Abstract
Clobetasol-17 propionate is a dihalogenated corticosteroid which, on the basis of the vasoconstrictor assay, is classified as the most potent of all topical steroids. Since 1973 it has been marketed specifically for the short term topical treatment of moderate and severe cortico-responsive dermatoses for its antinflammatory, immunosuppressive and antimitotic effects. Psoriasis, atopic dermatitis, chronic eczema, boullous disorders and itching dermatosis of the genital area are usually well controlled by clobetasol with a great improvement of quality of life in patients.
In this manuscript, we reviewed data concerning pharmacology, efficacy and safety profile of clobetasol-17 proprionate, updated studies on its use, efficacy of different formulations and impact on quality of life in corticosteroid-responsive dermatoses.
Introduction
In the last six decades topical corticosteroids have been the treatment of choice for many dermatoses, namely for those characterized by dry, scaly, crusted or ertythematous skin and usually associated with inflammation and pruritus. Psoriasis, atopic dermatitis, chronic allergic contact dermatitis, lichen simplex chronicus, prurigo or lichen sclerosus of the genital area usually affect patients’ quality of life and consume considerable economic and medical resources. When cortisone became available 60 years ago dermatologists quickly sensed the potential of the associated class of compounds since the parent compound, cortisone, unfortunatly demonstrated no topical activity because of its very slow rate of absorption in vivo.1,2 Shortly after negative trials with cortisone, hydrocortisone became available and its efficacy was immediately and firmly established in 1952 by Sulzberger and Witten for the treatment of several inflammatory diseases. 3 In the following years, increasingly potent topical steroids were experimented with an enthusiasm that was nevertheless tempered by the appreciation of potential side effects. The 1960s were the golden era for glucocorticoid with the introduction of the first synthetic congeners (e.g. prednisolone) followed by fluorinated derivates, (e.g. dexamethasone), possessing much more potency but in many cases these compounds have not been completely studied with regard to their exact mechanism of action.
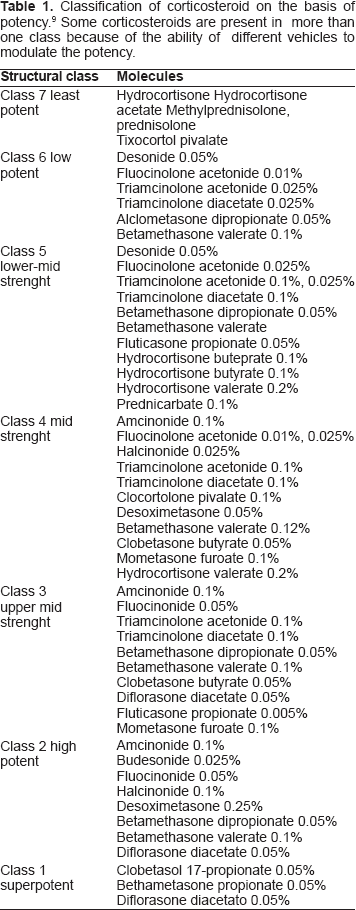
The vasoconstrictor assay measures the skin blanching effect of ethanolic extracts of corticosteroid preparations reflecting the intrinsic potency of steroid molecules. According to this assay the currently marketed topical steroids are classified on the basis of their relative potency and efficacy (Table 1).4–6 Demonstrated to be 1,800 times more potent than hydrocortisone, clobetasol 17-proprionate (CP), a fifthgeneration corticosteroid, is generally acknowledged to be the most potent topical steroid althought associated with a high potential risk of topical and systemic side effects.7–9
Classification of corticosteroid on the basis of potency. 9 Some corticosteroids are present in more than one class because of the ability of different vehicles to modulate the potency.
In this review we report data about its pharmacology and safety profile, we review updated studies on clinical efficacy and we discuss the favorable effects on impact on quality of life and on medical resources in corticosteroid-responsive dermatoses.
Pharmacology and Factors Influencing Clinical Activity
Clobetasol 17-propionate is a dihalogenated corticosteroid which has been marketed since 1973 specifically for the short term topical treatment of inflammatory and pruritic manifestations of moderate and severe cortico-responsive dermatoses. 10 Analog of prednisolone, it has an empirical formula of C25H32O5CIF with a molecular weight of 467; its chemical structure is shown in Figure 1. Studies in animals have demonstrated that approximately 50% of radiolabeled CP topically applied is sistemically absorbed with a very low peak plasma that occurs 48 hours after topical application; radioactivity in animals is still present after 168 hours with a return to negligible levels after 240 hours which equals 10 days. 11 In humans and in normal skin condition, the application of 35 mg of clobetasol is followed by absorption peaks after 5 and 15 hours with ointment, and 11 hours with cream, with a peak plasma levels of 0.6 to 0.7 μg/dl. 12
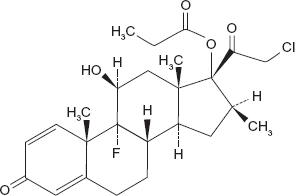
Chemical formula of clobetasol-17 propionate. Empirical formula of C25H32O5CIF with a molecular weight of 467.
The extent of percutaneous absorption is determined by many critical factors such as formulation, vehicle, anatomical district of application, integrity of the epidermal barrier, use of occlusive dressing and concentration and frequency of application. 13
Ointments were usually believed to be more potent in comparison to lotions and creams because of their occlusive effects and hydration of the stratum corneum. Nevertheless, this dogma is no longer accepted since it was demonstrated that the absorption can be determined by the overall ability of the vehicle to deliver the active agent and this depends on different variables: a) suspension of the molecule within the vehicle b) solubility of the vehicle and its partition coefficient with the skin c) lipophilicity and solubility of the steroid molecule d) the presence of penetration enhancer. 14 Clobetasol propionate is commercially available as 0.05% ointment, cream, gel, lotion, spray, emollient cream, foam and a new emulsion foam 0.05% formulation with a greater absorption rate (Table 2). 15 Its use is not indicated in children younger than 12 and it should be cautiously used in patients of any age on body areas of increased permeability such as the face, especially eyelids, skin folds or genital mucosa that usually clear faster and better in comparison to others such as dorsa of the hands, extensor surfaces of the knees, elbows, trunk, palms and soles.16,17 As already reported by Hehir et al, 10 inflammation and/or other pathological conditions which impair the epidermal barrier may enhance the percutaneous absorption. The application of 25 mg of CP on skin affected by atopic dermatitis and psoriasis lead to a rapid rise in plasma levels during the first three hours and peak levels between 0.6 and 15.8 ng/ml with return to baseline levels by 96 hours. By increasing CP concentration, an increased efficacy may be observed and this usually happens but not with a linear rate since a plateau of activity may be recorded at a relatively low concentration. 18 In these cases a method for intensifying the effectiveness up to tenfold 19 is, for example, the use of occlusive dressing by a plastic film. Owing to a “reservoir effect” clobetasol applied under occlusion can reside in the stratum corneum for up to two weeks. 20 The frequency of application can also influence the potency of clobetasol propionate, this has been investigated and will be reported forward in this paper.
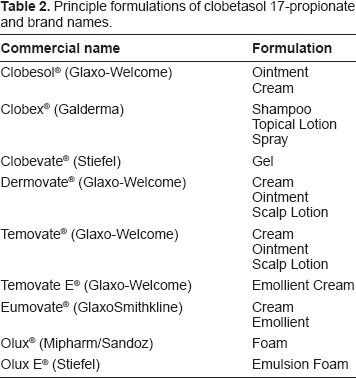
Principle formulations of clobetasol 17-propionate and brand names.
Mechanism of action of clobetasol propionate
Once applied on the skin and absorbed, CP exerts local and systemic effects, mainly vasoconstrictor, antinflammatory, immunosuppressive and antimitotic effects (Table 3). As for other corticosteroids, the specific biological activities of CP are brought about by the interaction with glucocorticoid receptor (CG-R) in the cytoplasm. 21 The unliganded receptor is a heterotetrameric complex comprised of heat-shock proteins (hsp), hsp70 e hsp90, and chaperone immunophilins. Once formed, the steroid-CG-R complex transolocates to the nucleus and interacts directly with DNA where by a conformational change of the receptor, it determines the shedding of the DNA-binding domain capping protein, hsp90. Exposure of the DNA-binding site allows the binding of the CG-R complex to the GC response element (GRE) that stimulates alteration in transcription of protein either negatively or positively. 22
Main actions of topical corticosteroids.

The lipocortin 1 (LC1), a member of the annexin 1 family and also known as annexin 1, is an important antinflammatory protein induced in this way by steroids. Lipocortin 1 seems to have different localisation in keratinocytes on the basis of physiological and pathological conditions, being localised preferentially on the cell membrane to counteract the actions of phospholipids and prostanoid production during inflammatory states. 23 By induction of LC1, CP suppresses the production of phospholipase A2 (PLA2) and cyclooxygenase-2 (COX2), both enzymes involved in the phospolipid-arachidonate lipid mediator cascade. A direct effect of clobetasol on the expression on the PLA2 gene that possess a GRE, has also been demonstrated. 24 The inhibition of PLA2 attenuate the production of leukotrienes and platelet-activating factor, other important mediators of inflammation. 25 The effect on COX2 seems to be due to a decrease in the stability of the mRNA rather than to an effect on transcription per se. 26
Clobetasol propionate and other glucocorticoids exert and important inhibitory action on two essential regulatory transcription factors for several genes involved in the inflammatory process, Activator Protein 1 (AP1), and Nuclear Factor kappa B (NFκB). The GC-R interacts directly with AP-1 and NFκB and indirectly upregulating IκB transcription, sequestering NFκB thereby depressing the levels of free NFκB available for binding to target inflammatory genes. 27 The immunosuppressive action of CP is induced via inhibition of inflammatory cytokines and subsequently by change of cellular population in the site of inflammation. Several studies have demonstrated that the transcription of IL2, IL6, IL8 is lowered by inactivation of AP1 and NFκB transcritption factors or in case of IL6 also by direct interaction, due to the presence of a GRE element in the gene of this interleukin and finally throught the enhancement of the mRNA instability of several cytokines.28–30
The modulation of mast cell number and activity has been proposed as a further antiflammatory mechanism of clobetasol with a consequent inhibition of degranulation of hystamine and other pro-inflammatory mediators into local environment, usually elevated in allergic contact dermatitis and in pruritus. 31 Clobetasol propionate modulates cutaneous immune reactions and the capacity of epidermal cells to present tetanus toxoid to antigen presenting cells and to augment concanavalin A mediated lymphocyte stimulation of autologous lymphocytes. This action was associated with a reduction in the number of HLA-DR/T6 positive LCs and with a marked decrease in Langerhans cell-dependent T lymphocyte activation. Therefore CP was able to inhibit not only the elicitation phase but also the induction phase of allergic contact dermatitis. 32
Nitric oxid (NO) plays a pivotal role in physiological and pathological conditions mediating vasodilatation, inflammation and immunomudulation and oxidative damage to cells and tissues. Synthetised by a family of enzymes that includes three isoforms, the endothelial eNOS, the neuronal nNOS, together know as collective cNOS, and the inducible iNOS, NOS is expressed in the skin and it is involved in basal dermal blood flow. It is elevated in several dermal pathologies and it has been postulated that it can contribute to the pathogenesis of erythema and oedema of psoriasis and atopic dermatitis. Clobetasol inhibits specifically the synthesis and production of iNOS whilst leaving cNOS activity untouched, and the resolution of inflammation induced by CP may be due, at least in part, to this action.33,34
Clobetasol has profound anti-proliferative effects on cultured cell lines. Studies in normal skin showed that topical steroids resulted in a significant decrease in epidermal mitosis in comparison with that in vehicle controls and similar findings have been shown in psoriatic skin. Studies using hairless mouse comparing the mitotic index and 3 H-thymidine uptake in normal and steroids treated skin suggest that the the action on cellular turn over can be a non specific cell cycle effect or a G1 interphase period. 35
The amount of CP sistemically absorbed after topical application binds to plasma protein in varying degrees, then it is metabolized primarily in the liver and it is excreted by the kidneys and in the bile.
Safety and tolerablity
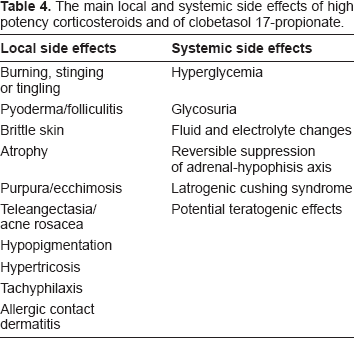
The very high potency of CP is accompanied by local and systemic side effects. In a meta-analysis of controlled clinical trials that included patients with psoriasis or eczema and healthy volunteers treated or tested with an emollient formulation of CP, adverse events judged to be possibly or probably related to CP emollient treatment occurred in only 1.1% of study subjects. 36 The local side effects have also been investigated during short-term (<3 weeks) application of clobetasol propionate without occlusion. Burning, stinging or tingling and less frequently pyoderma/folliculitis, brittle skin and/or “crasking” have been reported; after occlusion development of atrophy have been observed. 37 Very common side effects recorded with long treatment and/or chronic use are atrophy, onset of purpura and ecchimosis, teleangectasia, “acne rosacea”, hypopigmentation, hypertricosis, increase of intraocular pressure after periocular application and increased susceptibility to cutaneous infections. Allergic contact dermatitis has been described and probably it is thought to be more frequent than expected. 38 It should always been confirmed by patch test using high concentration of clobetasol (>1%). 39
As for the other high potency corticosteroids, CP can have systemic side effects such as hyperglycemia, glycosuria, fluid and electrolyte changes, and hypothalamic-pituitary-adrenal (HPA) suppression may occur. This event is more likely in children and when the topical treatment is prolonged more than two weeks and/or on extensive body areas.40,41 A teratogenic effect has been demonstrated in laboratory animals at relatively low dosage levels and after dermal application at very low doses. Its percutaneous absorption limits its use in nursing mothers since can be secreted into the breast milk and interfere with endogenous corticosteroid production. 42 Finally patients often tell about reduced effectiveness of topical steroids on repeated use. Tachyphylaxis to these agents has been demonstrated in humans for vasoconstriction and histamine-induced wheal suppression in normal skin and inflammed skin. 43 The side effects of corticosteroids are summurised in Table 4.
The main local and systemic side effects of high potency corticosteroids and of clobetasol 17-propionate.
Efficacy of Clobetasol Propionate in Corticosteroid-Responsive Dermatoses
Psoriasis
Psoriasis is a common multifactorial skin dermatitis with a strong genetic background whose prevalence is estimated to be 2%–2.8%. 44 The treatment of psoriasis is variable and depends on the extension and the clinical forms but it is estimated that 75% of patients with psoriasis has mild to moderate clinical form that can be satisfactorily managed on an outpatient basis with topical therapy. 45
Clobetasol propionate is the most common topical therapy used for psoriasis in many countries of the world. Its superior efficacy in comparison to other topical corticosteroids has been largely demonstrated.46–48 It can be used as monotherapy, in combined regimens or as adjunctive therapy with UVB and PUVA. 49 The concentration commonly applied is 0.05% but a concentration up to 8% can be reached in laquer for nail psoriasis. 50 Many different formulations are currently available: ointment, cream, gel, lotion, spray, foam, emulsion foam and shampoo. A special formulation is the one obtained with a hydrocolloid occlusion medication applied once a week that is associated with high effectiveness but also high rates of rebound.51,52
With regard to the monotherapy with CP, many studies, evaluating efficacy of different formulations on patients with moderate to severe plaque psoriasis, have been published in the last ten years. Decroix et al 53 compared the efficacy, safety, and cosmetic acceptability of CP lotion compared to its vehicle and CP cream in 222 patients. After 4 weeks of treatment, lotion was more efficient than vehicle and of equivalent efficacy as CP cream, but overall cosmetic acceptability was significantly better with lotion. A following multicentre, investigator-blind, randomized, active- and vehicle-controlled, parallel-group study on 192 subjects demonstrated the superiority and better remission profile of this last formulation in comparison to cream. 54 Two advanced formulations of CP 0.05%, spray and foam, were compared in a study of 77 randomized psoriasis patients. At the end of the treatment period (2 weeks for foam or up to 4 weeks for spray), patients treated with CP spray had a median 64% reduction in affected body surface area compared to a median 25% reduction in patients treated with the CP foam with improvements in quality of life statistically significantly greater at all time points to spray-treated patients. 55 Confirming previous results by Jarrat et al, 56 the Clobex Spray Community-Based Research Assessment (COBRA) trial, conducted on 1254 subjects, demonstrated the efficacy of CP spray 0.05% applied twice-daily as monotherapy. 57 A more recent work 58 compared the efficacy of CP spray 0.05% versus calcipotriene 0.005%-betamethasome diproprionate 0.064% (C-BD) ointment, showing a higher rating (75%) of clear or almost clear after 4 weeks of treatment in CP spray-treated patients. The foam formulation was used in a twice daily application in 279 patients of a multicenter, randomized, double-blinded, placebo-controlled study that showed the high effectiveness of CP foam 0.05% for scalp and non-scalp areas. The shampoo formulation was well tolerated and was more effective and better tolerated than calcipotriol solution with an improvement of quality of life in the short contact therapy of scalp psoriasis.59,60 Recently Warino et al 61 reviewed the current literature and assessed the relative potency of clobetasol propionate ointment compared to other clobetasol propionate preparations in the treament of psoriasis. They analyse only studies reporting the percentage of subjects that achieved desired efficacy endopoints and subjects’ mean change in symptoms from baseline, excluding studies before 1980 or with concomitant therapies. They found that the efficacy of clobetasol proprionate varied for the different vehicles ranging from 17% to 80% and they concluded that ointment preparations have similar efficacy to other preparations in clinical trial and that whichever vehicle patients find preferable should be chosen.
With regard to the different regimens of CP and its use in combined treatments, many studies have been reported in literature. In an open multi-centre trial, 316 psoriasis patients were treated with clobetasol propionate intermittently over a 14-day period. The treatment gave rapid clinical healing of the psoriasis infiltration in 62% of the patients. A total of 132 of these patients was subsequently put on maintenance treatment with two applications per week. This regimen kept the disease in remission throughout the observation period, which exceeded 4 months on average, in 75% of the patients. 62 A prospective randomized trial of anthralin in Lassar's paste compared with anthralin in 0.0125% clobetasol propionate showed a superior activty of this last regimen with a mean time to clearance of 14.9 days compared with 18.5 days, and with lower concentrations of anthralin. No significant difference was found in the rate of relapse (80% within one year) of the two treatment groups. 63 In a Norvegian randomised, double-blind study right- versus left-side comparison of two different ointments, CP and calcipotriol 50 microg/g bd, was carried out. The ointments were applied to two simmetrically-located areas for two weeks followed by a 4-weeks period of calcipotriol in 49 psoriasis patients. The two weeks of treatment with CP produced a significantly greater decrease in total symptom score than calcipotriol treatment (P < 0.0001) and this result was maintained throughout for the calcipotriol treatment period showing that the combined regimen was superior to calcipotriol ointment alone. 64
In a double-blind, randomized, parallel-group study 50 patients, with psoriasis on sites other than the face and scalp, were treated with tazarotene 0.1% gel plus clobetasol propionate 0.05% ointment for 6 weeks. The frequency of application of both medications was initially once daily and then was tapered to ensure gradual weaning. For the subsequent double-blind maintenance phase, patients were randomized to receive one of three maintenance regimens for 20 weeks respectively with tazarotene/clobetasol, tazarotene/vehicle, and vehicle. The first regimen showed to be superior in mantaining remission of psorasis. 65
Swinkels et al studied a dithranol regimen combined with CP in 12 patients with stable psoriasis vulgaris treating three comparable lesions on the extremities for 39 consecutive days. The first lesion was treated daily with short-contact dithranol cream followed by CP ointment 5 days per week. The second lesion was treated daily with short-contact dithranol cream followed by the vehicle of CP ointment. The third lesion was treated with CP ointment 5 days per week. Analysing several statistical parameters the authors concluded that intermittent addition of clobetasol-17-propionate ointment enhanced the antipsoriatic efficacy of short-contact, high-dose dithranol therapy in terms of clearing capacity and treatment duration, without shortening remission duration. 66 A prospective, single-center, open-label, right/left comparison of 28 lesion pairs in 15 patients was conducted by Bowman et al. They treated psoriasis patients for a 2-week period, followed by a 4-week post-treatment observation phase. Calcipotriene- and tazarotene-treated lesions showed nearly identical efficacy with their matched clobetasol-treated counterparts but erythema improved significantly more in clobetasol-treated lesions (P < 0.05). Differences became statistically insignificant during the post-treatment period (P = 0.20). All lesions worsened and plaque elevation returned somewhat more rapidly in calcipotriene- and tazarotene-treated lesions (P < 0.01), whereas other clincial parameters were not significantly different between the two treatment groups (P > 0.05). 67 Recently, we retrospectively reviewed the efficacy, and potential advantages and disadvantages of four regimens topically applied in a total of 666 psoriatic patients admitted for the first time over 15 years. 68 The regimen were respectively: 1) eosin 2% + clobetasone propionate on alternate days with eosin 2% + cade Oil; 2) eosin 2% + clobetasone propionate daily; 3) eosin 2% + clobetasone propionate on alternate days with eosin 2% + calcipotriene; 4) eosin 2% + vaselin daily. We calculated also the hospitalization time and we analysed the correlation with the response to the treatments. Even if a statistically significant response was recorded for every regimen, we found that the best combination was clobetasol propionate plus eosin on alternate days with eosin plus cade oil. The average length of treatment was of 7.5 days in all patients but no statistically significant difference among the different treated groups was recorded with respect to the length of hospitalization and PASI then a potential benefit due to hospitalization per se could not be excluded at all.
Atopic dermatitis
Atopic dermatitis (AD) is a common disease of childhood characterised by pruritus and xerosis that is marked by alternating periods of relapse and remission. Most AD is mild and can be managed with the use of emollients and standard therapy consists of topical corticosteroids or new topical calcineurin inhibitors. The efficacy of CP in treating AD has been evaluated in several studies. In children the application of one daily CP 0.05% without occlusion, as with other potent topical steroids, is accompanied by a fall in plasma cortisol. 69 In a double-blind, parallel-group, multicenter comparative trial in 127 evaluable patients with chronic, localized atopic dermatitis or lichen simplex chronicus, healing was reported in a higher percentage of patients treated with halobetasol propionate ointment than in those in the clobetasol propionate treatment group (65.1% versus 54.7%). Nevertheless the overall success rates were practically identical in the two treatment groups (93.7% versus 92.2%) with identical early onset of therapeutic effect within 3 days of the start of treatment and side effects. 70 Two studies demonstrated the high efficacy of CP emollient cream in moderate-to-severe atopic dermatitis also when compared with the vehicle.71,72
A comparative study between CP emollient cream formulation and lotion vehicle applied twice-daily for 2 weeks in the treatment of moderate to severe AD was carried out in 229 subjects. This multicenter, randomized, active- and vehicle-controlled, investigator-blinded study showed that lotion was effective, safe, well tolerated offering a better remission profile as compared to clobetasol propionate emollient cream. 73 Kimball and coll. evaluated safety and efficacy of CP emulsion foam 0.05% to treat steroid-responsive dermatoses in multiple age groups. A phase II open-label study evaluated the effect in 52 participants aged 6 years or older with mild-to-severe atopic dermatitis (AD). Another phase II open-label pharmacokinetic safety study was conducted in 32 participants aged 12 years or older with mild-to-moderate plaque-type psoriasis. Two phase III, randomized controlled studies assessed treatment success in participants aged 12 years or older with moderate-to-severe AD (N = 377) or mild-to-moderate plaque-type psoriasis (N = 497). In all studies, participants received study drug for 2 weeks. In the AD study and psoriasis study treatment success was determined using similar scales and improvement of at least two grades from baseline was considered remarkable. Significantly more participants achieved treatment success on clobetasol foam than vehicle foam and reversible HPA suppression was observed in 27% of participants aged 18 years or older and 47% in participants aged between 6 and younger than 12 years, but 0% in participants aged between 12 and younger than 18 years. These results renforced the findings that CP, like other class I topical corticosteroids, is not indicated in children under 2 years. 74
Allergic contact dermatitis and chronic eczema
A very few controlled studies have been carried out in the treatment of allergic contact dermatitis with 0.05% CP. The effectiveness of CP ointment was investigated in experimentally induced Rhus dermatitis and a rapidly decreased of the vesiculation, most prominent at the site to which clobetasol was applied the earliest (12 hours after exposure to Rhus extract) was recorded. 75 A comparative study evaluated the effect of CP in 21 volunteers with positive patch test reactions to nickel sulfate 5%. At six sites (three on each arm) different medications consisting in pimecrolimus 1% cream, tacrolimus 0.1% ointment, clobetasol propionate 0.05% ointment, and triamcinolone acetonide 0.1% ointment, as well as control preparations (Vanicream and petrolatum), were applied to one nickel site twice daily for 14 days. Most reactions were coded as resolved or as almost resolved by the end of the treatment regardless of the medication but a clear trend was observed for sites treated with active drug to do better than control sites. 76
Hand eczema is a common skin disease that tends to become chronic and may interfere with many types of work. Several etiological diagnosis are possible such as allergic contact dermatitis, irritant contact dermatitis, atopic dermatitis, unclassified eczema, nummular eczema, hyperkeratotic eczema or pompholyx. Emollients have been shown to be useful in reducing eczema activity and in the primary prevention. Protection of the hands is a fundamental aspect of the treatment but topical corticosteroids are still the mainstay. A few studies on their rational use, efficacy and side-effects have been conducted. The clinical effect of CP 0.05% versus flupredniden acetate was studied in the maintenance therapy of patients with chronic hand eczema. An initial regimen of continuous CP for 1 to 3 weeks which brought about healing in 90% of cases was chosen. In a subsequent double-blind left/right study, the capacity of the two corticosteroids for keeping the dermatitis in remission was compared using an intermittent schedule of 2 applications a week. The protocol was followed by 46 patients and the mean observation period was 138 days. Treatment with clobetasol kept patients free from relapses during the entire observation period in 70%, with flupredniden in 30%. Relapses occurred with clobetasol after a mean of 66 days, versus 36 days with flupredniden. 77 An open, randomised, third party blinded, left-right sided study compared the therapeutic efficacy of mometasone furoate cream 0.1% with CP cream 0.05% applied twice daily following a 3-week course of therapy in sixty consecutive patients with moderate to severe bilateral chronic eczema on the limbs. Overall, 53% of patients considered the mometasone furoate treated sites to be good or excellent vs. 88% for CP treated sites. 78 A double-blind, right to left, prospective, clinical trial studied a total of 47 patients randomly treated with either the ‘0.05% Clobetasol + 2.5% zinc sulphate’ cream or ‘0.05% Clobetasol alone’ cream twice daily for 2 weeks. The combination regimen was more effective than ‘0.05% Clobetasol alone’ cream (P < 0.05) with a significantly lower recurrences rate. 79
Lichen sclerosus genitalis
Lichen sclerosus (LS) is an inflammatory disease of unknown etiology affecting the anogenital skin in prepuberal girls, adult and postmenopausal females and old males. It can be associated with the development of squamous cell carcinoma in adult subjects. Super potent topical steroids are an effective treatment for many patients with lichen sclerosus, but concerns exist about their long-term safety. In girls the efficacy of a regimen based on CP ointment 0.05% was analysed in two small sized studies that showed excellent results with a 4-week period and 8-week period treatment but common recurrences and risk of atrophy obliged the physician to a continuing observation, every 6 to 12 months, in order to monitor recurrences and scarring complications.80,81
The treatment of vulval LS in adult females with CP have been reported since 1990.82,83 Subsequent studies investigated the efficacy of CP in comparison to testosterone proprionate 2%. A good efficacy was associated to testosterone in relation to symptoms even if no significant change was observed in relation to clinical aspects and histological alterations as observed on the contrary with CP without any noteworthy collateral effects. 84 Clobetasol 17-propionate 0.05% was also superior to testosterone for both remission induction and maintenance therapy in another study. 85
A randomized study was conducted on 79 patients with vulval LS treated for three months with four topical drugs, testosterone (2%), progesterone (2%), clobetasol propionate (0.05%) and a cream based preparation. Patients treated with clobetasol had a better response than the other treated groups. Remission of symptoms occurred in 75% of patients treated with CP compared to 20% treated with testosterone, 10% treated with progesterone and 10% treated with a cream based preparation. 86 Short-term and long-term treatment of vulvar LS with topical application of testosterone propionate 2% in petrolatum versus CP 0.05% were evaluated in two groups each of 20 patients. A comparable efficacy was recorded at the 3-month follow-up whereas a higher efficacy of CP at 1 year follow-up. 87 Cattaneo and coll found that after good results obtained with CP cream for 24 weeks, treatment with testosterone for further length of time appeared to have a negative effect. 88 A retrospective chart review study of 81 consecutive patients with biopsy-proven vulvar LS showed with clobetasol a 77% chance of complete remission of symptoms and a 47% chance of improvement in the clinical appearance of the vulva. 89 Treatment longer than 12 weeks with CP was studied in 137 women with severe vulvar LS submitted to a “as required” basis or to a regular basis treatment for six months. All patients were examined at two, three, six and 12 months following treatment. In the 6-month follow-up, 59% of the first group and 85% of the second had a complete response regarding their symptoms whereas on the 12-month follow-up, the respective numbers were 48% and 74%. No side-effects from the long-term use of CP 0.05% were recorded. 90 In a prospective study, conducted between 1981 and 2001, 83 women with vulvar LS were treated until complete clinical and histologic remission and followed up for evidence of clinical and histologic recurrence (median follow-up, 4.7 years) in order to evaluated the probability of developing a squamous cell carcinoma. Complete remission was obtained in 45 patients (54%). The probability of remission was significantly associated with age (P < 0.001). The estimated incidence of remission at 3 years was 72% in women younger than 50 years, 23% in women aged between 50 and 70 years, and 0% in women older than 70 years. The incidence of relapse was estimated to be 50% at 16 months and 84% at 4 years. Age had no effect on relapse prevalence. Treatment with a potent steroid cream seemed to improve but not to cure VLS in women older than 70 years, probably because of a long disease evolution. The 8 observed vulvar squamous cell carcinomas (9.6%) occurred in previously untreated or irregularly treated VLS lesions therefore although a protective effect from malignant evolution was suggested by the authors, the number of seemingly protected patients was too small to be statistically significant. 91 A large sized population study of two hundred and two patients with clinically typical or biopsy-confirmed vulvar LS and treated with CP was reviewed either at consultation (75%) or by retrospectively examining their chart. Topical CP was shown to be very effective but at least intermittent treatment was required long term in 85%. At follow up, 101 of 185 patients (56%) were asymptomatic but 22 (12%) continued to have moderate-to-severe symptoms. 92 A retrospective study was conducted on 83 women with vulvar LS associated with variable degrees of squamous cell hyperplasia (mixed disease). All patients were initially treated with topical fluorinated corticosteroids, and then 2% testosterone propionate in petrolatum or 0.05% clobetasol 17-propionate (44 (53%) versus 39 (47%)). The remission rates were 82 and 93% in the testosterone and clobetasol subgroups at the end of 6 months (p = 0.112), respectively. The disease recurred in 8% of the patients with higher recurrence rates in the testosterone arms although statistically significant differences were not obtained. 93 Topical treatment with clobetasol propionate represented a safe and effective therapy of penile LSA as well with no risk of epidermal atrophy but with some potential for triggering latent infections, especially human papillomavirus. 94
Oral lichen planus
Oral lichen planus (OLP) is a chronic inflammatory disease, currently considered an autoimmune disease of unknown aetiology, that causes bilateral white striations, papules, or plaques on the buccal mucosa, tongue, and gingivae. Erythema, erosions, and blisters may or may not be present. Comparative studied were conducted on the efficacy of CP 0.05% in this disorder. The treatment with CP 0.05% ointment in orabase was better that 0.05% fluocinonide ointment in orabase as measured by more rapid control of pain (within 7 days). 95 In another study CP has a comparable efficacy of mesalazine 5% that was then proposed as alternative to CP for the treatment of symtomatic oral LP. 96 A total of 60 patients with OLP were subdivided into three groups matched for sex and age and treated for 6 months respectively with 0.05% ointment or 0.05% fluocinonide ointment in an adhesive medium (4% hydroxyethyl cellulose gel) plus in each case antimycotic treatment consisting of miconazole gel and 0.12% chlorhexidine mouthwashes. A third placebo group, received only hydroxyethyl cellulose gel and antimycotic treatment as above. Considering complete responses, only clobetasol gave significantly better results than placebo with a resolution of lesion in 75% of cases versus 25% of fluocinonide and none in placebo group. After 6 months of follow-up, 65% of the clobetasol-treated group and 55% of the fluocinonide group were stable. Estimation of plasma cortisol levels showed no significant systemic adverse effects of clobetasol or fluocinonide. 97 Lo Muzio and coll evaluated 54 patients affected by vesiculo-ulcero-erosive oral lesions, 24 of them with OLP and 30 with aphthae, and they compared the efficacy of three different randomly administerd CP preparations: ointment, CP in an oral analgesic base, and CP in an adhesive denture paste. In all cases, the administration of the corticosteroid was effective in producing remission of symptoms in each group of patients. Clobetasol and adhesive denture paste correlated with an early remission of pain in lichen and apthous lesion. 98 The mouthwash of 0.05% clobetasol in aqueous solution over a 48-week period demonstrated to be a safe and efficacious option for the treatment of severe oral erosive lesions with a pain and ulceration disappearing in 93.3% of cases and 90% of patients reporting a full recovery in their daily life activities. 99 The comparison of regimens that included systemically administerd prednisone (50 mg/day), and afterwards clobetasol ointment in an adhesive medium plus antimicotics, versus CP plus antimycotics showed no significant differences between the two groups in terms of efficacy. Therefore the most suitable corticosteroid therapy in the management of OLP was the topical therapy, which is easier and more cost-effective than the systemic therapy followed by topical therapy. 100
New lipid microspheres loaded with 0.025% of clobetasol propionate compared with a commonly used formulation (a sort of dispersion of a lipophilic ointment in a hydrophilic phase) with the same amount of drug were tested in the topical treatment of OLP. Both formulations were found to be similar but a significant difference was registered for the first in terms of reduction in painful symptoms suggesting that the new topical drug delivery system may enhance, at least in terms of symptom remission and compliance, the effectiveness of clobetasol propionate at a dose of 0.025% in OLP therapy. 101 Different concentration of CP in OLP, 0.025% versus 0.05%, both placed in 4% hydroxyethyl cellulose bioadhesive gel, were investigated in a small sized study of 30 patiens. In all, 14 of the 15 clobetasol 0.025% patients (93%) and 13 of the 15 clobetasol 0.05% patients (87%), had symptoms improvement after 2 months of therapy (P = 0.001 in both groups). Also, 13 of the 15 clobetasol 0.025% patients (87%) and 11 of the 15 clobetasol 0.05% patients (73%) had clinical improvement after 2 months of therapy (P < 0.05 in both groups). Therefore no statistical differences were found in comparing the two formulations and a lower concentration is to be recommended. 102 Recent studies evaluated efficacy of clobetasol 0.05% in comparison to tacrolimus 0.1% ointment and different results were obtained, comparable efficacy or superior efficacy of tacrolimus.103,104
Boullous autoimmune skin dermatoses
Bullous skin diseases may be acquired or induced or they may be autoimmune in origin. The autoimmune bullous skin disorders are all characterized by the presence of autoantibodies that target distinct adhesion molecules of the epidermis and dermoepidermal basement membrane zone. The consequences of these antibodies are a loss of the targeted protein's adhesive properties, which leads, in turn, to the appearance of blisters and erosions. Like most autoimmune disorders, autoimmune bullous or vesiculo-bullous skin disorders are more likely to occur in women. The first report of efficacy of CP in autoimmune bullous disease was made by Westerhof in 1989. 105 Later Joly P. and coll carried out a study on 341 patients random treated either with topical clobetasol propionate cream (40 g per day) or oral prednisone (0.5 mg per kilogram of body weight per day for those with moderate disease and 1 mg per kilogram per day for those with extensive disease). Among the 188 patients with extensive bullous pemphigoid, topical corticosteroids were superior to oral prednisone (P = 0.02). The one-year survival rate was 76 percent in the topical-corticosteroid group and 58 percent in the oral-prednisone group. The results of the study confirmed that CP was effective for both moderate and severe bullous pemphigoid and superior to oral corticosteroid therapy for extensive disease with a rapid control of the disease and less severe complications. 106
Superpotent topical corticosteroids have also been demonstrated to improve bullous pemphigoid (BP) patients’ survival in a multicenter randomized controlled trial and stratified depending on the extent of BP in 312 patients. Two different regimens were compared: standard regimen (clobetasol propionate cream, 40 g per day initially, with CS tapering over 12 months) and mild regimen (10–30 g per day), with CS tapering over 4 months. A noninferior rate of BP control was obtained with the mild regimen 156/159 (98%) as compared with the standard regimen 150/150 (100%; P = 0.005). Event-free survival, that is, the combined outcome of deaths and life-threatening adverse events did not differ between the two treatment groups (P = 0.77). However, upon adjusting through the Cox model for age and Karnofsky score, a strong beneficial effect of the mild regimen was observed in patients with moderate BP, with an almost twofold decrease in the risk of death or life-threatening adverse events relative to the standard regimen. 107 The use of CP seems to improve clinical efficacy of systemically administerd dapsone in BP in conjunction with antibiotics and/or antimycotic ointments in Hailey-Hailey Pemphigus, and efficacious alone in mild pemphigus vulgaris with long remission in certain subgroup of patients.108–110
Other inflammatory and pruritic dermatosis
The potential use of CP has been noted in several different skin dermatoses that include seborrheic dermatitis, 111 chondrodermatitis nodularis chronica of helix, 112 vitiligo, 113 discoid lupus erythematous,114,115 parapsoriasis and mycosys fungoides, 116 but well controlled studies have been reported in only some of these disorders.
Discussion
Clobetasol 17-propionate is a widely used, superpotent topical steroid with proven efficacy in many chronic skin disease characterised by alternating periods of relapse and remission. It is topically applied to control inflammation and pruritus in psoriasis and atopic dermatitis, both of them affecting large children population. The results of studies conducted on HPA suppression demonstrated the safety of CP not only in adults but also in children older that 12 years, specifically when used within recommended doses (50 g/w) and short duration of therapy (2 consecutive weeks).
CP has demonstrated high efficacy in multiple clinical settings and in several formulations. Even if a recent review concluded that in comparable effects among different formulations, CP spray and foam seem to be associated with a high cosmetic acceptability along with high efficacy and consequent remarkable improvement of quality of life of patients, especially in psoriasis, in scalp and non scalp body areas. The ability of CP to induce a rapid clearing of skin lesions in inflammatory dermatoses, alone or with other topical agents, can lead to a reduction of the time of hospitalization. This aspect of the medical research was not deeply studied and it shoud be further investigated and correlate to rates and times of recurrences.
The applicability of CP to genital and oral mucosa for short periods offer a great therapeutical potential for several disorders affecting these particular anatomical areas whose pathological conditions were life threating in the past. The use of clobetasol has determined a remarkable improvement of patients survival with a parallel sparing of systemic corticosteroids and reduction of associated side effects. New delivery vehicles, as lipid microspheres, charged with lower concentration of clobetasol, are currently under consideration in oral lichen planus but they may be considered in the future for the treatment of genital disorders as well, since clobetasol, by reducing chronic inflammation, could have a protective effect from malignant evolution.
Conclusion
A review of the literature has shown that clobetasol is still the most potent topical corticosteroid with several mechanism of action that can properly control inflammation and pruritic manifestation of several chronic skin disorders. With regard to formulation differences when observed in the studies and considering also the quality of life whichever vehicle patients find preferable should be chosen. In psoriasis combination therapy seem to obtain best results in terms of efficacy and remission with considerable saving of medical resources in certain cases.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
