Abstract
Objectives
Review and summarize the mechanism of action of methylnaltrexone bromide (methylnaltrexone) and its effectiveness in the treatment of opioid-induced constipation.
Data Source
A multi-database search was conducted using PubMed and MEDLINE databases, in addition to electronic links to related articles and references.
Background
Opioids are effective medications for the management of moderate to severe pain, but they are associated with a number of side effects, especially within the gastrointestinal system. Constipation is a very common adverse reaction in patients with late-stage, adverse illness, who require long term administration of opioids on a chronic basis to help alleviate pain. In April 2008, the Food and Drug Administration approved the use of methylnaltrexone, a quaternary derivative of naltrexone which does not cross the blood brain barrier, for the management of patients with opioid-induced constipation. Methylnaltrexone acts as a selective peripheral Mu-receptor antagonist, without affecting the effects of opioids on central analgesia.
Conclusions
Studies have been shown that methylnaltrexone can be used safely in the treatment of opioid-induced constipation without either interfering with opioid effects on central anesthesia or precipitating opioid withdrawal.
Introduction
Opioids are commonly used in the management of moderate to severe pain in patients with late-stage, advanced illness. Opioids are potent analgesics that cross the blood brain barrier but they can cause side effects that affect patient quality of life. Constipation is one of the most common adverse reactions of opioid medications; their action is at the Mu-peripheral receptor, located in the smooth muscle (biliary system, bowel wall, bladder, etc.) producing relaxation and decreased motility.
Specific blockade of the adverse gastrointestinal effects of opioids has been tried for many years with opioid-receptor antagonists such as naloxone and nalmefene. These attempts have been met by the inability to separate the centrally acting beneficial effects of opioids (analgesia) from peripherally acting detrimental effects (gastrointestinal dysmotility) and thus could not provide a net clinical benefit for opioid-induced constipation in patients with late-stage, advanced illness.
A new and novel class of drug, methylnaltrexone, a peripherally acting Muopioid-receptor antagonist, approved by the FDA in April 2008, has accomplished specific activity at gut opioid receptors and blocks the adverse effects of administered opioids without inhibiting their centrally mediated analgesic effects or precipitating opioid withdrawal.
Place in Therapy and Indications
Methylnaltrexone is indicated as a therapeutic option in the treatment of opioid-induced constipation; in patients who are receiving opioids used in the management of moderate to severe pain, with late-stage, advanced illness, when response to laxative therapy has not been sufficient.1–3
Opioids are potent analgesics that cross the blood brain barrier, helping to alleviate pain. They can cause a series of side effects in the periphery, affecting the quality of life of the patient. Constipation is one of the most common adverse reactions of opioid medications.1,2,4
Methylnaltrexone blocks the peripheral action of opioids in the intestinal smooth muscle without interfering with the effects of central anesthesia or precipitating opioid withdrawal in patients with advanced illnesses. Most patients had an inadequate response to traditional laxatives.1,2,4
Mechanism of Action, Metabolism and Pharmacokinetics
Methylnaltrexone is a quaternary derivative of naltrexone;2,3,5 the compound is a white, odorless powder, freely soluble in water, 4 which does not cross the blood brain barrier due to its lower lipid solubility.1,6 (Figure 1)
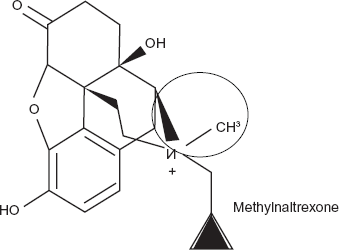
It is a peripherally acting Mu-opioid receptor antagonist, 1 which blocks opioid effects without affecting the central analgesic effect or precipitating opioid withdrawal.1,2,4 Studies have demonstrated the higher affinity of methylnaltrexone to the Mu-receptor compared to its lower affinity at the Delta and Kappa opioid receptor.1,4,7
The lack of central activity was demonstrated when methylnaltrexone did not cause withdrawal symptoms in opioid-tolerant dogs at higher doses compared to lower doses of naloxone which did cause symptoms.6,7
Methylnaltrexone only binds to peripheral receptors, unless it is administered directly to the brain. This was demonstrated by the inability of subcutaneous methylnaltrexone to reverse opioid-induced cataplexy in rats compared to intracerebral methylnaltrexone that did reverse at lower doses. 1 Also, the lack of central activity was evident by the ability of methylnaltrexone to reverse opioid-induced constipation without affecting opioid-induced analgesia or producing withdrawal signs.1,2,4,6
Nevertheless, published data suggest that the selectivity of methylnaltrexone in the periphery may be dependent to species. Rats and mice have a greater capacity than dogs and humans to N-demethylate the quaternary ammonium compound, N-methylnaltroxe to naltrexone, which easily penetrates the blood brain barrier. However no appreciable N-demethylation of methylnaltrexone was observed in dogs or humans.1,8 This may indicate, once again, the selectivity of methylnaltrexone exclusively in the periphery.
The action of methylnaltrexone in reversing opioid-induced constipation was demonstrated in isolated guinea pig ileum and human intestine by increasing bowel contractility and reducing gastrointestinal transit time. This can be achieved with IV, subcutaneous and oral administration of methylnaltrexone. 9
Intravenous or subcutaneous methylnaltrexone is rapidly absorbed in a dose-dependent manner. Its peak concentration is reached in 16 to 30 minutes and its half life is approximately 105 to 140 minutes while, orally, it is less well absorbed.2,3,7
The steady state of volume of distribution is 1.1 L/Kg, it is distributed to the tissue moderately and it is 11% to 15% bound to human plasma protein. 6
Methylnaltrexone is excreted in the urine primarily and is less so in feces as unchanged drug. Table 1 provides a brief overview of methylnaltrexone properties.2,3,7,10
Properties of methylnaltrexone.

Safety
Methylnaltrexone is contraindicated in patients with known or suspected bowel obstruction. The patient should discontinue methylnaltrexone if severe or persistent diarrhea occurs during treatment.2,3
Methylnaltrexone is a weak inhibitor of CYP2D6. 23 No significant drug interaction has been identified. Studies have shown that the most common adverse reactions of methylnaltrexone are abdominal pain (28.5%), flatulence (13.3%), nausea (11.5%), dizziness (7.3%), and diarrhea (5.5%). Most patients have reported gastrointestinal cramping and pain after administration of methylnaltrexone. They reported this as a mild to moderate symptom, very similar to defecation, and it disappears after a bowel movement6,11–14 Transient orthostatic hypotension can occur at surpratherapuetic doses (0.64 and 1.25 mg/kg) when methylnaltrexone is administered IV.1–3,13
Methylnaltrexone dosing is weight based. It is administered as one dose every other day, as needed, but no more than one dose should be given in a 24 hour period. The recommended dose is 8 mg for patients who weigh between 38 kg and 62 Kg or 12 mg for patients who weigh 62 kg to 114 kg; patients outside of this range should receive 0.15 mg/kg.2,3 Table 2 summarizes dosing recommendations of methylnaltrexone.
Dosing recommendation of methylnaltrexone.

One single dose of methylnaltrexone (12 mg/0.6 mL vial) costs $60.49 in the United States and about $50–55 in Europe. The methylnaltrexone kit, which contains 7 trays, costs about $300.57 in the United States.15,16
No dose adjustment is required in patients with mild to moderate renal or hepatic impairment; however, dose reduction is required by one-half in patients with severe renal impairment (creatinine clearance <30 mL/min).2,3
Methylnaltrexone is in pregnancy category B, safety and efficacy in children has not been studied. Differences in safety and efficacy in the elderly compared to younger patients have not been demonstrated.2,3
Efficacy of Methylnaltrexone in Clinal Studies
Preclinical studies have demonstrated the efficacy of methylnaltrexone in animals, effectively antagonizing opioid-induced inhibition of gastrointestinal motility.6,9,17–21 Its action is exclusively at the peripherally acting opioid-receptors vs. centrally acting opioid-receptors.6,17,19 In early studies, when methylnaltrexone was administrated subcutaneously, it antagonized opioid-induced inhibition of gastrointestinal transit in rats without affecting opioid effects on analgesia. However, when methylnaltrexone was administered intracerebrally, it did affect analgesia, showing independent action and selectivity of methylnaltrexone to centrally or peripherally acting opioid-receptors. 18 In another study, in isolated guinea-pig ileum and human small intestine, methylnaltrexone showed its efficacy in preventing opioid-induced dysmotility of gastrointestinal tract and, at the same time, enhanced the force of smooth muscle contraction. 9
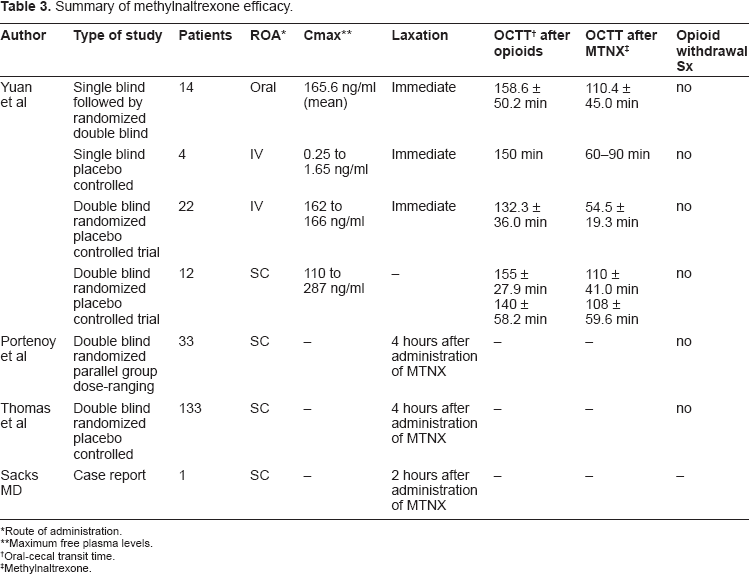
Oral methylnaltrexone efficacy was assessed in 14 healthy volunteers in 2 phases. In phase A, subjects received three increasing oral doses of methylnaltrexone (0.64, 6.4, and 19.2 mg/kg) to evaluate and obtain safety and tolerance information. In phase B, subjects received either: placebo + placebo, placebo + morphine 0.05 mg/kg IV or methylnaltrexone 19.2 mg/kg orally + morphine 0.05 mg/kg IV and the effects of oral methylnaltrexone on opioid-induced delay in oral-cecal-transit time were assessed. Doses of oral methylnaltrexone were well tolerated. After administration of morphine, an increase in gastrointestinal transit time was observed from 114.6 ± 37.0 to 158.6 ± 50.2 minutes (P < 0.001). Methylnaltrexone combined with morphine prevented delays in gastrointestinal transit (not significant) compared with baseline levels (110.4 ± 45.0 minutes, P = 0.28). 22
The efficacy of intravenous methylnaltrexone was evaluated in a single blind, placebo- controlled study in four patients with chronic opioid-induced constipation. On day 1, all patients received intravenous placebo followed by intravenous methylnaltrexone (0.05 to 0.45 mg/kg) twice a day. Laxation, oral-cecal-transit time and opioid withdrawal symptoms were observed and compared. Patients 1 and 2 showed immediate laxation after administration of methylnaltrexone (0.45 mg/kg). Patients 3 and 4 also showed immediate laxation after a reduced dose of methylnaltrexone IV (0.05 to 0.15 mg/kg). The reduction was made after patient 2 experienced severe abdominal cramping and he was discontinued from the study. Patients 1, 3 and 4 did not experience any adverse reactions after administration of methylnaltrexone. Oral-cecal-transit time in the three remaining patients reduced from 150 minutes (baseline) to 90, 60 and 60 minutes after administration of methylnaltrexone. 11
Intravenous administration of methylnaltrexone was also assessed in a double blind, randomized, placebo-controlled trial in 22 constipated patients who were enrolled in a methadone maintenance program. Patients were divided into 2 groups; 11 received placebo IV and 11 received methylnaltrexone IV in intervals for 2 days. Laxation, oral-cecal-transit time and opioid withdrawal symptoms were observed and compared. The 11 patients treated with placebo showed no immediate laxation. However, 10 of 11 patients treated with methylnaltrexone IV showed immediate laxation on day 1, and on day 2 all 11 patients achieved laxation (p < 0.001). Oral-cecal-transit time significantly decreased after administration of methylnaltrexone from baseline levels of 132.3 ± 36.0 to 54.5 ± 19.3 minutes, whereas oral-cecal-transit time after placebo administration remained unchanged. No opioid withdrawal symptoms were observed in any group. 12
Subcutaneous methylnaltrexone activity was assessed in a randomized, double blind, placebo-controlled trial, enrolling 12 healthy volunteers. Effects on oral-cecal-transit time, adverse reactions and pharmacokinetics were studied. The subjects were divided into 2 groups, each group received IV morphine 0.05 mg/kg and transit time was observed. In the first group, after 0.1 mg/kg subcutaneous methylnaltrexone, transit time decreased from 155 ± 27.9 (after morphine) to 110 ± 41.0 (after methylnaltrexone) minutes. In the second group, after administration of 0.3 mg/kg subcutaneous methylnaltrexone, oral-cecal-transit time decreased from 140 ± 58.2 to 108 ± 59.6 minutes. No adverse reactions were observed in the study. 5
Subcutaneous methylnaltrexone efficacy was described in a case report of a 56 year old woman with metastatic breast cancer to bone, liver and brain. The patient had not had a bowel movement for 10 days. Medical history reveled that she had been on a fentanyl patch for months. A bowel regimen was started and, following an inadequate response, subcutaneous methylnaltrexone was administered, producing laxation within 2 hours. 23
The effects of subcutaneous methylnaltrexone were also assessed in a double-blind, randomized, parallel group, dose-ranging study in 33 patients with advanced illness and opioid-induced constipation despite laxative therapy. Patients were treated with an initial dose-range of 1 mg, 5 mg, or 12.5 mg on days 1, 3 and 5. Then the initial dose was extended by including a 20 mg dose-group. The primary outcome was a laxation response within 4 hours after the first dose. Only 10% who received 1 mg dose experienced laxation within 4 hour; 43% of the 5 mg dose, 60% of the 12.5 mg dose, and 33% of the 20 mg dose experienced laxation within four hours of dosing. The median time to laxation was more than 48 hours for the 1 mg dose-group and 1.26 hours for patients receiving ≥5 mg (P = 0.0003). There was no apparent dose response with doses greater than 5 mg. 24
In a randomized double blind, placebo-controlled trial, 133 patients with advanced illness and opioid-induced constipation were treated with subcutaneous methylnaltrexone. Subjects received either placebo or 0.15 mg/kg subcutaneous methylnaltrexone for 2 weeks. Primary outcome was laxation within 4 hours after the first doses. The 48% treated with subcutaneous methylnaltrexone had laxation within 4 hours after the first dose, compared with 15% in the placebo group (P < 0.0001). Laxation occurred in more than 70% of the subjects 4 hours after at least one dose of methylnaltrexone. Patients in the placebo group reported the use of more laxatives compared to methylnaltrexone. No withdrawal symptoms were observed in this study. 13
A double-blind, randomized, placebo-controlled trial was conducted with 154 patients who had advanced-illness and opioid induced constipation in hospice and palliative care centers, during 2003–2005. Patients received a single dose of subcutaneous methylnaltrexone (0.15 mg/kg or 0.3 mg/kg) or placebo. Laxation within 4 hours was achieved in 62% and 58% for methylnaltrexone 0.15 mg/kg and 0.3 mg/kg, respectively, compared with 14% for placebo (P < 0.0001). Approximately half of the methylnaltrexone-treated patients defecated within 30 minutes of dosing. Most adverse events were related to the gastrointestinal system (abdominal pain and flatulence). Administration of methylnaltrexone did not change pain scores or result in opioid withdrawal. 14
The role of methylnaltrexone has been evaluated in clinical practice, with very surprising results. Besides its use in opioid-induced constipation in patients with advanced-illness, the attention of methylnaltrexone has been turned in the management of several clinical settings. In a randomized, double blind, placebo-controlled study, the efficacy of methylnaltrexone was evaluated in postoperative ileus in 65 patients who underwent segmental colectomy. Patients received either placebo or methylnaltrexone (0.3 mg/kg) very 6 hours, until discharge. Treatment was initiated after surgery. Patients who were treated with methylnaltrexone reported quicker response in bowel movement, compared with those receiving placebo (98 hours vs. 118.1 hours; P = 0.038) and earlier discharge eligibility (116.1 hours vs. 148.7 hours; P = 0.049). No significant differences were observed in time toleration of full liquid (P = 0.119), solid (P = 0.205), GI recovery (P = 0.263). More adverse reactions were observed in the placebo group. 25 This may represent an ideal opportunity to establish standardized interdisciplinary protocols that accelerate recovery, reduce postoperative morbidities and length of stay in the management of postoperative ileus.
Another beneficial effect of methylnaltrexone was also assessed in a randomized, double blind, crossover study, in patients with bladder dysfunction, due to its peripheral action in the smooth muscle. Infusion of remifentanyl (0.15 mcg/kg/min) was given to 13 healthy volunteers, followed by IV dose of methylnaltrexone (0.3 mg/kg), naloxone (0.01 mg/kg) or saline. Urodynamic effects were evaluated and pupil size was measured. Remifentanil infusion caused a decrease in detrusor pressure, complete urinary retention and miosis. Voiding was possible after methylnaltrexone, naloxone and saline infusion (P < 0.0001); miosis was reversed by naloxone but not methylnaltrexone or saline (P < 0.0001). 26 This data indicates that methylnaltrexone may play an important role for reversal of urinary retention without reversing central opiod effect.
There is some evidence that opioids may block cough reflex and peripheral mechanisms mediate it. This was demonstrated when methylnaltrexone (2.0 mg/kg) blocked opioid-induced cough suppression without affecting nocioception in guinea pigs. 27
It seems opioids also play an important role in the control of nausea and vomiting. Opiods receptor are located outside the blood brain barrier, at the chemo-receptor trigger zone (CTZ) in the area postrema, therefore is considered a physiologic peripheral compartment. Methylnaltrexone due to its peripheral action may help blocking opioid-induced emesis. In early studies, intramuscular (0.25 mg/kg) or intravenous (0.2 mg/kg) methylnaltrexone blocked the emetic effects of morphine in dose dependent manner, when administered in dogs (P < 0.05). 28
Yuan et al showed the efficacy of methylnaltrexone in a recent study in which rats reacted to nauseous and emetic stimulus by increasing their oral intake of non-nutritive substance like kaolin, after administration of oral ritonavir (20 mg/kg; P < 0.01). This effect was attenuated by intraperitoneal injection of methylnaltrexone in a dose dependent manner (P < 0.001). 29 This result shows potential benefit in decreasing nausea and vomiting in HIV patients who take ritonavir.
Conclusion
Opioids are commonly use in the management of moderate to severe pain, usually in nonmalignant medical conditions, for pain relief in the postoperative setting and for patients with late-stage advanced illness. Unfortunately, opioids are associated with a number of side effects, especially within the gastrointestinal system. Constipation is a very common adverse reaction in patient who requires administration of opioid on a chronic basis.
This has led to treating opiod-induced constipation as an extension of constipation in general, with the use of stool softeners, bulking agents, laxatives, lactulose, polyethylene glycol, etc; but none of them have been well studied to support the efficacy in patients. For this reason, attention has turned to blocking antagonizing opioid effects in the gastrointestinal system. Naloxone, a tertiary opioid-receptor antagonist, has been studied to alleviate the effect of opioids in the gut. Unfortunately, due to its central and peripheral action, naloxone reverses analgesia and precipitates opioid withdrawal.
A new class of drugs, peripherally acting Muopioid receptor antagonists, may help enhance multimodal management of opioid induced bowel dysfunction. Studies have shown that methylnaltrexone can be used safely in the treatment of opioid-induced constipation, due to its action on Mu-opiod receptors located in the periphery. It can be administered in many different ways (oral, intravenous and subcutaneous); it may antagonize the effects of opioid-induced constipation, previously described, producing laxation within 4 hours, without interfering with central anesthesia or precipitating opioid withdrawal (Table 3).
Summary of methylnaltrexone efficacy.
Route of administration.
Maximum free plasma levels.
Oral-cecal transit time.
Methylnaltrexone.
The effects of methylnaltrexone have been tested in other important implications for clinical practice, such as nausea, vomiting, gastroparesis, urinary bladder retention, etc; with very surprising results, but this promising effects need to be confirmed by additional clinical trials. It is clear that methylnaltrexone decreases opioid-induced constipation without affecting pain control or precipitating oipiod withdrawal, based on the available data. To assess effectiveness of this drug in other clinical settings, there is a need for studies assessing clinical practice.
Disclosures
The authors report no conflicts of interest. Dr. Saclarides is a speaker for Adolor/GSK.
