Abstract
Background:
Data on modalities of real-world use of androgen deprivation therapy, such as triptorelin, in men with prostate cancer (PCa) are lacking.
Objectives:
To describe the real-world management of triptorelin treatment over 12 months following treatment initiation in France, and to gain a better understanding of the choices being made about planned total treatment duration, formulation, and route of administration.
Design:
TALISMAN was a prospective, multicenter, longitudinal, non-interventional study.
Methods:
Patients with PCa eligible for triptorelin treatment for at least 12 months were enrolled and followed up for 12 months. The primary objective was to describe the proportion of patients treated continuously with triptorelin during the 12 months following treatment initiation. Planned total duration of treatment, formulation, administration route, and reasons for these choices were evaluated at baseline and at 6 and 12 months. Quality-of-life and safety data were collected.
Results:
Overall, 806 patients were enrolled, of whom 787 patients were included in the analysis. Mean (standard deviation) age was 74.0 (7.9) years, the main circumstance of prescription of triptorelin was a high-risk localized tumor (41.1%). Planned total duration of treatment at baseline was most commonly 24–36 months, and the main reasons for the choice of duration were Gleason score and guideline recommendations. Most patients (72.5%) received the 3-month subcutaneous formulation at baseline; main reasons for choice of formulation and route were physician preference and planned total duration of treatment. Overall, 85.3% (95% confidence interval (82.6, 87.7)) of patients received a continuous 12-month treatment with triptorelin, and 69.1% of patients undertook at least one supportive care pathway during the study period. Patients reported an increase in hormonal treatment-related symptoms and worsening of sexual activity and functioning. There were no new or unexpected safety findings.
Conclusion:
In France, compliance with continuous triptorelin treatment for 12 months is high and prescribing aligns with guideline recommendations.
Trial registration:
NCT04593420.
Plain language summary
Our study looked at how a hormonal treatment called triptorelin is being used to treat men with prostate cancer in France. Triptorelin lowers the patient levels of male sex hormones, which stops the cancer from growing. We looked at how long patients were getting their triptorelin treatment when it was prescribed for at least 12 months and we asked the doctors their modalities of treatment, in planned total duration of treatment and frequency of administration, as well as propositions of supportive care pathway. We also looked at the consequences of triptorelin treatment on patients’ quality of life. Our study included 787 men with prostate cancer who had an average age of 74 years and whose hormonal treatment was planned for at least 12 months. Most of the time, patients had a high risk prostate cancer, and doctors planned to treat their patient with triptorelin for 24 to 36 months, and explained their choice by the aggressiveness, with high risk features, of the cancer and the recommended treatment guidelines for hormonal treatment. Most of the time, doctors chose to give their patient a type of triptorelin that is taken every 3 months, and explained their choice by their personal preference and because it fit with how long they planned to treat their patient for. Most patients (671 out of 787; 85%) had treatment with triptorelin for the full 12 months that we followed them. 544 out of 787 patients (69%) took part in at least one supportive care pathway, like an extra assessment or treatment. When we looked at quality of life, patients said they had an increase in symptoms related to the hormonal treatment as well as sexual decline, both being usual with hormonal treatment like triptorelin. There were no new or unexpected safety findings. Overall, our study showed that most patients with prostate cancer in France are following their prescribed triptorelin treatment plan, and that doctors are prescribing in line with the recommended treatment guidelines.
Introduction
Prostate cancer (PCa) is the most commonly diagnosed cancer in men in France, with 57,357 new cases and 9264 deaths reported in 2022. 1 Treatment options vary depending on the stage of disease and the risk of progression; some cancers have high-risk features with a worse prognosis and are identified as more aggressive. 2 Androgen deprivation therapy (ADT) is widely used as a treatment option for men with high-risk localized, locally advanced, and/or metastatic PCa.3 –5 The aim of ADT is to reduce testosterone levels to <50 ng/dL4,5
The recommended duration of ADT depends on the aggressiveness of the disease, including its stage and high-risk features, and is also influenced by patient characteristics, such as frailty and the incidence and management of ADT side effects. 5 A long duration of ADT is recommended in high risk localized and locally advanced PCa, metastatic PCa, and in castration-resistant PCa, but ADT side effects may lead to shortening of treatment duration 6 or intermittent use of ADT.7,8 There are different types of ADT, including gonadotropin-releasing hormone (GnRH) agonists and GnRH antagonists; GnRH agonists are the most widely used among the ADT options. 9
Despite its widespread use, little is known about the modalities of use of ADT in the real world, particularly the planned total duration of ADT treatment at initiation, the formulation and administration route chosen, and the drivers behind these choices. Moreover, observational studies have highlighted the difficulty of maintaining treatment and follow-up in real life. In the French real-world CHRONO study conducted between 2010 and 2011, among 1438 patients with PCa receiving GnRH agonist treatment, 79.1% completed the 3- or 6-month evaluation and 70.8% completed the 12-month follow-up evaluation, 10 whereas in the real-world DEVENIR study conducted in 2011–2015 in France, 717 of 1301 patients (55.1%) completed the 24-month follow-up after initiating a GnRH agonist. 11
Triptorelin is one of the most frequently prescribed GnRH agonists in France and is available in 1- and 6-month intramuscular formulations, and in 3-month intramuscular and subcutaneous formulations.9 –14 Labeled indications for triptorelin include high-risk localized, locally advanced, and metastatic PCa, all of which have a long duration of ADT treatment (from 2 to 3 years up to lifelong treatment). The efficacy and safety of triptorelin have been demonstrated in clinical trials of patients with PCa, with a 3-, 6-, or 12- follow-up.15 –17 The purpose of the TALISMAN (Treatment of Aggressive prostate cancers in real Life: Initiation, Schedule, and MANagement of triptorelin treatment) study was to describe the real-world management of triptorelin treatment over 12 months following treatment initiation in France and to provide a better understanding of the choices being made about planned total treatment duration, formulation, and route of administration when prescribing triptorelin for patients with PCa and supportive care pathway provided.
Methods
Study design and oversight
TALISMAN (NCT04593420) was a prospective, multicenter, longitudinal, non-interventional study conducted in France from November 2020 to November 2022. Investigators (public and private practice physicians) were selected using the global OneKey® database, which includes all urologists, radiation oncologists, and medical oncologists involved in PCa management in France. Their selection was based on geographical representation within France. The investigators carried out the study without any additional intervention or change in their usual daily practice.
This study complies with the EU Directive 95/46/EC of the European Parliament and the Council of October 24, 1995, on the protection of individuals with regard to the processing of personal data and on the free movement of such data. This study was conducted in compliance with the recommendations of the Declaration of Helsinki, 2013 18 and the International Ethical Guidelines for Epidemiological Studies, Council for International Organizations of Medical Sciences (CIOMS), Geneva 2009. 19 Approval was obtained prior to study initiation from the Comité de Protection des Personnes Est-II (n° 2020-A02302-37, October 2020). This study adhered to all local regulatory requirements applicable to non-interventional studies. Patients were informed about the study, and consent to have their data collected and used was acquired verbally by the investigator prior to study enrollment and data collection.
Patients
Adults (aged ⩾18 years) with histologically confirmed PCa who were eligible for triptorelin treatment (according to the label) for a planned total duration of at least 12 months were enrolled. Investigators were asked to include (if feasible) all consecutive patients until the sample size was reached and/or enrollment was closed. Eligible patients were required to understand and complete a quality of life questionnaire, have been informed about the study, and agree to the use of their data. The decision to initiate triptorelin treatment was made by the investigator before inclusion in the study and was independent from the decision to enroll the patient.
Patients were excluded if they had received hormonal therapy within the previous 6 months, and simultaneous participation in a clinical trial was not permitted. Patients were withdrawn from the study when they stopped triptorelin treatment or if they withdrew their consent, with data collected up to the time of withdrawal.
Treatment
Triptorelin treatment was initiated for PCa with a planned total duration of at least 12 months.
The permitted formulations and administration routes of triptorelin were: Decapeptyl® LP 3 mg, a 28-day sustained-release formulation for monthly intramuscular injection 12 ; Decapeptyl® LP 11.25 mg, 3-month sustained-release formulation for 3-monthly subcutaneous injection or for 3-monthly intramuscular injection 13 ; or Decapeptyl® LP 22.5 mg, 6-month sustained-release formulation for 6-monthly intramuscular injection. 14
Data collection and procedures
Data were collected over 12 months by investigators for all enrolled patients, as part of their routine medical care. This included the collection of data at Visit 1 (V1; baseline visit), Visit 2 (V2; follow-up visit, 6 months), and Visit 3 (V3; end of follow-up, 12 months). If early study discontinuation occurred (i.e., before 12 months of follow-up), the reason for early discontinuation was recorded. The V1 procedures included a review and assessments of demographics, medical history, PCa history, prior PCa treatment, clinical and tumor measures, therapeutic history, supportive care pathway, quality of life, and adverse events (AEs). The V2 procedures included a review and assessments of clinical and tumor measures, therapeutic history, supportive care pathway, quality of life, and AEs. The V3 procedures included the same assessments as V2 and the collection of study discontinuation/completion information.
Quality of life was assessed using the health-related quality of life (HRQoL) European Organisation for Research and Treatment of Cancer (EORTC) Prostate Cancer Module (QLQ-PR25) questionnaire, which is made up of six subscales of urinary symptoms, incontinence aid, bowel symptoms, hormonal treatment-related symptoms, sexual activity, and sexual functioning. 20 All of the scales and single-item measures range in score from 0 to 100, with 100 corresponding to the worst HRQoL for symptom scales and the best HRQoL for sexual activity and sexual functioning scales. Patients recorded their answers to the QLQ-PR25 questionnaire; data were pseudonymized.
This was a non-interventional study; therefore, reporting of AEs and deaths followed regulations related to spontaneous cases. All AEs were collected throughout the study and classified by associated National Cancer Institute Common Toxicity Criteria for Adverse Events (NCI CTCAE version 5.0) worst grade and by drug relationship.
Objectives
The primary objective of the study was to describe the proportion of patients treated continuously with triptorelin during 12 months following treatment initiation.
Secondary objectives included: description of the planned total duration of triptorelin treatment at each visit and the reasons influencing the duration; identification of parameters associated with the planned total duration of triptorelin treatment at each visit; description of the formulation and administration route of prescribed triptorelin at each visit and the reasons influencing the choice at V1; identification of the parameters associated with the choice of formulation and administration route of triptorelin treatment at V1; change from baseline in each of the six subscales of the QLQ‑PR25; and description of the safety of triptorelin by review of AEs in the real-world setting.
Planned subgroup analyses were prespecified in the study protocol and included a description of data by the following subgroups: the circumstance by which patients were prescribed triptorelin treatment, planned total duration of treatment, formulation and route of triptorelin treatment, and specialty of the investigator responsible for inclusion of the patient. However, only two subgroup analyses are reported here (the circumstances of prescription of triptorelin and formulation and route of triptorelin).
Interim analyses of baseline data were planned when 50% of patients were enrolled (these data are not reported here because final data are more relevant, but have been previously presented at congresses and were in accordance with the final results presented here).21 –24
Statistical analyses
In total, 786 patients from 200 centers in France (i.e., four patients per investigator) were planned to be enrolled in this study. The sample size calculation was based on the precision of the proportion of patients treated continuously with triptorelin for 12 months following treatment initiation. An expected proportion of 80% of patients, with a precision of ±3% of the two-sided 95% confidence interval (CI) and taking into account a dropout rate of 15% led to a sample size of 786 patients.
The following analysis sets were included: enrolled population (all patients with informed consent), full analysis set (FAS)/safety population (all patients treated with at least one injection of triptorelin; described as the FAS throughout the rest of this article), and per protocol (PP; all patients in the FAS who had no major protocol violations/deviations and were followed for at least 300 days after V1, or withdrew before V3 because the treatment was stopped but had confirmed at V2 that the treatment was taken continuously).
Statistical analysis was performed using Statistical Analysis System (SAS®) version 9.4 or higher (SAS Institute Inc., Cary, NC, USA). The primary effectiveness analyses were performed using the FAS. The proportion of patients treated continuously with triptorelin for 12 months following treatment initiation was estimated and 95% two-sided Clopper–Pearson CI was calculated. Patients were only considered as “treated continuously with triptorelin during 12 months following treatment initiation” if the investigator confirmed the continuous treatment between V1 and V2 and between V2 and V3. Supportive analyses of the primary efficacy endpoint were based on the PP.
Secondary effectiveness and safety analyses were performed using the FAS. The planned total duration of triptorelin treatment, the primary reason for the choice of planned total triptorelin treatment duration at each visit, formulation and administration route of triptorelin prescribed, and the reasons for the choice of formulation and administration route at V1 were analyzed descriptively. Potential determinants of planned total duration of triptorelin treatment at V1 were identified using a multivariate regression model adjusted for the circumstance of prescription regardless of the significance (see Supplemental Material S1 for list of baseline variables tested). Potential determinants of, separately, the triptorelin formulation at V1 and the triptorelin administration route at V1, were identified using a multivariate regression model (see Supplemental Material S2 for list of baseline variables tested). For all multivariate analyses, all variables were first tested in a univariate model and those that were significant at the 20% level were selected. The correlation between all the selected variables was studied using Cramer’s V coefficient. In case of strong correlation, the variables were retained based on their clinical relevance. The stepwise method was used to determine the final model.
A qualitative description of each item of the QLQ-PR25 questionnaire at V1, V2, and V3 is presented. For the safety analyses, AEs will be described. For subgroups, only significant differences in which 95% CIs did not overlap were highlighted in the results. Quality of life was analyzed descriptively.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 25 The checklist is provided as Supplemental Material S3.
Results
Patient characteristics and baseline parameters
Overall, 806 patients from 126 centers were enrolled in this study; 709 (88.0%) completed the study, 97 patients (12.0%) did not complete the study and were withdrawn, main reasons for withdrawal were death (22.7%), lost to follow-up (21.6%) and other reason (39.2%).
In total, 787 patients (97.6%) were treated with at least one injection of triptorelin and included in the FAS (Figure 1). Of the 123 participating investigators responsible for the inclusion of patients in the FAS, 84 (68%) were urologists, 32 (26%) were radiation oncologists, and 7 (6%) were medical oncologists. Most patients in the FAS were enrolled by urologists (54.9% (432/787)) and radiation oncologists (42.6% (335)); a minority were enrolled by medical oncologists (2.5% (20)).

Patient disposition.
Baseline patient characteristics in the FAS are presented in Table 1. The mean (standard deviation, SD) age was 74.0 (7.9) years, and most patients (94.0%) had a Karnofsky score ⩾80. Overall, 73.0% of patients had at least one comorbidity. The three most commonly reported comorbidities were arterial hypertension (46.5%), dyslipidemia (23.4%), and diabetes (16.7%). At least one concomitant systemic treatment for comorbidities was reported by 65.4% of patients; the most common types of treatments were antihypertensives (46.0%), lipid lowering (22.9%), platelet aggregation inhibitors (18.6%), antidiabetics (14.7%), and anticoagulants (14.0%).
Baseline (Visit 1) patient characteristics—FAS.
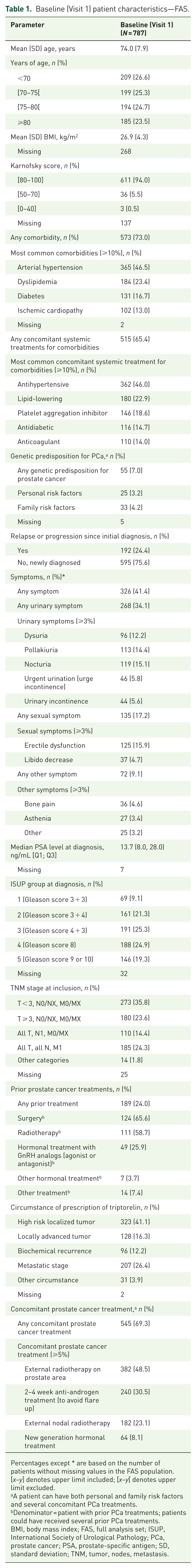
Percentages except * are based on the number of patients without missing values in the FAS population. [x–y] denotes upper limit included; [x–y[ denotes upper limit excluded.
A patient can have both personal and family risk factors and several concomitant PCa treatments.
Denominator = patient with prior PCa treatments; patients could have received several prior PCa treatments.
BMI, body mass index; FAS, full analysis set; ISUP, International Society of Urological Pathology; PCa, prostate cancer; PSA, prostate-specific antigen; SD, standard deviation; TNM, tumor, nodes, metastasis.
A genetic predisposition for PCa, not specified and left to the investigator’s understanding, was reported by the investigator for 7.0% of patients. Overall, 75.6% of PCa were newly diagnosed, while 24.4% of patients had relapsed or progressed since initial diagnosis. At diagnosis, the median prostate-specific antigen (PSA) concentration was 13.65 ng/mL (Q1, Q3: 8.00, 28.00) and most patients had a Gleason score >3 + 4 (International Society of Urological Pathology (ISUP) grade >2). At the time of inclusion, 35.8% of patients were classified as T < 3, N0/NX, M0/MX, 23.6% were T ⩾3, N0/NX, M0/MX, 14.4% had a cancer that had spread to the regional lymph nodes (all T, N1, M0/MX), and 24.3% had metastatic disease (all T, all N, M1).
Previous PCa treatment was reported in 189 patients (24.0%); the most common previous therapies were surgery (124/189; 65.6%), radiotherapy (111/189; 58.7%), and hormonal treatment with GnRH analogs (49/189; 25.9%). Most patients were prescribed triptorelin for treatment of high-risk localized tumors (HRLs; 41.1%), followed by metastatic stage (MS; 26.4%), locally advanced tumors (LAT; 16.3%), biochemical recurrence (12.2%), and other circumstances (3.9%). Most patients (69.3%) received at least one concomitant PCa treatment, mostly radiotherapy (prostate 48.5%, nodal 23.1%).
Primary endpoint: Proportion of patients treated continuously with triptorelin for 12 months
After 12 months, 671 patients (85.3%, 95% CI (82.6, 87.7)) had been treated continuously with triptorelin and 116 patients (14.7%) were not treated continuously; the latter included 45 patients who completed the study and 71 patients who did not complete the study owing to withdrawal. The proportion of patients treated continuously between V1 and V2 was 93.6% (737/787, 95% CI (91.7, 95.2)), and between V2 and V3 was 87.3% (687/787, 95% CI (84.8, 89.5)).
Main reasons for the 71 noncompletions were death (19 patients), lost to follow-up (18 patients), withdrawal by patient’s (3 patients) or investigator’s decision (3 patients), progressive disease (2 patients), and other reasons (26 patients); no patients withdrew owing to AEs. Similar results were reported in the PP.
Secondary endpoints
Planned total duration of triptorelin treatment
At baseline, more than one-third of patients (37.0%) had a planned total duration of triptorelin treatment of 24–36 months, and 25.9% had a planned total duration of life-time treatment (Table 2). The median planned total duration at V1 was 36.0 months (Q1, Q3: 18.0, 72.0).
Duration and prescribed formulation and route of triptorelin treatment and reasons for choice at baseline (Visit 1)—FAS.

[x–y] Denotes upper limit included; [x–y[ denotes upper limit excluded. Percentages are based on the number of patients without missing values in the FAS.
Only one reason could be provided for each patient.
Patients could have several main reasons for choice of formulation and administration route.
AFU, French Urological Association (Association Française d’Urologie); BMI, body mass index; cTN, clinical T and N stage; EAU, European Association of Urology; FAS, full analysis set; ISUP, International Society of Urological Pathology; PCa, prostate cancer; PSA, prostate-specific antigen.
The three most common reasons for the choice of planned total duration of triptorelin treatment at V1 were Gleason score/ISUP group (26.3%), French (French Urological Association), and/or international (European Association of Urology) guidelines (23.2%) and PSA level (15.8%; Table 2).
Following the multivariate analysis, age categories of 75–80 and ⩾80 years showed a statistically significant longer (p = 0.0412 and p < 0.0001, respectively) planned total duration of triptorelin treatment at V1 compared with the age category of 70–75 years. Patients with sexual symptoms showed a statistically significantly shorter (p = 0.0048) planned total duration of triptorelin treatment at V1 compared with those without sexual symptoms. Patients who were prescribed triptorelin treatment for MS or for other circumstances showed a statistically significantly longer (p < 0.0001 and p = 0.0002, respectively) planned total duration of triptorelin treatment at V1 compared with those who received triptorelin for the treatment of HRLs.
Few patients experienced a change in their planned total duration of treatment at V2 (1.6% (12/737)) or V3 (3.1% (21/687)); the new planned total duration was for life at V2 for 4/10 patients and ⩾24 months at V3 for 15/19 patients (Table 3). For the small number of patients who did experience a change in planned total duration, the most common primary reason at V2 was PSA response (50%; 6/12 patients), and at V3 it was tolerability of triptorelin (66.7%; 14/21 patients; Table 3). Following the multivariate analysis, disease evolution was the only parameter identified as a determinant of the change in the planned total duration of triptorelin. Disease progression statistically significantly increased the planned total duration of triptorelin treatment at V2 or V3 from V1 (p = 0.0301, compared with patients with disease control).
Duration and prescribed formulation and route of triptorelin treatment and reasons for the choice at Visit 2 and Visit 3—FAS.

[x–y] Denotes upper limit included; [x–y[ denotes upper limit excluded. Percentages are based on the number of patients without missing values in the FAS.
Questions related to the change of the planned total duration could only be answered for patients treated continuously.
Questions related to the change of triptorelin formulation and route at Visit 2 could only be answered for patients treated continuously with a change of the planned total duration at Visit 2.
Questions related to the change of triptorelin formulation and route at Visit 3 could only be answered for patients treated continuously with a change of the planned total duration at Visit 3.
AFU, French Urological Association (Association Française d’Urologie); EAU, European Association of Urology; FAS, full analysis set; PCa, prostate cancer; PSA, prostate-specific antigen.
Formulation and administration route of prescribed triptorelin
At baseline, 72.5% of patients were prescribed subcutaneous triptorelin every 3 months, with the remaining patients prescribed intramuscular triptorelin either every 6 months (14.6%) or every 3 months (11.8%; Table 2).
The most common reasons for the choice of formulation and administration route were the physicians’ own preference (50.8%), followed by planned total duration of treatment (28.1%), highly evolutive disease (14.6%), and potential impact on compliance (9.8%; Table 2).
For the choice of formulation at V1, multivariate analysis revealed that the specialty of the investigator and QLQ-PR25 sexual activity score were the significant determinants. The 6-month formulation was more frequently prescribed when the prescription was provided by a urologist (p = 0.0001, compared with a radiation oncologist) and in patients with high QLQ-PR25—sexual activity score (p = 0.0151, compared with QLQ-PR25—sexual activity score < median). For the choice of route of administration, multivariate analysis showed that the three significant determinants were anticoagulant treatment, prior PCa treatment, and planned total duration of treatment at V1. Intramuscular route was significantly more frequently prescribed at V1 in patients without anticoagulant treatment (p = 0.0279, compared with those with anticoagulant treatment), in patients with prior PCa treatments (p = 0.0310, compared with those without prior PCa treatment) and in patients with planned total duration of treatment [18–24[, [24–36], and >36 months or for life (p < 0.0129, compared with 12–18 months).
Very few patients had a change of their prescribed formulation or administration route at V2 (16.7% [2/12]) and V3 (4.8% [1/21]); for technical reasons, the question about change of formulation or route of triptorelin was displayed only for patients having a change of their planned total duration at V2 or V3 (Table 3).
Supportive care pathway
Supportive care pathways undertaken by patients were recorded; these included pretreatment and new treatment assessments, visits to a physician (planned or completed), and associated prescriptions or recommendations. In total, 69.1% of patients (544/787) undertook at least one supportive care pathway at V1, V2, or V3.
At V1, most patients (60.1%) undertook at least one supportive care pathway; 25.8% of patients had at least one pretreatment assessment, with lipid profile being the most frequent (20.2%), followed by glycemia (18.6%), and osteodensitometry (12.3%; Table 4). Almost a third (30.2%) of patients had, or had planned, at least one visit to a physician, with cardiologist being the most frequently visited physician (14.7%). Just under half (47.3%) of patients received at least one associated prescription or recommendation, with the most frequent recommendation being physical activity (37.1%), followed by dietary recommendation (29.0%), and vitamin D supplementation (19.7%).
Summary of supportive care pathways at Visit 1, Visit 2, and Visit 3—FAS.

Percentages are based on the number of patients without missing values in the FAS.
Patients could have several treatment assessments, several physicians, associated prescriptions, or recommendations.
FAS, full analysis set; G8, G8 geriatric screening tool; V1, Visit 1; V2, Visit 2; V3, Visit 3.
At V2 and V3, at least one new supportive care pathway was provided to 24.3% and 22.1% of patients, respectively. New treatment assessments were carried out in 6.5% and 9.5% of patients at V2 and V3, respectively. Lipid profile and glycemia assessments were the most common at both visits (V2: 4.0% and 4.0%; V3: 5.6% and 5.3%, respectively; Table 4). New visits to physicians (planned or completed) were reported for 11.6% and 9.2% of patients at V2 and V3, respectively. Ongoing or new associated prescriptions or recommendations were reported for 17.7% of patients at V2 and 16.4% of patients at V3, with the most frequent recommendation at both visits being physical activity (9.0% and 7.9%, respectively).
Quality of life evaluated by EORTC QLQ‑PR25
At V1, the mean (SD) score for the hormonal treatment-related symptoms subscale was 9.79 (10.48), indicating a low level of symptoms (Table 5). This is to be expected given that hormonal study treatment had not started at V1, and prior hormonal treatment in the past 6 months was an exclusion criterion.
Summary of the QLQ-PR25 at Visit 1, Visit 2, and Visit 3—FAS.

Values are presented as mean (SD). All of the scales and single-item measures range in score from 0 to 100. A high score for the urinary, bowel, hormonal treatment-related symptoms scales, and incontinence aid item represents a high level of symptomatology or problems, whereas a high score for the sexual activity and sexual functioning scales represents a high level of functioning. Incontinence aid item is only fulfilled if the patient wears an incontinence aid. The sexual functioning is not applicable if the answer to the question “To what extent were you sexually active?” in sexual activity is “Not at all.”
FAS, full analysis set; QLQ-PR25, Quality of Life Questionnaire—Prostate Cancer Module; SD, standard deviation.
Higher mean (SD) scores for hormonal treatment-related symptoms were reported at V2 (18.44 (14.03)) and V3 (19.07 (15.49)) compared with V1, indicating a deterioration in symptoms. This change was driven mainly by hot flushes, which were reported by 70.4% of patients at V2 and 68.6% at V3, whereas 13.4% reported having hot flushes at V1, with most hot flushes reported as occurring at a low frequency at all visits.
The mean (SD) score for the sexual activity subscale at V1 was 20.18 (23.66), indicating a low level of sexual activity (Table 5). The mean (SD) scores for sexual activity decreased from V1 to 11.12 (18.76) at V2 and 9.02 (16.44) at V3, showing a decrease in sexual activity. Of note, 81.4% of patients had no or little interest in sex at V1, and 89.7% were not at all or a little sexually active at V1. These values were, respectively, 88.2% and 96.8% at V2, and 91.2% and 97.9% at V3. Findings for the subscale sexual functioning should be interpreted with caution, as there was a high number of missing patients because the sexual functioning subscale was only applicable to patients who indicated at least some sexual activity. Sexual functioning worsened, indicated by a decrease from 65.20 (22.68) at V1 to 45.05 (24.89) at V2 and 45.28 (23.09) at V3.
There was also a high number of missing data for the incontinence aid subscale, as this section was only applicable to patients who reported using an incontinence aid. Urinary symptoms, incontinence aid, and bowel symptoms subscales were generally similar across the three visits (Table 5).
Symptoms
At V1, 41.4% (326/787) of patients presented with at least one clinical symptom, 34.1% (268/787) had one or more urinary symptoms, 17.2% (135/787) had one or more sexual symptoms, and 9.1% (72/787) had one or more other symptoms (mostly bone pain, 4.6%, and asthenia; 3.4%, other 3.2%).
A lower frequency of urinary symptoms was reported at V2 (24.5% (184/752)) and V3 (16.6% (119/715)) compared with V1. The distribution of urinary symptoms was similar across all visits, with the most frequent symptoms being pollakiuria and nocturia.
Compared with V1, the frequency of sexual symptoms was similar at V2 (18.1%; 136/752) but lower at V3 (11.9%; 85/715). Erectile dysfunction and libido decrease were the most frequent sexual symptoms at each visit (respectively, 15.9% and 4.7% at V1, 16.8% and 11.4% at V2, 10.6% and 6.4% at V3). A higher frequency of other symptoms was reported at V2 (29.4%; 221/752) and at V3 (25.0%; 179/715) than at V1, mostly due to asthenia (V2: 11.3%; V3: 7.8%) and other symptoms (not specified; V2: 21.7%; V3: 20.1%).
Subgroup analyses
Circumstances of prescription
Outcomes were assessed in subgroups of patients based on the circumstance by which patients were prescribed triptorelin treatment: HRL, LAT, biochemical recurrence after local treatment (BCR), MS, other circumstances (Table 6). Data for the other circumstances subgroup (n = 31) are not presented owing to the small sample size, which limits the clinical relevance of any findings.
Subgroup analysis by circumstance of prescription.

[x–y] Denotes upper limit included; [x–y[ denotes upper limit excluded. Percentages are based on the number of patients without missing values in the FAS population. Data presented by subgroups for 785 patients; two patients had missing results for circumstance of prescription and are not presented in this table. Patients could have only one main reason for choice of planned total duration. Patients could have several main reasons for choice of formulation and administration route. Data for the “other circumstances” subgroup (n = 31) are not presented. Bold indicates significant differences between subgroups, based on nonoverlapping of 95% CI, as detailed below.
HRL versus LAT and MS.
BCR versus HRL and MS.
HRL versus BCR and MS.
BCR versus MS.
LAT versus MS.
HRL versus LAT, BCR and MS.
MS versus HRL, LAT and BCR.
LAT versus BCR and MS.
MS versus HRL, LAT and BCR.
BCR versus HRL, LAT and MS.
LAT versus HRL and BCR.
AFU, French Urological Association (Association Française d’Urologie); BCR, biochemical recurrence after local treatment; CI, confidence interval; cTN, clinical T and N stage; EAU, European Association of Urology; FAS, full analysis set; HRL, high risk localized tumor; ISUP, International Society of Urological Pathology; LAT, locally advanced tumor; MS, metastatic stage; PSA, prostate-specific antigen.
Patients who were prescribed triptorelin for LAT and MS were most frequently enrolled by urologists. Patients with HRLs were most frequently enrolled by radiation oncologists. The planned total duration of triptorelin treatment at V1 was mainly between (24–36) months in the subgroups of HRL and LAT. For patients with metastatic tumors, the planned total duration was mainly for life.
The most common primary reason of choice of planned total duration at V1 was Gleason score/ISUP group, with significantly higher proportions of patients in the subgroups HRL (45.3%) and LAT (30.5%) reporting this as the reason, versus BCR (9.4%) and MS (3.4%). In the subgroup of BCR, the most common primary reason of choice was PSA level, with a significantly higher proportion of patients (22.9%) versus MS (8.7%). On the other hand, in the subgroup of MS, the most common reason of choice was number of bone metastases, with a significantly higher proportion of patients (39.1%) versus HRL, LAT, and biochemical recurrence (0.3%, 0, and 2.1%, respectively).
Prescribed formulations and administration routes of triptorelin were similar and consistent with the overall population. In all subgroups the most common reasons of choice were preferred investigator route/formulation and planned total duration of treatment, but planned total treatment duration was significantly higher in LAT versus MS, while preferred investigator route/formulation was higher in HRL and biochemical recurrence versus LAT.
The proportion of patients undertaking supportive care pathways was highest in the MS subgroup (69.6%) and lowest in the LAT subgroup (53.1%). No relevant differences were observed between subgroups in the proportion of patients treated continuously with triptorelin during the 12-month follow-up period, with ⩾83.6% of patients being treated continuously with triptorelin in all subgroups.
Formulation and route
Outcomes were assessed in subgroups of patients based on the formulation and route of administration of triptorelin: monthly, intramuscular; every 3 months, subcutaneous; every 3 months, intramuscular; and every 6 months, intramuscular (Table 7). Data for the subgroup Monthly, intramuscular (n = 8) are not presented because of the small sample size, which limits the clinical relevance of any findings.
Subgroup analysis by formulation and route.

[x–y] Denotes upper limit included; [x–y[ denotes upper limit excluded. Percentages are based on the number of patients without missing values in the FAS population. Data presented by subgroups for 785 patients; 2 patients had missing results for circumstance of prescription and are not presented in this table. Patients could have several main reasons for choice of formulation and administration route. Data for the subgroup monthly—intramuscular (n = 8) are not presented. Data not presented for the primary reason for choice of planned total duration at Visit 1 as no clinically relevant differences were observed between subgroups by formulation and route. Bold indicates significant differences between subgroups, based on nonoverlapping of 95% CI, as detailed below.
6mIM versus 3mSC and 3mIM.
3mSC versus 3mIM.
3mSC versus 6mIM.
3mSC versus 3mIM and 6mIM.
3mIM, every 3 months—intramuscular; 3mSC, every 3 months—subcutaneous; 6mIM, every 6 months—intramuscular; CI, confidence interval; FAS, full analysis set.
Patients in the 6-month formulation subgroup were more often enrolled by urologists versus radiation oncologists. Anticoagulant treatment was given significantly more frequently to patients receiving the 3-month subcutaneous treatment than those receiving the 3-month intramuscular formulation, but no difference was observed between patients receiving the 3-month subcutaneous treatment and those receiving the 6-month intramuscular formulation.
Across subgroups, the most commonly reported main reasons for choosing the specific formulation and administration route of triptorelin were investigator preference and planned total duration of treatment. Planned total duration of treatment was reported for a higher proportion of patients receiving the 6-month formulation than the 3-month subcutaneous formulation. Compared with the other subgroups, potential impact on compliance was reported more commonly as a main reason in the 6-month formulation subgroup and anticoagulant treatment was reported more commonly in the 3-month subcutaneous formulation subgroup.
No relevant differences between subgroups in planned total duration and reasons of choice of planned total duration were observed (data not presented).
No relevant differences were observed between subgroups in the proportion of patients undertaking supportive care pathways. No relevant differences were observed between subgroups in the proportion of patients treated continuously with triptorelin during the 12-month follow-up period, with ⩾84.7% of patients being treated continuously with triptorelin in all subgroups.
Safety
In the FAS, 109 patients (13.9%) reported 224 treatment emergent AEs (TEAEs) in total (Table 8). The most common TEAEs were hot flush (42 patients (5.3%)), diarrhea (15 patients (1.9%)), nocturia (14 patients (1.8%)), and dysuria (10 patients (1.3%)).
AEs in the FAS.

Format is number of patients (percent of patients) (number of events). Percentages are based on the number of patients in the FAS population.
AE, adverse event; FAS, full analysis set; TEAE, treatment emergent adverse event.
All TEAEs of hot flushes and one TEAE of nocturia were assessed by the investigator as related to the study treatment, while the other most commonly reported TEAEs were assessed as not related. In total, 80 TEAEs assessed by the investigator as related to the study treatment were reported in 57 patients (7.2%), and 144 TEAEs were assessed as not related in 70 patients (8.9%). The majority of TEAEs assessed as related were grade 1 (41 patients (5.2%)) and were consistent with the known safety profile of triptorelin in the male population.
Six patients (0.8%) reported nine TEAEs leading to study drug withdrawal. No AEs, no serious AEs, nor TEAEs led to study discontinuation. Overall, 33 patients (4.2%) reported 46 treatment emergent serious AEs (TESAEs) in total and 5 patients (0.6%) had TESAEs that were assessed by the investigator as related to the study treatment. In total 21 patients (2.7%) died, including 18 patients (2.3%) who died from a TESAE and 3 patients (0.4%) for whom death was not otherwise specified. Three deaths were assessed by the investigator as treatment-related but were then assessed as “not or unlikely related” by the sponsor, because the patient’s underlying disease of PCa or underlying comorbidities (cardiac and cerebrovascular disorders) were an alternative explanation. No new or unexpected safety concerns were identified in the study. The most frequent TEAE of hot flush is a well-known event with GnRH agonists. Management of this usual side effect was left to the investigator’s discretion, in accordance with their usual practice, and not collected.
Discussion
ADT is a common treatment option for men with high risk localized, locally advanced, and/or metastatic PCa, and GnRH agonists are the most widely used among the ADT options. 9 Triptorelin is one of the most frequently prescribed GnRH agonists in France; however, there is a lack of knowledge about the modalities of use in the real-world, particularly the planned total duration of ADT treatment at initiation, the formulation and administration route chosen, and the drivers behind these choices.
This large, prospective, non-interventional study, which included 806 patients and analyzed 787 patients enrolled by 123 investigators, demonstrated high compliance with triptorelin treatment in real-world practice in France. Overall, 85.3% of patients received continuous treatment with triptorelin for 12 months following treatment initiation. The proportion of deaths and TESAEs was low (21 and 5 patients, respectively) and, given that 69.1% of patients undertook at least one supportive care pathway (e.g., treatment assessment, referral to another physician, associated recommendation, or prescription) at V1, V2, or V3, these data suggest that effective management of AEs and effective supportive care pathway may help to maintain compliance with triptorelin treatment. Additionally, no new or unexpected safety concerns were identified and hot flushes, the most frequent AE reported, are well known to be associated with GnRH agonist treatment.12 –14
The proportion of patients who received continuous treatment with triptorelin for 12 months in TALISMAN (85.3%) is higher than in other observational studies conducted in France.10,11 In the CHRONO study, 1438 patients initiating GnRH agonist treatment were included and followed for 12 months. Of these patients, 5.7% discontinued treatment at 3 or 6 months, and 18.4% of the remaining patients discontinued treatment at 12 months. The reasons for discontinuation of ADT were not provided. 10 In the DEVENIR study, 1301 patients initiating GnRH agonist treatment were followed-up for 24 months, and 43.8% had their ADT treatment modified in the first 2 years of treatment. Reasons for modification were mostly missing owing to the observational design and long follow up of the study. 11 In the current study, 116 patients (14.7%) were not treated continuously during the 12 months following treatment initiation; these included 45 completers and 71 withdrawals. The main reasons for withdrawal were death (n = 19) and lost to follow-up (n = 18). None of the 116 patients stopped treatment owing to an AE.
Of the 787 patients in the FAS, the most commonly reported circumstances of prescription of triptorelin at V1 were for treatment of HRLs (41.1%), followed by MS disease (26.4%), LATs (16.3%), biochemical recurrence (12.2%), and other circumstance (3.9%). Treatment was generally prescribed in accordance with European and French guidelines in high risk PCa.3 –5 The planned total duration of treatment was most commonly between 24 and 36 months, and subgroup analyses demonstrated that treatment of this duration was most commonly prescribed to patients with HRLs (55.1%) or those with locally advanced PCa (42.2%). For most patients (66.7%) with MS PCa, triptorelin was prescribed for life.
The main reasons for choice of planned total duration of triptorelin treatment declared by the investigators at V1 were Gleason score, guideline recommendations, and PSA level. Tumor aggressiveness parameters were high risk features adapted to each stage (Gleason score for high risk PCa, PSA level for patients with biochemical recurrence, and number of bone metastases for patients with MS disease) and were the main factors influencing choice of planned total duration of ADT. Multivariate analysis identified that longer planned duration of treatment was associated with age ⩾75 years (vs age 70–75 years), circumstance of prescription (i.e., MS or other circumstance vs HRLs), and absence of sexual symptoms. Investigators may have planned a shorter total duration of ADT treatment for patients with sexual symptoms at baseline because of its well-known impact on sexuality. 5 We could not see any similarities between the parameters identified as determinants and the main reason of choice for the planned total duration. This is likely due to the limited list of reasons provided to the physicians and the fact that the study was not designed to compare these results. Changes in the planned duration of treatment at V2 or V3 were associated with disease progression; however, the proportion of patients who experienced a change in planned treatment duration was low.
Most patients (72.5%) received the 3-month subcutaneous formulation at baseline, main reasons for choice of formulation and route declared by the investigators were physician preference and planned total duration of treatment. For choice of the formulation, multivariate analysis identified that the 6-month formulation was more often prescribed by a urologist (vs radiation oncologist) and in patients with a higher QLQ-PR25 sexual activity sub-score. For choice of the route of administration, multivariate analysis identified that the intramuscular route was more often prescribed in patients without anticoagulant treatment, in patients with prior PCa treatment, and in patients with a longer planned total duration of treatment. There were a few similarities between the parameters identified as determinants of the choice of the route of administration and the reported reasons of choice of the prescribed formulation and route of triptorelin, with planned total duration of triptorelin treatment and anticoagulant treatment cited by physicians as the second (28.1%) and fifth (7.3%) most common reasons, respectively. In subgroup analysis, receipt of a concomitant anticoagulant treatment was more often a reason of choice for the 3-month subcutaneous formulation than the 3- and 6-month intramuscular formulations; however, concomitant anticoagulants were received by a similar proportion of patients in the 3-month subcutaneous and 6-month intramuscular formulation subgroups. The high proportion of patients receiving the 3-month subcutaneous formulation contrasts with findings from previous similar studies, which suggested that there may be a preference for intramuscular formulations in France10,11; however, these studies included a higher proportion of urologists than the TALISMAN study. Of note, in the present study, the 6-month intramuscular formulation was preferred by urologists.
One of the main findings of this study is the increase of the supportive care pathways provided by physicians managing ADT treatment, compared to previous observational studies conducted in France. In the CHRONO study conducted between 2010 and 2011, 1.4% (18/1312) of patients were given vitamin D and calcium supplementation, 10 compared with 19.7% and 10.8%, respectively, in this study, showing that prevention of bone loss has highly increased in the past 10 years. In the DEVENIR study conducted between 2011 and 2015, osteodensitometry was performed in only 1.1% (10/891) of patients at baseline, 11 compared with 12.3% in this study, showing that there was a notable increase in the assessment of bone loss at baseline in the past decade. Our data indicate that the risk of bone loss with GnRH agonist treatment is better known, assessed, and managed by the physicians who follow the French and European guidelines.
Findings from the analysis of quality of life, as evaluated by the EORTC QLQ-PR25 questionnaire, were generally in line with results reported in the literature and with the known tolerability profile of triptorelin and other ADTs.12 –29 Patients reported an increase in hormonal treatment-related symptoms, including treatment-related hot flushes, and a similar but less pronounced trend for feeling less masculine and having sore or enlarged nipples or breasts. Results from this study highlight the necessity for physicians to provide information to patients about the possible side effects of treatment and to help them cope with these side effects, to avoid lack of compliance. Patients in this study also reported decreased sexual activity and sexual functioning; hence, physicians should inform their patients of this possible side effect before initiation of treatment and provide psychosexual advice and drug and/or nondrug management of sexual dysfunction. However, urinary symptoms, incontinence aid, and bowel symptoms remained stable.
There were no new or unexpected safety findings. Overall, 21 deaths were reported during the study, including 3 that were assessed by the investigator as treatment-related; however, these deaths were assessed by the sponsor as “not or unlikely related.” Alternative explanations were underlying disease of PCa or underlying comorbidities, particularly cardiac and cerebrovascular disorders.
Observational studies may better represent routine clinical practice than randomized controlled trials. Strengths of the TALISMAN study include the prospective study design and the inclusion of a large number of patients with demographic and disease characteristics that reflect those of patients seen in real-world practice in France.10 –26 Given that this study was carried out according to each investigator’s routine practice, limitations include the potential for inherent bias and constraints in data collection and interpretation.
Conclusion
These data demonstrate that the proportion of patients who receive continuous treatment with triptorelin for 12 months in France is high and that prescribing is in line with guideline recommendations. Effective management of AEs and provision of supportive care pathways may help to maintain compliance with triptorelin treatment.
Supplemental Material
sj-docx-1-tau-10.1177_17562872251382970 – Supplemental material for Treatment of aggressive prostate cancer with triptorelin in real life in France: the TALISMAN study
Supplemental material, sj-docx-1-tau-10.1177_17562872251382970 for Treatment of aggressive prostate cancer with triptorelin in real life in France: the TALISMAN study by Thierry Lebret, Gilles Crehange, Nathalie Pello-Leprince-Ringuet, Valérie Perrot and Jérôme Rigaud in Therapeutic Advances in Urology
Footnotes
Acknowledgements
The authors thank all patients involved in these studies, as well as their caregivers, care team, investigators, and research staff in participating institutions. The authors thank Tamzin Gristwood (PhD) and Kirsty Walters (PhD) of Oxford PharmaGenesis, Oxford, UK, who provided medical writing support, which was sponsored by Ipsen in accordance with Good Publication Practice (GPP) guidelines.
Declarations
Article Note
The following updates have been made to this article:
*In the Treatment section, “DECAPEPTYL LP” has been corrected to “Decapeptyl® LP”.
*In the Treatment section, “3 3-month” has been updated to “3-month”.
*In Table 6 footnote, the sentence “Planned total duration ‘for life’ means planned total duration equals 72 months” has been removed, and “iMS versus HRL” has been updated to “iMS versus HRL, LAT, and BCR.”
*Following sentence has been removed “Planned total duration ‘for life’ means planned total duration equals 72 months” has been removed from Table 7.
*In the Discussion section, the phrase “previous nonobservational studies conducted in France” has been updated to “previous observational studies conducted in France.”
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
