Abstract
Because of the dramatic increase in severe infections caused methicillin-resistant Staphylococcus aureus (MRSA), including skin and skin structure infections (SSSI), and reports of vancomycin failures in the treatment of these infections, agents with better activity against MRSA are clearly needed. Daptomycin, the first cyclic lipopeptide, exerts its concentration dependent bactericidal activity against S. aureus through a calcium dependent formation of channels leading to disruption of the bacterial cell membrane potential. Daptomycin is 90% protein bound, has a half-life of 8–9 h, is eliminated through renal excretion, and has a good penetration into inflammatory skin blisters. This compound was shown to be non-inferior to the comparator in two double-blind randomized phase III studies including patients with complicated SSSI, using a dose of 4 mg/kg/day. In these studies, cure rates in the clinical and microbiology evaluable populations were 83.4% and 84.2% (95% CI, -4 to 5.6), and 84.7% and 85.9% (95% CI, -3.8 to 6.3) for daptomycin and comparator, respectively. Daptomycin also showed efficacy in prospective studies in patients with infective cellulitis or erysipelas, necrotizing soft tissue infection, and infected diabetic foot ulcers. Even though the standard dose of 4 mg/kg/day is considered appropriate for most SSSI, higher doses (e.g. 6 to 10 mg/kg/day) are currently suggested for patients with sepsis, necrotizing infection, prior glycopeptide failure, infections caused by vancomycin intermediate S. aureus (VISA) strains, renal insufficiency, burns, and in intravenous drug users. Overall, daptomycin is considered a well tolerated antibiotic. In clinical trials, drug discontinuation rate has been similar in patients receiving daptomycin compared with those treated with the comparators; however, muscular damage associated with increased serum creatine phopshokinase levels occurs in about 3% and 7% of the patients treated with 4 and 6 mg/kg/day, respectively. This reversible muscle toxicity appears to correlate with the daptomycin trough concentration and time of exposure.
Introduction
Skin and skin structure infections (SSSI) are among the most common bacterial infections in humans. In the United States, SSSI are associated with approximately 11 millions of consults in ambulatory care. 1 Staphylococcus aureus is the most common pathogen responsible for SSSIs. During the last few years there was a dramatic increase in cutaneous infections caused by community-associated methicillin-resistant S. aureus (CA-MRSA) strains. 2 Indeed, strains of CA-MRSA carrying the staphylococcal cassette chromosome mec (SCCmec) type IV (usually clone USA 300) and the Panton-Valentine leukocidin (PVL) have now been reported as the most prevalent cause of staphylococcal community onset SSSI, particularly purulent lesions. 3
Vancomycin has been for years the standard of care for patients with complicated SSSI due to MRSA. 4 However, several strains of MRSA with reduced susceptibility to vancomycin have appeared in the clinical arena. In addition, different centers have reported increasing minimum inhibitory concentrations (MICs) to vancomcyin among susceptible strains of MRSA (MIC creep). 5 In patients infected with MRSA a significant body of evidence indicates that higher MICs to vancomcyin are associated with worse clinical outcomes. 6 In this scenario new antibiotic options to treat patients with MRSA infection are needed.
Daptomycin
Mechanism of action, antimicrobial activity, and resistance
Daptomycin (Cubicin®) is the first member a new class of antimicrobials derivatives of Streptomyces roseosporus, called cyclic lipopeptides. Daptomycin was discovered in the 1980s and soon thereafter showed some efficacy in early clinical trials. However, its development was discontinued few years later due to skeletal muscle toxicity observed in phase II studies. This toxicity was shortly found to be related to the initial twice-daily dosing schedule. Subsequent clinical trials were resumed using once daily dosing which was associated with reduced muscle toxicity. Daptomycin was approved for clinical use in 2003 in the United States and in 2006 in Europe.
Daptomycin has a high molecular weight (1620 Da) due to the presence of a 13 member amino acid cyclic lypopeptide linked to a highly lipophilic decanoid acid side chain. The mechanism of action of daptomycin derives from the insertion of the lipophilic tail into the cell membrane of gram-positive organisms without entering the bacterial cytoplasm. This process, which is calcium dependent, leads to the formation of channels from which intracellular potassium is lost,7,8 disrupting the bacterial cell membrane potential and causing cell death. Daptomycin is unable to penetrate the outer membrane of Gram-negative bacteria and as a consequence the drug is inactive against Gram-negative microorganisms. 9 The bactericidal effect of daptomycin against S. aureus appears to take place without significant cell lysis, what correlates with the decreased release of proinflamatory mediators from infected macrophages found in vitro. 10
Daptomycin is active in vitro against a variety of gram positive organisms resembling the activity exerts by glycopeptides. Since the activity of daptomycin depends on the presence of calcium cation in the medium, the susceptibility test of this antibiotic must be determined by dilution methods using calcium-adjusted Mueller-Hinton broth medium (CAMHB). Calcium supplemented agar gradient strips (E-test), and automated and semi-automated systems for susceptibility are also reliable methods for determing daptomycin susceptibility 11
Daptomycin has shown very good in vitro activity against a wide variety of Gram-positive bacteria including S. aureus (methicillin susceptible and methicillin resistant strains), coagulase negative staphylococci, Streptococcus agalactiae, Streptococcus pyogenes, group C, group G, and group F β-hemolytic streptococci, Streptococcus pneumoniae (including penicillin-resistant isolates), Bacillus spp., Corynebacterium spp., viridans streptococci, and Streptotoccus bovis with MIC90s of ≤0.5 μg/mL. Against enterococci, MIC90s of daptomycin are usually of 1 or 2 μg/mL for Enterococcus faecalis and 4 μg/mL for Enterococcus faecium, regardless of the vancomycin susceptibility pattern. The current breakpoints for daptomycin susceptibility using CAMHB are ≤1 μg/mL for staphylococci and streptococci and 4 μg/mL for E. faecalis (breakpoints for E. faecium isolates has not been established yet). Daptomycin displays rapid and concentration-dependent bactericidal activity against staphylcococci, pneumococci, and E. faecalis and E. faecium,12,13 including MRSA, vancomycin-intermediate S. aureus, and vancomycin-resistant enterococci isolates.13,114 Daptomycin has shown in vitro activity against some gram-positive anaerobes (e.g. Peptostreptococcus spp., Clostridium perfringens, and Clostridium difficile) 15 but there are no clinical data on its use for the treatment of these microorganisms.
Daptomycin resistance is very rare in strains without prior exposure to this drug; a large survey found that only 0.04% of 10,000 S. aureus strains were resistant to daptomycin (≥2 μg/mL). 16 However, development of resistance to daptomycin while on therapy appears to be a more frequent phenomenon: 6% of patients participating in a large S. aureus bacteremia and endocarditis clinical trial had isolates that experienced increased in the daptomycin MICs to ≥2 μg/mL. 17 Of note, in most of these cases patients did not undergo for significant removal of the foci of infection. 17 The mechanisms underlying the development of resistance to daptomycin are not clearly understood, 18 although S. aureus strains with high daptomycin MICs appear to have an enhanced cell membrane fluidity, increased net positive surface charge, and decreased daptomycin surface binding, all of which derive in decreased daptomycin-induced depolarization and cell autolysis. 19 Other phenotypic characteristics such as heteroresistance to vancomycin and increased cell-wall thickness have also been found in a laboratory derived daptomycin non-susceptible strain. 20 In this regard, a report from the Centers for Disease Control and Prevention (CDC) suggested an association between decreased susceptibility to vancomycin and decreased susceptibility to daptomycin. 21 However, all strains of VRSA (van-A mediated) in this report were susceptible to daptomycin indicating that a mechanism other van-A may be responsible for the association. Vancomycin intermediate S. aureus (VISA) strains (vancomycin MICs of 4 to 16 μg/mL) had daptomycin MICs falling above the susceptibility breakpoint (≥2 μg/mL) more frequently than those with low vancomycin MICs.21,22 In addition, the development of daptomycin heteroresistance has been observed in heteroresistant VISA (hVISA) strains. 23 However, daptomycin displayed bactericidal activity against these hVISA strains 23 and was effective in a endocarditis model of hVISA. 24
Pharmacokinetics and pharmacodynamics properties related to SSSI
After a single intravenous dose (infused over 30 minutes) of 4 and 6 mg/kg of daptomycin, the mean peak serum concentrations is approximately 55 and 86 μg/mL, respectively, and the AUC24 at steady state for the same dosages is in the range of 500 and 750 μg·h/mL, respectively25,26 Daptomycin is about 90% protein bound and has a long terminal half-life (about 8–9 h) and, since it is distributed mainly into extracellular compartment, it has a small volume of distribution (~0.1 L/kg).25,26 The route of elimination is primarily through renal excretion, mostly as unchanged drug.
The penetration of daptomycin in inflammatory skin blister was evaluated in a small group (n = 7) of healthy volunteers receiving a 30-min infusion of 4 mg/kg/day of the drug. The maximum concentration (Cmax) reached by daptomycin in the blister fluid was 27.6 ± 9.5 μg/ml about 3 hs (Tmax) after the peak observed in serum; the mean overall penetration was 68% as measured by the area under the concentration-time curve over 24 hs. 27
Daptomycin should be administered intravenously, diluted in 0.9% sodium chloride, in once daily 30 minute-infusions. Daptomycin is not compatible with dextrose-containing solutions. In patients with a creatinine clearance of less than 30 mL/minute, the dosing interval should be modified and daptomycin has to be administered every 48 h. In patients on hemodialysis, daptomycin infusion should be delayed until the completion of the hemodialysis session because drug level can drop up to 35% if high permeability dialyzers are used. 28 Higher clearance of daptomycin occurs during continuous renal replacement therapies 29 and extended daily dialysis 30 suggesting that higher doses might be needed in this setting. Since daptomycin is not metabolized through the liver, no dosage adjustment is required in patients with moderate hepatic impairment (Child-Pugh class B). The intravenous administration of this compound in bolus has led to similar serum concentrations and safety profile as the 30-minutes infusion in healthy volunteers, which would significantly ease its use in the outpatient setting. 31 Obese subjects have higher Cmax and AUC concentrations (25 and 30%, respectively compared with non-obese patients) but still, dosage modification does not appear to be needed in this population. 32 Daptomycin is not approved for use in children, although it has been safely administered in 15 children (median age, 6.5 years) with invasive staphylococcal infection, most of which had CA-MRSA bacteremia. 33 Daptomycin was considered to be not teratogenic in animals and has been defined as pregnancy category B; however, because of the lack of sufficient clinical data in humans daptomycin should only be used in pregnant women when potential benefits outweigh potential risks. Same recommendation applies to lactating mother since it is unknown if daptomycin is present in human milk.
The peak/MIC ratio and the 24 h-AUC/MIC ratio were the in vivo parameters that best correlated with efficacy in a neutropenic mice thigh model of S. aureus infection. 34 In this model, the post-antibiotic effect was 5 h and, although daptomycin was shown to produce a post-antibiotic effect against S. aureus ranging from 1 to 6.3 hr. 12
Daptomycin has been approved for the treatment of complicated SSSI at a dose of 4 mg/kg/day and for S. aureus bacteremia (including right-sided endocarditis) at 6 mg/kg/day.17,35 However, based on its concentration dependent activity several experts have proposed a higher dose of daptomycin (i.e.: 8–10 mg/kg/day), particularly for S. aureus bacteremia and endocarditis. Theoretically, this off-label approach would also minimize the risk of development of resistance, esspecially in the setting of high inoculum infections. 36 In this regard and in the setting of cSSSI, doses higher than 4 mg/kg/day has been recommended in patients with sepsis, suspected necrotizing infection, in intravenous drug users, or patients with burn, diabetic foot infections, and in those with renal insufficiency. Higher doses may also be considered in patients with osteomyelitis, in patients with infection caused by VISA strains or in those patients who have experienced prior failure to glycopeptides. 37
Clinical uses of daptomycin in patients with skin and skin structure infections (Table 1)
Clinical studies on the use of daptomycin for skin and skin structure infections.
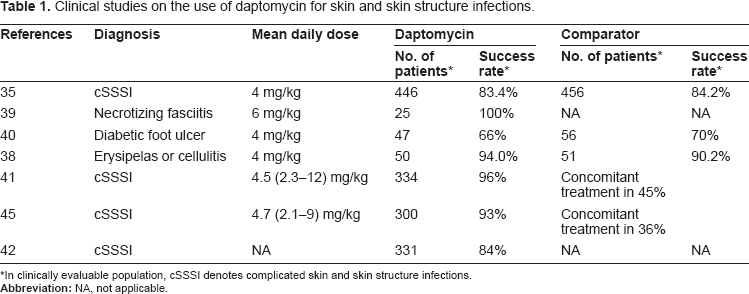
In clinically evaluable population, cSSSI denotes complicated skin and skin structure infections.
Prospective and comparative studies
Two identical phase III, double-blinded randomized trials in patients with complicated SSSI (cSSSI) compared daptomycin at 4 mg/kg every 24 h with standard therapy (vancomycin 1 g IV every 12 hrs or anti-staphylococcal semi-synthetic penicillins 4–12 g IV every 4 hrs). Randomization ratio was 1:1, antibiotic treatment was maintained for 7–14 days and the test of cure visit was conducted 6–20 days after the last dose of study medications. 35 Patients were allowed to switch to oral therapy if they fulfilled predefined criteria. A total of 1092 patients were enrolled and received at least ≥1 dose of study medication (intention to treat population). Wound infection and major abscesses accounted for more than 70% of the patients enrolled, and diabetes was present in about 30% of the subjects. The most common pathogens isolated were S. aureus (total = 628; MRSA = 87), S. pyogenes (n = 195), vancomycin-susceptible E. faecalis (n = 106), S. agalactiae (n = 71), viridans streptococci (n = 64), and Streptococcus dysgalactiae equisimitis (n = 27). No significant differences were found in the distribution of these pathogens between the treatment groups. More than one gram-positive pathogen was isolated in a significant proportion of patients (227 of 1092 subjects). Approximately one-quarter of patients received protocol allowed concomitant antimicrobials (e.g. aztreonam and/or metronidazole) and almost 30% of patients in each treatment group underwent for a surgical procedure (e.g.: drainage or debridement). Overall results from these phase 3 studies in patients with cSSSI indicated that daptomycin was non-inferior to the comparator. Cure rates in the clinical and microbiology evaluable populations were 83.4% and 84.2% (95% CI, -4 to 5.6), and 84.7% and 85.9% (95% CI, -3.8 to 6.3) for daptomycin and comparator, respectively. In patients infected with gram-positive organisms isolated at baseline response rates were also similar between the two treatment arms. In addition, in the clinically evaluable population the clinical success rate according to the baseline diagnosis was comparable in both treatment groups: wound infection 84% and 87%, major abscess, 92% and 88%, infected diabetic ulcer 66% and 70%, and non-diabetic infected ulcer 79% and 83% for, daptomycin and comparator, respectively. 35 Cure rates among 64 microbiologically evaluable patients infected with MRSA were 75% for daptomycin and 69% for the comparator (95% CI for the difference -28.5, 17.4).
About 10% of the intention-to-treat population had their parenteral therapy switched to oral antibiotics. A significantly higher proportion of patients in the daptomycin arm were considered cured with 4 to 7 days of intravenous therapy only (63% vs. 33%). This decrease in the requirement of intravenous treatment might impact the cost-effective analysis favoring daptomycin, although further studies are required. A relapse rate of 4% to 5% was seen in both treatment groups. 35
Daptomycin was evaluated in a prospective study in adult patients with infective cellulitis or erysipelas. 38 A total of 101 patients were randomized and received at least receive one dose of daptomycin (4 mg/kg/day) or vancomycin for 7–14 days. Most patients were female, white and had infections in the lower limbs. Obesity and diabetes were common in both treatment groups. S. aureus (particularly MSSA) was the most common pathogen isolated at baseline. Cure rates were similar in both study groups (94% vs. 90% for daptomycin and vancomycin, respectively; 95% CI for the difference -6.7, 14.3). Mean duration of study medication, median time to stabilization of the infection and to defervescence were also similar between treatment groups. 38
Daptomycin has been evaluated in a prospective study of patients with diagnosis of necrotizing soft tissue infection documented at surgery caused by gram-positive cocci. 39 Twenty-five patients received daptomycin (6 mg/kg/day) plus antianaerobic (metronidazol) and gram negative coverage (aztreonam or ciprofloxacin with/without an aminoglycoside). Patients with a creatinine clearance less than 30 ml/min were excluded from the study. Two-thirds of the patients had polymicrobial infections, 60% were diabetic, and isolated gram-positive organisms included S. aureus (n = 7), enterococci (n = 4), coagulase negative staphylococci (n = 6), S. pyogenes (n = 3), S. agalactiae (n = 4), and other streptococci (n = 10). A mean of 3 surgeries were required in each patient. All the clinically evaluable patients (n = 17) were considered cure at the test of cure after 3 to 28 days of daptomycin therapy. 39 Although more clinical data is needed daptomycin and antibiotics with activity against anaerobes and gram negative pathogens plus surgical intervention appeared safe and effective for the treatment of necrotizing soft tissue infections.
In a subset analysis of 103 clinically evaluable patients with infected diabetic foot ulcers that had been enrolled in the two phase III studies of cSSSI, the success rates were 66% and 70% for daptomycin and the comparator arm, respectively (95% CI, -14.4, 2.8). The most common isolated pathogen was S. aureus. 40
Retrospecive studies on the use of daptomycin
In a retrospective post-marketing registry on the use of daptomycin in 2004 (CORE-2004), 1160 were included in the analysis; 41 the outcome was determined by the investigator at the clinical site. The most common indication was SSTI with 45% of the cases (n = 522). From this 188 were uSSSI (uncomplicated cellulitis, simple abscess, erysipelas, furuncles, acne, and impetigo) and 334 were cSSSI. The most important diagnosis among cSSSIs included were surgical site infections (n = 142), wound infections (n = 64), mayor abscesses (n = 62), diabetic foot infections (n = 39), infected ulcers (n = 25), and necrotizing infections (n = 10). The most common pathogen was MRSA with 57% of the isolates, then MSSA (12%), coagulase negative staphylococci (13%), and VRE (11%). The outcome was determined to be clinical success (cure or improved) in 96% of patients with cSSSI and 98% among those with uSSSI. The response rate was similar between all the pathogens isolated at baseline. The median time of clinical response was 4 days (range 1–32 days) in patients with cSSSI. The majority of the patients with bacteremia (13 of 14) were successfully treated with daptomycin.
In a retrospective, non-comparative, and unblinded report of data on post-marketing use of daptomycin (EUCORE 2007), from 2006 until 2008, 1127 patients were included in the analysis. 42 The overall initial dose was 6 and 4 mg/kg for 47% and 32% of the patients, respectively. The most common indication for use was cSSSI with 331 patients. S. aureus was the predominant pathogen, isolated in 34% of patients. Among patients with cSSSI, 39% were considered cured, 45% improved, 7% failed and 9% were deemed non-evaluable. A recent retrospective assessment of clinical outcomes of patients enrolled in this cohort found that, in the univariate analysis, outpatient treatment and the presence of SSTI were associated with clinical success. 43 In the same study, endocarditis, bacteremia, renal insufficiency (creatinine clearance less than 30 ml/min), and diabetes mellitus were indepedently associated with daptomycin clinical failure. 43
In a small pilot randomized study including 96 patients with cSSSI, most of which were caused by MRSA, a high dose (10 mg/kg/day) of daptomycin given for only 4 days was compared with standard of care (vancomycin or semisynthetic penicillins) given for as long as needed. 44 This study showed that the comparator group had higher success rate than the daptomycin high-dose short duration arm, although not statistically signficant (87.5% vs. 75%; 95% CI: -27.9, 2.9); the median duration of treatment in the comparator arm was 8 days.
The activity of daptomycin against most of the gram-positive organisms associated with SSSI and its once-daily administration makes of daptomycin a very suitable agent for outpatient treament. In this regard, in the CORE registry 2005 similar results were reported in the outpatient setting compared with those who received inpatient treatment. 45
Safety profile in clinical studies of SSSI
Overal, daptomycin is considered a well tolerated antibiotic. In clinical studies, the rate of drug discontinuation was similar in patients receiving daptomycin compared with those treated with the comparators. Soon in its clinical development, the finding of muscular skeletal toxicity was of concern; this toxicity was later found to be related to the frequency rather than the total dose of daptomycin administered. 46 Although infrequent muscular toxicity can also occur with once-daily dose of daptomycin. The muscular damage caused by daptomycin is associated with a degenerative-regenerative process within the myofibers, causing increased serum creatine phopshokinase (CPK) levels, a useful clinical marker. Even though the exact mechanism of skeletal muscle damage is unknown, a correlation with the daptomycin Cmin concentration and time of exposure exists. In patients with SSSI treated with 4 mg/kg/day dose, the level of serum CPK was similar in those receiving daptomycin or the comparator (2.8% vs. 1.8%). 35 Only two patients in the daptomycin group had to discontinued the drug because the elevation in the serum CPK level. However, in clinical trial of patients with S. aureus bacteremia or endocarditis with daptomycin at 6 mg/kg/day, 17 more patients in the daptomycin group experienced CPK increase than those in the comparator arm (6.7% versus 0.9%, respectively); however, only 2.5% of the patients required discontinuation of daptomycin. It appears that muscular toxicity usually takes place after at least 7 days of treatment with daptomycin.
In a more recent pilot study, 48 patients with cSSSI were randomized to receive daptomycin in a dose of 10 mg/kg for 4 days. 44 The safety profile of daptomycin in this study was similar to that reported in prior studies using lower doses. More recently, a retrospective analysis of 61 patients, 14 of which had cSSSI, treated with daptomycin at a mean dose of 8 mg/kg/day (range, 7–11 mg/kg/day) for a median of 25 days (range, 14–82 days) developed CPK elevation rate similar to that seen in prior clinical trials using lower doses. Three of the 61 subjects (4.9%) developed grade III CPK elevation (>1000 U/L) on day 24–28 of treatment, associated with muscular complaints, all of which resolved after discontinuation of daptomycin. 47 It should be noted than in healthy volunteers, daptomycin was well tolerated in doses up to 12 mg/kg/day for 14 days. 26 Therefore, it is recommended that patients who receive daptomycin have CPK determined at least weekly, and the antibiotic stopped in the presence of unexplained muscle pain and CPK increased at least five times upper normal limit or an isolated CPK elevation at least 10 times upper normal limit.
Another symptoms that were reported more frequently in subjects receiving daptomycin at 6 mg/kg/days than in the comparator arm were those related to the peripheral nervous system such as paresthesias, dysesthesias and peripheral neuropathies. These symptoms were mild to moderate in severity and most resolved during treatment. 17
No significant drug interactions are expected with daptomycin since this drug is not metabolized through the cytochrome P450 system. In patients taking other drugs which are associated with muscle toxicity such as HMG-CoA reductase inhibitors, CPK levels should be monitored more frequently when they are coadministered with daptomycin or these drugs should be momentarily discontinued.
Conclusions
SSSIs are frequently challenging infections that are commonly caused by MRSA strains, both nosocomial and community acquired. The efficacy of daptomycin in this setting was shown in well designed clinical trials. The use of daptomycin in a dose of 4 mg/kg/day appears as appropriate for most SSSIs; however, many investigators are recommending now higher doses of daptomycin in view of its concentration dependent activity and the risk of resistance development in severe staphylococcal disease. The once daily dosing regimen of daptomycin is a clear advantage for outpatient management of complicated SSSIs. Daptomycin in patients with SSSIs should be considered in rapidly progressing infections, prior failure or intolerance to glycopeptides, and SSSIs caused by MRSA with high vancomycin MICs.
Disclosures
MES is a consultant to Theravance Inc., has received a research grant from Theravance Inc. and honoraria from Astellas in the past. ECN has received honoraria and a research grant from Theravance Inc. and Johnson & Johnson. This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
