Abstract
Nonalcoholic fatty liver disease (NAFLD) is on the rise and has become a major etiology for chronic liver disease. It is frequently associated with obesity, insulin resistance, hypertension, and dyslipidemia and is considered the hepatic manifestation of metabolic syndrome. In this review, we present a summary of the epidemiology and pathogenesis of NAFLD, and discuss the clinical evaluation and stratification of NAFLD patients into low, intermediate, and high risk with respect to liver-related outcomes. While diet and exercise are the cornerstone of treatment in all patients, the low rate of adherence and inadequacy of these recommendations necessitate pharmacologic intervention, especially in intermediate- and high-risk patients. We discuss vitamin E and pioglitazone which are often used as first-line therapy by many practitioners, with pentoxifylline and liraglutide as backup agents. Several drugs are in advanced-phase clinical trials and will likely change the landscape for management of NAFLD in the very near future.
Keywords
Introduction
Nonalcoholic fatty liver disease (NAFLD) defines a spectrum of liver disease ranging through simple steatosis, nonalcoholic steatohepatitis (NASH), liver fibrosis, and liver cirrhosis.1,2 It is the most common etiology of chronic liver disease, 3 with the worldwide prevalence estimated between 11% and 46%.4–6 In the United States, NAFLD is currently the third most common indication for liver transplantation. 7
NAFLD is characterized by hepatic steatosis, defined as accumulation of fat (triglyceride) in greater than 5% of hepatocytes in the absence of other causes of steatosis including excess alcohol intake and congenital errors of metabolism. Whereas simple steatosis is characterized by liver lipid accumulation without inflammation and often carries a relatively favorable clinical course, 8 NASH, which occurs in about 25%–40% of NAFLD patients,9–12 involves hepatocellular injury and liver inflammation and is a significant risk factor for cirrhosis and hepatocellular carcinoma (HCC).1,9,13 In the United States, NASH has been estimated to account for over 13% of HCC cases.14,15
Within the spectrum of NAFLD, NASH is considered especially worrisome as it signifies hepatocellular injury and liver inflammation, leading to other hepatic and extrahepatic complications. Day and James 16 proposed the initial theory for NASH pathogenesis involving the two-hit hypothesis. According to this hypothesis, the first hit of NASH is simple steatosis resulting from insulin resistance and excessive fatty acids, sensitizing the liver to a second hit, likely involving oxidative stress, mitochondrial dysfunction, and lipid peroxidation and leading to inflammation and hepatic fibrosis. However, it appears that steatosis may not be a necessary prerequisite for NASH and liver inflammation. 17 Subsequently, the multiple parallel hit hypothesis by Tilg and Moschen 18 proposed that NASH results from a culmination of various factors in parallel, including disrupted lipid metabolism, lipotoxicity, altered cytokines and adipokines, oxidative stress, endoplasmic reticulum (ER) stress, mitochondrial dysfunction, gut-derived endotoxin, and genetic predisposition. Here, we review the epidemiology and pathogenesis of NAFLD and discuss risk stratification of NAFLD patients. We also highlight pharmacological agents for NAFLD currently in phase 2 and 3 trials.
Epidemiology
NAFLD remains a major etiology for chronic liver disease, with prevalence ranging between 10% and 35% based on the study population and method of diagnosis. Some of the prevalence reports are based on liver biopsy, which is considered the gold standard for diagnosing NAFLD. In the United States, liver biopsy of potential liver transplant donors indicated that 20% of these potential donors were ineligible for donation due to steatosis >30%. 19 In presumably healthy 589 consecutive potential liver transplant donors undergoing liver biopsy in Korea, a 51% prevalence of NAFLD was reported. 20 Furthermore, a recent study conducted in Greece showed that almost 40% of postmortem liver biopsies had histologic evidence of steatohepatitis. 21 Some NAFLD prevalence reports are based on imaging, which is less invasive than biopsy and more practical for population-based assessments. Abdominal ultrasound in randomly selected patients from healthcare centers in Spain showed that the prevalence of NAFLD was 33% in men and 20% in women. 22 Data from the Dallas Heart Study, which used proton magnetic resonance spectroscopy to determine liver fat content and steatosis, estimated that about a third of Dallas County residents in Texas, US, had steatosis based on hepatic triglyceride level greater than 5.5%. 23
NAFLD is associated with obesity, insulin resistance, hypertension, and dyslipidemia and is considered the hepatic manifestation of metabolic syndrome.24–27 In a study comparing the impact of obesity, insulin resistance, and fatty liver on the development of incident type 2 diabetes mellitus (T2DM) in 12,000 South Korean individuals, each risk factor was independently associated with doubling of T2DM risk. 28 Individuals with all three risk factors had a 14-fold increase in the risk of having T2DM. Resolution of fatty liver was associated with a reduction in incident T2DM to a level comparable to someone who had never had NAFLD, 29 and patients in whom NAFLD worsened over the five-year study period had significantly increased risk of T2DM compared to those with NAFLD improvement.
Other than T2DM, NAFLD is associated with several other complications, including cardiovascular disease, cirrhosis, and HCC.9,30–35 In a recent study of 2804 subjects, both men and women with NAFLD were found to be at significantly higher risk of developing cardiovascular disease, compared to those without NAFLD. 36 Although many NAFLD patients do not develop cirrhosis, NAFLD as etiology for end-stage liver disease has been on the rise. Between 2004 and 2013, NAFLD as etiology for end-stage liver disease in new liver transplant waitlist registrants increased by 170%, compared to 14% for Hepatitis C virus (HCV)-related end-stage liver disease, and 45% for alcohol-related liver disease. 37
The prevalence of NAFLD varies between various races and ethnic groups. Using magnetic resonance spectroscopy to assess hepatic steatosis, NAFLD was evident in 45% Latinos, 33% Whites, and 24% African-Americans. 23 In a recent study, longitudinal data collection among a cohort of 215,000 patients in California and Hawaii showed the highest prevalence of chronic liver disease and cirrhosis among Japanese Americans (6.7%), followed by Latinos (6.7%), Whites (4.1%), African-Americans (3.9%), and Native Hawaiians (3.9%). 38 Although NAFLD was the most common etiology for chronic liver diseases across all ethnic groups, Japanese Americans and Native Hawaiians were significantly more likely to have NAFLD-related chronic liver disease than Whites. 38 The variation among races may be attributed to various factors, including insulin resistance, lifestyle and diet, distribution of adiposity, and genetic factors.
Pathogenesis and Natural History
NASH is thought to be a multifactorial disease with multiple parallel hits, including disrupted lipid metabolism, lipotoxicity, altered cytokines and adipokines, oxidative stress, ER stress, mitochondrial dysfunction, gut-derived endotoxin, and genetic predisposition. Hepatocyte steatosis, evidenced by accumulation of lipid droplets primarily triglycerides within hepatocyte cytoplasm, is a histopathological feature of NASH. It reflects an accumulation of lipids due to lipid input (from dietary fats, circulating free fatty acids (FFAs) from adipose tissue lipolysis, and de novo lipogenesis [DNL]) exceeding lipid output. 39
The liver uptakes FFAs from circulation depending on the concentration of transport proteins including fatty acid transport proteins, fatty acid binding proteins (FABPs), and fatty acid translocase (FAT/CD36), as well as the concentration of FFA in the blood.40–43 NAFLD is associated with increased circulating FFA in the blood and increased hepatic expression of proteins involved in FFA transport. Moreover, DNL, which utilizes metabolic precursors including acetyl-CoA to synthesize fatty acids, has been shown to be upregulated in NAFLD, thus enhancing fatty acid influx into the liver.14,44–47
Hepatic FFA provides energy to the liver through oxidation by mitochondria. It has been shown that in NAFLD, excess mitochondrial oxidation due to increased FFAs eventually results in impaired mitochondrial respiration.48–50 Moreover, increased hepatic FFA as seen in NAFLD leads to stimulation of oxidation in peroxisomes and microsomes in the ER. These hepatic FFAs can also be re-esterified into triglycerides that are assembled into very low-density lipoproteins (VLDL) that can be secreted or stored in lipid droplets. Studies have shown that secretion of VLDL is increased in NAFLD.43,51–53 It appears that in conditions of FFA overload, hepatic steatosis occurs when FFA oxidation and VLDL secretion are unable to utilize the excess FFAs, resulting in esterification into triglycerides and storage in lipid droplets. Therefore, hepatic steatosis can result from increased liver influx of FFA, or perturbation in any of the pathways that usually acts to compensate for excess FFA, including FFA oxidation or VLDL secretion.
Insulin resistance is a major factor underlying hepatic steatosis. Binding of insulin to its receptor leads to activation of phosphoinositol-3-kinase and protein kinase B, initiating the insulin signaling pathway. Under normal conditions, insulin inhibits hepatic gluconeogenesis and enhances hepatic glucose uptake and DNL. Under conditions of insulin resistance commonly seen associated with NAFLD, inhibition of hepatic gluconeogenesis is lost. This leads to increased glucose, which stimulates DNL through the carbohydrate response element-binding protein (ChREBP), while insulin simultaneously retains DNL stimulation via sterol regulatory element-binding protein 1c (SREBP-1c). In NAFLD patients, insulin resistance occurs not only in the liver but also in adipose tissue and skeletal muscle, leading to adipose tissue resistance to the antilipolytic effect of insulin and to reduced uptake of glucose into skeletal muscle, respectively.25,43,54–56 Together, these perturbations caused by insulin resistance lead to steatosis. In addition to insulin resistance in adipose tissue, adipose tissue enlargement and hypertrophy, altered secretion of adipokines including adiponectin and leptin, have been seen in patients with NAFLD.
The gut microbiota has also been shown to play a role in hepatic steatosis. Under healthy conditions, the gut microbiota plays an important role in energy homeostasis in the host individual. 54 There are three main bacterial phyla within the human gut: gram-negative Bacteroidetes, gram-positive Firmicutes, and Actinobacteria. It appears that the composition of gut microbiota is altered in NAFLD in both rodent and human studies, with increased Firmicutes/Bacteroidetes ratio. The gut microbiota also affects body fat and hepatic triglyceride accumulation. Exposure of germ-free mice, which have less total body fat compared to controls with a normal gut microbiota, to gut microbiota derived from conventionally raised mice resulted in insulin resistance and a 60% body fat increase in these originally germ-free mice.57,58 Furthermore, changing the gut microbiota with antibiotics resulted in reduced triglyceride accumulation in mice fed a high fat diet. 59 Mechanisms through which the gut microbiota may affect liver energy metabolism include regulation of short-chain fatty acids, ethanol production, and choline level.
Genetic susceptibility also contributes toward the development of NAFLD. The patatin-like phospholipase domain-containing protein 3 (PNPLA3) I148M variant is a strong determinant of hepatic fat content and predisposes to HCC in the presence of triggering metabolic risk factors including obesity. 60 Two independent genome-wide association studies were the first to link the common rs738409 C > G single-nucleotide polymorphism, which encodes for the I148M variant of PNPLA3 with hepatic fat content, steatosis, and alanine aminotransferase (ALT) levels.61,62 The 148M allele, which results in an amino acidic substitution next to the catalytic domain, decreases PNPLA3 enzymatic activity toward glycerolipids and leads to the development of macrovesicular steatosis.63–65 Individuals with familial hypobetalipoproteinemia, a rare disorder of lipoprotein metabolism, have reduced plasma levels of total cholesterol, low-density lipoprotein cholesterol (LDL-C), and apolipoprotein B with consequently reduced hepatic export of VLDLs, leading to hepatic steatosis. Other genetic determinants associated with NAFLD include the transmembrane 6 superfamily member 2 (TM6SF2) E167K gene variant 66 and the human telomerase reverse transcriptase (TERT) gene. 60 In addition, genome-wide association studies or candidate gene studies have identified a number of genetic variants associated with increased susceptibility to NAFLD.67–70
Recent studies show that microRNAs contribute to pathogenesis of NAFLD/NASH. The most abundant miRNA in the liver, miR-122, is involved in fatty acid biosynthesis and lipid metabolism, cell cycle regulation, and HCV replication.71–74 MiR-122-deficient mice had lower serum triglyceride, total cholesterol, LDL-C, and high-density lipoprotein cholesterol (HDL-C).75,76 Correlation between miR-122-5p and unfavorable lipid profile has been observed in humans. 77 In livers from NASH patients, 23 miRNAs were found to be under- or overexpressed when compared to normal livers. 78 The targets of those differentially expressed miRNAs were predicted to be in cell proliferation, apoptosis, inflammation, oxidative stress, and metabolism. There is currently growing interest in identifying miRNA biomarkers that would distinguish simple steatosis from steatohepatitis, and steatohepatitis from fibrosis. Some microRNA-based therapies, including miR-122 antagonists, are currently being investigated in preclinical studies.
Epigenetic mechanisms also play a role in NAFLD/ NASH pathogenesis. Histone deacetylase 3 (HDAC3) regulates hepatic lipogenesis in murine liver, and its depletion results in lipid synthesis and storage in droplets.79,80 Aberrant DNA methylation, involving DNA methyl transferase (DNMT), is one of the cardinal features of carcinogenesis. In humans, DNMT levels were higher in NASH patients compared to those with simple steatosis and associated with NAFLD activity score. 81 Analysis of 100 human frozen liver sections showed that functionally relevant differences in methylation could distinguish between advanced and mild NAFLD. These findings suggest that differential methylation contributes to differences in pathogenesis of NAFLD. 82
Clinical Assessment
Over the last few years, there has been considerable refinement in the clinical approach to individuals with suspected NAFLD. NAFLD is suspected in individuals with risk factors such as obesity, hypertension, T2DM, and dyslipidemia along with elevation of liver enzymes. The disease, however, does not require the presence of elevated liver enzymes for its diagnosis. When NAFLD is suspected, the presence of hepatic steatosis can be confirmed from a computed tomography (CT) scan, magnetic resonance imaging (MRI), or the continuation attenuation parameter of the fibroscan. The CT scan is sensitive and specific but exposes the subject to radiation. MR-based proton density fat fraction is currently the gold standard for noninvasive evaluation of hepatic steatosis but is also the most expensive method available. 83
From a liver perspective, the risk of a clinical outcome (development of variceal bleeding, hepatic encephalopathy, ascites, or liver-related death) is linked to the development of cirrhosis. The presence of steatohepatitis and the severity of underlying fibrosis are the best predictors of progression to cirrhosis. Steatohepatitis can only be diagnosed by a liver biopsy; consequently, it does not lend itself to widespread application. Substantial progress has been made in the assessment of hepatic fibrosis using noninvasive methods.
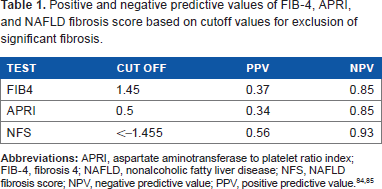
There are three major groups of noninvasive tests that are used for the assessment of hepatic fibrosis. The first relies on commonly used laboratory tests and indices developed from them. The three most robust of these include the fibrosis 4 (FIB-4) index, aspartate aminotransferase (AST) to platelet ratio (APRI), and the NAFLD fibrosis score. 26 Of these, FIB-4 and APRI are etiology-agnostic markers of fibrosis. These tests have a very high negative predictive value84,85 and can exclude fibrosis with accuracy (Table 1). While FIB-4 > 3.2 and APRI > 1.5 can detect advanced fibrosis with a positive predictive value of 0.8–0.85,86–88 their ability to diagnose cirrhosis is limited. All three tests have been shown to predict liver-related outcomes and all-cause mortality in those with NAFLD. 89
Positive and negative predictive values of FIB-4, APRI, and NAFLD fibrosis score based on cutoff values for exclusion of significant fibrosis.
Specific laboratory tests to evaluate hepatic fibrosis, including lysyl oxidase levels, extended liver fibrosis panel, fibrometer, hyaluronic acid, and procollagen III n-terminal peptide, are currently under further study. These tests have not yet been convincingly shown to be superior to the panels based on simple laboratory measures described above.
The best noninvasive tests to evaluate hepatic fibrosis are based on elastography. Vibration-controlled transient elastography, also known as fibroscan by Echosens, is the most studied of these ultrasound-based methods. Vibration-controlled transient elastography can identify fibrosis stage with an area under the receiver operating characteristic curve (AUROC) of 0.8–0.9 and can also detect cirrhosis with an AUROC greater than 0.9. 90 It is, however, operator dependent and is affected by obesity, hepatic inflammation, hepatic congestion, cholestasis, and postprandial state. MR elastography is the gold standard for detection of fibrosis noninvasively and can identify fibrosis stage with an AUROC greater than 0.9. 91 However, it is substantially more expensive than other modalities, and up to 5% of subjects are unable to tolerate the procedure due to size or claustrophobia.
Aside from progression of liver disease, NAFLD patients are also at increased risk for cardiovascular disease and cancer.9,31,32 Thus, a careful assessment of cardiovascular risk is recommended, and appropriate therapy is provided to minimize risk. Similarly, it is recommended that patients follow all current guidance on cancer screening. Risk stratification and management in NAFLD requires a multidisciplinary approach often involving gastroenterologists, hepatologists, primary care physicians, endocrinologists, and nutritionists/dietitians.
Based on the presence of risk factors, evidence of steatosis, and markers of fibrosis, subjects with NAFLD can be categorized into three categories with respect to their risk of liver outcomes (Fig. 1). (I) Low-risk individuals have one or no risk factors for NAFLD, with laboratory panels (APRI, FIB-4, NAFLD fibrosis score) and liver stiffness measures below the threshold of detection of fibrosis. (II) Intermediate-risk individuals have multiple risk factors and elevated ALT but do not have other features of advanced disease. In selected cases of intermediate phenotype or when the diagnosis is in doubt, a liver biopsy is performed to confirm the diagnosis and assess the risk of developing cirrhosis. (III) High-risk individuals have advanced fibrosis and are characterized by an AST:ALT ratio >1, platelet count <150,000/mm3, and liver stiffness measurements indicative of bridging fibrosis or cirrhosis. Typically, such patients are older (age >50 years) and have multiple features of the metabolic syndrome.
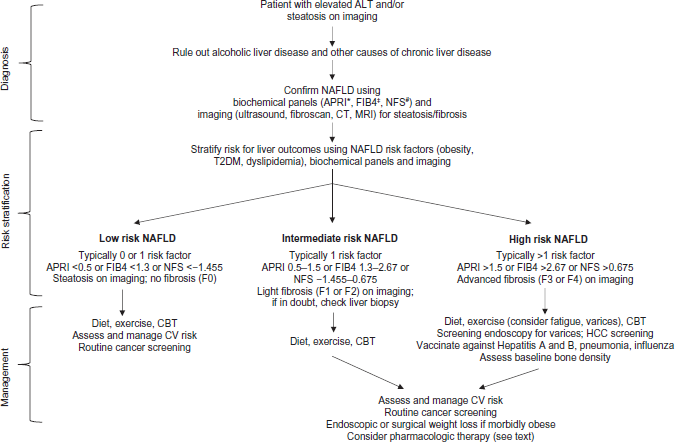
Assessment and management of NAFLD. Patients with NAFLD typically come to the clinician's attention due to elevated alanine aminotransferase (ALT) or steatosis on imaging usually done for unrelated indications. These patients should undergo evaluation to rule out alcoholic liver disease and etiologies other than NAFLD that could cause chronic liver disease. Diagnosis of NAFLD is confirmed using biochemical panels and imaging studies aimed at assessing steatosis and fibrosis. These confirmatory studies, together with NAFLD risk factors, are used for patient stratification into low-, intermediate-, and high-risk categories for liver-related outcomes. Recommendations are provided for management of patients in the different risk categories.
Treatment of NAFLD
Low-risk NAFLD subjects
NAFLD or NASH patients without fibrosis are at low risk of liver-related outcomes over a 10-year time frame, 92 hence, any therapeutic intervention must have a very high safety profile since the benefits for the group as a whole will be modest over this time frame. Currently, diet, exercise, lifestyle modifications, and engagement in healthy living and wellness form the cornerstone of care for such individuals. While their liver-related outcomes may be low, they are still at risk for cardiovascular disease and development of cancer. Therefore, attention must be paid to the patient's cardiovascular risk and routine cancer screening.
Many diets have been prescribed for NAFLD and are derived largely from the obesity literature. However, there is a paucity of high-quality long-term trials to demonstrate the beneficial effects of diet on NAFLD. Many subjects are unable to engage in lifestyle changes and to sustain such changes over a long period of time, greatly limiting the feasibility of long-term studies. This may be partly attributable to the failure of many patients to perceive their lifestyle as a factor contributing to their clinical condition. Recent data suggest that many subjects are in a precontemplation stage within the spectrum of change with respect to lifestyle and behavior. 93 In addition, active eating disorders including binge eating is overrepresented in this population. The biological impact of binge eating and nocturnal eating disorder are yet unknown. The best data to support the beneficial effects of lifestyle change on NAFLD come from studies in which reduction in caloric and fat intake with or without physical activity led to improvement in steatohepatitis.94–96
The potential impact of exercise on NAFLD has also not been evaluated systematically. It is currently recommended that activity guidelines from the American Diabetes Association or American Heart Association be followed in this population. The authors also find that the use of pedometers and setting modest achievable goals, coupled with repeated positive reinforcement, are key elements to the success of lifestyle interventions. The effect of yoga, tai chi, and other exercises that are combined with meditation on long-term weight loss, improvement in metabolic status, and liver disease are areas for future research.
Intermediate-risk NAFLD subjects
These are typically individuals with steatohepatitis with some fibrosis. At a noninvasive level, they are likely to have features of the metabolic syndrome, progressive weight gain, steatosis on imaging, fibrosis markers above the threshold for fibrosis, and elastography data suggestive of some fibrosis but not cirrhosis. Intermediate-risk NASH patients also need to be assessed for their cardiovascular status and cancer risk management. Lifestyle intervention remains a cornerstone of care and is essential. In addition, these subjects may be considered for pharmacological therapy. It is important to note that no drugs are currently approved for NAFLD. At present, all pharmacological recommendations for NAFLD represent off label or experimental use and should be administered only with complete understanding of risks/benefits by both provider and patient. Current treatment guidelines propose vitamin E as first-line treatment for NASH with diabetes or cirrhosis.
Vitamin E has been studied in phase 2A and 2B trials.97–99 At a dose of 400–800 IU/day, it consistently improves steatosis and steatohepatitis but has not yet been shown to improve fibrosis stage for the group as a whole. It does not affect insulin resistance, body weight, or the cardiovascular risk factors such as LDL-C or HDL-C. A recent meta-analysis, presented in abstract form only, demonstrated that it is effective in diabetic subjects with NASH as well. 100 Vitamin E works in only 40%–45% of subjects and most subjects are nonresponders. It is therefore currently recommended that therapy be reserved for those with a well-defined risk profile for disease progression and that the benefits of treatment be monitored with histologic assessment of the liver. A simple assessment of liver enzymes is inadequate to evaluate whether vitamin E has resolved NASH. That being said, it has been shown that those who lose weight and normalize ALT levels on vitamin E have a high probability of NASH resolution. 101 At the same time, those with weight gain and increasing ALT are unlikely to have histological improvement. 101 Levels of indole propionic acid, an intestinal bacterial product, have been shown to identify those who are likely to respond to vitamin E. 102
Pioglitazone is a peroxisome proliferator-activated receptor (PPAR)-γ agonist that has also been shown to improve NASH,97,103,104 and in one meta-analysis, it was found to improve hepatic fibrosis. 105 It is an insulin sensitizer and improves insulin resistance. At a dose of 30 mg/day, it improves steatosis, ballooning, inflammation, and possibly fibrosis. However, pioglitazone use is associated with weight gain and osteopenia. There is also concern for increased risk of bladder cancer,106,107 although this risk seems to be lower than previously thought. 108 The potential for weight gain and fluid retention have been the principal reasons why it is used as a backup or second-line agent.
Liraglutide is a GLP-1 agonist that is administered subcutaneously. In a recent trial, it had a remarkable benefit on steatohepatitis within one year compared to placebo-treated subjects. 109 Although this was a multicenter study, the number of patients was fairly small and the placebo response rates were much lower than those reported in literature. Therefore, while promising and provocative, these data need to be replicated in large-scale multicenter trials before any recommendations about its use can be made. Pentoxifylline is another agent that has been studied in small pilot studies.110–113 While the data appear to be promising, they need to be confirmed in long-term trials. It is currently used mainly as a second-line agent, with nausea being a common side effect. Ursodeoxycholic acid has been studied and improves liver enzymes and markers of insulin sensitivity.114–116 It improves steatosis but its effects on features of steatohepatitis are not clear-cut and thus cannot be recommended as a primary therapy for NASH. Metformin, an insulin sensitizer that presumably works by improving adenosine monophosphate-activated protein kinase (AMPK) activity in the liver, 117 has also been studied in several clinical trials and shown to improve insulin sensitivity in the short term and even reduce steatosis. However, it does not improve steatohepatitis and so cannot be recommended as a treatment for NASH.
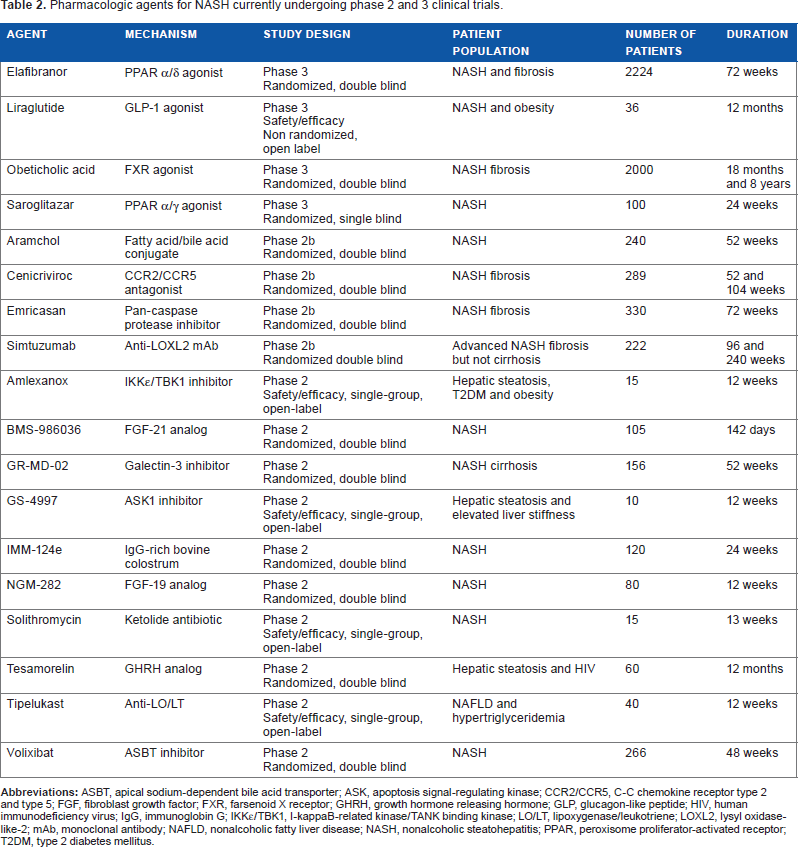
Several other agents are currently in clinical trials. Of these, obeticholic acid (OCA) and elafibranor are currently in phase 3 trials. OCA is a farsenoid X receptor (FXR) agonist that has been shown to improve insulin sensitivity in subjects with T2DM and suspected NASH. In a large phase 2B trial, it convincingly improved steatosis, inflammation, ballooning, and fibrosis. 118 It demonstrated a trend for improvement in steatohepatitis. However, it increases LDL-C and also causes pruritus in some subjects. The long-term implications of the increase in LDL-C are unclear and remain to be clarified, as this is important in assessing the long-term utility of OCA. Elafibranor is a PPAR α/δ agonist that increases lipid oxidation and inhibits macrophage activation.119,120 In the GOLDEN trial, 121 it improved hemoglobin A1C, LDL-C, and HDL-C. Although it missed the a priori primary histological improvement endpoints, a post hoc analysis demonstrated that an improvement occurred in those with NASH and NAFLD activity score >4. Several other agents that work through various mechanisms are in clinical trials at this time (Table 2).
Pharmacologic agents for NASH currently undergoing phase 2 and 3 clinical trials.
High-risk NAFLD subjects
These are subjects with cirrhosis; thus, management of cirrhosis, which is largely etiology agnostic, must be considered in addition to NASH-specific therapy. All subjects should have screening endoscopy for varices and regular hepatic imaging for HCC screening.122–124 Baseline bone density assessment and vaccinations for hepatitis A and B, pneumonia, and influenza should be considered. 122 Similar to low- and intermediate-risk subjects, lifestyle management is the cornerstone of care for high-risk NAFLD subjects, with the caveat that the severity of fatigue and the presence of varices should be factored into the types of recommendations for exercise. 125 Appropriate screening for encephalopathy should be instituted. 122 Attention to overall salt intake is important, given its role in development of ascites. There are currently no specific drugs recommended for treatment of NASH-related cirrhosis. Studies focused on lysyl oxidase 126 are nearing completion and other studies to inhibit galectin, a profibrogenic molecule, are under way.
Summary
NASH is a very common disease. Knowledge about NASH is rapidly evolving and the pathways for evaluation are becoming clearer, with a greater emphasis on noninvasive tools. The treatment of NASH currently involves lifestyle management and optimizing weight along with judicious use of vitamin E and pioglitazone, with pentoxifylline and liraglutide as backup agents. Of these pharmacologic agents, the data for pioglitazone are far more robust. Many drugs are in advanced phase trials, and the results of these trials are likely to influence pharmacological treatment for NASH in the near future.
Author Contributions
Wrote the first draft of the manuscript: BAB, AJS. Contributed to the writing of the manuscript: BAB, AJS. Agree with manuscript results and conclusions: BAB, AJS. Jointly developed the structure and arguments for the paper: BAB, AJS. Made critical revisions and approved final version: BAB, AJS. Both authors reviewed and approved of the final manuscript.
