Abstract
Mixed hyperlipidemia is a frequent atherogenic dyslipidemia characterized by elevated triglycerides, low HDL cholesterol levels, and excess of small, dense LDL particles, often with elevated apolipoproteinB and non-HDL cholesterol concentrations. Although statins are the drug of first choice, many high-risk patients with mixed hyperlipidemia do not achieve the recommended goals with statin monotherapy. Due to the complementary effects of ezetimibe and fenofibrate on the components of atherogenic dyslipidemia, the combination of these two drugs is an alternative treatment option for patients with mixed hyperlipidemia. However, the potential benefit of ezetimibe-fenofibrate combination therapy in cardiovascular risk reduction strategies remains to be established and this combination therapy should only be considered as a second-line therapy and appears to be particularly useful for patients with a poor response or an intolerance to statin monotherapy.
Introduction
As the epidemic of metabolic syndrome and type 2 diabetes continues to progress, the prevalence of mixed hyperlipidemia is markedly increasing across the world. This dyslipidemia is characterized by elevated levels of triglycerides (TG) and decreased levels of high-density lipoprotein cholesterol (HDL-C). Although plasma levels of low-density lipoprotein cholesterol (LDL-C) may be only slightly above target levels, an increase in atherogenic small, dense LDL is a common feature, as well as elevated non-HDL cholesterol (non-HDL-C) and apolipoproteinB (ApoB) concentrations. All these metabolic abnormalities usually named as atherogenic dyslipidemia or atherogenic lipoprotein phenotype can contribute to increase the risk for cardiovascular disease. 1 Moreover, postprandial lipaemia has emerged as a contributor to cardiovascular risk, and a delayed clearance of chylomicrons and their remnants is usually observed in patients with mixed hyperlipidemia.
In high risk patients with mixed dyslipidemia, an ideal treatment should normalize the atherogenic lipid triad (high non-HDL-C and/or LDL-C, low HDL-C and high TG). In clinical practice, mixed hyperlipidemia is one of the must difficult lipid abnormalities in which to adequately achieve recommended LDL-C and non-HDL-C targets. LDL-C is the established primary target of treatment by both the US2,3 and European4,5 guidelines. Beyond lowering LDL-C, the National Cholesterol Education Program Adult Treatment Panel III (NCEP ATP III) introduced a secondary target of therapy, non-HDL-C, in patients with elevated TG or the metabolic syndrome to take into account the burden of atherogenic apoB100 lipoproteins.2,3 A consensus conference has recently suggested treatments goals for LDL-C, non-HDL-C and apoB in patients with cardiometabolic risk and lipoprotein abnormalities. 6
Statin therapy has been shown to decrease cardiovascular morbidity and mortality, primarily through their recognized efficacy in reducing LDL-C levels. 7 So, in all the guidelines, statin therapy is recommended as first-line pharmacotherapy of mixed hyperlipidemia. But some patients with mixed hyperlipidemia are intolerant or non-responder to statin therapy, and there is a need for other alternative therapies.
Fibrate therapy is more effective than statins to decrease TG and increase HDL-C levels, but less effective regarding LDL-C reduction. 1 In addition, fibrates affect the LDL size and density profile, promoting a change from small-dense to large-buoyant LDL particles. 8 Even if clinical trials using fibrates have reported conflicting results, several sub-group analyses from VAHIT,9,10 BIP11,12 and FIELD 13 have shown that the benefit of fibrates mainly appear for patients with atherogenic dyslipidemia (high TG-low HDL-C lipid profiles) associated with type 2 diabetes and/or metabolic syndrome.
Based on their respective lipid-modifying activity, the combination of a statin and fibrate would be a logical choice to improving achievement of lipid targets. 14 However, when fibrates are used in combination with statins, attention must be paid to the risk for myositis and rhabdomyolysis, 15 even if this risk is less with fenofibrate compared to gemfibrozil.16,17 Whether the strategy of the combined fenofibrate-statin therapy translates to reduction in cardiovascular risk is unknown and currently evaluated in the large prospective ACCORD study. 18 Moreover this strategy cannot be proposed to patients with poor tolerance to statin therapy. For these patients other combined therapies should be used and the combination of ezetimibe plus fenofibrate can be one of the new alternatives for the management of mixed hyperlipidemia.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Ezetimibe monotherapy
The concentration of plasma cholesterol is maintained by biosynthesis through the endogenous pathway and by absorption of dietary and biliary cholesterol through the exogenous pathway.
19
Ezetimibe is the first number of a class of selective cholesterol absorption inhibitors. Ezetimibe is a synthetic 2-azetidinone whose chemical name is 1-(4-fluorophenyl)-3(
Originally developed as ACAT inhibitors, it soon became clear that 2-azetidinones did not inhibit ACAT2 in a relevant manner, but do block the intestinal absorption of cholesterol. Ezetimibe effectively inhibits the intestinal absorption of cholesterol and plant sterols without affecting absorption of triglycerides, fatty acids, bile acids or fat-soluble vitamins. 20
It was been reported that ezetimibe inhibits at least two different types of sterol transporters in the mouse small intestine. 21 The first is the Niemann-Pick C1-Like 1 (NPC1L1) protein which is a critical mediator of cholesterol and phytosterol absorption and an essential component of the ezetimibe-sensitive pathway.22,24 The second potential target of ezetimibe is a protein complex of annexin-2 and caveolin-1 which seems to be also involved in intracellular sterol trafficking. 25 NPC1L1 is undoubtedly a key protein in enterohepatic absorption and ezetimibe blocks the sterol-induced internalization of NPC1L1. 26 After in vivo administration, ezetimibe is rapidly absorbed, glucuronidated and recycled by the enterohepatic circulation, resulting in repeated delivery to the site of action in the intestine and thus limiting the peripheral exposure.20,27,28 This recirculation may explain the long half-life (≍22 h) of ezetimibe, which allows for once-daily dosing of the drug. Ezetimibe localizes in the intestinal wall, mainly as the glucuronide. Ezetimibe glucuronide inhibits the internalization of NPC1L1 and cholesterol as potently as ezetimibe.29,30 Ezetimibe is mainly excreted in the feces (as parent drug), with a small proportion in the urine (as glucuronide). 31
There was no clinically significant effect of food on the oral bioavailability of ezetimibe. 31 Ezetimibe is unlikely to cause drug interactions with common CYP450 substrates.27,31 No changes in the pharmacokinetics of statins, warfarin, digoxin, ethinyl estradiol or glipizide were observed when these agents were administered with ezetimibe. Coadministration of cholestyramine reduced the area under the curve (AUC) of ezetimibe by up to 80%. Cyclosporin increases the bioavailability of ezetimibe by approximatively fourfold, but there was no significant effect of ezetimibe on cyclosporin levels in cardiac transplant recipients. 32 Due to the unknown effects on the increased exposure to ezetimibe in patients with moderate or severe hepatic insufficiency, ezetimibe is not recommended in these patients. 27 The mean AUC for total ezetimibe is increased approximately 1.5-fold in severe renal disease (creatinine clearance ≤30 ml/min/1.73 m2). However, non adjustments are currently recommended for patients with renal insufficiency.
A study conducted in hypercholesterolemic subjects confirmed the mode of action of ezetimibe as an inhibitor of cholesterol absorption in humans. 33 This double-blind, placebo-controlled, crossover study in 18 men with mild to moderate hypercholesterolemia evaluated the effects of 2 weeks of treatment with ezetimibe 10 mg on intestinal absorption. Ezetimibe decreased fractional cholesterol absorption by 54% and LDL-C levels by 20% compared with placebo. This was associated with a compensatory increase of cholesterol hepatic synthesis, as reflected by increased in the lathosterol-to-cholesterol ratio. Ezetimibe also reduced plasma concentrations of campesterol and sitosterol by 48% and 41% respectively. 33
Fenofibrate monotherapy
Fenofibrate belongs to a class of lipid-modifying drugs used in the treatment of patients with hypertriglyceridemia, mixed hyperlipidemia, metabolic syndrome and/or diabetic dyslipidemia.34,35 This class of drugs exert their effects by activating the peroxisome proliferator-activated receptor α (PPARα). 36 Fenofibrate has been available since 1975 and the pharmacological properties of fenofibrate have been reported in many reviews.37–39
Fenofibrate is the isopropyl ester of 2-[4-(4-chlorobenzoyl)-phenoxy]-2-methyl propanoic acid and is a prodrug that is converted in the pharmacologically active metabolite fenofibric acid. After the original 300 mg standard formulation, several formulations of the drug have been developed to improve its bioavailability: a capsule formulation of micronized fenofibrate available in 67 mg, 200 mg, and 267 mg doses, a microcoated tablet formulation available in a 160 mg dose and a new nanoparticle tablet formulation available in a 145 mg dose.
Activated PPARα stimulates the expression of genes encoding various enzymes that regulate fatty acid and lipoprotein metabolism.35,39 Fenofibrate promoted the β-oxidation of fatty acids and, by consequence, reduced the availability of fatty acids for very-low density lipoprotein (VLDL) synthesis and secretion. Another key mechanism for the triglyceride-lowering effects of fenofibrate is the promotion of intravascular lipolysis. Fenofibrate increased the expression of the gene for lipoprotein lipase and decreased apolipoprotein (Apo) CIII expression in the liver. In fact, fenofibrate altered both the synthesis and the catabolism of the triglyceriderich lipoproteins.36,39 Moreover fenofibrate treatment reduced the proportion of small, dense LDL, with the formation of larger, less dense LDL particles with a higher affinity for the LDL-receptor and thus catabolized more rapidly. 40 PPARα activation with fenofibrate also increased ApoAI and ApoAII synthesis, decreased the cholesteryl ester transfer protein-mediated transfer of cholesterol from HDL to VLDL, enhanced cell cholesterol efflux by induction of ABCA1 expression and decreased SR-B1 in the liver. All these effects contribute to the increase of plasma HDL-C concentrations.41,42
In addition to the lipid-modifying activity, fenofibrate also had numerous pleiotropic effects mediated by PPARα activation,36,40 such as effects on vascular inflammation, fibrinogen or plasminogen activator inhibitor-1.38,39
Fenofibrate is rapidly hydrolyzed to its active metabolite fenofibric acid. The half-life of fenofibric acid is 20 hours, allowing for once-daily administration. Fenofibrate is mainly excreted in the urine, as fenofibric acid and fenofibric acid glucuronide. 39 The clearance of fenofibrate is greatly reduced in patients with renal dysfunction and dose reduction is recommended in patients with renal impairment. 39
Fenofibrate has a low potential for drug interactions, with the exception of cyclosporin with a potential increase of the nephrotoxicity of cyclosporin in patients receiving fenofibrate. 39 Concomitant administration of fenofibrate has no clinically significant effect on the pharmacokinetics of simvastatin, rosuvastatin or atorvastatin, and only modest effects on the exposure of pravastatin. The pharmacokinetics of fenofibrate are not significantly modified by the concomitant administration of statins.1,39 No pharmacokinetic studies of fenofibrate are available in patients with hepatic impairment. Fenofibrate has also been shown to potentiate the effect of coumarin-type anticoagulants.
Ezetimibe-fenofibrate combination therapy
The pharmacodynamic and pharmacokinetic interaction between ezetimibe and fenofibrate has been evaluated in healthy subjects with primary hypercholesterolemia in a 2-week placebo-controlled, parallel-group study comparing ezetimibe-fenofibrate combination therapy, ezetimibe alone, fenofibrate alone and placebo: 43 ezetimibe did not significantly affect the pharmacokinetics of fenofibrate. The concomitant administration of 200 mg micronized fenofibrate and 10 mg ezetimibe resulted in a significant (≍50%) increase in steady state total ezetimibe exposure. The mean maximum observed plasma concentration (Cmax) and the AUC of total ezetimibe were significantly increased by approximately 64% and 48%, respectively. However, this increase in total ezetimibe exposure was not considered to be clinically significant considering the flat dose-response of ezetimibe: for LDL-C decrease and safety in the range 10 to 40 mg/day. 44
Clinical Studies
Ezetimibe monotherapy
The efficacy and safety of ezetimibe administered as single agent therapy at a dose of 10 mg/day in patients with primary hypercholesterolemia have been assessed in several double-blind studies. A meta-analysis of eight randomized, double-blind, placebo-controlled trials showed that, compared with placebo, ezetimibe 10 mg monotherapy significantly reduced LDL-C and TG levels by respectively 18.58% and 8.06%, and increased HDL-C levels by 3.00%. 45 Ezetimibe also reduced non-HDL-C and apoB concentrations. Subfractionation of apoB containing lipoproteins showed that the reduction in LDL-C was due to a fall in all the LDL subfractions. 46 The impact of ezetimibe in monotherapy in modulating LDL size seems relatively small. 47 Ezetimibe produced mainly quantitative rather than qualitative changes in LDL. Ezetimibe affected the metabolism of apolipoproteins B48 and B100 in males with mixed hyperlipidemia, with significant decreases in triglyceride rich lipoprotein apoB48, VLDL apoB100, and LDL apoB100 pool sizes and increases in VLDL and LDL apoB100 fractional catabolic rates. 48 Moreover ezetimibe improved post-prandial hyperlipidemia in patients with mixed hyperlipidemia. 49
In all the phase II and phase III studies, ezetimibe monotherapy was well tolerated, with an adverse event profile of ezetimibe similar to that in the placebo group. 45 There are no end point trials evaluating the benefit of ezetimibe monotherapy to reduce risk for cardiovascular morbidity and mortality. Ezetimibe has only been evaluated until now in combination with simvastatin in a specific population of patients with aortic stenosis: 50 in SEAS trial, simvastatin and ezetimibe reduced the incidence of ischemic cardiovascular events, but not events related to aortic-valve stenosis. Several trials have examined the effect of ezetimibe alone 51 and in combination with statins52–54 on the evolution of carotid intima-media thickness (IMT): the neutral findings observed in ENHANCE study could be due to normal carotid IMT at baseline. 52 Two other trials SANDS 53 and VYCTOR 54 have shown a positive effect of ezetimibe combined with a statin in populations with abnormal carotid IMT. In a small study, 51 ezetimibe improved carotid IMT in Japanese patients with mixed hyperlipidemia and high carotid IMT at baseline.
Fenofibrate monotherapy
The potential of monotherapy with fenofibrate in the treatment of patients with primary dyslipidemia has been well established in numerous placebo-controlled and comparative trials.37–39 The effect of fenofibrate on the concentration of plasma lipids is largely influenced by the baseline lipid levels. Fenofibrate therapy was consistently associated with a substantial decrease of serum TG by 20% to 50%, usually directly proportional to the baseline TG levels. Fenofibrate increased HDL-C levels by 5% to 20%, to a degree directly dependent of baseline TG and HDL-C levels. Fenofibrate had widely variable effects on LDL-C levels mainly dependent of the type of dyslipidemia. For example, fenofibrate may decrease LDL-C by 20% in patients with normal TG levels, but generally produces less reduction in LDL-C in patients with mixed hyperlipidemia. Patients with severe hypertriglyceridemia and low levels of LDL-C may raise their LDL-C levels during fenofibrate treatment, possibly as a result of an accelerated catabolism of triglyceride-rich lipoproteins, leading to an increased LDL conversion and an increased LDL particle size. Treatment with fenofibrate also reduced the postprandial increase in triglycerides and remnant lipoprotein particles,1,55 particularly in patients with mixed hyperlipidemia. 56
Two recent trials57,58 provided data on the efficacy of fenofibrate monotherapy in large populations of patients with mixed dyslipidemia: in these trials, TG decreased by 43.2% and 41.3%, HDL-C increased by 18.8% and 18.2%, LDL-C decreased by 5.5% and 15.7%, and non-HDL-C decreased by 16.2% and 21.0%. The effects on apoB were in agreement with the evolution of non-HDL-C levels: apoB levels decreased by 15.2% and 20.1%. ApoA-I levels were increased by 8.4% and 10.8%.57,58 Finally these trials have confirmed a large and significant shift in LDL size with a decrease in the proportion of small, dense LDL during fenofibrate treatment.
Fenofibrate has also numerous pleiotropic effects:35,39 in patients with dyslipidemia, fenofibrate therapy reduced plasma fibrinogen, CRP levels, pro-inflammatory cytokines (interleukin-6, tumour necrosis factor-α) and monocyte chemoattractant protein-1. 39 Fenofibrate also improved endothelial dysfunction.59,60 Among fibrates, only fenofibrate significantly reduced uric acid levels.39,61 At the opposite, fenofibrate therapy induced two potential deleterious effects: an increase in creatinine levels39,62 and a significant elevation in homocysteine levels.39,63 Whether these effects on creatinine and homocysteine are of clinical importance remains to be determined. Creatinine rises was not associated with an alteration in renal function, as measured by glomerular filtration rate. 64 The creatinine elevation associated with fenofibrate treatment is fully reversible within a few weeks of ceasing therapy, 65 also suggesting an absence of permanent renal damage.
In summary, fenofibrate has a beneficial action on all the lipoprotein abnormalities present in patients with mixed hyperlipidemia. This pattern of mixed dyslipidemia being usually observed in type 2 diabetes and presumably to follow-up on the favourable effects of gemfibrozil treated diabetic patients in VA-HIT study, 10 the potential role of fenofibrate for cardiovascular disease (CVD) risk reduction has only been evaluated in type 2 diabetes, but not specifically type 2 diabetic patients with mixed dyslipidemia.
In the DAIS study, 66 418 diabetic men and women were randomized to fenofibrate or placebo for 3 years. Baseline lipids were LDL-C of 132 mg/dL, TG of 221 mg/dL, and HDL-C of 40 mg/dL. Fenofibrate lowered LDL-C by 6% and TG by 28%, and raised HDL-C by 7%. Fenofibrate slowed the angiographic progression of coronary atherosclerosis: the progression of focal coronary atheroma was 40% less in the fenofibrate group compared with placebo, without significant effect on diffuse atheroma. Interestingly, although the study was not powered to look at clinical events, there was a non significant 23% reduction in CVD events. These effects seemed to be explained not only by the changes in HDL-C, LDL-C and TG levels, but also by a significant increase in LDL particle size. 67 Additionally, fenofibrate reduced the incidence of microalbuminuria by 54%. 68 In DAIS, the increase of homocysteine did not alter the beneficial effect of fenofibrate. 69
The FIELD study was the first large landmark cardiovascular event-based trial with fenofibrate. 65 FIELD was a 5-year, randomized, placebo-controlled, double-blind study of the efficacy and safety of fenofibrate 200 mg/day in 9795 type 2 diabetic patients with (22%) or without (78%) previous CVD. Mean baseline lipids were lipids were LDL-C of 119 mg/dL, TG of 153 mg/dL, and HDL-C of 43 mg/dL. Unfortunately, only 21% of patients had a marked atherogenic dyslipidemia defined (for comparison with other fibrates trials) by TG ≥ 200 mg/dL and low HDL-C < 40 mg/dl in men and <50 mg/dL in women. 13 At 4 months and study close, fenofibrate decreased respectively TG by 29% and 22%, LDL-C by 12% and 6% and increased HDL-C by 5% and 1%. Fenofibrate treatment did not have a significant effect on the primary end point (CHD death or non-fatal MI) reduced by only 11% (p = 0.16). There was, however, a significant reduction (-11%, p = 0.035) in the secondary end point of total CVD events (a composite of CVD death, MI, stroke, and coronary or carotid revascularization), largely driven by significant reductions in non-fatal MI (-24%, p = 0.01) and coronary revascularization (-21%, p = 0.003). There was a non-significant increase in coronary death in the fenofibrate group.
The FIELD investigators also reported a significant 20% reduction of all MI events (p = 0.006) and significant 36% and 47% reductions in the risks of first amputation and minor amputation events. 70 Fenofibrate treatment was also associated with significant reductions in the need for retinal laser therapy 71 and decreased progression/increased regression of albuminuria. 65
Some explanations can be proposed to explain the global results of FIELD: the higher rate of statin use in the placebo group may play a role. Among fenofibrate patients not receiving off-trial lipid treatment, the CVD composite endpoint was significantly reduced by 19%. The poor effect of fenofibrate in reducing CVD events, more particularly in secondary prevention, could also be explain by the significant increase of homocysteine levels. It has been shown that gemfibrozil increased plasma homocysteine less than fenofibrate63,72 and this difference could explain the better clinical benefit of gemfibrozil in VA-HIT9,10 and HHS. 73 Another hypothesis to explain the FIELD results is the modest effects of fenofibrate on lipid parameters. Particularly the effect on HDL-C decreased over time in FIELD, but the mean HDL-C at baseline was normal, higher than in DAIS and the effect of a fibrate on HDL-C levels depends mainly of patients’ lipid profiles. The declining effect of fenofibrate on HDL-C over time could also be due to the fact that elevated homocysteine has been reported to reduce the ApoAI expression in mice and humans. 74
More convincing, the relative lack of favourable effects in FIELD may be related to the selected population. Fibrates reduce CVD effectively in patients with insulin resistance/overweight, high TG and low HDL-C. 75 In FIELD, the baseline TG was lower and the HDL-C was higher than in HHS or VA-HIT. New complementary analyses recently reported 13 have provided important data on the clinical utility of fenofibrate in mixed hyperlipidemia: in the subgroup of 2,014 patients with low HDL-C and TG > 200 mg/dL, fenofibrate treatment induced a significant 27% reduction of CVD events (Fig. 1). This highly significant effect corresponds to a 4.3% absolute risk reduction, with a number need to treat of 23 to avoid 1 or more CVD events. The beneficial effect of fenofibrate on macrovascular events appears larger among type 2 diabetic subjects with mixed dyslipidemia. 13
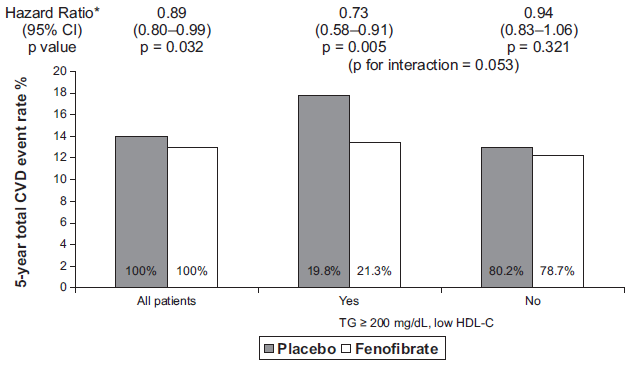
Post-analysis of FIELD: 13 greater CVD risk reduction with fenofibrate in patients with atherogenic dyslipidemia (High TG/Low HDL-C).
In the treatment of dyslipidemia, fenofibrate was generally well tolerated with few side-effects. The most common side-effects were gastrointestinal disturbances, liver function test abnormalities and increase creatine phosphokinase. Fenofibrate also increased plasma creatinine levels by an unclear mechanism. 39 In patients with type 2 diabetes who participated in the FIELD trial, a slight, but significant increase in pancreatitis (0.8% in fenofibrate group vs. 0.5% in placebo group) and pulmonary embolism (1.1% vs. 0.7%) and a non significant increase in deep vein thrombosis (1.4% vs. 1.0%) were observed. The excess of pancreatitis may be due to the increased lithogenicity of bile. The increased risk of venous thrombotic events may be related to the increased homocysteine level, a risk factor for thrombosis. 76
Ezetimibe-fenofibrate combination therapy
The efficacy of ezetimibe plus fenofibrate combination therapy has been evaluated in specific populations of patients with mixed hyperlipidemia.57,77 Indeed, due to the complementary biological efficacy of these two drugs, mixed hyperlipidemia appears the best choice for this combination therapy.
Patients with mixed hyperlipidemia have been recruited and included in two randomized trials.57,77 The inclusion criteria, main baseline characteristics and efficacy results are reported Table 1: the populations appear comparable regarding age, sex or BMI for example. The major differences between these two trials were the presence or not of metabolic syndrome (mandatory criteria in study 2), the lipid inclusion criteria and the absence of placebo controlled group in the study 2. 77 The patients recruited in the smallest study 2 have baseline lipid criteria non typically seen in metabolic syndrome, with a normal HDL-C and high LDL-C levels.
Summary of the two clinical studies evaluating the efficacy of ezetimibe/fenofibrate combination therapy.
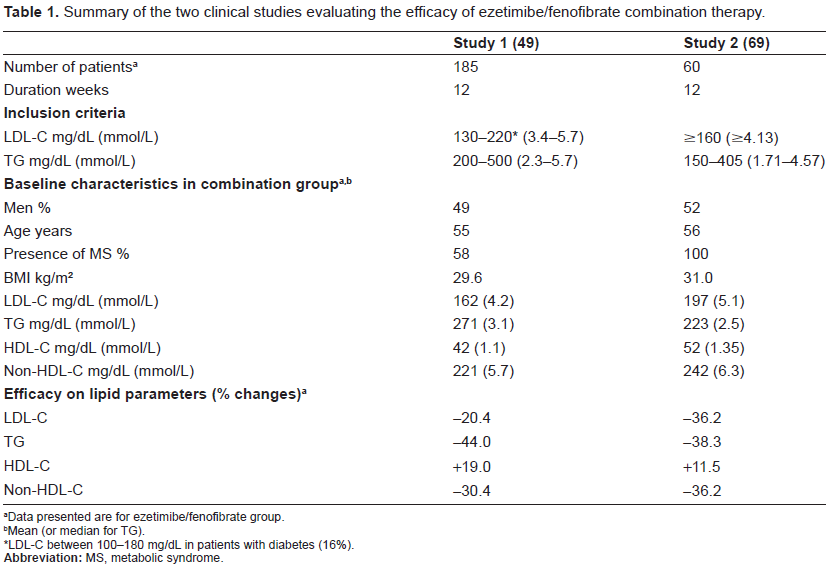
Data presented are for ezetimibe/fenofibrate group.
Mean (or median for TG).
LDL-C between 100-180 mg/dL in patients with diabetes (16%).
In the study 1, the efficacy and safety of ezetimibe co-administered with fenofibrate was compared with that of ezetimibe alone, fenofibrate alone and placebo during 12 weeks. 57 A total of 625 patients were randomized to receive one of the four daily treatments: placebo, ezetimibe 10 mg, fenofibrate 160 mg, and ezetimibe 10 mg + fenofibrate 160 mg. The coadministration therapy reduced LDL-C by 20.4%, non HDL-C by 30.4%, TG by 44.0% and increased HDL-C by 19.0%. The change in LDL-C was influenced by baseline TG levels: greater LDL-C lowering was noted in all active treatments in patients with TG ≤ 3.1 mmol/L (median). At baseline, ≥70% of patients in each treatment group had an atherogenic LDL size pattern B (preponderance of small, dense LDL particles). After treatment, a greater proposition of patients on ezetimibe plus fenofibrate (64%) and fenofibrate alone (62%) treatments shifted from a more atherogenic LDL size pattern to a larger, more buoyant and less atherogenic size pattern. Ezetimibe/fenofibrate combination therapy increased LDL peak particle size 78 and produced favourable changes in lipoprotein subfractions as assessed by the Vertical Auto Profile II method. 78 Depending on the study variable, the effects of the combination of ezetimibe plus fenofibrate were either additive (LDL-C, total cholesterol, non-HDL-cholesterol and apoB) or fenofibrate-dependent (TG, HDL-C, ApoAI, hsCRP, fibrinogen and LDL size pattern shift). Finally, LDL-C and non-HDL-C goal attainment was greater with coadministration than with either single treatment: about 60% of patients treated with ezetimibe plus fenofibrate reached LDL-C and non-HDL-C targets.
After completing the 12-week randomized, double-blind base study, 576 patients entered into a 48-week, double-blind, extension study during which they received fenofibrate (n = 236) or ezetimibe plus fenofibrate (n = 340). 79 Improvements from baseline in LDL-C (-22.0% vs. -8.6%), non-HDL-C (-31.6% vs. -19.4%), apoB (-25.2% vs. -16.2%), TG (-46.0% vs. -41.8%) and HDL-C (20.9% vs. 17.8%) levels were significantly greater with ezetimibe-fenofibrate combination therapy than with fenofibrate alone.
In the study 2, 77 180 patients were randomized to receive one of the three treatments: ezetimibe 10 mg, fenofibrate 145 mg or ezetimibe 10 mg + ezetimibe 145 mg. Ezetimibe/fenofibrate treatment resulted in greater reduction in LDL-C (-36.2%), smaller increase in HDL-C (+11.5%) than in study 1 (Table 1), with a trend to less decrease in TG levels. This may be explained by differences in the baseline lipid profile with higher baseline LDL-C, lower baseline TG and normal HDL-C in study 2 compared to study 1. Globally the baseline lipid profile in study 1 is more typical of the atherogenic lipid profile usually seen in mixed hyperlipidemia.
The combination of ezetimibe plus fenofibrate was well tolerated during both 12-week studies,57,77 and during the long term extension of study 1. 79
Place in Therapy
Despite the benefits conferred by statin therapy across various patient populations, a significant residual cardiovascular risk remains in high-risk patients, sometimes even if the LDL-C goals are achieved. Residual cardiovascular risk is undoubtedly multifactorial, but there is recent evidence for an important contribution to cardiovascular risk of lipid abnormalities beyond LDL-C, such as high TG and low HDL-C levels. These abnormalities classically noted as “atherogenic dyslipidemia” are typically seen in patients with mixed hyperlipidemia and frequently observed in patients with type 2 diabetes and/or metabolic syndrome.
Although statins are indicated as first-line therapy for patients with mixed hyperlipidemia, statin treatment may be limited by intolerance or poor-response in monotherapy with a failure to reach LDL-C and non-HDL-C goals. Fenofibrate is a second choice treatment for patients with mixed hyperlipidemia, but the benefit of fenofibrate monotherapy on macrovascular events in the global population of FIELD study is smaller that the cardiovascular benefit observed during statin treatment. This result can be explain by the small effect of fenofibrate monotherapy on LDL-C and/or non-HDL-C parameters. The combination of ezetimibe with fenofibrate offers an alternative strategy for patients with atherogenic mixed hyperlipidemia. The complementary efficacy of ezetimibe and fenofibrate affect all components of atherogenic dyslipidemia observed for these patients with significant decreases of LDL-C, non-HDL-C, ApoB and TG and increase of HDL-C. Moreover, a large proportion of patients with mixed hyperlipidemia can simultaneously reach LDL-C and non-HDL-C goals with this combination therapy. Even if no direct comparative data with statin monotherapy are available, the global improvement of lipid profile suggests that the combination of ezetimibe and fenofibrate could be an alternative of statin monotherapy.
The limitation of the use of this combination of ezetimibe plus fenofibrate is mainly due to the absence of data regarding the long term clinical benefit and safety profile. Until then this combination should be considered as a second-line therapy.
Nevertheless, the ezetimibe plus fenofibrate combination therapy offers an important treatment alternative to patients with mixed hyperlipidemia who respond inadequately or are intolerant to statin therapy.
Conclusion
Mixed hyperlipidemia is an important risk factor for the development of cardiovascular disease. In clinical practice, the management of patients with mixed hyperlipidemia is often more difficult than the treatment of patients with pure hypercholesterolemia, due to the necessity to normalize all the components of atherogenic dyslipidemia.
The combination of ezetimibe and fenofibrate is an additional alternative approach to improve the overall atherogenic lipid profile of patients with mixed hyperlipidemia. This combination therapy can be proposed as a second-line therapy, and appears to be particularly useful for patients with a poor response or an intolerance to statin monotherapy.
Disclosures
The author reports having received grant/research support and/or honoraria from a consultant for Astra-Zeneca, Genzyme, Kowa, Merck & Co, Merck-Schering-Plough, Novartis, Pfizer, Sanofi-Aventis, SMB, Solvay and Takeda.
