Abstract
Treatment for hepatitis B (HBV), a global disease affecting over 400 million persons, has changed over the past 15 years. Many of the current treatments are oral therapies which suppress HBV viral load and improve liver-related outcomes. However, differences exist in the currently available therapies both in terms of potency and resistance. The newest HBV nucleotide to be approved for the treatment of hepatitis B is tenofovir disoproxil fumarate that when phosphorylated to its active form leads to DNA chain termination. Tenofovir is active against human immunodeficiency virus (HIV) and was approved for its treatment in 2001. It is also active against HBV and was approved for its treatment in 2008. Tenofovir has emerged as an effective and potent therapy against lamivudine resistant strains as well as wild type HBV. Tenofovir resistance to HBV is rare. Long-term use of tenofovir has been associated with renal and bone marrow toxicity in some HIV-infected patients. To date, this is the only oral therapy which is both potent and does not lead to viral resistance in both treatment naïve and experienced patients. Further studies in HBV mono-infected persons are needed to evaluate long-term impact of tenofovir use.
Introduction
Hepatitis B (HBV) is the leading cause of liver disease world-wide, with 400 million people infected. 1 Transmission of HBV has decreased substantially in the US due to vaccination efforts, especially in children. However, approximately 51,000 new HBV infections occurred in 1995, mostly in adults. 2 HBV occurs at the highest frequency in unvaccinated adults who have multiple sexual partners, use injection drugs, or are men who have sex with men. 2 Although HBV infection when acquired in adulthood may be self-limited, 15%-20% of acutely infected adults will develop chronic liver disease. The complications of HBV infection include cirrhosis of the liver, hepatocellular cancer, and portal hypertension, which can present as variceal bleeding, encephalopathy, and ascites.
In order to reduce the burden of disease and its related complications, many therapeutic options have become available. In the last 15 years, treatment for HBV has changed significantly. Initially, treatment for HBV was limited to interferon therapy. Interferon, given daily or three times weekly for 4-6 months did lead to hepatitis B e antigen (HBeAg) seroconversion and in some cases, hepatitis B surface antigen (HBsAg) loss,3–6 especially after years of follow-up. HBeAg seroconversion or loss were both endpoints that were considered desirable as the virus was no longer actively replicating.
Although interferon-based therapies can be given for a finite period of time and lead to viral clearance in some patients, there are significant adverse side effects including: flu-like symptoms, depression, myalgias, fatigue, and cytopenias. Furthermore, fear of injections may make some patients reluctant to be treated. Because of these issues, other oral treatments were developed. The first oral nucleoside therapy to be used for the treatment of HBV was lamivudine (Epivir®; GlaxoSmithKline). Lamivudine was initially approved by the Food and Drug Administration (FDA) for the treatment of human immunodeficiency virus (HIV) at the dose of 300 mg per day. Studies in patients with HBV found that 100 mg was effective and led to viral suppression of HBV DNA. 7 Initial studies examined histological endpoints to see if treatment would improve hepatic fibrosis and delay the emergence of complications. Lamivudine showed both histological improvement and decrease in viral load.8,9 However, once the drug was stopped, most patients’ HBV viral load would resurge. Thus, one year of treatment was insufficient and longer duration of therapy was needed, which led to the emergence of lamivudine resistant virus. Long-term monotherapy, especially in the setting of incomplete viral suppression, can lead to the emergence of resistance variants which have mutations that allow the virus to replicate leading to increase in HBV viral load despite being on therapy. Subsequently, other nucleosides/nucleotides (nucleos(t)ides) such as adefovir (Hepsera®; Gilead Sciences) became available for the treatment of lamivudine resistant strains. Adefovir at the dose of 10 mg was well tolerated and was effective against lamivudine resistant virus. Subsequently, other potent nucleos(t)ide inhibitors have been developed for the treatment of HBV such as entecavir (Baraclude®; Bristol-Myers Squibb Co.) and telbivudine (Tyzeka®; Idenix Pharmaceuticals Inc. and Novartis Pharmaceuticals Corp.).
Tenofovir disoproxil fumarate (Viread®; Gilead Sciences), a second generation nucleotide, was developed for the treatment of HIV and its chemical structure was modified in order to reduce renal side effects. It was then co-formulated with emtricitabine (Emtriva®; Gilead Sciences) and marketed as Truvada® (Gilead Sciences). In this review, we will discuss the pharmacology and safety of tenofovir for the treatment of HBV and how it compares to other nucleos(t)ides in terms of efficacy.
Review of Pharmacology, Mode of Action and Pharmacokinetics
Mechanism of Action
Tenofovir is an analog of adenosine monophosphate, an endogenous purine that plays a vital role in many physiological processes including DNA replication. All purines, pyrimidines, and nucleos(t)ide analogs must undergo a series of phosphorylations by intracellular kinases to become physiologically active. Tenofovir is classified as an acyclic nucleoside phosphonate, or nucleotide analog, because it has the first of three needed phosphonate groups bonded to its alkyl side chain. The monophosphate group bonded at the 5’ position to tenofovir provides a potential advantage over other nucleoside analogs, such as lamivudine, since tenofovir contains the first of three required phosphonate groups making it a potentially more active compound. Once phosphorylated to the active triphosphate form, tenofovir competes with endogenous deoxyadenosine 5‘-triphosphate at HIV-1 reverse transcriptase and HBV polymerase to be incorporated into the growing strand of DNA. Since tenofovir lacks the hydroxyl group in the 3’ position found on deoxyadenosine 5‘-triphosphate another DNA base pair cannot be linked to tenofovir which results in DNA chain termination. All nucleos(t)ide analogs, including tenofovir, are weak inhibitors of human DNA polymerases alpha, beta, and mitochondrial DNA polymerase gamma. Therefore all nucleos(t)ide analogs carry the potential risk for lactic acidosis and severe hepatomegaly with steatosis resulting in an FDA mandate that a black box warning be included in their respective package inserts. Tenofovir is weaker than other nucleoside analogs at inhibiting mitochondrial DNA and therefore has less mitochondrial toxicities compared with other nucleos(t)ide analogs.10,11
Pharmacokinetics
Tenofovir was FDA approved in 2001 in adults for the treatment of HIV and in 2008 for the treatment of HBV. Most of the pharmacokinetic data available for tenofovir was gathered during clinical trials in HIV infected patients. Emtricitabine is structurally similar to lamivudine, and while not FDA approved for the treatment of HBV, does possess activity against HBV. Patients using co-formulations of emtricitabine and tenofovir (Truvada®) are receiving dual nucleos(t)ide therapy for HBV, which may decrease the emergence of resistance.
Tenofovir has poor oral bioavailability and is commercially available as a diester prodrug, tenofovir disoproxil fumarate, which is hydrolyzed in vivo to tenofovir and subsequently phosphorylated to its active triphosphate form. In addition to improving bioavailability, the diester prodrug also improves potency. 12 Neither tenofovir nor tenofovir disoproxil fumarate are substrates for cytochrome P450 (CYP450) enzymes and no dose adjustments are needed for patients with hepatic impairment. Each commercially available form of tenofovir contains 300 mg of tenofovir disoproxil fumarate, which is equivalent to 245 mg of tenofovir, and is administered once daily for patients without renal impairment for both HIV and HBV indications. The dose of tenofovir used for HIV and HBV treatment is the same, unlike lamivudine and adefovir where lower treatment doses for HBV are used, possibly contributing to the lower potency and differences in response rates. The oral bioavailability of tenofovir in the fasted state is 25% versus 40% when taken with a high fat meal (~700 to 1000 kcal containing 40 to 50% fat). Approximately 7% of tenofovir is bound to serum proteins with a serum half life of 17 hours after a single dose which extends to beyond 24 hours with continued dosing. This allows for convenient once daily dosing without regard to food. 10
Seventy to 80% of tenofovir is recovered as unchanged drug in the urine representing both passive filtration at the glomerulus and active secretion by renal tubular cells. Renal impairment reduces tenofovir clearance and extends its duration of activity. Dose adjustments for tenofovir are recommended based on calculated creatinine clearance (CrCL). The dose of tenofovir remains fixed at 300 mg and the interval is extended for varying degrees of renal insufficiency (see Table 1).
Tenofovir dosage adjustments in renal insufficiency. 10
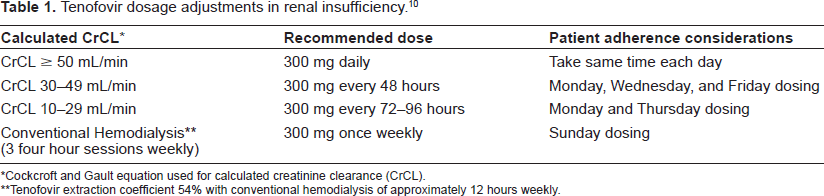
Cockcroft and Gault equation used for calculated creatinine clearance (CrCL).
Tenofovir extraction coefficient 54% with conventional hemodialysis of approximately 12 hours weekly.
Pediatric and Pregnancy Considerations
Tenofovir is not FDA approved for children less than 18 years of age. Studies conducted by investigators at the National Institute of Health in HIV infected children showed significant declines in bone marrow density (BMD) in children 11-17.5 years of age treated with tenofovir.13,14 The FDA has given tenofovir a pregnancy category B rating as animal studies have shown tenofovir to cause hepatic adenomas in mice and decreased fetal growth and reduction in fetal bone porosity in monkeys at exposures that exceed the levels anticipated in humans. The Department of Health and Human Services included cautionary statements in their most recent publication of the perinatal HIV treatment guidelines on the use of tenofovir because of limited data regarding fetal bone effects; therefore, tenofovir should only be used as a component of a maternal combination HIV regimen after careful consideration of other alternatives. Furthermore, maternal function should be carefully monitored. 15 It is currently unknown to what extent a decline in renal function or BMD will occur in HBV mono-infected patients treated with tenofovir for chronic viral suppression but expanding tenofovir's use to this population may provide both investigators and clinicians with the first definitive way to estimate tenofovir nephrotoxicity as HIV and other antiretroviral medications will be eliminated as confounding factors.
Efficacy Studies
Nucleos(t)ide Therapy for the Treatment of HBV
In order to understand the impact of tenofovir on the treatment of HBV, it is useful to review the drugs which are currently available for treatment (see Table 2). Initial studies with lamivudine, adefovir, and emtricitabine8,9,16–18 compared different doses of drug to placebo because no nucleos(t)ide therapy was considered standard of care. All of these studies which compared active drug to placebo found improvement in histology, decrease in log HBV DNA, and a higher proportion with undetectable HBV DNA in those on active drug. As lamivudine became the standard of care, the comparison group became lamivudine.19–22 Both entecavir and telbivudine were superior to lamivudine with regards to log HBV DNA decrease and proportion achieving undetectable viral load. Entecavir compared to lamivudine had a higher proportion with histological improvement in both HBeAg-positive and negative groups but for telbivudine the improvement in histology was only seen in HBeAg-positive group. Pegylated interferon in combination with lamivudine did show higher level of undetectable virus; and both groups using peginterferon showed significantly higher levels of HBeAg loss, HBeAg seroconversion, and HBsAg loss compared to lamivudine alone. 22 With increasing concern over lamivudine resistance, adefovir had become the first line therapy in naïve patients. The most recent treatment trials have used adefovir as the comparison drug.23–25 Entecavir, telbivudine, and tenofovir were all superior to adefovir with regards to log HBV DNA decrease and proportion with undetectable viral load at 1 year.
Outcomes with HBeAg-positive and HBeAg-negative patients treated for HBV with currently available nucleos(t)ides.
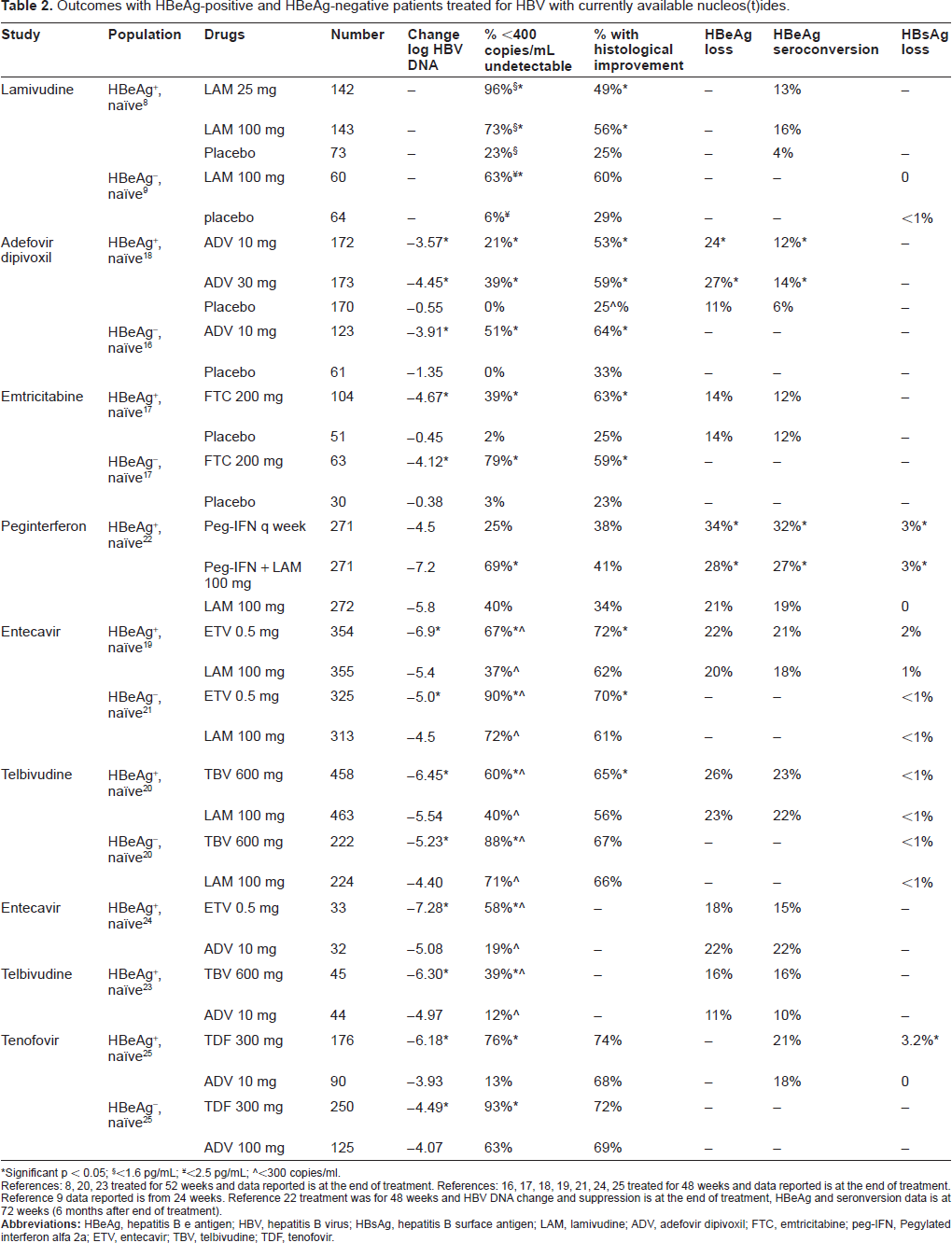
Significant p < 0.05;
<1.6 pg/mL;
<2.5 pg/mL;
<300 copies/ml.
References: 8, 20, 23 treated for 52 weeks and data reported is at the end of treatment. References: 16, 17, 18, 19, 21, 24, 25 treated for 48 weeks and data reported is at the end of treatment. Reference 9 data reported is from 24 weeks. Reference 22 treatment was for 48 weeks and HBV DNA change and suppression is at the end of treatment, HBeAg and seronversion data is at 72 weeks (6 months after end of treatment).
Until the recent study comparing adefovir to tenofovir, 25 prior studies with tenofovir have been mostly in those with HIV-infection as the drug has been approved for HIV treatment since 2001. Studies evaluating efficacy of tenofovir in combination with lamivudine were first reported in HIV/HBV co-infected patients.26,27 Further studies examined the efficacy of tenofovir in lamivudine resistant populations showed effectiveness against lamivudine resistant strains.28–31 Retrospective studies have examined lamivudine compared to lamivudine and tenofovir finding higher HBV DNA suppression in combination therapy. 32 Other studies comparing the combination of lamivudine and tenofovir to tenofovir have found similar levels of HBV DNA suppression with combination compared to tenofovir. 33 Tenofovir was associated with higher rate of achieving undetectable HBV viral loads 34 in HIV/HBV co-infected patients on antiretroviral therapy. However, tenofovir viral breakthrough has been reported 35 but only one reported case of viral resistance has been associated with tenofovir use. 36
There are two prospective studies of interest in HIV/HBV. First, a randomized study in HIV/HBV co-infected patients found tenofovir to be non-inferior to adefovir; however, this study was underpowered 37 and is in contradiction to the findings in a larger randomized trial of adefovir to tenofovir in HBV monoinfected patients. 25 Second, in a prospective, randomized trial in HIV/HBV naïve patients which compared lamivudine to lamivudine plus tenofovir to tenofovir as part of the HIV antiretroviral therapy, the investigators found a higher proportion of patients achieved HBV DNA viral suppression in the treatment arms containing tenofovir. 38 Thus, tenofovir may be very effective either as monotherapy or in combination with lamivudine.
Studies examining tenofovir in HBV mono-infected patients also demonstrated the efficacy of tenofovir in lamivudine resistant strains.39–42 Others have found tenofovir to be effective in patients with adefovir resistance or those with inadequate response to adefovir.43,44 Comparison of adefovir to tenofovir for lamivudine resistant variants found higher rates of undetectable HBV viral loads in those on tenofovir.40,42 Patients who achieved undetectable HBV viral loads on tenofovir than switched to adefovir underwent viral breakthrough in several case reports.45,46 Thus, several studies suggest that tenofovir is superior to adefovir. Finally, a large randomized trial comparing adefovir to tenofovir demonstrated that tenofovir was significantly superior to adefovir in terms of HBV DNA suppression and histological improvement. 25 No significant differences were noted between tenofovir and adefovir with regards to histological improvement, but when the combined endpoint of histological improvement and viral suppression was used, tenofovir was superior. 25 Interestingly, tenofovir showed a significantly higher proportion of HBsAg loss compared to adefovir. 47 The rate was similar to what has been observed with peginterferon at the end of treatment. 22 However, lifelong suppressive therapy with oral nucleos(t)ides may be necessary as clinical cure defined by HBeAg seroconversion and HBSAg loss does not always occur with current nucleos(t)ide therapy.
Use of Tenofovir in Cirrhosis and Transplant Patients
Although the studies of tenofovir in cirrhotic patients are limited, there are case reports of improvement of decompensated liver disease in patients with lamivudine resistance treated with tenofovir 48 and in improvement of liver function tests, and other parameters of end-stage liver disease. 49 Furthermore cirrhotic patients with adefovir resistance have also had improvements when treated with tenofovir.50,51
Although the data on safety of tenofovir in transplant patients is limited, there are reports of successful use of tenofovir for lamivudine resistant virus following liver transplantation. 52 In the post-transplant setting, tenofovir is being examined as a prophylaxis without the use of hepatitis B immune globulin. 53 A long-term follow-up study of HBV patients post heart transplant found 5 of the 9 deaths occurred due to lamivudine resistance, but successful rescue occurred in 4 patients who were treated with either adefovir or tenofovir. 54 However, these reports did not examine the impact of tenofovir on renal function especially in the setting of immunosuppressive medications. Tenofovir may lead to increase in serum creatinine levels which has also raised concerns about changes in Model for EndStage Liver Disease (MELD) score, a scoring system currently used to allocate livers for transplantation. 55 Further studies are needed in patients who are treated with tenofovir prior to transplantation and post-transplant to evaluate long-term efficacy and safety.
Viral Dynamics of Tenofovir
Studies examining viral dynamics with tenofovir are limited to those in HIV/HBV co-infected patients.56–58 All studies demonstrated a biphasic response to nucleos(t)ide therapy similar to that seen with HIV and hepatitis C. A faster first phase decline was associated with high HBV viral load and HBeAg-positivity. 57 The investigators also found time to reaching undetectable HBV viral load was longer in those with higher HBV viral load (150 days if HBV DNA < 108 log versus 316 days when HBV DNA > 108 log) and HBe Ag-positive. 57 A study comparing wild type and lamivudine resistance actually found a slower first phase decline in those with lamivudine resistance. 56 A larger study of HIV/HBV patients comparing viral kinetics in those on tenofovir, lamivudine, or both found no differences in kinetic parameters by treatment group. Overall treatment effectiveness was 98%, half-live of infected virions was 1.2 days, and median life of infected cells was 7.9 days. The authors reported longer infected half-life of hepatocytes in HBeAg-positive compared to HBeAg-negative patients. 58 The viral dynamic studies suggest that tenofovir dynamics are similar to other nucleos(t)ides. Furthermore, no differences in viral dynamics are evident between monotherapy vs. combination therapy. There may be lower antiviral effectiveness of tenofovir in lamivudine resistance, but this seems transient. No data to date shows if HBV genotypes affect viral dynamics.
Resistance
One of the problems with nucelos(t)ide therapy is that long-term use is associated with development of resistance. These nucleos(t)ides have begun to be used for longer periods of time, similar to the HIV model of treatment. Lamivudine resistance for HBV has been reported to occur at a rate of 20% per year 59 and in one study occurred in over 90% of HIV/HBV co-infected patients treated for 4 years. 60 Viral resistant populations have a characteristic mutation in the reverse transcriptase (RT) polymerase (see Table 3). For example, lamivudine resistance is associated with genotypic resistance in RT polymerase at L180M or M204V/I.61,62 Both in vitro and in vivo studies have found adefovir and tenofovir to be active against lamivudine resistant virus.39,40,45,63,64 Telbivudine, although highly potent in terms of HBV viral load suppression, resistance does occur at the same site as lamivudine. 20 Adefovir resistance is associated with emergence of rtA181V or rtN236T. 61 Tenofovir has also been shown to be effective in those who have suboptimal response to adefovir. 44 In addition there are case reports of patients with adefovir resistance who were treated successfully with tenofovir.50,65 Entecavir has been found to have a high genetic barrier leading to a cumulative failure of 0.8% at 4 years in naïve HBV patients. 66 However, once a person has lamivudine resistance, then developing entecavir resistance is easier. Thus, resistance to entecavir occurs more frequently in patients who are lamivudine experienced with a cumulative failure rate of 40% in lamivudine experienced patients on entecavir for 4 years. 66 Mutations in the RT polymerase seen with entecavir are at rtM204V + rtL180M plus other variants or rtM204I plus other variants. 61 There are case reports of salvaging entecavir resistant virus with tenofovir. 67 However, in vitro constructs of entecavir resistant virus suggests that there is a 4 fold decreased susceptibility to tenofovir. 62 Tenofovir resistance has been reported in one case 36 associated with rtA194T mutation. A recent study constructed the rtA194T mutation alone or in addition to lamivudine resistance and demonstrated reduced replication efficiency compared to wild type. Viral fitness was restored in precore and basal core promoter mutations. Furthermore, clones harboring rtA194T had partial resistance to tenofovir in vitro but remained susceptible to telbivudine and entecavir. 68
Nucleos(t)ide therapy and potential mutations in RT polymerase gene.

Combination vs. Monotherapy
Monotherapy with drugs such as lamivudine has lead to the development of resistant HBV. Borrowing from the HIV model where combination therapy has been shown to be effective in reducing the emergence of resistant variants, studies in HBV have focused on using combination therapy to improve both viral dynamics and reduce the emergence of resistant variants. Combination therapy trials have evaluated lamivudine plus adefovir versus adefovir 69 in lamivudine resistant patients; lamivudine plus adefovir versus lamivudine 70 in naïve patients, or both 64 (see Table 4). However, these studies failed to show improved antiviral effect compared to appropriate control populations, except for the study by Peters and colleagues which only showed differences when lamivudine was compared to adefovir or combination of lamivudine and adefovir. However, the patients in this study were lamivudine experienced and had resistant variants that would not respond to lamivudine treatment. Other studies examining emtricitabine vs. emtricitabine plus clevudine; 71 lamivudine plus famciclovir vs. lamivudine 72 also did not show improved antiviral effects with combination therapy.
Outcomes from studies comparing monotherapy to combination therapy for HBV treatment.
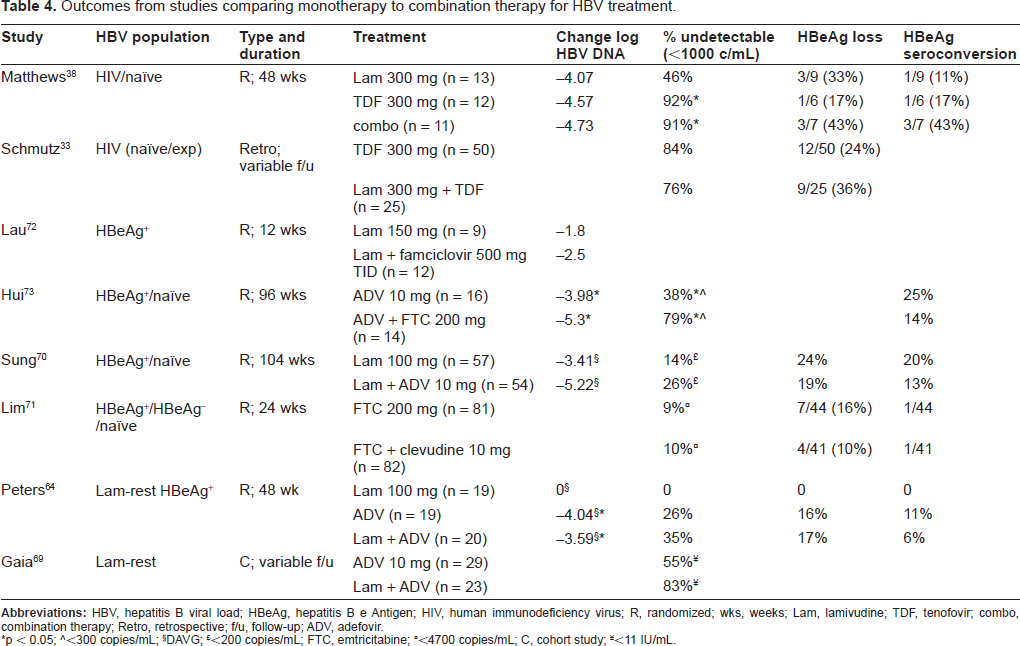
p < 0.05;
<300 copies/mL;
DAVG;
<200 copies/mL; FTC, emtricitabine;
<4700 copies/mL; C, cohort study;
<11 lU/mL
A recent study comparing adefovir vs. adefovir plus emtricitabine in HBeAg+ naïve patients 73 show improved viral decline in HBV DNA (-5.3 vs. -3.98, p = 0.05) and viral suppression <300 copies/mL (79% vs. 38% p = 0.03) in combination therapy versus monotherapy. Sung et al also showed that in HBeAg+ naïve patients on lamivudine had a higher proportion developing viral breakthrough compared to those treated with the combination group of lamivudine and adefovir (45% vs. 19%, p = 0.018). 70 Both studies70,73 focused on naïve patients and followed them for an extended period of time to evaluate for efficacy and resistance. Other studies may have been unable to show differences because of short time of follow-up71,74 or examining a population with lamivudine resistance64,69 which in effect was equivalent to receiving one active drug. Thus, combination therapy when given over a prolonged period of time in HBV naïve patients appears to be more potent and reduces the emergence of resistant variants.
However, both lamivudine and adefovir are less potent than drugs such as entecavir, tenofovir and telbivudine. Highly potent drugs may not need to be used in combination with other drugs. Most of the studies evaluating combination therapy initially were reported in HIV/HBV co-infected patients. The studies using tenofovir and lamivudine or tenofovir and emtricitabine are limited because of retrospective design, inclusion of both naïve and experienced patients,32,33 lack of an appropriate control population,26,30,75 or small number of patients. 27 To date, these studies suggest improved efficacy with tenofovir alone or in combination. However, a recent study by Mathews and colleagues 38 randomized HIV/HBV treatment naïve patients to receive lamivudine versus tenofovir versus lamivudine and tenofovir as part of the antiretroviral therapy (ART). This study showed a significantly higher proportion of patients achieved <1000 copies/mL in the tenofovir and combination arm compared to the lamivudine alone regimen (92% vs. 91% vs. 46%, respectively, p = 0.013). Thus, nucleos(t)ides with a high genetic barrier to viral resistance and high level antiviral efficacy such as entecavir and tenofovir may be able to be used as monotherapy in treatment naïve patients. However, drugs such as lamivudine, telbivudine, and adefovir are probably best used in combination therapy.
Safety and Tolerability
Nephrotoxicity
Tenofovir is one of three nucleotide analogs which include adefovir, approved for HBV treatment, and cidofovir, approved in 1996 for the treatment of cytomegalovirus (CMV) retinitis. It is interesting to note that, all three nucleotide analogs share nephrotoxicity as an adverse effect. In fact, cidofovir (Vistide®; Gilead Sciences) carries a black box warning regarding renal impairment with strict criteria for use to reduce the incidence of nephrotoxicity. 76 Adefovir was initially developed for the treatment of HIV at a dose of 60 mg daily and was halted due the development of nephrotoxicity. A lower 10 mg dose of adefovir was found to be active against HBV with an acceptable balance of efficacy and safety and received FDA approval for HBV treatment in 2002. 77 The mechanism of nephrotoxicity is mediated by the active uptake of nucleotides from the systemic circulation by human renal organic anion transporter 1 (hOAT1) causing the accumulation of the parent nucleotide or metabolites in the proximal renal tubular cells resulting in mitochondrial DNA depletion and cell toxicity. 78 Tenofovir, unlike cidofovir and adefovir, is efficiently transported by hOAT1 but has not been shown to produce the degree of nephrotoxicity seen with the other nucleotide compounds. 78 A recent study found that tenofovir caused ultrastructural abnormalities to mitochondrial DNA in proximal renal tubules 79 but not to other organs, which suggests that tenofovir toxicity is mostly associated with tubulopathy.
Overall, the degree of nephrotoxicity seen with tenofovir is low and is estimated to range from 0.3 to 2% per year.80,81 Rather than overt cytotoxicity to the renal proximal tubule epithelial cells, a milder form of nephrotoxicity is associated with tenofovir. Because tenofovir was initially approved for HIV, safety data on long-term tenofovir use is mostly available from use of tenfovir for HIV-infected patients. Tenofovir has been associated with Fanconi's syndrome, 82 nephrogenic diabetes insipidus, 83 renal failure, 84 and a decrease in glomerular filtration rate. 85 Fanconi's syndrome, a disorder in which the proximal tubule function is impaired, results in the loss of electrolytes including phosphate, bicarbonate, amino acids, and glucose into the urine. The loss of bicarbonate may result in metabolic acidosis. Winston and colleagues showed a statistically significant increase in anion gap of 0.78 mmol/L and a decline in CrCL of 6.8 mL/min in a retrospective review of 948 HIV patients with no baseline renal impairment. Patients experiencing tenofovir induced nephrotoxicity may have serum creatinine and phosphorus levels within established normal ranges. 86 In the registration trial for tenofovir in HIV infected adults, 600 ART naïve HIV patients treated with either tenofovir or stavudine and the same background ART regimen were followed for 144 weeks without developing statistically significant changes in serum creatinine or hypophosphatemia between treatment groups. 87 However, a recent study of HIV patients treated with tenofovir compared to those on other nucleosides or on no medications found evidence of tubular dysfunction without impairment of glomerular filtration rate (GFR). Furthermore, in a multivariate model tenofovir use and older age was independently associated with increased risk of tubular dysfunction. 88 Thus, patients on long-term tenofovir should be monitored for serum creatinine and phosphate levels and urinalysis samples to monitor for evidence of glucose and phosphorus.
The incidence of hypophosphatemia varies among clinical studies and has been reported at 7% in ART naïve HIV patients, 15% in the HIV Outpatient Study (HOPS) cohort by investigators at the Center for Di sease Control (CDC), and up to 31% in a United Kingdom cohort.87,89,90 Two reports concluded that moderate, asymptomatic hypophosphatemia is frequently seen in HIV infected patients treated with ART but often the cause of renal dysfunction cannot be entirely accounted for by the use of tenofovir91,92 as all three studies did not show statistical differences in the incidence of hypophosphatemia between treatment groups. Some associations with nephrotoxicity and hypophosphatemia that have been established in tenofovir treated HIV patients include: older age, African-American race, co-morbidities that also lead to renal dysfunction such as diabetes and hypertension, concurrent nephrotoxic medications such as NSAIDs, low body mass index, medications known to increase tenofovir exposure (lopinavir/ritonavir (Kaletra®; Abbott Laboratories), duration of ART use, and potentially genetic haplotype differences.90,93–95 There does not appear to be a definitive time course relationship to the development of tenofovir induced nephrotoxicity as patients may experience nephrotoxicity at any time point during tenofovir therapy. Fortunately, renal function usually returns to baseline within four to six weeks of discontinuation of tenofovir.
Bone Marrow Toxicity
Phosphorus is a critical electrolyte to the function of the human body and is carefully balanced by an elegant feedback system involving the kidney, parathyroid gland, and skeletal system. Increased urinary excretion of phosphorus is a sensitive marker of proximal renal tubular dysfunction and causes the parathyroid gland to release parathyroid hormone stimulating catabolism of bone by osteoclasts in order to maintain serum phosphorus levels at physiologic levels. This can result in osteopenia or osteomalacia with loss of BMD placing individuals at risk for fracture. In the registration trial for tenofovir in HIV infected adults, there was a significantly greater mean percentage decrease from baseline in BMD at the lumbar spine (-2.2% tenofovir vs. -1.0% stavudine, p = 0.001) but only a trend toward a difference at the hip (-2.8% tenofovir DF vs. -2.4% stavudine, p = 0.06) in tenofovir treated study participants at week 144. 96 Case reports of bone fracture due to osteomalacia have been reported in HIV patients.97,98 Unfortunately, HIV infection itself has been associated with a decline in BMD creating treatment challenges for clinicians. Long term safety data with tenofovir in patients with HBV mono-infection is not available. Further studies and monitoring are needed in this population to help determine how much of the renal and bone toxicity associated with tenofovir may be confounded by concomitant medications used to treat HIV or HIV itself.
Non-Renal Adverse Events
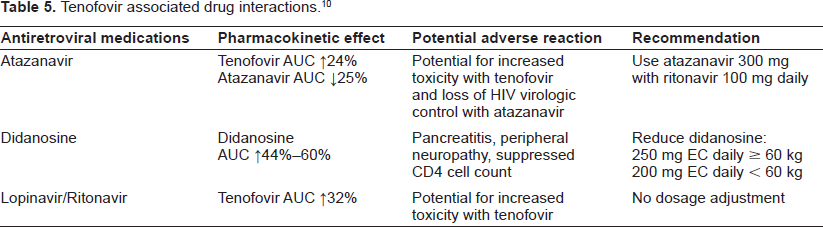
Over 12,000 patients have received tenofovir in clinical trials and expanded access studies where the most common adverse reactions of Grade 2-4 severity occurring in ≥10% of study patients included rash, diarrhea, headache, pain, depression, asthenia, and nausea. 10 In addition to these side effects, tenofovir may interact with other medications, most of these are HIV medications, and the dose may require adjustment (see Table 5).
Tenofovir associated drug interactions. 10
Patient Focused Perspective
Tenofovir is a nucleotide analogue that is highly potent for the treatment of HBV. There is little evidence for development of tenofovir resistance to date, but this remains a possibility. In addition, tenofovir has been shown to affect HBsAg clearance 25 at the end of treatment, which may turn out to be of importance especially if long-term follow-up studies show increasing rates of HBsAg clearance. Unlike interferon-based therapy which requires weekly injections and has many side effects, tenofovir is well tolerated. However, long-term use of tenofovir may lead to kidney and bone toxicity. The use of the drug requires monitoring of serum creatinine and phosphorous levels as well as urinalysis. The drug can be taken once daily making it a simple regimen.
Conclusions and Place in Therapy
Tenofovir is a potent drug for the treatment of HBV and leads to significant viral suppression. Both entecavir and tenofovir in naïve patients rarely lead to viral resistance which makes both these drugs attractive as first line therapies. In the case of entecavir, we know that multiple mutations are needed to lead to entecavir resistance. Telbivudine, another potent drug for HBV treatment, is limited by the emergence of lamivudine resistance making it a less desirable drug for long-term therapy. Tenofovir, has activity against lamivudine resistant strains, and to date only one case report has shown possible tenofovir resistance. Thus, the drug of choice in lamivudine experienced patients would be tenofovir. In HBV treatment naïve patients, entecavir remains a viable alternative. In the HIV-infected patients, Truvada® has become the drug of choice. Yet, there remain populations in which tenofovir may not be suitable including those with renal insufficiency.
However, if the goal of therapy is HBsAg loss, a clinical cure, and not viral suppression, than peginterferon is more likely to produce these results. 99 HBeAg seroconversion and HBsAg loss is also more likely with peginterferon 22 than nucleos(t)ide.16–21,25 Recent data suggests that tenofovir may be more likely to lead to HBsAg seronconversion. 25 Further studies are needed to evaluate the impact of tenofovir in combination with peginterferon.
Tenofovir is a potent and relatively safe drug that can suppress HBV DNA. Data on long-term therapy is limited but to date viral resistance is uncommon. Safety data are limited to patients with HIV-infection, and it remains unknown if the long-term renal and bone toxicity will be similar to that seen in HIV-infected patients and if it will limit long-term use for HBV treatment. Since the risk of toxicity is low compared to the potential benefit of viral suppression, HBeAg seronconversion, and in some patients HBsAg loss, tenofovir has become a very important drug in the arsenal of therapeutic options available for the treatment of HBV.
Disclosures
The authors report no conflicts of interest.
