patient characteristics, treatments, and outcomes. It is generally advisable to maximize oral hygiene and avoid unnecessary invasive dental procedures before or while receiving bisphosphonates. Management of BAONJ is largely supportive focusing on pain-control and prevention of infection; the use of oral antimicrobial rinses, minimal surgical debridement, and antibiotic therapy if superinfection is present. Recently, concerns have been raised about a potential increased risk for atrial fibrillation in patients treated with either zoledronic acid or alendronate. However, the risk of atrial fibrillation from bisphosphonates is small, if it exists at all.115,116 The FDA recommends that healthcare professionals not alter their prescribing patterns for bisphosphonates and patients not stop taking their bisphosphonates. 117
Safety data on the long-term use of bisphosphonates exists up to 10 years in the general population, but unfortunately limited in patients on hormone ablation therapy for breast or prostate cancer. Another long-term concern with bisphosphonates is that over-suppression of bone remodeling might weaken bone rather than strengthening it. 118 In one report, 9 patients with non-vertebral fracture who had taken alendronate for 3-8 years showed evidence of severe suppression of bone formation on bone biopsy. 119 Additional research is needed to better define safety concerns with long-term administration of bisphosphonates in this patient population.
Clinical Practice Guidelines for Cancer Treatment-Induced Bone Loss
Unless screening tests are performed, CTIBL is not diagnosed until it is complicated by a clinical fracture. For women in the general population, the U.S. Preventive Services Task Force recommends routine dual-energy x-ray absorptiometry (DXA) for those aged 65 and older, or 60 and older if they are at increased risk of osteoporotic fracture. 120 In contrast, clinical practice guidelines for men are less evidence-based. The International Society for Clinical Densitometry and the National Osteoporosis Foundation recommend routine DXA for all men aged 70 and older.121,122 The American College of Physicians recently concluded that DXA screening would not be cost-effective in the U.S. male population younger than 80 years of age.123–125 DXA screening is recommended only for “men who are at increased risk for osteoporosis” and candidates for drug therapy. The guideline identified ADT as an important risk factor for low BMD-mediated fractures. There is a growing concern that current guidelines that rely on BMD may fail to identify patients with increased fracture risk. The World Health Organization Collaborating centre for Metabolic Bone Disease at Sheffield has developed an algorithm to calculate a person's 10-year fracture risk (FRAX). 126 The purpose of FRAX is to help physicians decide which patients with known femoral neck BMD should be given osteoporosis medications. It is a multivariate model that incorporates not only femoral neck bone mineral density (BMD) but also nine clinical risk factors (i.e. age, sex, body mass index, long-term use of oral glucocorticoids, a parent history of hip fracture, a prior fragility fracture, current smoking, high alcohol consumption, and secondary osteoporosis, such as rheumatoid arthritis) to calculate a person's 10-year fracture risk. It was derived from nine cohorts from North America, Europe, Asia, and Australia and validated in 11 independent cohorts with a similar geographic distribution. Currently, FRAX does not consider hormone ablation therapy as a secondary cause of osteoporosis. The external validity of FRAX in cancer patients receiving hormone ablation therapy has yet to be determined. Additional studies are needed to better define an individual's absolute risk of fracture.
Al-induced bone loss in women with breast cancer
The ASCO has suggested an algorithm for the management and treatment of AI-induced bone loss. 111 In patients with a history of breast cancer, postmenopausal women receiving AIs are considered as “high-risk” and recommended to undergo annual DXA assessment of the spine and hip, and receive calcium and vitamin D supplements. Currently, the guideline only recommends bisphosphonates (alendronate, risedronate, and zoledronic acid) for women with osteoporosis (T-score ≤ -2.5). Those with BMD above the T-score threshold for a diagnosis of osteoporosis (T-score > -2.5) are reassured and monitored in an annual basis. Other guidelines suggest a slightly more cautious level for intervention (T-score < -2.0 or the rate of bone loss exceeding 4% per year in women with baseline osteopenia) and the use of bone-targeted treatment in patients with multiple risk factors for fracture regardless of BMD. 127,128 There are no FDA-approved therapies for the treatment or prevention of AI-induced bone loss; however, treatment options are approved for postmenopausal osteoporosis. Oral alendronate 70 mg weekly, oral risedronate 35 mg weekly, one 75 mg tablet taken on 2 consecutive days once a month, or 150 mg once a month, oral ibandronate 150 mg monthly, intravenous ibandronate 3 mg every 3 months, intravenous zoledronic acid 4 mg every 6 months, are all considered appropriate. The dose of zoledronic acid used in postmenopausal osteoporosis is typically 5 mg annually given by intravenous route. The 4 mg dose every 6 months, which has been used to prevent AI-induced bone loss in a clinical trial setting, is considered off-label. Based on Z/ZO-FAST data, the U.K. researchers reported the use of upfront bisphosphonate (i.e. intravenous zoledronic acid) is cost-effective in women receiving AIs for early breast cancer with an estimated incremental cost-effectiveness ratios (ICERs) that ranged from £17,900 to £30,000 per quality-adjusted life-year (QALY) gained. 129 In contrast, a recent cost-effectiveness analysis conducted by the author demonstrated that the use of upfront bisphosphonate (i.e. oral risedronate) is not reasonably cost-effective with an estimated ICER of $195,300 per QALY gained. The study concluded that DXA screening followed by risedronate for those with a T-score ≤ -1.0 (i.e. osteoporosis or osteopenia) is a cost-effective use of resource in this patient population. 130
ADT-induced bone loss in men with prostate cancer
At present, there are no consensus guidelines for the screening and treatment for ADT-induced bone loss in men with prostate cancer. Most men on ADT do not receive adequate screening or therapy aimed at preventing or treating ADT-induced bone loss.131–134 In the absence of consensus guidelines regarding fracture prevention in men receiving ADT, a number of experts have recommended a case-finding approach–-measuring BMD by DXA before starting ADT and administering bisphosphonates to men have low BMD (i.e. osteoporosis).135–139 Others have advocated the upfront use of bisphosphonates regardless of baseline BMD.140,141 No oral or intravenous bisphosphonates has been FDA-approved to prevent or treat ADT-induced bone loss. Treatment options for ADT-induced bone loss may be weekly oral alendronate70 mg, 3-monthly intravenous pamidronate 60 mg, and intravenous zoledronic acid 4 mg one time or every 3 months for up to 1 year. Among them, only alendronate and zoledronic acid (5 mg every 12 months) havebeen FDA-approved for the prevention and treatment of male osteoporosis in general. The use of pamidronate or zoledronic acid for the treatment of ADT-induced bone loss is considered off-label. The author recently conducted a cost-effectiveness analysis of DXA screening followed by alendronate therapy for men starting ADT for localized prostate cancer. 142 The study concluded that DXA screening followed by alendronate for those with a T-score ≤ -1.0 (i.e. osteoporosis or osteopenia) is a cost-effective use of resources. Upfront alendronate therapy without DXA screening was not reasonably cost-effective with an estimated ICER of $316,700 per QALY gained.
Conclusions
Institute of Medicine's recent cancer survivorship report states that the long-term sequelae of a cancer diagnosis and its treatment have largely been ignored. 143 Bone loss and its associated increased risk of fracture, exacerbated by systemic cancer therapy, is a serious long-term health issue for breast and prostate cancer survivors that cannot be ignored. Comparative effectiveness research of antiresorptive agents for more than 1 year is needed to provide recommendations for the most optimal agent and regimen. The long-term results of ongoing clinical trials may provide additional guidance regarding treatment duration, and these treatment guidelines will be updated accordingly.
There has been growing interest in a human monoclonal antibody against RANKL, denosumab. Treatment with denosumab was associated with a 24-month BMD increase in both women receiving AIs for breast cancer and men receiving ADT for prostate cancer.144,145 Denosumab significantly reduced the incidence of new vertebral fractures by 62% and showed a trend toward a positive effect on nonvertebral fractures in men receiving ADT for localized prostate cancer. 146
In addition to pharmacotherapy, we cannot overemphasize the role of multidisciplinary approach.147,148 Optimal prevention and treatment include strategies to minimize bone resorption and to reduce falls, as well as comprehensive geriatric assessment to reduce risk factors, especially calcium and vitamin D supplementation, smoking cessation, moderate alcohol consumption, and weight-bearing exercise. With the growing number of older breast and prostate cancer survivors, efforts should be made to advance closer collaboration between oncologists and geriatricians in patient care. Additional research is needed to improve our understanding of pharmacotherapy against CTIBL (Table 7).
Research agenda regarding bisphosphonates use in cancer patients on hormone ablation therapy.

Disclosure
The author reports no conflicts of interest
