Abstract
The purpose of this article is to review the effectiveness and tolerability of pioglitazone and glimepiride combination treatment for type 2 diabetes, particularly in the context of metabolic control and cardiovascular prevention. We reviewed studies of pioglitazone and glimepiride combination treatment through literature in the internet based Medline. The search was conducted using the search terms pioglitazone, glimepiride, glycemic control, combination therapy, and type 2 diabetes mellitus. The current evidence shows that pioglitazone in association with glimepiride is an effective option in the treatment of type 2 diabetes. Fixed dose combination permits a better compliance to therapy in patients with type 2 diabetes. More studies are needed to establish a role of pioglitazone and glimepiride in atherosclerosis and cardiovascular prevention beyond glycemic control.
Keywords
Introduction
Every year, evidences and recommendations are published about treatment of type 1 and type 2 diabetes mellitus and their complications. 1
Pharmacological treatment is commenced when diet and physical exercise are no longer sufficient to achieve therapeutic goals: that is reduction of glycated haemoglobin (HbA1c < 7%). Two great classes of drugs are currently used for the treatment of diabetes: insulin-sensitizers and secretagogues. At this point in time, no drug exists that allows glycemic control for an undetermined period and combination therapy and/or insulin is recommended when treatment goals are not achieved. 2
In patients with type 2 diabetes mellitus, two pathophysiological issues are usually addressed: insulin-resistance and insulin secretory dysfunction. In addition, patients with type 2 diabetes mellitus and poor glycemic control are at high risk of development of atherosclerosis and cardiovascular disease. 3
Combination treatment with pioglitazone and glimepiride is a promising therapeuticl strategy, effective in addressing both pathophysiological defects of type 2 diabetes mellitus. Moreover, a large number of studies have demonstrated efficacy and safety of both drugs. Among insulin-sensitizing agents, pioglitazone significantly improves glycemic control, to a similar extent of that of metformin and rosiglitazone, but appear more beneficial for lipid metabolism than rosiglitazone. Glimepiride provides a similar improvement in glycemic control, compared to other sulphonylureas (SUs), but presents better tolerability and safety, in particular in patients with a history of acute myocardial infarction (AMI), because of the demonstrated neutrality of this molecule on the so-called “myocardial pre-conditioning”. The need and benefits of multiple drug therapy in patients with type 2 diabetes mellitus has been well documented in literature. In clinical practice, compliance to a multi-drugs therapy is a common problem that easily translates into poor compliance to medical prescription. Combination treatments have been demonstrated to be a good tool to increase patients’ adherence to therapy. Despite a rapid effect on glycemic control, which is commonly considered an advantage for patients with high cardiovascular risk, the mechanism of action of SUs is related to a more rapid decline in β-cells function, which is usually impaired in patients with recent diagnosis of type 2 diabetes mellitus. Conversely, pioglitazone has been demonstrated to preserve β-cells function, although glycemic control may be achieved more slowly than with SUs.4,5 Both pioglitazone and glimepiride present a number of so-called “complementary effects” beyond glycemic control, including vascular benefits and lipid profile improvement. 6 Fixed dose administration of these components appears a reasonable choice to start a combined pharmacological treatment in high cardiovascular risk patients who need both a rapid reduction of glycemic values and a sustained metabolic control over time.
In this manuscript we highlight efficacy and safety of combination treatment with pioglitazone, a thiazolidinedione (TZD), and glimepiride, a second-generation SU, in improving glycemic control, reducing cardiovascular risk and maintaining patients’ adherence to therapy.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Pioglitazone: mechanism of action
Thiazolidinediones interact with a group of nuclear receptors, known as peroxisome proliferator-activated receptors-γ (PPAR-γ) thus improving insulin-sensitivity in special tissues. PPARγ are mainly expressed in the adipose tissue, and, at low levels, in the liver and the vascular tissue. Activation of PPARγ affects both glucose and fat metabolism. Particularly, activation of glucose metabolism seems to be the result of modifications in free fatty acid (FFA) metabolism.7,8 As a PPAR-γ agonist, pioglitazone (Fig. 1) reduces insulin-resistance by enhancing the effect of insulin on peripheral tissues. 9 In the adipose tissue, pioglitazone increases glucose mobilization, up-take and utilization and stimulates differentiation of large adipose cells to a smaller pattern, which increases tissue insulin-sensitivity. 10
Pioglitazone acts mainly on adipose tissue, liver and muscle through activation of PPAR-γ receptors, a nuclear transcription factor, which is activated by its ligand and modulates transcription of genes of proteins involved in metabolism of both glucose and lipids. Moreover, pioglitazone induces secretion of adipocitokines, which are produced by adipose tissue. Pioglitazone reduces inflammatory citokines levels, as tumor necrosis factor (TNF-α), improves adiponectin (ADN) production and reduces resistance (r).11,12
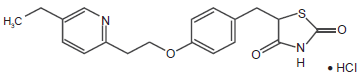
Pioglitazone chemical structure.
Pioglitazone promotes a normo-lipidemic profile by reducing triglycerides (Tg) levels, FFA and lipids peroxidation, improves low density lipoproteins (LDL-C) composition by increasing the number of larger LDL (LDL 1), is less atherogenic, and reduces small dense LDL particles. In regard to high density lipoproteins (HDL-C), concentration of HDL 1, and more protective particles, is induced by pioglitazone. In the liver, pioglitazone reduces apolipoprotein C III (Apo C III), which correlates with Tg concentration and increases apolipoprotein AI (Apo A I), a component of HDL. In the muscle, pioglitazone reduces activation of protein-C kinasis and reduces sfingomielinasis (enzymes that catalyze sfingomielina hydrolysis in cells membrane) activity. 13
Pioglitazone: pharmacokinetics
Following oral administration, in the fasting state, pioglitazone is first measurable in serum within 30 minutes, with peak concentrations observed within 2 hours. Food slightly delays the time to peak serum concentration to 3 to 4 hours, but does not alter the extent of absorption.
Pioglitazone is an extensively protein bound (>99%) in human serum, principally to serum albumin and is metabolized by hydroxylation and oxidation; the metabolites also partly convert to glucuronide or sulfate conjugates. Following oral administration, approximately 15% to 30% of the pioglitazone dose is recovered in the urine. Pioglitazone is excreted primarily as metabolites and their conjugates.
The mean serum half-life of pioglitazone and total pioglitazone ranges from 3 to 7 hours and 16 to 24 hours, respectively. 14
Glimepiride: mechanism of action
Glimepiride is a second-generation SU primarily activating specific receptors (SURs) on pancreatic β-cell membrane. When SUs are bound to SURs, ATP-sensitive potassium channels close, thus resulting in an augmented trans-membrane calcium flux and insulin release from β-cells. Among SUs, only glimepiride is internalized in the cell and is able to stimulate both first and second phases of insulin secretion. 15
In the UK Prospective Diabetes Study (UKPDS) 16 intensive treatment with SUs or insulin reduced (-12%) all outcomes with a reduction of 15% of microvascular complications; incidence of AMI was reduced by SUs of 16%, even if this result did not reach statistical significance (p = 0.052). According to data from a number of clinical studies, glimepiride presents a moderately favourable effect on total cholesterol (TC) levels in subjects with type 2 diabetes mellitus and metabolic syndrome. 17
The main side effects of using SUs are hypoglycemia, which is more common in patients with renal impairment or poor adherence to dietary advice, and weight gain (mean weight gain 2 Kg), partially explained by improvement of glycemic control.
Treatment with SUs has been suggested for Beta-cell exhaustion. However, results of UKPDS showed that a progressive reduction of insulin store is due to the natural progression of diabetes independent of treatment, and the potential role of SUs in this context is still under investigation. 16
As with any other glucose-lowering agent, glimepiride is recommended in association with diet and physical exercise. Glimepiride reduces fasting plasma glucose (FPG) and HbA1c in different percentages according to dosage of administration 18 Moreover, glimepiride is able to modify atherosclerosis markers, such as plasminogen activator inhibitor-1 (PAI-1), lipoprotein (a) [Lp(a)] and homocysteine (HCT). 19
Glimepiride: pharmacokinetics
After oral administration, glimepiride (Fig. 2) is completely absorbed in the gastrointestinal tract and its bioavailability is approximately 100%. Studies with healthy volunteers and patients with type 2 diabetes showed that peak serum of glimepiride is achieved around 1 hour after oral administration while maximum plasmatic concentration occurs within 2-3 hours. Glimepiride is transformed in the liver through a hepatic oxidative process into two active metabolites. The cytocrome involved is P 450 2C9. Pharmacokinetics of glimepiride is directly related to dosage in a therapeutic range between 1 e 8 mg/day and is well tolerated in both healthy volunteers and diabetic patients independently of food ingestion. Half-life is 5-8 hours. Comparison between a single daily dose and fractionated administration does not show significant differences in pharmacokinetic, with a small variability between individuals. Pharmacokinetic data is similar in subjects of different ages and/or sex. In patients with impaired renal function, clearance of glimepiride appears accelerated and mean plasmatic concentration is reduced. This is probably due to increased elimination caused by a reduction of the quote bound to proteins.20,21
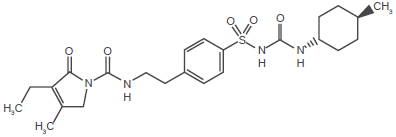
Glimepiride chemical structure.
Pioglitazone/glimepiride fixed dose combination
Bioequivalence studies have been conducted for single dose of pioglitazone/glimepiride 30 mg/2 mg and 30 mg/4 mg fixed dose combination in healthy subjects. Results obtained showed the single tablet combination was bioequivalent to pioglitazone and glimepiride association. 22
Clinical Studies and Efficacy in Metabolic Control
In current clinical practice, pioglitazone is usually administered in association with other drugs, in either double or triple therapy. 23 Data is available about a significant improvement of glycemic control in patients treated with pioglitazone in association with a SU.24,25 Hanefeld et al investigated 639 subjects treated with glimepiride. In these patients, the addition of pioglitazone to glimepiride significantly improved HbA1c, FPG and fasting plasma insulin (FPI). Compared with the association of metformin and glimepiride, combined treatment using pioglitazone and glimepiride provided significant reduction of Tg (p < 0.008), significant increase of HDL-C (p < 0.001), while LDL-C underwent an increase of 2% with the pioglitazone group and a reduction of 5% with the use of metformin. Treatment with pioglitazone and a SU led to a reduction of the albumin/creatinine ratio of 15%, while the same parameter was increased by 25% in subjects treated with pioglitazone and metformin (p < 0.017). 26
In a recent study by Derosa et al effects of pioglitazone and rosiglitazone in association with glimepiride were compared in 87 subjects with type 2 diabetes mellitus and metabolic syndrome. Both associations led to a significant reduction of HbA1c (1.3%, p < 0.01), FPG, FPI and PPI, without significant differences between treatments. Pioglitazone in association with glimepiride determined a significant reduction of TC (-11%), LDL-C (-12%) and Apo B (-10.6%) and a significant increase of HDL-C (+15%). In contrast; an observation was made of a significant impairment of the same parameters with rosiglitazone plus glimepiride. 27
Roberts et al evaluated efficacy and tolerability of glimepiride in association with metformin and pioglitazone or rosiglitazone in subjects who had not achieved optimal glycemic control with biguanide in association with pioglitazone or rosiglitazone. Results from this study confirmed that association of pioglitazone in patients treated with glimepiride and metformin provides a significant improvement of glycemic parameters with a reduction of HbA1c of 1.3%. Lipids parameters results were comparable between groups. Triple therapy tolerability resulted in an acceptable increase in the occurrence of hypoglycemia, but there was no noted increase in severity. This was observed in subjects treated with glimepiride compared to placebo. 28
A possible favourable role of association therapy with pioglitazone and glimepiride has been suggested in head-to-head studies. In a 12-month study comparing pioglitazone, glimepiride and metformin monotherapy in 114 subjects with type 2 diabetes mellitus, all agents showed equal effectiveness in reducing HbA1c. 29 Different effects have been elucidated for pioglitazone and glimepiride patterns of glycemic control, as demonstrated by Tan et al in another 12-month study, involving 244 Mexican patients. In this study, pioglitazone (titrated from 15 mg to 45 mg/day) provided a significant reduction of HbA1c, compared to glimepiride (titrated from 2 to 8 mg/day). Moreover, Homeostasis Model Assessment index (HOMA), Quantitative Insulin Sensitivity Check Index (QUICKI) and FPI were significantly ameliorated in subjects treated with pioglitazone. This data demonstrated that even if glimepiride induces a more rapid reduction of HbA1c, pioglitazone provides more efficacious and lasting glycemic control and is able to improve insulin-sensitivity. 30
Recent data suggests the association of pioglitazone and glimepiride might favourably affect lipid profile. Pioglitazone monotherapy benefits on lipid profile are well established in literature. The main effect of pioglitazone is a consistent increase of HDL-C and reduction of Tg. Even if a moderate increase in LDL-C has been reported in clinical trials, this is associated with a qualitative improvement of LDL particles which are shifted to a larger, less atherogenic pattern.31–34
Pioglitazone efficacy on lipid metabolism has been observed also in studies of combined therapy. In a 2-years study, pioglitazone in association with gliclazide provided sustained improvement in glycemic control, significant reduction of Tg and increase of HDL-C (p < 0.001). 35
In studies with glimepiride, data is available about favourable effects of this SU on lipid metabolism in patients with type 2 diabetes mellitus. 36
In a 12-month study, glimepiride in association with metformin provided a slight improvement in total cholesterolemia, which was not observed in the rosiglitazone-treated patients (p < 0.05). 17
The possible effects of pioglitazone and glimepiride on the so called “non-conventional” components of cardiovascular risk have simulated new lines of research among the experts. Recently published results showed that glimepiride improves plasma adiponectin (ADN) levels, especially in subjects with type 2 diabetes mellitus with low ADN concentrations before treatment, and may directly contribute to improvement of HDL-C. 37
In a recent study, involving 124 patients with type 2 diabetes mellitus, treatment with glimepiride was associated with a significant reduction of lipoprotein (a) [Lp(a)], PAI-1 ed homocysteine (HCT) after 6 months (p < 0.05), compared to repaglinide. 38 In a subsequent study, 91 type 2 diabetic subjects treated with glimepiride (4 mg/day) were randomized to pioglitazone (15 mg/day) o rosiglitazone (4 mg/day) as an add-on therapy. After 12 months, a significant improvement of HbA1c, FPG, PPG, FPI and HOMA index was observed in both groups. Levels of plasminogen activator inhibitor-1 (PAI-1) were significantly improved after 12 months in both groups, however no changes in t-PA and fibrinogen (Fg) concentrations were obtained with any treatment. 39
Furthermore, available data suggest a possible role of some glucose-lowering and insulin-sensitizing agents in reducing blood pressure (BP). Choosing an association therapy may be favourable in patients with type 2 diabetes mellitus and hypertension.
Thiazolidinediones were recently shown to be effective in reducing BP values in association with metformin compared to glimepiride. 40 In another study, 87 subjects with type 2 diabetes mellitus and metabolic syndrome treated with glimepiride (4 mg/day) were randomized to pioglitazone (15 mg/day) o rosiglitazone (4 mg/day). After 12 months of combined treatment, significant reduction of systolic blood pressure (SBP) and diastolic blood pressure (DBP) were observed (SBP -3.8 mmHg and DBP -3.2 mmHg for pioglitazone and glimepiride; SBP -4.4 mmHg for SBP and -4.8 mmHg for DBP for glimepiride and rosiglitazone) (p < 0.05). Both treatments provided significant reduction of HbA1c, FPG, PPG, FPI and PPI and HOMA index (p < 0.01, respectively). 41
Microalbuminuria represents another CV risk marker in type 2 diabetes and metabolic syndrome. 42
Association of pioglitazoneand glimepiride seems a promising therapeutic tool in the prevention of cardiovascular events in patients with microalbuminuria and/or moderate renal impairment, which is commonly observed in patients with recent diagnosis of type 2 diabetes mellitus. Combination treatment with pioglitazone and SU has been shown to be more effective in reduction of urinary albumin/creatinine ratio (-15%) compared to combination treatment of pioglitazone and metformin (+2%). 26 This may be partly explained by amelioration of insulin-sensitivity induced by TZDs. 43 To our knowledge, no studies have evaluated the possible effects of pioglitazone plus glimepiride combination treatment in patients with end-stage renal disease.
The possible role of glimepiride in reducing albumin/creatinine ratio has not yet been established. However, results from a study by Rosenkranz et al suggest that glimepiride administered to patients with moderate renal impairment is safe, according to pharmacokinetics of this molecule. 44
Both pioglitazone and glimepiride have been demonstrated to provide significant improvement in the coagulative state of patients with type 2 diabetes mellitus and metabolic syndrome.
Antithrombotic effects of glimepiride (2 mg/day) in association with metformin (1500 mg/day) have been compared to those of rosiglitazone (4 mg/day) and metformin in 95 subjects with type 2 diabetes. After 12 months, both treatments produced, a significant reduction of HbA1c, FPG, PPG, FPI, PP. The HOMA index was significantly ameliorated only in the group treated with rosigliazone. Moreover, glimepiride and rosiglitazone in association with metformin determined a significant reduction of PAI-1. 45
Safety and Tolerability
Oedema
Oedema represents one of the most common and undesirable side effects of treatment with a TZD. Oedema seems more common when TZD's are used in combination therapy. An increased incidence of oedema has been reported in studies with pioglitazone insulin. 46
There is evidence that the association of pioglitazone and glimepiride increase the risk of peripheral oedema. 30 Recent data shows that administration of combination therapy of pioglitazone and glimepiride is substantially safe and a correct management of patients, and is effective in the prevention of oedema. 47
Hypoglycemia
Hypoglycemia remains a key-issue in treatment with a SU. Glimepiride appears to have a lower risk of hypoglycemia, compared to the older SUs. 48 In a recent study Anwar et al compared repaglinide and glimepiride treatment in a group of Muslim patients with type 2 diabetes practicing fasting during Ramadan period. Repaglinide and glimepiride presented a similar risk of hypoglycemia during treatment. Moreover, the longer half-life of glimepiride provided a better control of glycemic values during 13.5 fasting hours. 49
In fasting type 2 diabetic patients, the risk of hypoglycemia with glimepiride appears comparable to repaglinide and insulin glargine, as recently demonstrated. 50
Osteoporosis
As with other TZDs, pioglitazone increases the risk of osteoporosis. The mechanism of how pioglitazone reduces bone mass density (BMD) is not yet well understood, however if this phenomenon is probably due to action of TZDs on differentiation of osteobasts. 51 Further studies are needed to better understand the mechanisms of bone loss associated with using pioglitazone. No studies are available that demonstrate a possible increase in risk of osteoporosis with glimepiride monotherapy, therefore impairment in bone metabolism with fixed dose combination treatment with pioglitazone and glimepiride can be considered comparable to that observed in clinical studies with pioglitazone monotherapy.
Hepatotoxicity
Hepatic dysfunction was a serious issue which led to withdrawal of troglitazone during clinical use; however cases of hepatotoxicity with pioglitazone have been few in number and less severe in consequence when compared to troglitazone. 52
Results from clinical studies of association of TZDs and SUs are encouraging in regard to the risk of liver injury. 53 The report of occasional cases of liver injury in literature suggests a small risk of hepatotoxicity with glimepiride. 54 Data from clinical studies suggest that pioglitazone in association with glimepiride present a safe profile of hepatotoxicity 55 and results of a recent long-term clinical trial has shown that pioglitazone improves markers of liver function in patients with type 2 diabetes. 56 To date, no evidence is available about an increase risk of liver injury in patients treated with fixed dose of pioglitazone and glimepiride. In 2005, an open, phase III, multicentric study was conducted to determine the efficacy and tolerability of the triple drug combination glimepiride 2 mg/day plus pioglitazone hydrochloride 15 mg/day plus metformin SR 500 mg/day in 101 Indian patients with type 2 diabetes mellitus. The combined treatment allowed achievement of recommended goals in glycemic control and lipid metabolism. There were no cases of serious adverse events and drug interactions reported. 57
Body weight
A possible side effect of combination treatment with pioglitazone and metformin is represented by an increase in body weight, which may be not tolerated by patients. Increase in body weight for pioglitazone is accompanied by amelioration in insulin-sensitivity and body fat pattern, with a reduction of visceral, atherogenic, fat and increase of subcutaneous fat. 58
Sulphonylureas induce amoderate weight gain, primarily due to a reduction of urinary loss of kilocalories, which improves with better glycemic control. Weight gain presents individual variation and in a study on 1000 patients with type 2 diabetes mellitus treated with glimepiride, optimal glycemic control was obtained without significant increase in body weight. 59 Moreover, increase in body weight is easily contained through compliance to dietary and life-style advice. 60
Beta-cells Functionality
One of the most discussed problems in pharmacological treatment of type 2 diabetes is preservation of pancreatic β-cell function. Sulphonylureas mechanism of action implicates a progressive exhaustion and desensitisation of β-cells. This phenomenon has been definitively demonstrated for glibenclamide.61,62 In addition, prolonged exposure to SUs renders β-cells less responsive to subsequent sulfonylurea stimulation. 63 Some data suggest that, glimepiride, as with other SUs, stimulates reactive oxygen species production via protein kinase C-dependent activation of NAD(P)H oxidase thus leading to β-cell apoptosis in vitro. 4
In regard to pioglitazone, data from several sources indicate the role of pioglitazone in preservation of β-cells, through a direct way and action on other metabolic components as FFA concentration. In vitro and in vivo studies demonstrate that pioglitazone restores insulin secretion impaired by lipotoxicity and can protect β-cell from apoptosis induced by high levels of glucose.5,64
Moreover, results from clamp studies and studies investigating the oral glucose tolerance test, as well as indirect measures of β-cell function such as HOMA index, confirm improved β-cells function with pioglitazone.65,66 In conclusion, TZDs and SUs exert differential effects on β-cell performance over time and, according to available data, association of pioglitazone and glimepiride may be more beneficial for β-cells functionality compared to glimepiride monotherapy; conversely, however, starting therapy with glimepiride allows a more rapid achievement of glycemic control than pioglitazone monotherapy.
Conclusions
Cardiovascular risk and TZD
As previously described, both pioglitazone and glimepiride have been demonstrated to be effective in achieving glycemic control and ameliorating lipid profile in type 2 diabetes mellitus. In the last two years, reviews of the literature have developed a still open question about the cardiovascular risk with TZDs. We have considered this issue in the present manuscript as it has developed interesting discussion about pioglitazone safety and efficacy in patients at high cardiovascular risk.67,68 Pioglitazone efficacy in prevention of cardiovascular events has been largely demonstrated in the Secondary prevention of macrovascular events in patients with type 2 diabetes study (PROactive). 69 Data from the PRO-active research, as explained above, demonstrated a favourable impact of this molecule on cardiovascular risk. In particular, long term treatment with pioglitazone seems to reduce the risk of AMI. In a recent meta-analysis, pioglitazone results associated a reduced risk of death, AMI or stroke among a diverse population of patients with type 2 diabetes. However some research data indicated that the risk of heart cardiac failure is increased with pioglitazone, without an increase in mortality from heart failure. 70
A recent meta-analysis of 42 randomized control trials in among 28443 patients treated with rosiglitazone has shown an increase in the risk of AMI in subjects treated compared to controls (p = 0.03). 67 A subsequent meta-analysis has included randomized clinical trials with a specified intention to evaluate CV events in diabetic and non-diabetic subjects (pre-diabetic). Studies included had almost a 12-months follow up period and provided numerical data of CV side effects. Results of this analysis, according to Nissen et al has shown that rosiglitazone increases the risk of AMI (p < 0.001) without raising the risk of CV mortality (RR 0.90; CI 95%:0.63-1.26; p = 0.53). Limitations of collecting data from the studies included in the meta-analysis and the methodological limitations have been analyzed in the paper. 68 After having considered this data, the rosiglitazone label was modified by the FDA association in August 2007. 71 In a recently published review of 20 case reports and 10 uncontrolled clinical studies, it was observed that, among the cohort analyses, rosiglitazone was associated with an increased incidence of lower extremity oedema, pulmonary oedema, and patients required treatment for heart failure. In regard to the risk of AMI, in one study published in the same year, no difference was found in the hazard risk of AMI between patients treated with rosiglitazone, alone or in association with insulin, metformin or SUs. 72 In long-term studies looking at the association of pioglitazone plus glimepiride and rosiglitazone plus glimepiride, no cardiovascular side effects were observed among participants, and in general no adverse effect resulted in study withdrawals. 39
In a study by Roberts et al add-on therapy with glimepiride did not produce serious adverse effects among participants who were previously treated with metformin plus pioglitazone or rosiglitazone, compared to a placebo, but no direct comparison between TZDs was performed. 28
In conclusion, despite mounting evidence in literature and additional analyses of clinical studies, the data supporting the cardiovascular safety of rosiglitazone, alone or in association with metformin or SU, is still inconclusive. 73
Cardiovascular Risk and Glimepiride
As previously reported in this manuscript, in patients with a history of AMI, treatment with SUs might impair the so-called “cardiac preconditioning” that is the exposure of myocardial tissue to brief, repeated periods of vascular occlusion in order to render the myocardium resistant to the deleterious effects of ischemia or reperfusion. The risk of impaired myocardial preconditioning has been confirmed for some first-generation SUs such as glibenclamide. Glimepiride and, in general, second-generation SUs present a lower affinity for myocardial SURs with an increased safety profile in subjects with type 2 diabetes mellitus and coronary disease. 74 The role of glimepiride and SUs in CV prevention is still being debated, in particular after the contrasting results obtained in two large clinical trials, the Action in Diabetes and Vascular disease: PreterAx and DiamicroN Modified Release Controlled Evaluation (ADVANCE) and the Action to Control Cardiovascular Risk in Diabetes (ACCORD). 75 Moreover, there is evidence that SUs with greater selectivity for β-cell receptors, such as glimepiride and gliclazide, are associated with a lower mortality when used in combination with metformin in comparison with glibenclamide even if further investigation are needed in this field. 76 Recently published results of the Pioglitazone Effect on Regression of Intravascular Sonographic Coronary Obstruction Prospective Evaluation study (PERISCOPE) clearly demonstrated that treatment with pioglitazone results in a significantly lower rate of progression of coronary atherosclerosis compared with glimepiride. 77 This information about a possible role of glimepiride in ischemic precondition is available from results obtained in experimental studies with animal models. 78 There are only few important studies 79 that evaluated the effect of glimepiride in ischemic preconditioning in real-life myocardial ischemia, so that the clinical relevance of these findings has still to be elucidated.
Among sulphonylureas, glimepiride showed a minimal influence on risk of blockage of calcium channels in myocardial cells and data about the safety of use of glimepiride in patients with coronary artery disease is available. Abbink et al showed that treatment with glimepiride (6 mg/day) produced similar effects of metformin (1500 mg/die) and glibenclamide (15 mg/die) on brachial vasodilatatory response to diazoxide, acetylcholine, al dipyridamole and ischemia. 80 In addition, glimepiride seems to enhance PPAR-γ activity. 81
Place in Therapy
In conclusion, combination therapy with pioglitazone and glimepride appears a promising alternative in type 2 diabetic patients at high cardiovascular risk. The complementary action on reduction of HbA1c of these two drugs allows a more rapid achievement of glycemic targets, and subsequently, a more sustained glycemic control over time. This probably is related both to a potential magnification of pioglitazone PPAR-γ activity by glimepiride and a better effect of pioglitazone on β-cells functionality. In regard to amelioration of lipid abnormalities, data from clinical trials is not conclusive, even if a substantial improvement is usually observed both with glimepiride and pioglitazone in monotherapy and association trials, as reported above. To date, no studies have been published that directly compared fixed dose association of TZDs and glimepiride or TZDs and metformin in reducing lipid alterations or cardiovascular risk. Since cardiovascular efficacy and safety of TZDs is still being debated among the experts, the role of fixed combination of pioglitazone and glimepiride is to be elucidated in further studies.
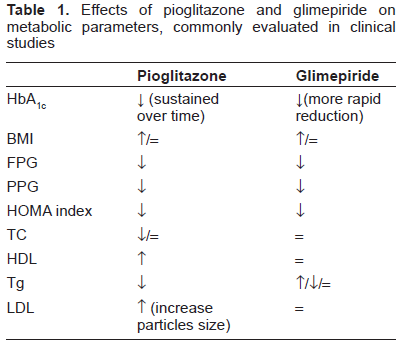
Furthermore, both evidence from literature and clinical experience suggest that combination treatment with pioglitazone and glimepiride is generally well tolerated and safe in patients with type 2 diabetes mellitus at a high cardiovascular risk when label instructions are considered (Table 1).
Effects of pioglitazone and glimepiride on metabolic parameters, commonly evaluated in clinical studies
Patients Perspectives
Non-adherence to oral glucose-lowering agents is a common problem in clinical practice, and more than 50% of patients are considered non-adherent.82,83
Pioglitazone plus glimepiride is available in tablets (30/2 mg and 30/4 mg) and fixed-dose combination therapy appears more suitable for type 2 diabetic patients who usually are treated with multiple tablets. So that, in addition to the complementary pharmacological effects of drug components, a better glycemic and metabolic control is more probable as patients compliance is increased. 84
Funding Disclosure
The authors certify that have no affiliation with, or financial involvement in, any organization or entity with a direct financial interest in the subject matter or materials discussed in the manuscript.
Disclosure
The authors report no conflicts of interest.
