Abstract
Atrial fibrillation (AF) is the most common cardiac arrhythmia with increasing prevalence. AF is associated with considerable morbidity and mortality. Cardioembolic stroke results as a consequence of thrombus formation within the left atrial appendage (LAA) in patients with AF and has a significant impact on our health system. Oral anticoagulation with either vitamin K antagonists or the novel anticoagulants is an effective medical therapy for prophylaxis against strokes in patients suffering from AF. Unfortunately, bleeding complications and compliance issues limit their use. A need for newer approaches have thereby been developed and studied such as percutaneous occlusion of the LAA. Several LAA occlusion devices have been developed, and with improved operator experience, they have been successful in stroke prevention in patients with nonvalvular AF and fewer periprocedural complications. This article reviews the use of percutaneous LAA occlusion devices in the prevention of cardioembolic stroke.
Keywords
Introduction
Atrial fibrillation (AF) is the most common cardiac arrhythmia, affecting 1% of the general population, 6% of people older than 65 years, and 9% of people older than 80 years.1,2 With aging of the population, the prevalence of AF and its impact on the health system is projected to increase substantially.3,4 It is also likely that the true prevalence of AF is underestimated as it can be difficult to detect paroxysmal AF.
AF is associated with considerable morbidity and mortality. AF is an independent risk factor for stroke, occurring in up to 19% of patients with stroke older than 70 years. 5 After adjusting for other risk factors, AF is associated with a four- to fivefold increase risk of ischemic stroke. 5 Paroxysmal AF also carries a similar risk of stroke as patients with persistent AF. 6 Furthermore, AF has been noted to be underestimated in cryptogenic ischemic strokes, and the utilization of implant-able cardiac monitors will help diagnose clinically silent paroxysmal AF. 7 Cardioembolic strokes are generally more severe than other types of ischemic stroke and have been associated with higher 30-day and 1-year mortality.8,9
In the United States, there are about 800,000 strokes each year, with 1.5% of those attributed to AF younger than 59 years and 23% of those older than 80 years. 5 Stroke is the third leading cause of death and the number one cause of major morbidity in the United States and estimated to cost the healthcare system ~30 billion dollars each year.
Anticoagulation is the mainstay treatment for reducing the risk of stroke in the presence of AF. This, however, comes with considerable risks of bleeding, and therefore, there is a need for other methods to prevent embolic strokes such as percutaneous left atrial appendage (LAA) closure devices. In this article, LAA closure and effectiveness of preventing stroke are reviewed.
AF and the Atrial Appendage
LAA is a remnant of the embryonic left atrium. 10 It forms a finger-like multilobulated pouch. The LAA wall is thin and its surface is irregular because of trabeculations of pectinate muscles (Fig. 1). 11 The LAA is separated by the main body of the left atrium by an orifice that is on average ~1.1 cm in diameter. In the setting of long-standing AF, there is an increase in volume secondary to progressive stretching and dilatation.
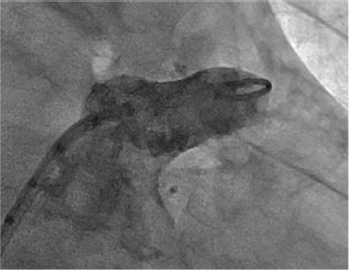
Left atrial appendage. Fluoroscopic image of the LAA as seen on angiography.
AF is associated with mechanical dysfunction of atrial tissues. Loss of contractility in the LAA can result in local stasis and thrombus formation. Echocardiographic and autopsy studies have demonstrated the LAA to be a source of thrombus formation in >90% of patients with nonvalvular A F. 12 Virchow's triad assists in the understanding of probable mechanisms for thrombogeneis in the LAA.13–15 The combination of abnormal blood flow from stasis (because of diminished contractility of the LAA) and increased LA and LAA volume, abnormal blood constituents (because of underlying cardiovascular disease results in activation of coagulation cascade), and abnormal changes of vessel wall all contribute in increasing the likelihood of thrombogenesis.16–19 This may explain that slow flow as seen by low LAA inflow velocities or spontaneous echo contrast has been associated with increased thrombus formation and increased stroke risk in patients with AF. 20
The LAA has been categorized into various shapes, and the four most commonly utilized classifications are chicken wing (LAA with a bend in the body), windsock (single lobe without bend), cauliflower (short main lobe with multiple sublobes in various directions), and cactus (multiple secondary lobes). 21 Studies have noted that different morphological/structural features of the LAA may influence the propensity for stroke risk. Beinart et al. 22 have reported that a larger LAA neck diameter and LAA depth are associated with a higher prevalence of prior stroke in patients with AF. Other shape descriptions and risk of stroke have also been studied with poor reproducibility. In two studies, chicken wing appeared to correlate less with prior embolic events as compared to cauliflower, cactus, or windsock shapes, but this was not reproduced by another larger study.21,23–25 LAA morphology may not be a reliable predictor of stroke risk. However, the extent of trabeculations noted within the LAA may be a better predictor of thromboembolic events. 25
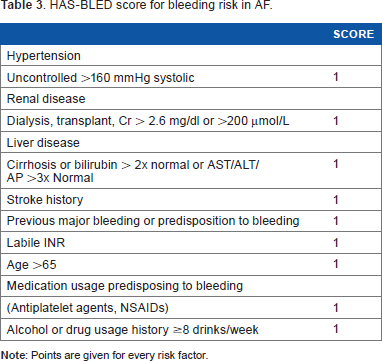
Stroke is usually not as a result of a single entity but from the influence of a multitude of factors. As mentioned earlier, the LAA is the most common site for thrombus formation in patients with AF. As many as 25% of strokes in AF can be linked to intrinsic cerebrovascular disease. 26 The CHADS2 and CHA2DS2-VASc scores assist in estimating an individual's risk of yearly thromboembolic events based on his/her comorbidities, while the HAS-BLED score assists in estimating the risk of bleeding (Tables 1–3).27–29 This led to the guidelines of which patients would derive clinical benefit from anticoagulation. Hypertension and aortic atherosclerosis have been associated with increased prevalence of LAA thrombus, spontaneous echo contrast, and low LAA velocities. 30 The mechanistic connections of other risk factors, such as the LAA, and the risk of thromboembolic stroke remain not well understood. Despite these ambiguities, the LAA remains a consistent and predictable site for thrombi formation resulting in embolic events, and thus a target for prophylaxis.
CHADS2 score model for thromboembolic risk in AF.
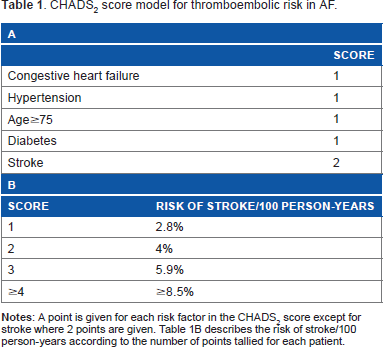
CHA2DS2-VASc score model for thromboembolic risk in AF.
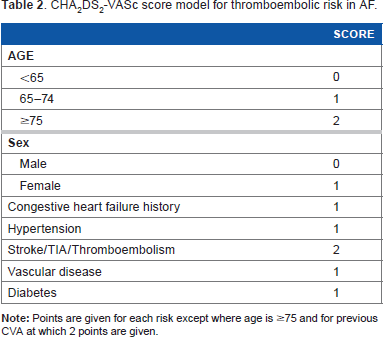
HAS-BLED score for bleeding risk in AF.
Oral Anticoagulation: Benefits and Limitations
The effectiveness of medical therapy targeting rate and/or rhythm control and stroke risk reduction is modest at best and not without side effects. Anticoagulation remains the mainstay therapy for patients suffering from AF and at risk of stroke. In particular, warfarin anticoagulation reduces the relative risk (RR) of stroke by ~64%. Furthermore, the risk of major bleeding with warfarin use is 0.5%–1% per year in patients older than 80 years; this risk can rise to as high as 13% in the first year. 31 Warfarin itself has several barriers for achieving a therapeutic range, including frequent laboratory monitoring, interactions with food and other medications, and its narrow therapeutic range.
Other alternatives to warfarin have been explored. Briefly, aspirin by itself has been widely studied, and trials have shown that against placebo, it has a 22% RR reduction, but, when compared to warfarin, there was a 36% higher stroke rate.32–34 Even the dual antiplatelet combination of clopidogrel and aspirin fared worse compared to warfarin with a 44% higher rate of vascular events and a 30% increase in major bleeding.35,36 Newer anticoagulants have now been introduced and widely used. The RE-LY trial studied dabigatran, a direct oral thrombin inhibitor, in two different doses against warfarin. The 150 mg dose was associated with superiority, and a 35% lower rate of stroke and systemic embolism but similar bleeding, while the 110 mg dose was associated with no significant difference of stroke and systemic embolism but a 20% lower rate of major hemorrhage. 37 The ARISTOTLE trial studied apixaban, a direct factor Xa inhibitor, and showed superiority to warfarin in terms of stroke and systemic embolism with a 21% reduction. It also showed a 31% reduction in the risk of major hemorrhage and 11% reduction in death. 38 Finally, ROCKET AF that studied rivaroxaban, also an oral factor Xa inhibitor, demonstrated noninferiority to warfarin in stroke and systemic embolism (21% reduction) without any significant difference in the risk of major bleeding. 39 Rivaroxaban also has the benefit of once daily dosing.
Short of open-heart surgery, the only alternative to current anticoagulation therapy is percutaneous catheter-based closure devices, which results in occlusion of the LAA and reduces the risk of thromboembolic strokes. Clearly with an aging population and the risk of bleeding with anticoagulation, there is a critical need for other methods of reducing embolic stroke.
Surgical LAA Ligation
Regarding open-heart surgery, surgical ligation or exclusion has been used for many years though there is little evidence regarding its effectiveness. In 1949, Madden first described LAA resection in two patients with recurrent arterial emboli. 40 It then started to gain momentum until a review article in 1970 suggested that LAA ligation was not effective in reducing embolic events. 41 It started to gain interest again slowly in the 1990s following several surgical techniques and devices being developed. These include oversewing the orifice from the epicardial surface, suturing the LAA orifice from the endocardial surface, excising the LAA, mechanically stapling across the orifice, and most recently, using the AtriClip (Atricure) to atraumatically clip the LAA orifice.
Surgical excision LAA results have been previously reviewed. Briefly, there have been two major randomized trials of surgical LAA closure. The Left Atrial Appendage Occlusion Study (LAAOS) I trial randomized 77 patients in a 2:1 ratio to LAA closure or no occlusion. 42 A suturing method was initially performed but subsequently a stapling method was adopted given the high incidence of persistent leaks. Thirty-four percent of patients had residual flow. In the 30-day postoperative period, 3.8% had embolic events in the treatment group, while none in the control group. Patients were followed up for a mean of 13 months, and there were no further embolic events reported.
The LAAOS II was performed later and randomized 52 patients with AF and at least one risk factor for stroke to LAA closure vs no closure. 43 They reported 100% successful closures of LAA as determined by intraoperative transesophageal echocardiography (TEE). Follow-up was for one year, and there was no difference in primary outcome of death, myocardial infarction, stroke, or major bleeding. Of note, there was a trend to higher reoperation for bleeding in the closure group (8% vs 3.9%). This was, however, nonsignificant.
The AtriClip was evaluated in the EXCLUDE study. 44 It consists of two parallel titanium tubes and two nitinol springs with a knot-braided polyester fabric. Seventy-one patients were included in the registry where the LAA was excluded at the time of open-heart surgery. Successful exclusion was achieved in 96% of the patients. No late leaks or complications related to the device were reported at 1-year follow-up. Stroke or TIA occurred in 3.1% of the patients; 30% of those were on warfarin at that time. Even though the device appears to be safe, there remains limited evidence on its efficacy with reducing thromboembolic events. The AtriClip and the Tigerpaw (Maquet) systems have both been approved by the FDA for LAA closure during open-heart surgery.
The application for open-heart surgical LAA closure is limited, given that the majority of patients with AF do not require open-heart surgery. Surgical suturing or stapling of LAA appears to be inadequate, and LAA clipping, while safe, has no randomized controlled trials. This has led to the need for percutaneous closure devices.
Transcatheter Cosure Devices
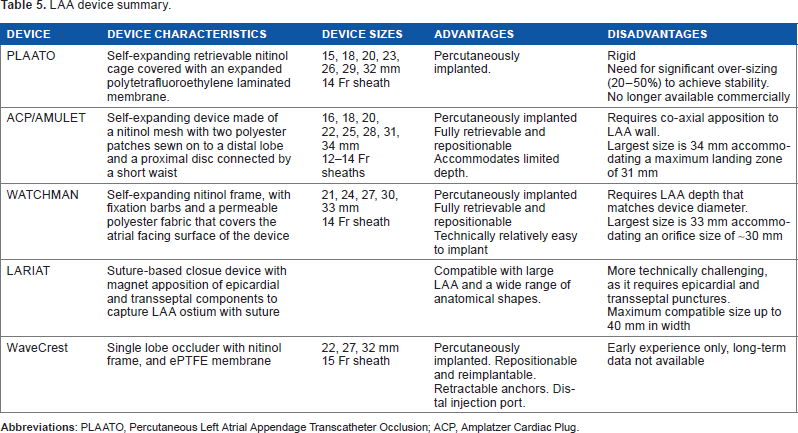
Several devices have been developed for percutaneous LAA closure. The first device developed was the Percutaneous Left Atrial Appendage Transcatheter Occlusion (PLAATO) system. 45 It consists of a self-expanding retrievable nitinol cage covered with an expanded polytetrafluoroethylene membrane. The membrane is laminated directly to the outer frame allowing direct contact with the LAA wall. The device was evaluated in a nonrandomized North American multicenter trial of 64 patients who had contraindications to warfarin but were at a high risk of thromboembolic events. 46 The trial proved insightful, revealing a high rate of anatomic closure at the time of procedure (residual flow ≤3 mm in 98% of cases), with excellent safety. At a mean of 3.75-year follow-up, there was a 3.8% per year risk of stroke or TIA, while it would have been expected to be at 6.6% per year with the CHADS2 score.
Bayard et al described the European PLAATO experience where 180 patients with AF and contraindications to warfarin were studied. 47 Ninety percent of patients had a successful LAA occlusion. The reported stroke risk was only 2.3% per year compared to the expected 6.6% per year at a mean follow-up of 9.6 months. It appeared to be safe with 1.1% mortality, and 3.3% reported pericardial tamponade requiring surgical intervention. The use of this device was limited by some design flaws, being rigid and the need for significant oversizing (20%–50%) to achieve stability. The device is now no longer in development but was the basis for further devices to be developed and implemented in clinical use.
The more recent devices such as the WATCHMAN, Amplatzer™ Cardiac Plug (ACP) and the Amulet™ are more flexible and require less oversizing (10%–20%). It is important to understand that the LAA orifice is typically oval, and the tissue is softer than in the ventricle; therefore, a more flexible device would allow more successful occlusion and less risk of perforation. The PLAATO device also necessitated a deeper LAA for better occlusion, whereas the newer ACP/Amulet allows for occlusion of a short proximal portion and early separation into lobes. Given the different number of lobes, length, and orifice size of the LAA, each individual's anatomy is unique and can sometimes be referred to as a fingerprint. 48
Watchman
The WATCHMAN device was evaluated in the Watchman Left Atrial Appendage System for Embolic Protection in Patients with AF (PROTECT AF) trial (Fig. 2). 49 It consists of a self-expanding nitinol frame, with fixation barbs and a permeable polyester fabric that covers the atrial facing surface of the device. The fabric of the WATCHMAN device is permeable to blood, and for this reason, patients require to continue on conventional thromboembolic prophylaxis until the device is endothelialized (~45 days postimplantation). A TEE is conventionally done prior to ceasing anticoagulation to ensure endothelialization. After 45 days if TEE was satisfactory, the anticoagulant was ceased and replaced with clopidogrel for a further 4.5 months, at which point aspirin was continued indefinitely. PROTECT AF was a noninferior randomized trial enrolling 707 patients with nonvalvular AF with a CHADS2 ≥1 in a 2:1 ratio. The WATCHMAN device was noninferior to warfarin for the primary end point of cardiovascular/unexplained death, any stroke, or systemic embolization at 1065, 1588, and 2621 patient-years of follow-up (Table 4).49–51 Of note, the noninferiority margin was set at 2, ie, the WATCHMAN device needed to have less than twice the events (in the control arm) to satisfy noninferiority of which was achieved in all the follow-ups mentioned earlier. At 2621 patient-years (3.8 years), the WATCHMAN device was superior to warfarin. The safety of the device was concerned. In particular, there was a 1.7% risk of cardiac perforation, and a 3% risk of pericardial effusion with tamponade. There is an important learning curve effect, which was supported by two registries carried out later.50,52 The rate of complication within seven days was 3.7% compared to 7.7% in the initial randomized trial.
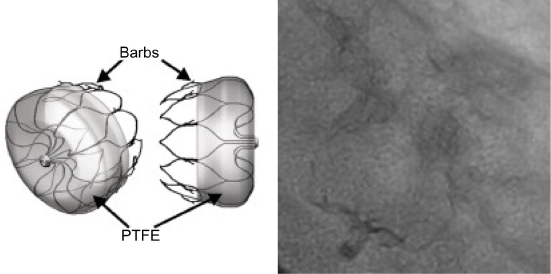
The WATCHMAN device. The WATCHMAN device by Boston Scientific is a self-expanding nitinol frame covered with a 160° μm polyethylene terephthalate membrane. The device is preloaded within a delivery catheter. It has 10 active fixation anchors and available in five sizes (21, 24, 27, 30, and 33° mm). The diagram is © 2016 Boston Scientific Corporation or its affiliates. All rights reserved. Used with permission of Boston Scientific Corporation.
Outcomes of randomized WATCHMAN trials.

Some confounding factors in the PROTECT AF trial were the low CHADS2 score, the ongoing use of warfarin in the device group, and the discretionary use of antiplatelet agents. The PREVAIL trial was therefore designed to answer a few of these concerns. It was also designed as a noninferiority trial using Bayesian statistics. 53 A total of 269 patients were randomized to WATCHMAN plus short-term warfarin and 138 patients to dose-adjusted warfarin. The mean CHADS2 score was 2.6. The deployment success rate was high at 95.1%, and at 45 days postimplantation, 92.3% of patients were able to successfully cease warfarin following TEE. The primary end point margin was not met as the upper bound of the credible interval was 1.89, exceeding the noninferiority margin set at 1.75 [RR 1.07 (0.57 to 1.89); Table 4]. Event rates for the primary end point favored the control group, with the rate of ischemic strokes being 1.94 in the WATCHMAN vs 0.71 in the control group. Regarding the secondary end point (ischemic events after the seventh procedural day), noninferiority was achieved on the basis of the 18-month rate difference. The third primary end point evaluating safety of WATCHMAN achieved noninferiority. Compared to PROTECT AF, safety events related to the procedure, including the incidence of pericardial effusions requiring surgical drainage and procedural stroke, were significantly reduced.
Recently, a meta-analysis of PROTECT AF and PREVAIL was presented at TCT by Holmes et al 54 . The analysis showed comparable efficacy for WATCHMAN with a hazard ratio (HR) of 0.79 (P = 0.22). Similarly, there was a difference in all-cause stroke or systemic embolization, major bleeding, or all-cause death. Although there were more ischemic strokes noted in the warfarin arm (HR = 1.95, P = 0.05), WATCHMAN had significant reduction in hemorrhagic stroke (HR = 0.22), which resulted in reduction in disabling stroke by half with WATCHMAN. Moreover, the WATCHMAN device fared better with regard to major bleeding beyond seven days (HR = 0.51) and cardiovascular mortality (HR = 0.48).
Residual leaks remain a significant component of the WATCHMAN device. The clinical significance of the leaks is unknown. In PROTECT AF, flow around the device detected by TEE was noted in 41% and 32% of patients at six-week and one-year follow-up, respectively. When a post hoc analysis was performed, there was no association between clinical events and the presence of peridevice leak, regardless of the size of leak and whether warfarin was continued or not. Given the small number of events, this analysis had limited power.
Patients in whom anticoagulation is contraindicated is challenging. ASA Plavix Feasibility Study With Watchman Left Atrial Appendage Closure Technology (ASAP) was a prospective, multicenter, observational trial that studied 150 patients with AF and contraindications to anticoagulation. 55 Following implantation of the device, the patients were treated with dual antiplatelet therapy for six months, and then aspirin indefinitely. The observed rate of stroke or systemic embolism was only 2.3% per year at a mean follow-up of 14.4 months, significantly less than the predicted 7.3% per year based on the CHADS2 score. This study was small and non-randomized; therefore, more studies are required to clarify this further. Moreover, the ACTIVE Writing Group published in 2006 showed that aspirin and clopidogrel was associated with a bleeding hazard similar to that of warfarin. 35
Amplatzer Cardiac Plug
The ACP (St Jude Medical) uses a similar technique to the PLAATO device (Fig. 3). It is a self-expanding nitinol mesh that consists of a lobe placed in the LAA and a disk that sits at the orifice in the left atrium. A polyester patch is sewn on each and they are connected by a short central waist. The distal lobe contains six pairs of barbs designed to increase its stability within the LAA.56,57 This device is different from the WATCHMAN with the additional disk that sits in the left atrium, while the WATCHMAN is required to be inside the appendage entirely, with requirement for depth similar to the ostial diameter. Thus, the ACP device can accommodate LAA that is limited by depth.
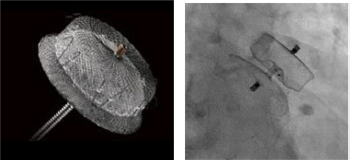
Amplatzer cardiac plug. ACP is a self-expanding nitinol mesh that consists of a lobe placed in the appendage and disk that sits at the orifice. A polyester patch is sewn on each, and they are connected by a short central waist. The distal lobe contains six pairs of barbs designed to increase its stability within the appendage.
There are no randomized trials published, but there are multiple observational studies reported. Animal data published has demonstrated uncomplicated device delivery with complete occlusion of the appendage at 30-day and 90-day follow-ups. 58 One of the larger observational trials was an investigator-initiated retrospective preregistry to evaluate procedural feasibility and safety up to 24 hours after implantation of the ACP. 56 A total of 143 LAA closures in patients were attempted and successfully performed in 132 (96%) patients. Serious complications occurred in 10 (7.0%) patients (three patients with ischemic stroke; two patients experienced device embolization, both percutaneously recaptured; and five patients with clinically significant pericardial effusions requiring drainage). In the subsequent European multicenter postmarket observational study, 96.5% of the studied 197 patients underwent successful ACP implantation. Eleven (5.6%) procedure-related adverse events occurred within seven days post-procedure consisting of one AV fistula, three serious pericardial effusions, one cardiac perforation, one hematoma, two cases of periprocedural thrombus that resolved during the procedure, and three device embolizations. 59 An abstract presented at ESC reported that at six-month follow-up there were two ischemic strokes, and out of the 47 patients who had reached two-year follow-up, there was a further ischemic stroke, with a 1.24% per year risk of stroke compared to a predicted 5.33% per year based on the CHADS2 score. 60 Given the limited follow-up, more data should be awaited.
A pooled ACP experience from 21 European and Canadian centers was presented by Tzikas at Transcatheter Cardiovascular Therapeutics. 61 A total of 1047 patients with a mean CHA2DS2-VASc score of 4.5 and HAS-BLED score of 3.1 were included in this analysis. Implant success was 97.3%, and closure rate at follow-up TEE (<3 mm residual flow) was 98.1%. The reported periprocedural major adverse events were 4.3%. The observed annual stroke rate was 2.3%, a 59% reduction in stroke rate as compared to an expected 5.6% stroke rate based on the CHA2DS2-VASc score.
Overall, the observational registry studies with ACP showed reasonably good safety and efficacy data. The frequency of procedural success, serious pericardial effusions, and embolizations appear similar to that of the WATCHMAN device. Although these findings are encouraging, randomized controlled trials comparing the different devices are required to firmly assess the safety and efficacy of the ACP device.
Lariat Procedure
The Lariat device (SentreHeart) was developed to enable approximation for soft tissue but has been used clinically to ligate the LAA through the delivery of a surgical suture via a combined transseptal and subxiphoid approach. 62 It requires pre-procedural computed tomography to assess anatomic feasibility of the LAA orifice. Appendages that are posteriorly orientated having lobes behind the pulmonary artery or having a diameter >40 mm should not have been applied. 63
Clinical experience has been limited to observational studies. The first was reported in 2011 where 13 patients underwent LAA ligation using this device (11 percutaneous and ywo had open chest ligation). 62 The first prospective experience was published by Bartus et al 64 . This was a single-center prospective study where patients with AF and were poor candidate or ineligible for warfarin underwent LAA ligation. Out of 119 patients who fulfilled inclusion criteria, 92 were included while the rest were excluded because of unfavorable CT dimensions or thrombus present on TEE. Successful closure was achieved (<1 mm leak) in 92.3% of cases. Procedural complications occurred in five patients with three significant pericardial effusions and two patients developed pericarditis. At one-year follow-up, there were no thromboembolic events and persistent closure with no or new leaks. Of note, there is no long-term efficacy data available and there was no control group.
A further multicenter, retrospective study was published to assess the efficacy and safety of the Lariat device. 65 In this study, 154 patients were reviewed who underwent the procedure in the United States. The device success was reported at 94% defined as successful ligation and <5 mm leak by post-procedural TEE. With regard to complications, 10.4% significant pericardial effusions and 9.1% of procedure-related major bleeding were observed. Other reported complications included late leaks and thrombus at the site of the Lariat ligation.
There are no data on the need for anticoagulation or dual antiplatelet therapy following this suture ligation procedure. With the small number of observational studies, the Lariat appears to be effective but does have significant procedural complications associated with it, including major bleeding, pericardial effusions, and pericarditis. A more robust, randomized clinical trial is required to assess the long-term efficacy and safety of this procedure.
LAA device summary.
Perspective
Placing this in context, there are no randomized surgical studies demonstrating efficacy of the LAA closure at the time of surgery. There is an ongoing LAAOS III randomized prospective study (n = 4700), which will hopefully provide some directions on the role of surgical LAA closure. In the meanwhile, FDA clearance is available for the AtriClip and Tigerpaw devices for LAA closure. The efficacy of these devices against oral anticoagulation is unknown.
The only two published randomized trials for percutaneous LAA closure were the PROTECT AF demonstrating superiority of LAA closure and the PREVAIL that did not achieve noninferiority of LAA closure compared to warfarin. The device has recently received FDA approval. Of particular note, there were concerns of higher ischemic strokes with in the device arm, although hemorrhagic stroke events were significantly reduced with device. In addition, this was in the era prior to the newer oral anticoagulant therapy and therefore the comparison of WATCHMAN to new oral anticoagulants is unknown. These newer agents are less likely to result in hemorrhagic cerebral bleeding/major bleeding and do not require dose adjustments. The other potential issue is that device-associated thrombus may occur in 2%–4% of cases with various devices 52 ; however, the risk of ischemic stroke related to device-associated thrombus is felt to be low (0.7%). 55
Patient selection is an important consideration, and currently, the majority of LAA closures performed worldwide are for patients with contraindications or who are perceived poor candidates for long-term anticoagulation. Therefore, perhaps the best candidates for LAA closure are those with moderate-to-high risk of stroke and who have moderate-to-high risk of bleeding with oral anticoagulation. The 2012 European Society of Cardiology guidelines for the management of AF provided a Class IIB recommendation (Level of evidence B) for LAA closure with those at high risk and contraindication to anticoagulation. 66 The CAP registry suggests that operator experience affects rates of complications, and therefore, it is expected that less complications will occur as operator skills increase. 52 Moreover, patients with a longer life expectancy are likely to benefit more from this procedure as the net clinical benefit of this procedure is likely to increase over time. Patient preference and understanding of the procedure should be taken into consideration as well.
Conclusion
AF is an increasing burden on society with an increasing prevalence worldwide. While OACs are the mainstay treatments in the prevention of embolic events, they have several limitations and not all patients can tolerate them long term. Percutaneous LAA closure is a feasible option and appear to be a safe and efficacious alternative, but only limited randomized trials are currently available and only for patients who are candidates for long-term anticoagulation. Extrapolation of randomized trials with the WATCHMAN device to other devices may not be appropriate, as there are mechanical differences with these devices. Further randomized controlled trials comparing between devices, involving different patient population (ie, patients with contraindications/relative contraindications to anticoagulation), and comparing novel oral anticoagulants should be pursued to evaluate the safety and efficacy of percutaneous LAA closure.
Author Contributions
Conceived the concepts: PF, JS. Analyzed the data: PF, JS. Wrote the first draft of the manuscript: PF, JS. Contributed to the writing of the manuscript: PF, JS. Agree with manuscript results and conclusions: PF, JS. Jointly developed the structure and arguments for the paper: PF, JS. Made critical revisions and approved final version: PF, JS. Both authors reviewed and approved of the final manuscript.
