Abstract
Globally, the incidence of tuberculosis (TB) is declining but the proportion of drug-resistant cases has increased. Strains resistant to both isoniazid and rifampin, and possibly other antibiotics, called multidrug-resistant (MDR), are particularly difficult to treat. Poorer outcomes, including increased mortality, occur in patients infected with MDR strains and the costs associated with treatment of MDR-TB are substantially greater. The recent recognition of MDR-TB and strains with more complex resistance patterns has stimulated the development of new TB medications including fluoroquinolones, oxazolidinones, diarylquinolines, nitroimidazopyrans, ethylenediamines, and benzothiazinones. Bedaquiline, a diarylquinoline, was approved for the treatment of MDR-TB in 2012. Addition of delamanid to WHO-approved treatment improved outcomes for MDR-TB and for extensively drug-resistant TB in a large randomized, controlled phase II clinical trial and is undergoing evaluation in a large international phase III study. This review will focus on MDR-TB and the role of delamanid in its treatment.
Introduction
The recently published global tuberculosis (TB) report by the World Health Organization (WHO) was cautiously optimistic.
1
Between 2010 and 2011, the incidence of TB decreased 2.2%.
1
Since 1990, TB mortality declined 41% and the 2015 millennium development goal of reducing the 1990 TB mortality rate by 50% will likely be achieved.
2
Unfortunately, multidrug-resistant (MDR) cases, patients with
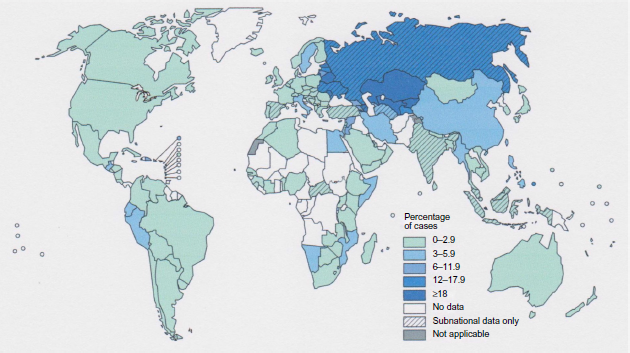
Percentage of new TB cases with MDR-TB.
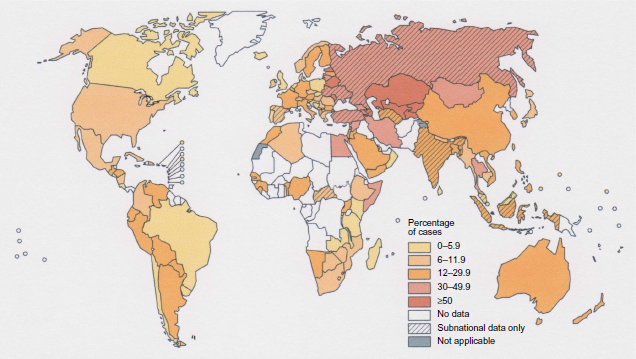
Percentage of previously treated TB cases with MDR-TB.
The aim of this paper is to review the potential role of delamanid in the treatment of MDR-TB. To identify citations, PubMed searches were performed with the terms ‘treatment of multidrug-resistant tuberculosis’, ‘new drugs for the treatment of tuberculosis’, ‘nitroimidazopyrans’, ‘OPC-67683’ and ‘delamanid’. The bibliographies of the relevant references were also searched.
Treatment of multidrug-resistant tuberculosis
The WHO advises that patients with drug-sensitive (DS) TB be treated in directly observed therapy, short term (DOTS) programs with four drugs, generally isoniazid, rifampin, ethambutol, and pyrazinamide for 2 months followed by thrice weekly isoniazid and rifampin for a further four months.4,5
Previous TB treatment is the greatest risk factor for MDR-TB. 6 Unfortunately, treatment is considerably more difficult and outcomes are significantly worse in patients with MDR-TB and especially in patients with XDR-TB. 7 Patients with drug-resistant (DR)-TB require more complex drug regimens for longer periods and treatment of DR-TB is a great deal more expensive; an added burden in high prevalence countries with limited resources. 7
Poor outcomes, high treatment failure rates, recognition that inadequate treatment of MDR-TB results in more complex drug resistance patterns including XDR-TB, and the perceived effectiveness of DOTS programs, inspired the development of DOTS plus.8,9 These are multidimensional programs that specialize in the management of MDR-TB to provide appropriate laboratory support and medical personnel with the necessary expertise to treat MDR-TB. 8 They require adequate financial support, often from international agencies if unavailable locally, to offer care that is substantially more expensive and complex than required to treat DS-TB, and the resources to monitor performance of the program to facilitate quality improvement. 8 DOTS plus programs must provide reliable laboratory services to dependably diagnose pulmonary TB, perform drug susceptibility testing (DST) on MTB isolates against all first-line and ideally also against all second-line agents, and monitor sputum culture and DST results serially through the obligatory 20 month or longer MDR-TB treatment period. Medical services and medications should be made available free of charge, including reliable access to the required 2nd-line medications. Medications should be dispensed within a DOT strategy. Even in the USA, interruptions in the availability of 2nd-line drugs occur. 10 Appropriately-trained medical personnel, physicians and nurses with the necessary expertise, including the experience to properly manage adverse drug reactions with established algorithms, are essential for an effective DOTS plus program. 8
Treatment of MDR-TB is resource intensive. In South Africa, MDR-TB cases comprise 2.2% of the total, yet account for 32% of the national TB management budget. 11 Per patient cost for DS-TB treatment averaged $257, compared to means of $6,772 and $26,392 per MDR- and XDR-TB case, respectively. 10 Treatment is even more expensive if patients require hospitalization. 12 MDR-TB treatment is expensive relative to the gross domestic product of a particular country. In Peru, the estimated average cost per MDR-TB case is $2,423 and $14,657 in the Tomsk region of Russia. 13 Despite the expense of treating MDR-TB, the cost per disability-adjusted life year (DALY) is reasonable at $143 in the Philippines, $243 in Peru, and $745 in Tomsk, Russia. 13 In the USA, the median treatment duration for patients with MDR-TB is 766 days. 14 Estimates of cost or cost per DALY in the USA have not been published but undoubtedly are substantial.
Sputum culture conversion, ie, microbiological cure, is less likely in patients with MDR-TB if the sputum mycobacterial smear is positive at treatment initiation, the isolate is also resistant to pyrazinamide, fluoroquinolones, injectables, or a thioamide, the patient experienced a poor TB treatment outcome previously, or suffers from alcoholism or drug addiction.15–18 Even in the optimal setting of a DOTS plus program, only 65% are cured or complete therapy, 11% die, 14% default, and 7% fail therapy. 19 American data confirm that both 1st- and 2nd-line MTB drug resistance contribute to treatment failure rates and mortality. 20 As expected, treatment outcomes are better in HIV-negative patients.
The WHO treatment recommendations for MDR-TB are based on case series, retrospective chart reviews, and expert opinion because of the costs and difficulties undertaking clinical trials in these patients.21–23 The heterogeneity of drug resistance profiles, high prevalence of HIV co-infection, need for prolonged treatment courses, and resource limitations in high prevalence countries often jeopardize successful therapy. Even in controlled trials, management is difficult and patients often fail to complete treatment. 24 Intensive regimens containing a minimum of five effective tuberculosis antibiotics, including an injectable and a fluoroquinolone, reduce the risk of recurrent disease and all-cause mortality.25,26 The WHO 2008 MDR-TB treatment recommendations were updated in 2011.7,27 The 2011 recommendations endorse treatment for a minimum of 20 months, including an intensive phase with a minimum of 8 months treatment with pyrazinamide. A fluoroquinolone, a 2nd-line injectable antibiotic (capreomycin, kanamycin or amikacin but not streptomycin which is considered a 1st-line drug), a thioamide (either ethionamide or prothionamide) and cycloserine, terizidone (a compound of 2 cycloserine molecules and terephtalaldehyde) or p-aminosalicylic acid should be given through the full treatment period. 27 If the MTB strain is not sensitive to enough 2nd-line drugs to provide a minimum of five effective medications, additional drugs may be chosen from the 3rd-line options (clofazimine, linezolid, amoxicillin/clavulanate, clarithromycin or imepenem) to complete the regimen. 28 Thiacetazone is a 3rd-line option but contraindicated in HIV co-infected patients because of the risk of potentially fatal cutaneous hypersensitivity reactions. 29 Table 1 lists the drugs available for treatment of DS- and DR-TB.
Drugs used in the treatment of drug-sensitive and drug-resistant tuberculosis.

Considered a 1st line agent.
The 2011 WHO guidelines suggest that ethambutol may be used but recommend against its inclusion in the standard treatment regimen for MDR-TB.27,30 Data review concluded that ethambutol does not improve outcomes and there are concerns about the reproducibility of DST with ethambutol. 30 When patients are treated without benefit of DST results, treatment should include at least 2 drugs not used previously.
Pending approval of new drugs, strategies to combat DR-TB, including MDR-TB and XDR-TB, consist of higher dosing of isoniazid and rifampin, substitution of rifabutin for rifampin for the minority with rifabutin-sensitive MDR-TB, the use of antibiotics generally used to treat other bacterial infections, eg, clarithromycin, amoxicillin/clavulanate, and linezolid, and other classes of medications that inhibit MTB such as phenothiazines. 28
Thioridazine inhibits DR-MTB, including XDR-TB, independently of antibiotic activity. It suppresses nicotinamide adenine dinucleotide dehydrogenase (NADH) activity, an important respiratory chain enzyme, interferes with calcium-calmodulin binding disrupting bacterial resistance mechanisms, and inhibits bacterial drug efflux mechanisms.28,31–33 Several case series indicate that thioridazine is effective in patients with XDR-TB but neuropsychiatric side effects, although less severe than with other phenothiazines, will limit its use.31–33
Patients with MDR-TB are more likely to fail therapy, develop more complex drug resistance, including XDR-TB, and are more likely to die from TB. 12 Regimens to treat DR-TB have their limitations, either an only modest effect on MTB or side effects.34,35 Second-line drugs are associated with more frequent and more severe side effects further complicating treatment. 36 There is an urgent need for effective drugs with acceptable side effect profiles to effectively treat MDR-TB and XDR-TB, shorten treatment for both DS-TB and DR-TB, and rapidly sterilize latent tuberculosis infection (LTBI) in contacts of MDR- and XDR-TB patients.36–45
Latent tuberculosis infection
Cases of active TB only represent the tip of the iceberg. Although there are 8 to 9 million new cases of TB annually, TB skin test (TST) data suggest that one-third of the global population, over two billion people, are infected. The immune system stabilizes MTB infection within the macrophages of healthy patients, a condition called LTBI. 45 Individuals with LTBI represent a large reservoir of people with the potential to develop active disease.
The lifetime risk of LTBI progressing to active disease is approximately 10%; higher in patients immunosuppressed by HIV infection or receiving iatrogenic immunosuppression for a constellation of autoimmune diseases or organ transplantation.34,46 Other conditions that increase the risk of developing active disease include diabetes, previous gastrectomy, renal failure, being underweight, or silicosis. The risk of active disease is also greater in patients with brisker reactions to TST, children less than 5 years, recent TST converters, and patients with chest x-ray evidence of healed tuberculosis. 46 In LTBI, the hypoxic intracellular environment inhibits MTB metabolism making treatment difficult. 47 Drugs that effectively target intracellular organisms would reduce the risk of future disease.
Identification and treatment of LTBI are important TB prevention strategies in wealthy low prevalence countries. Unfortunately, the resources to identify and treat LTBI are unavailable in most high prevalence countries. Preventive treatment with isoniazid is effective for LTBI due to DS-MTB. Efficacy estimates range from 60% to 90%. 48 LTBI treatment is also effective in patients at higher risk of developing active disease.49–51 Rifampin or combination isoniazid and rifampin are both more effective and allow treatment to be shortened to four and three months, respectively.52–54 Attempts to further reduce LTBI treatment to 2 months with rifampin and pyrazinamide were limited by hepatotoxicity; fatal in some cases. 55
Hopes that MDR-TB and XDR-TB are less contagious are unfounded. Approximately 5% of household contacts of MDR-TB or XDR-TB cases developed TB and most were MDR-TB.56,57 There is no consensus on the best management for MDR-TB contacts. Some suggest treating contacts whereas others suggest withholding therapy, ongoing observation, and only treating if they become ill. Unfortunately, the literature is limited and recommendations are based on a few case series treated with various regimens. 58 A South African study reported that treating children less than 5 years exposed to pulmonary MDR-TB with individualized regimens based on the DST results of the suspected index case was beneficial. 59 From 1995 to 2010, 47 cases of MDR-TB, including 22 with smear positive pulmonary disease, were identified in Victoria, Australia. 60 Forty-nine of 570 contacts were suspected to have been infected by the index cases. None treated for LTBI developed active disease, whereas 2 of the untreated contacts did. 60 Other studies have not shown benefits to treating patients exposed to MDR-TB.61,62 Medical surveillance rather than treatment is recommended for XDR-TB contacts. 58 New TB antibiotics without cross-resistance to existing drugs would provide treatment options for MDR-TB and XDR-TB contacts.
Need for new treatment options
After a hiatus of nearly 50 years, new classes of tuberculosis antibiotics are under development.36–45,63 The increasing prevalence of MDR-TB and XDR-TB, and the linkage between the TB and HIV epidemics highlight the need for new, more effective drugs. Drugs that act synergistically to rapidly eradicate MTB infection and shorten treatment regimens, do not interfere with the pharmacokinetics of other medications especially antiretroviral therapy (ART), are active against dormant organisms, and will eradicate LTBI would be valuable.36–40 Representative molecules in the new drug classes undergoing commercial development are listed in Table 2. They include two oxazolidinones (AZD5847 and sutezolid). The new drug classes and their lead molecules are: nitroimidazopyrans (PA-824 and delamanid), ethylenediamines (SQ-109), and benzothiazinones (BTZ043). On December 31, 2012, bedaquiline, a diarylquinoline, became the first potentially 1st-line drug approved by the FDA since rifampin in 1966.64,65
New molecules in new drug classes in clinical development.
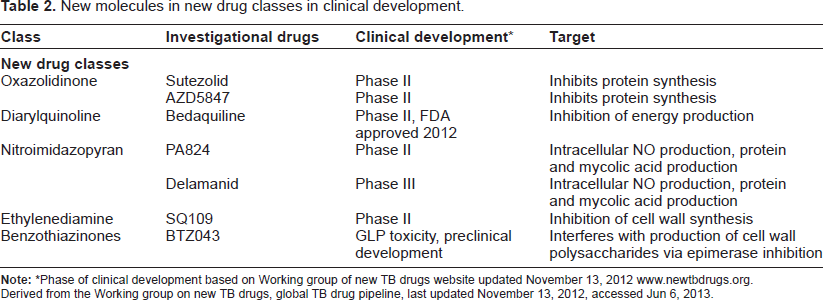
Phase of clinical development based on Working group of new TB drugs website updated November 13, 2012 www.newtbdrugs.org.
Derived from the Working group on new TB drugs, global TB drug pipeline, last updated November 13, 2012, accessed Jun 6, 2013.
The results of a promising phase IIB clinical trial with delamanid were recently published and an international, multi-center phase III trial was started in September, 2011. Final data collection for the primary outcome measure, the proportion of patients achieving sputum culture conversion at 2 months, will be completed in September, 2013, and the estimated study completion date is September, 2015. 66
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Nitroimidazoles, also called bicyclic nitroimidazopyrans, are chemically related to metronidazole. Two drugs, PA-824, a nitroimidazo-oxazine, and delamanid, previously named OPC-67683, a nitrodihydro-imidazo-oxazole, are undergoing commercial development.37,39,67,68 PA-824 was discovered first and stimulated interest because of its good anti-mycobacterial activity and unlike earlier nitroimidazopyrans, was not mutagenic. 69 Japanese researchers investigating the properties of the nitro-dihydroimidazooxazoles, recognized that delamanid had superior activity against MTB than other closely related compounds. 70
PA-824 and delamanid have similar antimycobacterial actions; inhibiting mycolic acid biosynthesis.
71
Mycolic acids are long chain fatty acids that bestow hydrophobicity to the mycobacterial cell wall impeding drug penetration into mycobacteria.72,73 Disrupting cell wall metabolism and facilitating better drug penetration may allow for shorter effective drug regimens.
21
Shorter regimens should improve treatment completion and success rates.
74
Both PA-824 and delamanid are active against DS- and DR-MTB strains.70,75 They also demonstrate activity against
Its activity requires the mycobacterial deazaflavin F420-dependent glucose-6-phosphate dehydrogenase (G6PD), Fgd 1, and resistance to delamanid is conveyed by mutations of either F420 or Fgd 1.75–80 Delanamid is a prodrug that must be reduced by the deazaflavin-dependent nitroreductase to its des-nitro metabolite to be active.39,76 Mutations of Rv3547, the gene coding for the deazaflavin-dependent nitroreductase, also convey mycobacterial resistance to delamanid.39,76
Both PA-824 and Delamanid promote intracellular generation of microbiocidal nitrogen oxidative intermediaries including nitric oxide (NO).69,75 Since dormant MTB do not undergo mycolic acid biosynthesis, the activity of PA-824 and delamanid are better explained by intracellular NO generation, toxic even to dormant MTB.77,78
Delamanid has minimal effects on CYP in concentrations up to 100 μM since it is not metabolized by the cytochrome P450 enzymes (CYP).39,76 Therefore, interactions with other drugs should not be a problem.39,76 In patients co-infected with HIV, the lack of interaction with ART is a major advantage.76,81 Delamanid can be administered with rifampin without either drug affecting the metabolism of the other. 67 This is an important advantage over bedaquiline which is metabolized by CYP. 82
Caution is required in patients receiving other medications that also cause QT prolongation or hypokalemia. 84
Systemic exposure to delamanid increased in a greater than dose-proportional fashion between 5 and 400 mg in healthy volunteers and high fat meals increased absorption. 36 Diacon and coworkers studied the pharmacokinetics of delamanid in 42 patients with smear positive pulmonary TB without other serious co-morbid medical conditions. 83 HIV-positive patients not receiving ART could participate in the study if they had CD4+ lymphocyte cell counts greater than 350X10 6 /liter. 83 Patients were randomized to treatment with delamanid 100 mg, 200 mg, 300 mg or 400 mg daily for 14 days. To serve as positive controls, six patients received standard therapy with isoniazid, rifampin, ethambutol, and pyrazinamide. 83
Plasma concentrations of delamanid were determined by mass spectroscopy. The pharmacokinetic data after treatment with 100 mg, 200 mg, 300 mg and 400 mg daily for 14 days from the study by Diacon and coworkers is shown in Table 3. Delamanid exposure did not increase proportionately with the daily dose and exposure overlapped between patient groups being greatest in those receiving 300 mg daily. The small number treated with each dose and their anthropomorphic differences may account for the variability in drug concentrations. 83 Delamanid exposure increased with food administration. 81
Mean plasma pharmacokinetic parameters of delamanid on day 14.
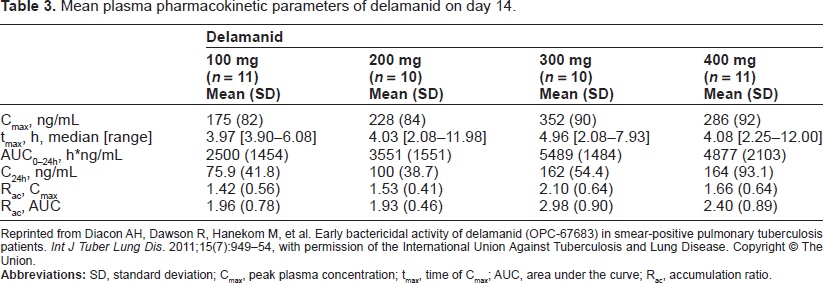
Reprinted from Diacon AH, Dawson R, Hanekom M, et al. Early bactericidal activity of delamanid (OPC-67683) in smear-positive pulmonary tuberculosis patients.
In another study, patients received 100 mg or 200 mg twice daily with meals, 10 hours apart. 84 The following represent the mean and coefficient of variation, as a percentage in parentheses. After 8 weeks, the mean Cmax was 414 (39.9) ng/mL and 611 (35.6) ng/ mL after the morning dose and 400 (40.5) ng/mL and 588 (36.2) ng/mL after the evening dose in the 100 mg BID and 200 mg BID-treated groups, respectively. 84 Mean trough values (Cmin) were 304 (42.2) ng/mL and 460 (36.6) ng/mL with 100 mg BID and 200 mg BID, respectively. 81 The mean AUC0–24h values were 7,925 (37.5) ng/mL-hr and 11,837 (33.6) ng/mL-hr with 100 mg BID and 200 mg BID, respectively. 84
The Cmax values for delamanid in the Gler study were approximately twice the values determined by Diacon and colleagues. Protocol design and anthro-pometric differences likely contributed to the differences in the pharmacokinetic data between the studies. In the study by Diacon and colleagues, medication was given once daily while fasting and all patients were South African. 83 Patients took the medication twice daily with food and the majority of patients were Asian, the remainder American or European, in the other study. 84 Differences in duration of the two studies, two and eight weeks, respectively, may also explain some of the differences in pharmacokinetics.83,84
Preclinical Data
Delamanid inhibits mycobacterial G6PD, primarily in the MTB complex species. It is inactive against other bacteria and eukaryotes, and does not inhibit G6PD in man. 80 In one study, delamanid showed dose-dependent killing of DS-TB superior to isoniazid and similar to rifampin. 85 Susceptibility testing against both DS- and DR-MTB strains demonstrated MICs ranging from 0.006 to 0.012 mcg/mL. 76 All strains tested were more susceptible to delamanid than to rifampin, isoniazid, ethambutol, or streptomycin. 76 Delamanid was more active, with lower MICs against both DS-MTB and DR-MTB strains than PA-824. 76 Moreover, there is no antagonism with rifampin, isoniazid, ethambutol or streptomycin. 80
Delamanid is highly active against intracellular MTB in human macrophages. 76 Delamanid is as effective as rifampin at one-third the concentration and more effective than either isoniazid or PA-824. 76 Its potent effect against dormant intracellular MTB may be important for the treatment of LTBI and allow shorter regimens for treatment of active disease.
Delamanid was effective in a murine model of pulmonary TB. 76 Bacterial counts in the lungs of mice infected with MTB declined in a dose-dependent fashion. The concentration required to reduce colony forming units (CFU) by 95% was considerably less than the required concentrations of any first line drug. 76 It was also effective in immunodeficient BALB/c nude mice. 75 Administration of delamanid with standard therapy resulted in only a single MTB colony being cultured in one of 6 mice at 3 months and none at 4 months, compared to persistence of MTB colonies in 4 of 5 mice after 6 months of standard therapy. 76
Clinical Studies
The early bacterial activity (EBA) of delamanid, 400 mg daily, was modest for the first 4 days but subsequently the number of CFU in cultured sputum decreased progressively to day 14. 36 In another pulmonary TB study in man, the number of MTB colonies declined steadily with all doses of delamanid over 14 days. Although the differences were not statistically significant, there was a trend to a greater effect with increasing daily doses between 100 mg and 300 mg. 83
An international, randomized, double-blind trial of the effects of delamanid in patients with pulmonary MDR-TB was recently published. 84 Subjects were stratified, based on the presence or absence of cavities on chest x-ray. Patients whose sputum cultured MDR-TB were treated with an optimal background regimen (OBR) according to the 2008 WHO guidelines and one of delamanid 100 mg, delamanid 200 mg, or placebo, given twice daily for 2 months. The primary outcome, sputum culture status at 2 months, correlates with the risk of relapse. 85 Both doses of delamanid chosen for this study were adequate to provide plasma levels (AUC0–24h) that exceeded the threshold for maximal bactericidal activity. 84 There were a small number of HIV-positive study participants with CD4+ lymphocyte counts of 350 or greater and who were not receiving ART. 84 In addition to the expected exclusion criteria, treatment with moxifloxacin was forbidden and patients were excluded if they were being treated for arrhythmias, had QT prolongation or other ECG conduction abnormalities. The study sites were in high, medium, and low prevalence countries, and patients were hospitalized for the 8 week study period. Patients were monitored for a further 4 weeks to confirm their sputum culture status while they continued the WHO-approved OBR. 7
The OBR included any 1st-line drugs that the patients’ sputum culture isolates were susceptible to, an injectable antibiotic, a fluoroquinolone, and supplemental 2nd-line drugs to provide an effective 4 or 5 drug regimen. 7 All patients received the WHO-recommended intensive phase regimen for 6 to 8 months, including a minimum of 4 months treatment after sputum culture conversion. 7 The continuation phase lasted 12 to 18 months, including a minimum of 16 weeks treatment after sputum culture conversion. 7 Although treatment regimens were personalized, they included treatment with 4 to 6 drugs for 18 to 24 months. Treatment with delamanid or placebo, and the follow up period were completed within the WHO-recommended intensive treatment phase. 83 The trial drugs were taken in a DOT fashion 10 hours apart and with food to increase systemic exposure.
To determine the response to treatment, sputum was obtained for culture the day before treatment started, the day treatment began, and then weekly through the remainder of the treatment period and the 4 week follow-up period. 84 Samples were handled in the standard fashion and cultured both on solid and in liquid media. Testing was done locally according to the Clinical and Laboratory Standards Institute guidelines for sputum processing, microscopy, culture, mycobacterial identification, and DST. To qualify as culture negative, sputum was culture negative on day 57 and all subsequent cultures performed weekly through day 84 had to remain negative. 84 As expected, liquid culture was more sensitive than solid media culture. 84 Just under 30% of the patients treated with OBR and placebo achieved sputum-culture conversion by day 57 as determined by culture in liquid media, compared to 45.4% and 41.9% treated with delamanid 100 mg and 200 mg BID, respectively. The results with both doses of delamanid were statistically significant compared to placebo but were similar to each other. 84
After completion of the blinded trial, patients could participate in an open label trial. Those who entered the open label trial received a further 6 months of delamanid, either 100 or 200 mg, twice daily. 86 Subsequently, the outcomes of all patients that participated in either the blinded trial or in both trials, were reported. 86 For each patient, data collection ended 24 months after entry into the randomized trial or when therapy was completed, whichever happened first. 86 The open label trial included 213 of the 481 patients from the blinded trial.
Treatment with delamanid was interrupted for a minimum of 4 weeks in patients entering the open label trial to accommodate the 4 week follow-up period of the blinded trial. In the open label trial, delamanid was started at a dose of 100 mg BID, and after 2 weeks, the local investigator had the option of increasing to 200 mg BID. Medication was administered by DOT. Patients were hospitalized for the first 2 weeks of treatment and for a further 2 weeks if the dose was increased to 200 mg BID. The remaining treatment was administered in the outpatient setting unless the patient's medical condition warranted hospitalization.
Data from 421 of the 481 patients entered into the blinded study and consented to participate in the observational study were included in the analysis. Only solid media culture data were reported in the longer observational study. The WHO-defined treatment outcomes were used for analysis.
7
In the observational study, 66% were male with a median age of 34 years, range 18 to 63 years. Only 4 patients were HIV-positive and 56 (13.3%) had confirmed XDR-TB.
86
Three-quarters of the patients treated with delamanid for a minimum of 6 months had favorable outcomes, ie, cured or completed treatment.
86
One quarter died, failed treatment, or defaulted.
86
Among patients treated with delamanid for 2 months or less, only 55% experienced favorable outcomes (
Caution is required when comparing results between the studies since the primary outcome in the blinded study was the liquid media culture results and the primary outcome in the open label study was the culture results on solid media.84,86 Thirty-eight Chinese patients enrolled in the Gler study were also reported separately. 87 There was a trend to a better sputum conversion rate with delamanid but the cohort was too small to draw any other conclusions except that results were similar to those of the rest of the study population.
Ongoing trial
A phase III international trial comparing the addition of delamanid or placebo to OBR in MDR-TB patients is underway in 8 countries, including the 3 Baltic republics, Moldova, India, Peru, the Philippines, and South Africa. 66 Delamanid 100 mg BID for 2 months followed by 200 mg BID for 4 months or placebo will be added to OBR as recommended in the 2011 WHO treatment guidelines. The recruitment goal is 390 MDR-TB patients stratified into 2 groups; including a group of HIV-positive patients on ART. Patients who meet the WHO criteria for XDR-TB will be excluded.
Trials with and comparing delamanid to other new drugs
There are no published trials comparing delamanid to other new drugs but its MICs (0.006–0.024 μg/mL) with both DS-TB and DR-TB, are lower than bedaquiline (0.030–0.12 μg/mL), SQ-109 (0.16–0.63 μg/mL), or PA-824 (0.015–0.25) μg/mL. 36 An eight week, placebo-controlled phase II study in MDR-TB found that the addition of bedaquiline to OBR reduced the time to sputum culture conversion and at eight weeks, more of the bedaquiline-treated subjects had converted-to-negative.88,89 The sputum culture conversion-to-negative rate in liquid media was 48%, similar to the eight-week liquid media data when delamanid was added to OBR.84,88
A recently published trial compared the 14 day EBA of PA-824 in various combinations with other drugs. 90 The 14 day EBA of the combination of PA-824, moxifloxacin, and pyrazinamide, was significantly greater than that of bedaquiline, combination bedaquiline and pyrazinamide, or the combination of bedaquiline and PA-824. Although there was a trend towards a higher 14 day EBA, the difference was not significantly greater than the results with the combination of PA-824 and pyrazinamide. 90 Similar studies with delamanid would be interesting since it has greater EBA than PA-824. 76 If delamanid has similar or greater potency than PA-824 in combination with other drugs, it may improve outcomes and shorten treatment regimens for MDR-TB and XDR-TB.
Safety
Some bicyclic nitroimidazoles increase tissue radio-sensitivity and are mutagenic.91,92 Development of some molecules in this class was stopped because of their potential mutagenicity. 76 An earlier nitroimidazo-oxazole, CGI-17341, demonstrated good activity against MTB but was mutagenic. 76 Delamanid, although structurally similar, is not mutagenic, as demonstrated by the bacterial reverse mutation test, but retains potent activity against MTB. 76
In early EBA studies, doses of delamanid up to 400 mg were well tolerated. 36 Delamanid had no effect on CYP liver enzymes at concentrations up to 100 μg/mL. 76 In the study by Gler et al, the rates of serious adverse events were similar in the delamanid-treated patients and the placebo group. 84 Despite excluding patients taking antiarrhythmic medications, or with QT prolongation or other conduction abnormalities, QT prolongation developed in 10% and 13% treated with delamanid 100 mg and 200 mg BID, respectively, compared to 4% in the placebo group. 84 None of the patients with QT prolongation experienced clinical symptoms such as syncope or arrhythmias. The rate of serious adverse events was similar in the delamanid 100 mg BID and placebo groups, 9.9% and 8.8%, respectively. The rate was slightly higher in the group that received delamanid 200 mg BID, 12.5%. QT prolongation was the only severe adverse event that was more common in the delamanid-treated patients. Discontinuations due to adverse events were similar across all three treatment groups: 2.5% in the delamanid 100 mg BID arm, 3.8% in the delamanid 200 mg BID arm, and 2.5% in the placebo arm. 84 Adverse events are common in MDR-TB patients since the majority of OBR drugs have significant side effect profiles. In the Gler study, adverse events were reported by 94.4%, 91.3% and 94.4% of patients in the placebo, 100 mg BID and 200 mg BID treatment arms, respectively. 84 Rates of nausea and vomiting were similar in the placebo and Delamanid 100 mg BID groups but higher in the 200 mg BID group. 84 Other adverse event rates were similar in the delamanid and placebo groups. 84
Potential Roles in Therapy
Similar to other TB drugs, there are delamanid-resistant MTB strains emphasizing the need to manage this drug appropriately in supervised settings and in combination with other drugs. 78 Most importantly, the addition of delamanid to MDR-TB treatment regimens improved outcomes.84,86 Hopefully, future trials combining delamanid with other new drugs such as bedaquiline and moxifloxacin will improve on outcomes seen in the trials with PA-824. 90
The introduction of delamanid offers potential advantages in the treatment of both DS- and DR-TB. If the faster and more complete eradication of MTB seen in the murine pulmonary TB model is duplicated in man, shorter treatment regimens may be possible. Although no studies have been published yet, delamanid should be an effective medication for extrapulmonary TB. It should not be used for TB meningitis until it is known whether it adequately penetrates the blood-brain barrier. 90 Its activity against intracellular MTB in human macrophages may provide an option for effectively treating contacts of MDR-TB and XDR-TB cases and to reduce the burdens of these difficult, costly, and life-threatening conditions.
Conclusions
Several new drugs with activity against MDR-TB are undergoing commercial development. Bedaquiline was the first new 1st drug approved by the FDA for the treatment of TB since 1966. Unfortunately, a recent report has raised concerns about its use. 94 Although the convert-to-negative rate at 6 months was 79% with bedaquiline versus 58% in the patients who received placebo, 10 of 79 bedaquiline-treated patients died compared to 2 of 81 placebo-treated patients. 94 Five of the 10 deaths among the bedaquiline-treated patients and the 2 deaths in the placebo arm were due to TB. 94 The concerns recently raised about bedaquiline emphasize the need to continue developing other drugs for the treatment of MDR-TB. Hopefully, delamanid will prove to be effective and safe and will be available in the next few years.
Delamanid is currently in phase III trials. 66 It has excellent activity against both actively replicating and dormant MTB including those resistant to the currently available 1st-line drugs. Moreover, it is more active against MTB, both DS and DR strains, than the currently available 1st-line drugs. An important advantage over other medications is that it is not metabolized by CYP enzymes and can be given in combination with ART, rifampin, or with other drugs metabolized by CYP enzymes, including bedaquiline.64,65 QT prolongation is the most concerning adverse effect and could potentially limit its use in patients with co-morbid heart disease. Two recently published trials showed that its addition to WHO-recommended OBR improved outcomes in patients with MDR-TB. The results of a placebo-controlled, phase III trial of treatment with delamanid, 100 mg BID for 2 months followed by 200 mg BID for 4 months, added to OBR in MDR-TB patients will be available in 2015 and will hopefully provide further evidence for its use in the treatment of MDR-TB. 66
Author Contributions
Analyzed the data: SKF. Wrote the first draft of the manuscript: SKF. Contributed to the writing of the manuscript: SKF. Agrees with manuscript results and conclusions: SKF. Jointly developed the structure and arguments for the paper: SKF. Made critical revisions and approved final version: SKF. The author reviewed and approved of the final manuscript.
Funding
SKF has no financial competing interests with the contents of the manuscript.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the author has provided signed confirmation of compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
