Abstract
Myelodysplastic syndromes (MDS) are marked by progressive cytopenias and risk of transformation to acute myeloid leukemia. Supportive care with transfusions, antibiotics, and hematopoietic growth factors has long been the mainstay of therapy for MDS, given that most patients are not eligible for more intensive chemotherapy. The hypomethylating agent 5-azacitidine (AZA) was the first chemotherapeutic agent approved by the U.S. Food and Drug Administration for the treatment of MDS, and it represented a real advance in the management of the disease. In Phase III trials, azacitidine demonstrated a higher response rate and a longer overall survival compared to supportive care alone. Importantly, it is a well-tolerated drug that can be given IV or SC in various outpatient schedules. Future studies are expected to evaluate the activity of AZA in combination with other epigenetic modifying agents and to establish the relative efficacy of azacitidine and decitabine. This review summarizes the current treatment landscape in MDS and specifically addresses the role of azacitidine in the management of MDS.
Introduction
Myelodysplastic syndromes (MDS) are a spectrum of diseases characterized by ineffective hematopoiesis, cytopenias and risk of progression to acute myeloid leukemia (AML). Many different strategies and agents have been used for the treatment of MDS, but until recently, no therapy had been proven to alter the natural history of the disease. Currently, allogeneic stem cell transplant is the only therapy with the possibility of a cure, but because patients with MDS are generally elderly with other comorbid illnesses, very few are eligible for a transplant. Among those patients that do pursue transplant, the treatment related mortality remains high. Therefore, most patients will benefit from a well tolerated treatment that improves cytopenias and delays progression to AML. Only a handful of drugs have been approved by the FDA for the treatment of MDS, and, of those, 5-azacitadine (Vidaza™) has been shown to increase survival when compared to supportive care alone. We will review here the role of 5-azacitadine (AZA) in the treatment of MDS.
The prevalence of MDS has likely been underestimated in the past. Most publications quote the number of new cases in the US to be around 10,000 per year, but a recent review of Medicare reimbursements suggested that there were 4-7 times that number of cases of MDS diagnosed in the United States in 2003. 1 Although this review was not precise in its diagnostic criteria for MDS, undoubtedly the impact of the disease on society has been underestimated. MDS has been difficult to categorize because of the relative lack of distinct findings on bone marrow biopsy, the varied number of cell lines affected, and the variable clinical course. The most commonly referenced classification schemes are the French-American-British (FAB) classification and the more recent WHO classification, 2 both of which incorporate dysplastic changes and percentage of blasts in the bone marrow (Table 1). These stratifications, however, have not been reliable in terms of prognosis. The prognosis of MDS can be estimated using the International Prognostic Scoring System (IPSS, Table 2), 3 which takes into account variables not included in the above classification schemes, including number of cytopenias and cytogenetic abnormalities.
WHO classification of MDS.
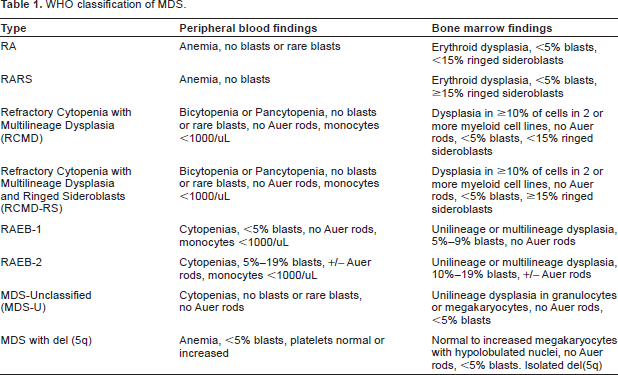
International prognosis scoring system in MDS.
The pathophysiology of MDS is poorly understood; however, it is thought to be caused by inappropriate bone marrow apoptosis. 4 This leads to the typical and somewhat conflicting findings of a hypercellular bone marrow combined with peripheral blood cytopenias. Patients with MDS can present with a host of different symptoms, and many present asymptomatically with the finding of one or more cytopenias on routine blood work. In the later stages of MDS, patients can develop life threatening infections as a result of neutropenia, bleeding complications secondary to thrombocytopenia, and severe fatigue with concomitant anemia.
Treatment Modalities
In the past, clinicians managed MDS patients with supportive care only as treatment options were lacking. Red blood cell (RBC) transfusions can provide symptomatic relief for patients with fatigue due to their anemia but cause secondary complications from iron overload. Some patients respond to erythropoietin with short lived improvements in anemia, but erythropoietin can cause thrombotic complications. In low risk patients, erythropoietin used within established guidelines can provide durable responses and may even result in a small increase in survival.5,6 G-CSF and GM-CSF have been used in neutropenic populations; however patients with MDS have a predictable poor response to these agents given their dysfunctional bone marrow environment. Antibiotics are often necessary to fend off bacterial infections, and along with RBC transfusions, are the mainstay of supportive care for many patients.
Some of the available therapies for MDS are active only in specific subsets of MDS patients. The immunosuppressive agents cyclosporine and antithymocyte globulin (ATG) have shown response rates approaching 50% in small studies, but responses are generally limited to patients with hypoplastic MDS or HLA-DR15 positivity. 7 Lenalidomide, a thalidomide derivative, has been approved for use in the small subset of MDS patients who have 5q- cytogenetics. 8 Lenalidomide has the most activity in those with 5q- who do not have neutropenia or severe thrombocytopenia and have low or intermediate risk MDS. This agent does cause a significant degree of drug-related cytopenias and should therefore be used cautiously in patients without the above mentioned characteristics. The only therapy that has shown promise for curing the disease is allogeneic stem cell transplant. Unfortunately only an estimated 5% of patients with MDS are eligible for allogeneic stem cell transplant at some point during their disease due to donor availability and the significant mortality and morbidity of the intervention.
Epigenetic Modifying Agents: 5-Azacitidine
Agents that affect DNA methylation can change gene transcription activity without altering the DNA itself. In the case of patients with MDS, it is thought that DNA hypermethylation leads to transcriptional silencing of genes involved in cellular proliferation and differentiation, which may account for a hypercellular bone marrow filled with cells that do not have normal functional ability. DNA methyltransferase inhibitors have been shown to restore the normal demethylated state of several types of tumor suppressor genes such as p16, E-cadherin, hMLH1, VHL, and p15, both in vitro and in vivo.9–12 The recognition of abnormal gene methylation patterns in leukemia and MDS led to the initial trials using DNA hypomethylating agents in MDS.13–16
AZA and decitabine (5-aza-2‘-deoxycytidine) are the two DNA methyltransferase inhibitors currently approved for use in patients with MDS. AZA was initially approved in 2002 after a phase III study demonstrated efficacy in all classes of MDS. 17 AZA is a cytosine nucleoside analog with an altered heterocyclic ring, which is thought to give the compound its hypomethylating ability in gene regulatory regions. 18 It is cytotoxic to cells in S phase and exerts its action on rapidly dividing cells. The drug is phosphorylated into a triphosphate form after its entry into the cell. It is then integrated into RNA and is incorporated into the DNA in place of cytosine. 19 DNA methyltransferases become trapped at the sites of the cytosine analog, and DNA methylation is thereafter reduced. Tumor suppressor genes are again able to function normally and the disease is halted. In addition to its DNA hypomethylating effects, the drug likely has other effects in the cell, such as histone deacetylation and immune regulation, which may contribute to its efficacy. AZA has a half-life of approximately 41 minutes after sub-cutaneous administration and 20 minutes with intravenous use. It is primarily excreted in the urine.
Phase II Studies
Initial phase II studies performed by the Cancer and Leukemia Group B (CALGB) demonstrated efficacy of AZA in MDS.20, 22 In the CALGB 8421 trial, patients received a continuous intravenous infusion of AZA 75 mg/m2 daily for 7 days, repeated every 4 weeks. Twelve percent of patients had a CR (defined as normalization of bone marrow and blood counts) and 25% had a PR (>50% restoration of the deficit from normal for all three peripheral blood lines, elimination of transfusion requirements, and a decrease in percentage of bone marrow blasts by greater than or equal to 50%). Overall, 21 of the 43 patients (49%) had a measurable response to the treatment. Sixty-three percent of patients reported mild to moderate nausea and/or vomiting, and 33% of patients required dose reduction secondary to myelosuppression. The second phase II CALGB study demonstrated similar response rates with subcutaneous administration of AZA (75 mg/m2/day for 7 days every 4 weeks). Twelve of the 70 patients had a CR (17%), and 16 had hematological improvement. Toxicity was similar to that reported with IV administration, with the addition of occasional skin reactions at the injection site. In both studies, the median time to response was 3 cycles (range 1-17).
Phase III Studies
191 patients with MDS were recruited for a phase III study performed by the CALGB group between 1994 and 1996. 17 MDS was established based on the FAB criteria (<30% bone marrow blasts), and those patients with RA or RARS had to have one of the following: symptomatic anemia requiring RBC transfusions for at least 3 months before study entry, thrombocytopenia ≤50,000 or bleeding requiring platelet transfusions, or neutropenia with an ANC < 1 × 109/L and an infection requiring intravenous antibiotics. Therapy-related MDS patients were included if they were cancer-free for 3 years and had not had radiation or chemotherapy for 6 months.
The patients were stratified by FAB classification and randomly assigned to receive AZA or supportive care. AZA was given as a subcutaneous injection (75 mg/m2 per day) in 7 day cycles on days 1, 29, 57, and 85. After 4 cycles of AZA (113 days), the two groups were evaluated by bone marrow biopsy. Patients who had a CR at 4 months continued on therapy for 3 more cycles, those who had a PR or showed improvement in peripheral cell lines were continued on the drug until a CR was achieved or until relapse occurred. 60% of those in the AZA arm had a measurable response manifest by a CR, PR, or either improvement in peripheral cell lines or ≥50% decrease in transfusion requirement from baseline. Only 5% of the supportive care arm achieved one of these goals. 7% of those in the AZA arm achieved a CR and 16% achieved a PR in contrast to the supportive care arm in which there were no patients who achieved either a CR or PR. The median time to initial response was 64 days (cycle 3), and the median time to best response was 93 days (cycle 4). In addition, the median duration of response for those who achieved CR, PR, or HI was 15 months (Table 3).
United States phase II and III studies of AZA in MDS.

Potentially skewing the results, there was a large amount of crossover between the two groups. In fact a total of 49 patients eventually crossed from the supportive care group to the AZA group, and these patients were evaluated in the AZA group. Median overall survival was 20 months for those that were given AZA as compared to 14 months for patients who received supportive care. In the overall survival (OS) calculation, patients who did cross from the supportive care arm into the AZA arm were included in the supportive care data, and the difference in OS was not significant (p = 0.10). The investigators further analyzed the results to compare three groups of patients: supportive care patients who never crossed into the AZA group or those that crossed over after 6 months, supportive care patients who crossed over in the first 6 months, or patients initially randomized to receive AZA. In this calculation, the effect on mortality was significant when comparing the first group listed above with the third group (11 months vs. 18 months, p = 0.03).
The median time to progression to AML or death in the AZA group was 19 months, significantly longer than the 8 months seen in the supportive care arm (p = 0.04). The most common toxicity was myelosuppression, which was seen in >50% of patients. Myelosuppression was often transient, and the authors reported that patients usually recovered in time to receive their next treatment cycle. Nausea, vomiting, and infection were also reported side effects.
At the beginning of the study, there were 65 patients receiving RBC transfusions in the AZA arm, 29 of those (45%) were not receiving transfusions at the conclusion of the study. Quality of life was assessed over the course of the study by telephone interviews and patients randomized to receive AZA reported significantly decreased fatigue and dyspnea and improved physical functioning and positive affect. 23 Of course, this was not a blinded study and patients knew whether or not they were receiving treatment, so it is possible that some of the effect seen on QOL was attributable to a placebo effect.
The diagnostic criteria for MDS and AML have changed over the years as more has been discovered about the diseases and their prognoses. Therefore, the definition of CR and PR has fluctuated over the last 20 years. In an effort to appropriately compare past trials with more current trials using AZA, a retrospective analysis was done in 2006 on the two CALGB phase II trials reported above and the CALGB phase III trial. 22 Using the current WHO classification, a total of 103 of the patients enrolled actually would have been classified as having AML. The results of the patients with various classifications of MDS treated with AZA validated the previously reported data listed above. The updated response data is shown in Table 3.
A second phase III study was a larger, multi-center European trial conducted from 2004 to 2006. 24 It compared AZA to several conventional care regimens in high risk MDS (IPSS intermediate-2 or high risk). This was an intriguing study because it compared the three most common options available to clinicians when evaluating patients with MDS. It enrolled 358 patients and randomized 179 patients to receive AZA and 179 patients to receive conventional care. Patients in the AZA group were treated with 75 mg/m2 sc per day for 7 days every 28 days for at least 6 cycles. The doses were delayed if necessary for myelosuppression in certain instances. The conventional care group was further stratified so that 105 patients were assigned to best supportive care, 49 to low-dose cytarabine, and 25 to intensive chemotherapy. The patients were assigned to the various treatment modalities by investigators based on their ECOG performance status, age, and comorbidities. Supportive care included blood product administration, antibiotics, and G-CSF as needed. Low dose cytarabine was administered 20 mg/m2 per day subcutaneously for 14 days for at least 4 cycles, and intensive chemotherapy consisted of induction with cytarabine 100-200 mg per day by continuous intravenous infusion for 7 days, plus 3 days of either intravenous daunorubicin (45-60 mg/m2 per day), idarubicin (9-12 mg/m2 per day), or mitoxantrone (8-12 mg/m2 per day). Patients who achieved CR or PR after induction received one or two consolidation courses with reduced doses of the cytoxic drugs used for induction followed by best supportive care. After the assignment to one of the treatment group modalities, patients were then randomized to receive either AZA or the conventional care regimen.
The overall survival favored patients who received AZA as compared with the combined group of patients who received conventional care regimens. The median survival was 24.5 months vs. 15 months (p < 0.001), and the hazard ratio was 0.58. The subgroup analysis showed that the AZA group lived an average of 9.6 months longer than their counterparts in the best supportive care group and 9.2 months longer than the group that received low dose cytarabine (p = 0.0045 and p = 0.0006 respectively). Although there was a trend towards increased survival in the AZA group when compared with the group who received intensive chemotherapy, the number of patients in each group was small and the difference did not reach statistical significance. The results of the study, stratified by treatment assignment, are summarized in Table 4.
Results of phase III European study.
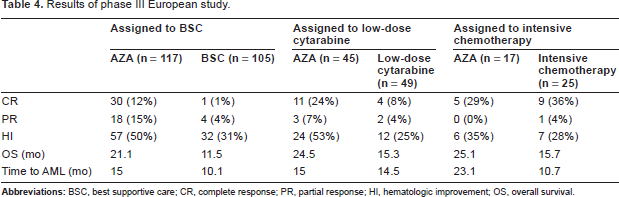
Patients receiving AZA had more complete and partial remissions based on the International Working Group (IWG) response criteria. 25 There were 51 remissions in the AZA group (31 CR and 20 PR) as opposed to 21 total remissions in the conventional care group (14 CR and 7 PR), p = 0.001. The difference was primarily seen when compared to the best supportive care group and the low dose cytarabine group. Median time to transformation to AML was 17.8 months in the AZA group compared to 11.5 months (p < 0.001) in the conventional care group, and 45% of patients initially dependent on RBC transfusions at baseline in the AZA group became transfusion independent, while only 11.4% transfusion dependent patients in the conventional care group achieved this goal.
The most severe adverse reactions were related to peripheral cytopenias, a common problem experienced by those in the conventional care group as well. The patients in the AZA group also experienced nausea, vomiting, fatigue, and diarrhea. Risks of infection were similar across the groups but infections necessitating intravenous antimicrobials were lower in the AZA group.
This study revealed several important practical points regarding the treatment of subgroups of patients with MDS. For example, the survival benefit from AZA was seen across all types of cytogenetics, including those with numerical or structural abnormalities of chromosome 7, who historically have a very poor outcome with other treatment modalities. Also the median number of AZA treatment cycles was nine, and patients who were continued on for longer periods generally did better on the drug. Patients in the intensive chemotherapy arm trended to experience more complete responses than those in the AZA group, and although the number did not reach significance, it suggests that patients who are candidates for allogeneic stem cell transplant may be more suitable for intensive chemotherapy if the transplant is anticipated in the near future. Importantly, a survival benefit at one year was observed in AZA-treated patients in all response categories, including those whose best response was hematologic improvements or stable disease, suggesting that CR or PR is not required for a patient to derive benefit from treatment with AZA. 26
Largely based on the results of the two phase III studies, the National Comprehensive Cancer Network recommends in the Practice Guidelines for Oncology for 2009 that AZA should be considered for MDS patients with progressing disease or relatively high-risk disease. 27 If patients are candidates for allogeneic stem cell transplant but are expected to have a delay in the procedure, perhaps because of unavailability of a donor or for time to improve the functional status of the patient, they may benefit from use of the agent. Those patients with IPSS low or INT-1 category disease can be considered for AZA if they do not respond to standard supportive care interventions such as erythropoietin.
The best dosing schedule for AZA has not been firmly established, and, in general practice, physicians choose from a variety of schedules. In the Phase II and III studies discussed above, AZA was given at 75 mg/m2 for 7 consecutive days every 28 days, but the 7 day treatment can be logistically difficult to administer. A community-based, Phase II study randomized patients with MDS, most of whom had low risk disease, to one of three dosing schedules of AZA, each given SC every 4 weeks: 75 mg/m2/d for 5 days, followed by 2 days off, followed by 2 additional days of treatment; 50 mg/m2/d for 5 days, followed by 2 days off, then another 50 mg/m2/d for 5 days; or 75 mg/m2/d for 5 days. 28 The rates of hematologic improvement (44%-56%) and transfusion independence (50%-64%) appeared similar in the three arms, but the study was not designed to detect statistically significant differences between the dosing schedules. A larger study would be required to determine whether efficacy is compromised with a dosing schedule that skips doses over the weekend.
Decitabine
Decitabine is the other DNA methyltransferase inhibitor currently approved for the treatment of MDS, and it is generally regarded to have efficacy similar to AZA. Decitabine is a cytosine analog that is incorporated into DNA and then inhibits DNA methylation in a similar process as AZA. Two Phase II studies of decitabine for the treatment of MDS demonstrated overall response rates of 42% to 54% with CRs in 20% to 28% of patients.29,30 This led to a large, phase III randomized study that compared decitabine to best supportive care. 31 One hundred seventy patients were enrolled, of whom approximately 70% were in the IPSS Intermediate-2 or High risk groups. Patients in the treatment arm received decitabine 15 mg/m2 intravenously every 8 hours over the course of 3 days, and cycles were repeated every 6 weeks. Seventeen percent of those treated with decitabine had either a CR or PR, versus 0% of those on the supportive care arm. The median duration of the response was 10.3 months. Patients in the decitabine arm had a trend to longer median time to AML or death than those in the supportive care arm (12.1 vs. 7.8 months, p = 0.16). This difference reached statistical significance for the subgroup of patients with IPSS intermediate-2 or high risk disease (12.0 vs. 6.8 months, p = 0.03). However, the intent to treat analysis demonstrated no difference in overall survival of the two groups (14.0 months vs. 14.9 months, p = 0.636). Of note, only 46 of the 89 patients randomized to the decitabine arm completed more than 2 courses of treatment, and only 23 patients completed six or more cycles of therapy. Toxicities were generally related to marrow suppression and infection, with 15 patients in the decitabine arm developing pneumonia (18%), compared to 9 in the supportive care arm (11%).
A second Phase III study, sponsored by the EORTC, reported a response rate of 34% and an improvement in progression free survival in MDS patients treated with decitabine versus those treated with supportive care alone. 32 However, there was no difference in overall survival or time to AML between the two arms.
Future Directions
Although AZA is approved for both SC and IV administration, the efficacy of a short IV infusion of AZA has not been established. Preliminary results of a small phase II study that treated MDS patients with a 20 minute IV infusion of AZA on 7 consecutive days of a 4 week cycle demonstrated responses and a tolerable safety profile, but further follow-up is needed. 33 In addition, an oral formulation has been developed and is being tested in preliminary studies. 34 The duration of treatment using AZA in MDS has not been clarified. The analysis of the European phase III AZA study indicated that continued treatment with AZA is indicated when tolerated. 35 The median number of cycles prior to response was 3, and 43% of responders improved their response with continued treatment. These results underline the value of delivering multiple cycles to establish a response and continuing therapy beyond the time of first response, but the best duration of AZA therapy in responding and non-responding patients is unknown.
The combination of epigenetic modifying agents also shows promise. Histone deacetylase inhibitors (HDACi) such as valproic acid and SAHA (suberoylanilide hydroxamic acid) have shown efficacy in phase I and II studies, and regimens combining HDACi's and AZA may prove to be synergistic in MDS. Several small studies have been completed, including one phase I/II study that enrolled 53 patients with MDS and AML and treated patients with a combination of AZA, valproic acid and ATRA.36,37 The overall response rate in this study was 42%, and further studies looking at combination therapies for MDS are ongoing.
The relative benefits of AZA and decitabine have yet to be established, and, in clinical practice, patients are often eligible to receive both agents in sequence. Sequential use of the two agents has not been extensively studied, but in one small study 14 patients were treated with decitabine after failure of azacitidine therapy. 38 Five of the patients achieved response by the IWG criteria, (3 CRs and 1 PR). These results demonstrate that some patients respond to hypomethylating agents given in succession. However, it's worth noting that the median duration of remission in the above study was only 5.3 months and the median survival was 6 months. A Phase III study currently in development is expected to establish the relative efficacy of AZA and decitabine in MDS.
Conclusion
The DNA methyltransferase inhibitor AZA has been shown to increase survival for a number of patients with MDS and generally has been well tolerated. As MDS is often a disease of the elderly, treatments such as AZA may become the mainstay for providing relief from repetitive transfusions, limiting hospitalizations, and providing an increased survival time for many patients. It does not provide a cure for patients with MDS but certainly is a valuable treatment resource for patients who previously had very limited options.
Disclosures
The authors report no conflicts of interest.
