Abstract
Levetiracetam is a second-generation antiepileptic drug which first came to the United States market in 1999. It has a mechanism of action that is not well elucidated. However, it is a very favorable antiepileptic drug due to its reliable pharmacokinetics, minimal drug interactions, seizure efficacy and good tolerability. It is an agent that has established efficacy as an adjunct therapy agent for partial and refractory seizures. As a monotherapy agent, levetiracetam also appears to be an attractive agent with observed efficacy and tolerability. Since levetiracetam has recently become available intravenously, it is also being reviewed as an agent for acute status epilepticus. In pediatrics, levetiracetam is widely used with efficacy seen in small clinical trials for a variety of seizure types. Levetiracetam is well tolerated: the most common adverse effect being somnolence and behavioral effects. Overall, levetiracetam is a notable antiepileptic drug that has added significantly to the current antiepileptic armamentarium.
Introduction
Levetiracetam (Keppra; UCB Pharma, Inc., Smyrna, Ga., USA) is one of the new second generation antiepileptic drugs (AED) which first received approval from the United States Food and Drug Administration (FDA) as adjunctive treatment for partial-onset seizures in adults in November of 1999. 1 In June 2005, it received approval for use as adjunctive therapy in children 4 years and older with partial-onset seizures. In 2006, levetiracetam was granted approval as adjunctive therapy for myoclonic seizures in patients 12 years of age or older. It also received approval for treatment of primary generalized tonicclonic seizures in children 6 years of age and older in March of 2007. Levetiracetam has data in patients with benign partial epilepsy and status epilepticus, but there is no FDA indication at this time.2,3 While it is primarily used for epilepsy, other off-label uses for levetiracetam include bi-polar disorder, migraine prophylaxis, neuropathic pain, postherpetic neuralgia, and myoclonus. 4
Levetiracetam
Levetiracetam is an S-enantiomer pyrrolidine derivative. Although the exact mechanism of action of levetiracetam is not known, studies speculate it may involve inhibition of N-type voltage dependent calcium channels, reduction of GABA and glycine mediated inhibition, reduction of potassium currents, and/or synaptic protein binding which alters neurotransmitter release.5,6 The protein-binding site of levetiracetam has been discovered and is thought to be bound to a neuronal synaptic vesicle (SV2A). 7 Unfortunately, the exact molecular action of SV2A is unknown. Structurally, levetiracetam is similar to piracetam, a nootropic agent that has shown efficacy against myoclonic epilepsy. 8
Levetiracetam is a unique antiepileptic because its pharmacokinetic profile is noticeably different from a majority of other AEDs;6,8,9 including favorable linear pharmacokinetics and less drug interactions compared to the first generation antiepileptics. Levetiracetam has rapid absorption properties and is 100% bioavailable. Administering with food may delay absorption but does not affect the extent of absorption. It has a relatively quick onset of action ranging from 20 minutes to about 2 hours depending on the formulation. Levetiracetam has low protein binding, <10%, and a volume of distribution similar to total body water (Vd = 0.5-0.7 L/kg). The low molecular weight and protein binding suggest that the embryo and fetus could be exposed to the drug. It is primarily metabolized by enzymatic hydrolysis with inactive metabolite formed; UCB L057 a carboxylic acid metabolite and 2 minor metabolites which are products of the 2-oxo pyrrolidine ring. Levetiracetam follows the linear pharmacokinetic model and elimination is primarily renal through glomerular filtration (66%) with small amounts undergoing tubular reabsorption. Therefore, dosage adjustments should be made for patients with renal impairment and supplemental doses should be administered after dialysis. The pharmacokinetic profile of levetiracetam in children has been reported to be similar to adults; 10 however, the half-life in children is slightly shorter (6.0 ± 1.1 hours) compared to adults (6-8 hours). In a small study of neonates, the half-life was found to be 18 hours. 11 In addition, total body clearance is approximately 30%-40% faster in children compared to adults based on single-dose studies of 20 mg/kg in children ages 6-12 years. 12 Thus, higher maintenance doses may be required in children compared to adults. A higher elimination has been found in 12 pregnant women as plasma concentrations decreased throughout trimesters; clearance increased from 124.7 (±57.9) L/day (baseline) to 427.3 (±211.3) L/day (in the third trimester, p < 0.0001). 11 The therapeutic range of levetiracetam is not well defined and therefore serum concentrations monitoring is not routinely done. Levetiracetam concentrations have been reported between 6-20 mg/L, 13 but have not been well correlated with clinical efficacy. Although, some patients may have levels drawn to verify compliance.
One benefit of levetiracetam is the lack of drug interactions. Several clinical studies have assessed levetiracetam for pharmacokinetic drug interactions with other AEDs and have not seen any clinically significant interactions with carbamazepine, gabapentin, lamotrigine, phenobarbital, primidone, or valproic acid.14,15 However, increases in phenytoin levels of 27%-52% have been noted in a small number of patients with the addition of levetiracetam. 14 Furthermore, a pharmacodynamic interaction has also been observed where carbamazepine toxicity has occurred. 16
Levetiracetam has a pregnancy category of C: as animal data suggests a risk and few human studies are available. 17 Low levels of levetiracetam have been found in breast milk. 11 The milk:plasma ratio has been found to be 1.05 in a study of 11 lactating women. 11 The relative infant dose in this study was 2.4 mg/kg/day and plasma concentrations of the infant were 4-20 μmol/L.
It is additionally challenging to specifically study levetiracetam's use in pregnant women as pregnant women with seizures are often on many concomitant AEDs. In pregnant rats, an equivalent of the maximum recommended human dose (MRHD), 3000 mg, was studied throughout pregnancy. 17 An increased incidence of retarded growth prenatally and postnatally, as well as, minor skeletal changes were found. Increased mortality and behavioral changes were seen at six times the MRHD. Doses of twelve times the MRHD during organogenesis resulted in decreased weight of the fetus and increased skeletal changes. When six times the MRHD was administered during the third trimester no fetal changes or harm were noted. Skeletal changes and mortality were also seen with pregnant rabbits. Human data is sparse. A United Kingdom pregnancy register reported major congenital malformations (two children having spinal bifida, 1 having pyloric stenosis) in three infants of 78 women taking levetiracetam and other antiepileptics. 18 In the 39 women taking levetiracetam alone, four infants had a low birth weight. Women currently taking levetiracetam can be enrolled in the following registries for levetiracetam: the Antiepileptic Drug Pregnancy Registry (1-888-233-2334) or the Keppra pregnancy registry (1-888-537-7734).9,17
Levetiracetam dosing (oral and intravenous) in adolescents (≥16 years) and adults for partial onset seizures is usually initiated at 500 mg twice daily with increases of 500 mg/dose occurring every 2 weeks.9,13 The recommended maximum in this age group is 1500 mg twice daily but doses greater than 3000 mg have been utilized in trials. The extended release oral levetiracetam is only approved for children 16 years and older with partial onset seizures. 1 The recommended initial dose is 1000 mg daily with possible increases by 1000 mg every two weeks. The recommended dose of levetiracetam for partial onset seizures in children (4-15 yrs of age) ranges from an initial dose of 10 mg/kg/dose twice daily with biweekly increases of 20 mg/kg/day to maintenance doses of approximately 60 mg/kg/day in twice daily dosing. For myoclonic seizures, oral dosing is initiated at 500 mg twice daily, with increases every two weeks by 500 mg/dose. A maintenance dose of 1500 mg twice daily is recommended, with higher doses not showing efficacy. Some physicians may initiate therapy at lower doses and titrate to maintenance therapy doses.
Levetiracetam (Keppra) is available as a grape-flavored, oral solution (100 mg/ml) and as 250 mg, 500 mg, 750 mg, or 1000 mg tablets.9,13 In September 2008, Keppra XR was approved as a 500 mg extended release tablet. 1 A generic formulation of levetiracetam became available for the 250 mg, 500 mg, and 750 mg tablets in November 2008. 1 An intravenous (IV) formulation of levetiracetam (Keppra) (500 mg/5 ml) was approved by the FDA in July of 2006 and is the first of the second-generation antiepileptics to have an IV formulation. 1 It is recommended that the tablet be swallowed whole and may be taken with or without food. The IV formulation should be infused over 15 minutes and may be mixed with normal saline, lactated ringers or 5% dextrose in water. The solution is then stable for 24 hours in polyvinyl chloride (PVC) bags which are at room temperature.
Use for Adjunct Therapy in Adults
The efficacy of levetiracetam for adjunct treatment of epilepsy is well established. There were three non-published multicenter, randomized, double-blind, placebo-controlled clinical trials used to obtain FDA approval. A total of 904 patients who received either placebo or levetiracetam1000 mg, 2000 mg, or 3000 mg (doses divided twice daily) were evaluated. Effectiveness was determined by a reduction in weekly partial seizure frequency (%) and by responder rate (≥50% reduction in weekly seizures from baseline). In the first of 3 studies levetiracetam at doses of 1000 mg (n = 97) and 3000 mg (n = 101) daily was evaluated and compared to placebo (n = 95) over a study period of 18 weeks. The reduction in seizure frequency over placebo was 26.1% in the 1000 mg group and 30.1% in the 3000 mg group. Moreover, the responder rates were 37.1% and 39.6% for the 1000 mg and 3000 mg groups, respectively. A second study evaluated the efficacy of levetiracetam doses of 1000 mg (n = 106), 2000 mg (n = 105), and placebo (n = 111) over a treatment period of 16 weeks. Results showed a 17.1% and 21.4% reduction in seizure frequency over placebo for the 1000 mg and 2000 mg groups, respectively. A responder rate of 20.8% and 35.2% was seen in the 1000 mg and 2000 mg groups, respectively (p = 0.02). A third study evaluated levetiracetam 3000 mg (n = 180) versus placebo (n = 104) over a 16 week treatment period. A 23% reduction in seizures was seen, with a responder rate of 39.4% for the levetiracetam group. 19 All results were stated to be statistically significant by UCB, but no p-values were documented for the primary endpoint.
Among peer-reviewed published trials, the efficacy of levetiracetam as an adjunct agent at different dosages (1000 mg, 2000 mg, 3000 mg, 4000 mg) was evaluated by multiple multicenter, randomized, placebo-controlled trials.20–22 In a majority of these trials, the most common concomitant AEDs were carbamazepine, phenytoin, valproate, gabapentin, and vigabatrin. The results of a 14 week treatment trial, after a 4 week titration period, by Cereghino and colleagues demonstrated a 20.9% seizure frequency reduction over placebo (n = 95) in the 1000 mg group (n = 98), and 27.7% reduction over placebo in the 3000 mg group (n = 101) (p < 0.001). 18 Responder rates were 33%, 39.9%, and 10.8% for the 1000 mg, the 3000 mg, and placebo groups respectively (p < 0.001). 20 In a 24 week study, Betts and colleagues evaluated placebo (n = 39) or levetiracetam 2000 mg (n = 42) and 4000 mg (n = 38) daily without titration to desired maximum dosage. 21 The responder rates at 24 weeks were 48.1% (p < 0.05), 28.6% (NS), and 16.1% for the 2000 mg, 4000 mg, and placebo groups, respectively. The difference was statistically significant between the 2000 mg and placebo groups. Seven patients were seizure free at 24 weeks (4-2000 mg; 2-4000 mg; 1-placebo). A response rate was seen in 43% of patients who completed the additional 24 week open-label phase of the study. 21 Interestingly, the 4000 mg daily dose was observed to have less efficacy and more somnolence associated with use. Shorvon and colleagues evaluated placebo (n = 112), levetiracetam 1000 mg daily (n = 106), or levetiracetam 2000 mg daily (n = 106) in a 12 week trial. 22 Percent reductions in seizure frequency from baseline compared to placebo were 16.4%, and 17.7% for the 1000 mg and 2000 mg groups, respectively (p = 0.003). The responder rate was also significant for levetiracetam when compared to placebo with 22.8% of patients in the 1000 mg group (p = 0.019) and 31.6% patients in the 2000 mg group (p < 0.001). 22 The results of all these studies demonstrated the efficacy of levetiracetam at doses of 1000 mg, 2000 mg, and 3000 mg. 21
A multicenter, randomized, double-blind, parallel-group, responder-selected study conducted by Ben-Menachem and colleagues evaluated the efficacy and tolerability of levetiracetam 1500 mg twice daily (n = 181) as monotherapy versus placebo (n = 105). 23 Prior to being placed on levetiracetam monotherapy, levetiracetam was added on to a baseline AED regimen to determine if patients would respond to levetiracetam therapy. In the 16 week add-on phase of this trial, the median partial seizure frequency was decreased significantly in the levetiracetam patients compared to placebo: median percentage reduction in partial seizure frequency from baseline to addon was 39.9% for levetiracetam versus 7.2% for placebo (p < 0.001). The responder rate in the addon phase was significantly higher with levetiracetam (42.1%) compared to placebo (16.7%) (p < 0.001) and 14 of 171 (8.2%) levetiracetam patients remained seizure free compared to only one placebo patient (p = 0.012). The number needed to treat to obtain one responder was 3.9 and one seizure free patient was 13.9. The patients who responded continued on into a monotherapy phase of this trial which is reviewed in the next section. 23
Privitera analyzed pooled data from three phase III clinical trials20,22,23 of adjunctive levetiracetam for partial-onset seizures with or with-out generalization. 24 Levetiracetam doses of 1000-3000 mg were utilized in the studies and all patients were titrated over 4 weeks. Results indicated that patients treated with levetiracetam had a 31.3% median decrease in seizure activity when compared to placebo (p < 0.001). Patients receiving levetiracetam had an overall response rate to treatment of 35% compared to placebo (9.4%) (p < 0.001). The response rates by dose were 28.6%, 35.2%, and 39.5% for 1,000 mg, 2,000 mg, and 3,000 mg respectively. The author concluded that levetiracetam is effective as adjunctive therapy for reducing seizure frequency at doses of 1000-3000 mg daily. 24
Boon and colleagues conducted a continuation trial from the Cereghino and colleagues trial in which a dose-response effect of levetiracetam was evaluated by cross over design (n = 324). 25 Patients with partial seizure were randomized to placebo versus levetiracetam 1000 mg or 2000 mg daily for 16 weeks and were then crossed over to the alternative treatment for 16 weeks. Statistically significant seizure reductions of 16.9% and 18.5% were observed in the 1000 mg and the 2000 mg group versus placebo(p < 0.001), respectively. There was no statistically significant difference seen between the two levetiracetam groups (p = 0.59). Secondarily, a response rate of 26.2% (p = 0.004) and 34.3% (p = 0.001) was seen in the 1000 mg group and 3000 mg group, respectively, versus placebo (12.2%). A reduction in seizure frequency of at least 75% was observed in 13.7% (p = 0.043) of the 1000 mg patients and 20% (p < 0.001) of the 2000 mg group, while only 4% of placebo patients saw a 75% reduction. The results of this trial demonstrated a dose-dependent decrease in seizure frequency with levetiracetam. 25
Larger, open-label studies such as the KEEPER trial and the SKATE trial have further confirmed levetiracetam's efficacy at doses of 1000-3000 mg for partial seizures in adults. Morrell and colleagues conducted an open-label community-based (United States) study of levetiracetam as add-on therapy for partial seizures (KEEPER trial). 26 Patients were started on levetiracetam 500 mg twice daily for two weeks and then evaluated for seizure control. If not controlled, then the dose was increased by 1000 mg daily every two weeks to a maximum dose of 3000 mg daily. Patients were followed for a total of 16 weeks. A total of 1030 patients formed the intention-to-treat population and 767 of those completed the trial (72.5%). The median reduction in partial seizure frequency was 62.3%. A response rate was observed in 57.9% of patients, and 40.1% of patients had at least a 75% reduction in partial seizures over the 16 weeks study period. A 100% reduction in partial seizures was seen in 20.1% of the patients. The authors concluded that levetiracetam was an acceptable choice for add-on therapy for partial seizures. 26
The Safety of Keppra as Adjunctive Therapy in Epilepsy (SKATE) is an open-label, community-based phase four trial of levetiracetam in Europe and Australia. 27 This trial was designed similarly to the KEEPER trial; there were 1541 patients enrolled who received levetiracetam for 16 weeks at similar doses and titration schedule. Levetiracetam was evaluated for use as add on therapy for refractory partial seizures. 27 The median reduction in seizure frequency over the treatment period was 51.4% for partial seizures and 50.2% for all seizures. A response rate was seen in 50.2% of patients (767/1529) and at least a 75% reduction was seen in 32.3% of patients (492/1529). There were 16.2% of patients (247/1529) who were seizure free for the entire trial period. For all seizures, 50.1% (766/1530) and 32.5% (498/1530) of patients experienced at least a 50% or 75% reduction, respectively. Seizure freedom was seen in 15.8% of patients for all seizures (242/1530). At the end of the trial, 3.8% of patients had a moderate or marked deterioration of their epilepsy, as determined by the investigator. Quality of life was also evaluated in several population subsets of this trial. The results of the Patient-weighted Quality of Life in Epilepsy questionnaire (QOLIE-10-P) for 981 patients showed an overall increase in mean scores after starting levetiracetam. 27
Levetiracetam has been used for adjunct therapy at doses of 1000 mg, 2000 mg and 3000 mg for treatment of refractory partial onset seizures for several years (see Table 1). It is effective for this indication and should be considered for all patients requiring additional seizure control.
Leveitracetam use for adjunctive therapy.
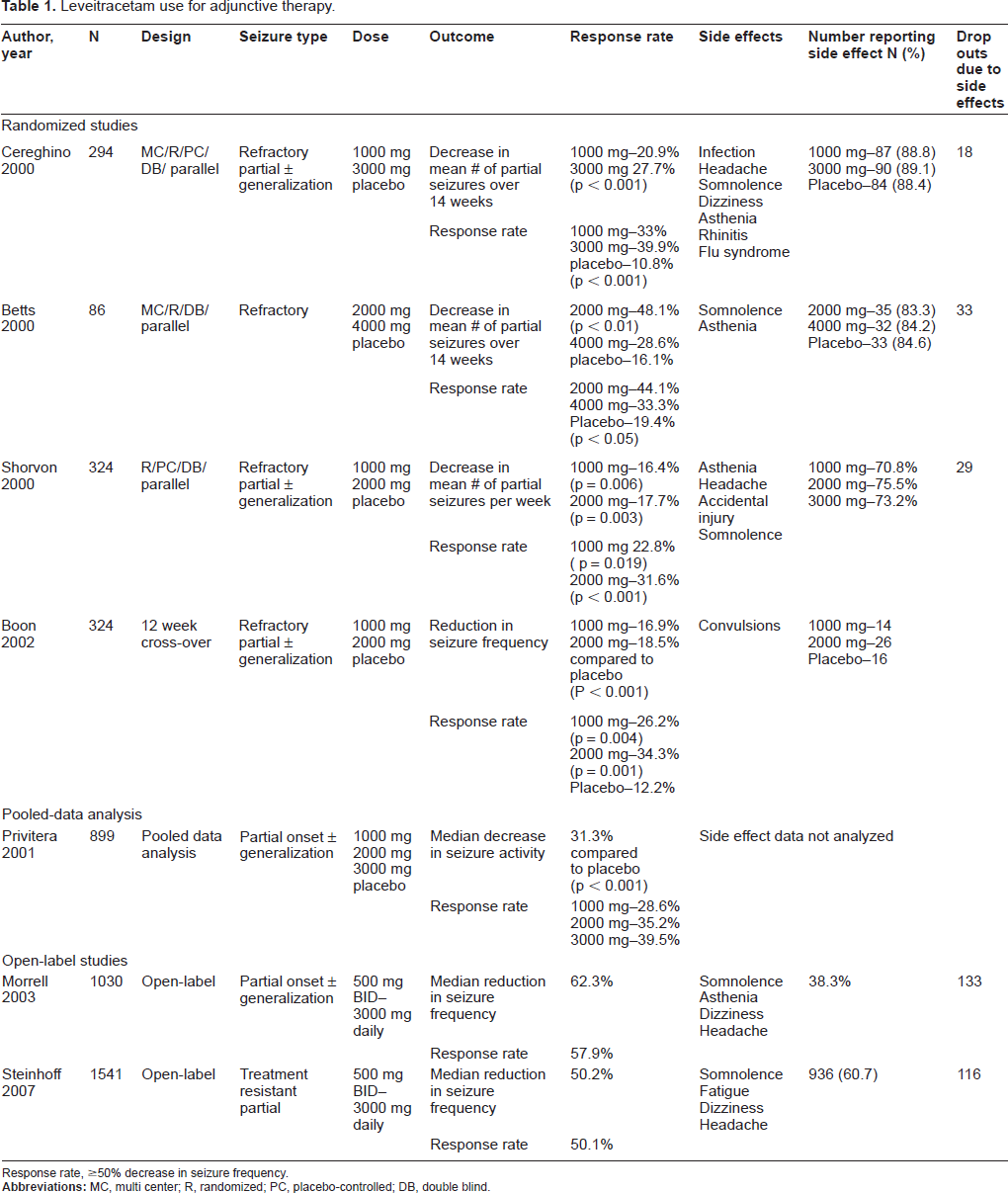
Response rate, ≥50% decrease in seizure frequency.
Use for Monotherapy in Adults
For most of the second generation antiepileptics, including levetiracetam, efficacy as a monotherapy agent has not been FDA approved or rigorously studied in randomized double-blind, placebo-controlled fashion. Despite this, levetiracetam and many of the new AEDs are used clinically as monotherapy agents.
There are several studies that have been conducted demonstrating levetiracetam efficacy as a monotherapy agent.14,23,28–30 A majority of these studies were primarily designed to assess levetiracetam's efficacy as adjunctive therapy, but separately analyzed a subset of patients on monotherapy.23,29,30 Although there were a few prospective studies,14,23 a majority were retrospective or observational in nature.28,30,31 Similar efficacy outcomes were observed, such as reduction in mean weekly seizure frequency and response rate. A few studies also looked at patients who were seizure free and length of time patients were seizure free.28,30
Ben-Menachem and colleagues conducted a monotherapy evaluation of 86 (17 placebo, 69 levetiracetam) patients who had positive benefits during an adjunct therapy phase. Of the 69 levetiracetam patients (80%), 49 were successfully down-titrated to monotherapy (71%), 8 placebo patients were switched to levetiracetam for ethical reasons. 23 Median absolute reduction in partial seizure frequency was 0.61 from baseline to evaluation (p = 0.012). Median percentage reduction in partial seizure frequency was 73.8%. Responder rate was 59.2%, with a slight increase in seizure frequency seen over the end of the add-on phase. This study was conducted in an early stage of drug development to determine if monotherapy with levetiracetam was effective for partial seizures. Use of levetiracetam monotherapy appears to be supported by this data. 23
A retrospective review of elderly (62-92 years old) patients with partial seizures with or without generalized seizures was conducted by Alsaadi and colleagues. 28 Fourteen patients were identified and seizure frequency was tracked using a seizure diary, 8 patients were newly diagnosed with epilepsy. Thirteen patients continued on levetiracetam for at least 6 months. Eight patients became seizure free; 2 patients had >50% but <75% seizure reduction and 2 patients had >75% but < 100% reduction of seizures. The mean dose of levetiracetam was 1839.2 mg per day (range, 500-3000 mg/day). The authors concluded that in newly diagnosed or chronic elderly patients with epilepsy levetiracetam was as safe and effective as monotherapy. 28
Levetiracetam is FDA indicated as adjunctive therapy for myoclonic seizures in patients with Juvenile Myoclonic Epilepsy (JME). Although there is no current FDA indication for its use as a monotherapy agent for JME, there is published data that support its use as a monotherapy agent for this indication in both adults and adolescents.30,32–36 A few open-label observational studies have been conducted on patients with JME (n = 32; n = 48)32,35 and with various generalized epilepsies characterized by myoclonic seizures (n = 35). 30 Patients received levetiracetam in doses of 1000-3000 mg daily as either monotherapy or add-on therapy. Verotti and colleagues observed 29 of their 32 patients (90%) achieving seizure freedom at the 12-month follow-up period. 35 Whereas the two other studies reported seizure freedom of 33.3% and 43% of their patients.30,32 Response rates from these studies ranged from 75% for the monotherapy group to 83% of all patients 30 and 100% from the Verotti study. 35 Labate and colleagues observed better results in patients with juvenile myoclonic epilepsy (n = 21), while worse or unchanged results were observed in patients with refractory generalized epilepsy (n = 6). 30
Based on these studies (Table 2), levetiracetam can and has been used effectively as monotherapy for the treatment of epilepsy and should be considered for treatment where monotherapy is indicated. It appears that levetiracetam is very effective as monotherapy for myoclonic seizures. Further larger studies are being conducted to evaluate the use of levetiracetam as monotherapy in many various seizure types.
Levetiracetam use as monotherapy.
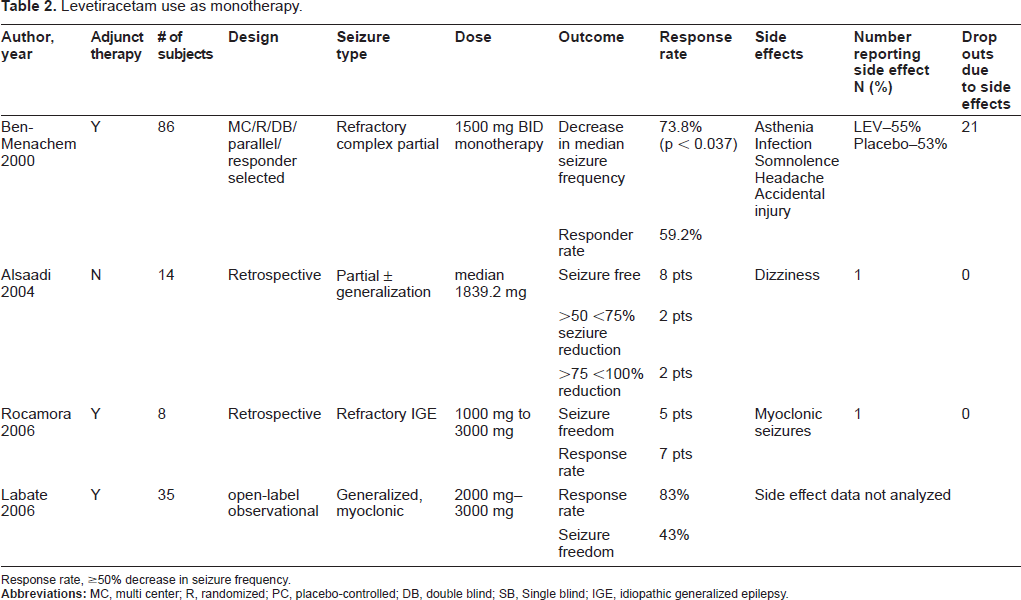
Response rate, ≥50% decrease in seizure frequency.
Long-term Therapy in Adults
The efficacy of levetiracetam for long-term therapy has been evaluated by several studies.37–43 Most of these studies were done up to one year, with the exception of Bauer and colleagues who looked at patients for up to 5 years (mean = 3 years). 41 These studies all aim to demonstrate that levetiracetam can maintain seizure remittance over an extended period of time.
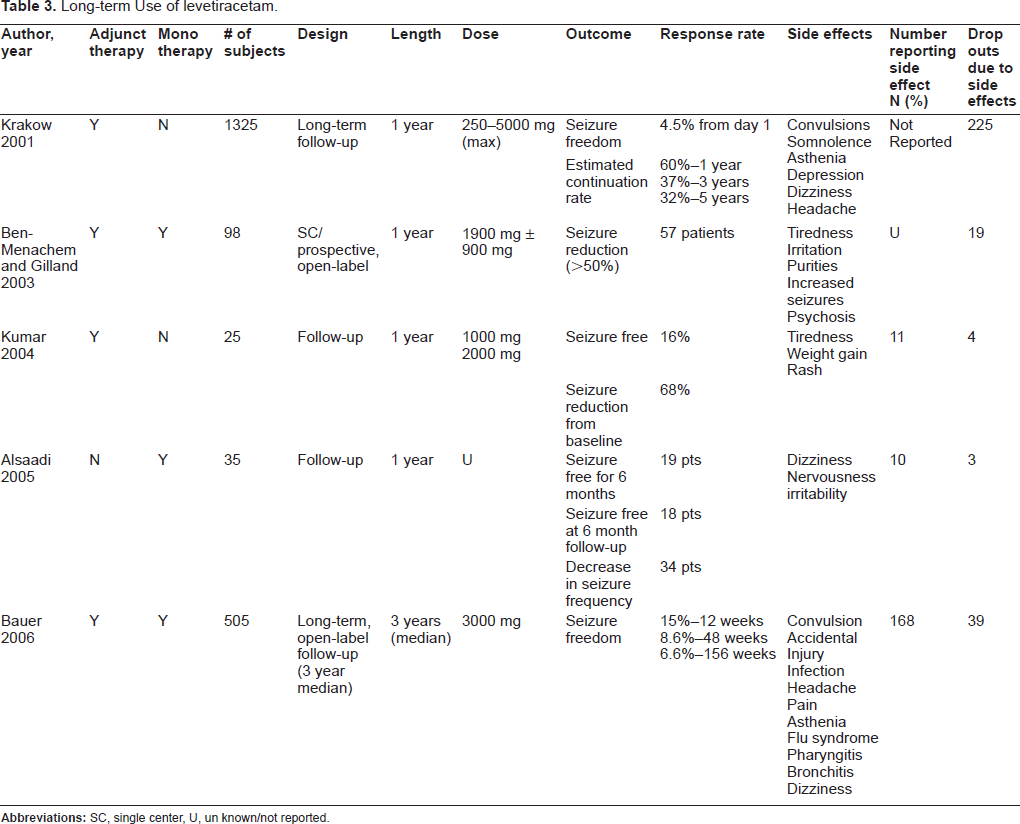
Krakow and colleagues evaluated the long-term follow-up of patients who were enrolled in premarketing levetiracetam clinical trials. 37 The authors evaluated seizure freedom as opposed to seizure reduction. The median starting dose was 1000 mg daily (250-4000 mg), with a median daily dose of 3000 mg daily (250-5000 mg). Of 1,325 patients evaluated, 13% were seizure free at 6 months and 8% were seizure free at 1 year. From the first day of levetiracetam therapy (Day 1), 4.5% of patients were seizure free. The estimated continuation rate of levetiracetam was 60% for 1 year, 37% for 3 years and 32% for 5 years. There were 34.2% of patients who withdrew from levetiracetam due to adverse events (convulsions and somnolence) or lack of efficacy. 37
Ben-Menachem and Gilland conducted a singlecenter, prospective, add-on, open-label long term (1 year) follow-up study (n = 98). 38 Results showed a 62% response rate at the end of the year. Thirteen patients (13.2%) were seizure free at the end of the evaluation period. Side effects were reported by 42 patients (43%) while taking levetiracetam, with tiredness being reported most commonly. This was the first conducted study which looked at long-term efficacy post-marketing. 38
Kumar and colleagues retrospectively evaluated 25 consecutive patients with generalized epilepsies taking adjunct levetiracetam therapy. 39 All patients had been on levetiracetam at least 6 months prior to review. Patients received levetiracetam titrated to 1000-2000 mg daily. Four of 25 patients (16%) became seizure free and 17/25 (68%) had some improvement over baseline. Significant side effects were reported by 11 patients (44%). 39
Alsaadi and colleagues evaluated patients who had started levetiracetam either as monotherapy or had been converted to monotherapy in this single center study. 40 Forty-six patients were identified by the authors; however, only thirty-five patients were evaluated for 1 year. At 1 year, 19 (54%) patients were seizure free for at least the past six months (8 monotherapy). A response rate was seen in 37.1% of patients (>50% but <75% seizure reduction in 25.7%; >75% seizure reduction in 11.4%). Seventeen patients (94%) who were seizure free at 6 months remained seizure free at 1 year. Three patients discontinued levetiracetam after the first two weeks of treatment due to feeling nervous and irritable. 40
Bauer and colleagues conducted an open-label study evaluating the long-term use of levetiracetam for epilepsy. 41 Five hundred five patients enrolled in the study with 274 completing successfully. The primary efficacy variable was the total seizure frequency per week. The median treatment duration with levetiracetam was almost 3 years (24 days to >7 years) with a median dose of 3000 mg (250-6000 mg). The median seizure frequency per week was 0.8 at baseline and 0.3 at 5 years. Seizure freedom from the first dose of medication was seen in 7 subjects into the fifth year. Moreover, other rates of seizure freedom were observed as 15.0% at 12 weeks, 8.6% at 48 weeks, and 6.6% at 156 weeks. Early discontinuation occurred for 138 patients due to lack of efficacy; 67 patients were able to be down-titrated to levetiracetam monotherapy for at least 3 months. 41
There have only been a couple of comparative trials published with levetiracetam. Brodie and colleagues evaluated levetiracetam (1000 mg to 3000 mg daily; n = 285) monotherapy against carbamazepine controlled-release (CBZ-CR) (400 mg to 1200 mg daily; n = 291) monotherapy. 42 Blood samples were drawn to assess the plasma concentration of both study drugs for correct assignment and adherence. The primary endpoint was ≥6 months of seizure freedom as stated in the European regulatory guidelines. Per-protocol analysis was used to assess the primary endpoint (n = 237 for levetiracetam; n = 235 for CBZ-CR at 6 months). Follow-up at 1 year was examined as a secondary endpoint. Results showed that 73% of levetiracetam patients and 72.8% of CBZ-CR patients obtained ≥6 months of seizure freedom at the last evaluable dose (adjusted absolute difference, 0.02%; 95% CI -7.8% to 8.2%). At 1 year, seizure freedom was seen in 49.8% (142/285) and 53.5% (155/291) for levetiracetam and CBZ-CR, respectively. Tolerability of study medication was similar between groups. Levetiracetam was concluded to be non-inferior to CBZ-CR based on the above data. 42
A comparison of levetiracetam (n = 304) and topiramate (n = 470) was conducted by Bootsma and colleagues. 43 The objective was to evaluate the long term effects of the medication between the two drugs on partial onset and generalized seizures. A follow-up period of 24 months was used, with evaluation every 6 months. At 6 months, no significant difference was seen between the retention rates of the drugs. The retention rate after 1 year was better in the levetiracetam treated patients at 65.6% versus 51.7% for the topiramate patients (p = 0.0015); at 24 months levetiracetam showed a retention rate of 45.8% compared to 38.3% for topiramate (p = 0.0046). Seizure freedom rates were not statistically different between the two medications at any time point evaluated and varied from 11.1% to 14.3% for levetiracetam and 11.6% and 20% for topiramate. Due to neurocognitive side effects, twice as many patients stopped taking topiramate as levetiracetam during the study. 43
Additional studies are being conducted that evaluate long-term efficacy of levetiracetam. Based on the data available at present (Table 3), levetiracetam appears to be safe and effective long-term treatment (> 1 year) and can be safely continued for epilepsy treatment. In the comparator studies that are available, levetiracetam appears to be as efficacious as other antiepileptic agents.42,43
Long-term Use of levetiracetam.
Use in Status Epilepticus
Due to a relatively rapid onset of action, the efficacy of levetiracetam has been evaluated for use in treating refractory status epilepticus. None of the studies reviewed were large multi-center randomized trials; a majority were either case-control or retrospective chart reviews. In a majority of the studies, status epilepticus was defined as seizures lasting longer than 30-60 minutes despite appropriate treatment;2,44–47 and\or repetitive seizures2,46,48 However, Uges and colleagues defined status epilepticus as continuous seizure for 5 minutes or more or two or more seizures with incomplete recovery of consciousness. 49 Non-convulsive status epilepticus (NCSE) was confirmed with electroencephalography (EEG).44,49–51 Most of the status epilepticus studies available have been conducted using the oral form of levetiracetam2,44–46,48 but a few studies have evaluated the efficacy of intravenous (IV) levetiracetam47,49,52 Moreover, there have been a few case reports that have detailed the use of IV levetiracetam for treatment of status epilepticus.50,51 It is important to note that patients who were able to take oral levetiracetam may not have been as sick as those who received the IV formulation in the various studies; therefore patients taking oral levetiracetam may have had a better prognosis from the start, making it difficult to directly compare the outcomes of the oral and IV studies.
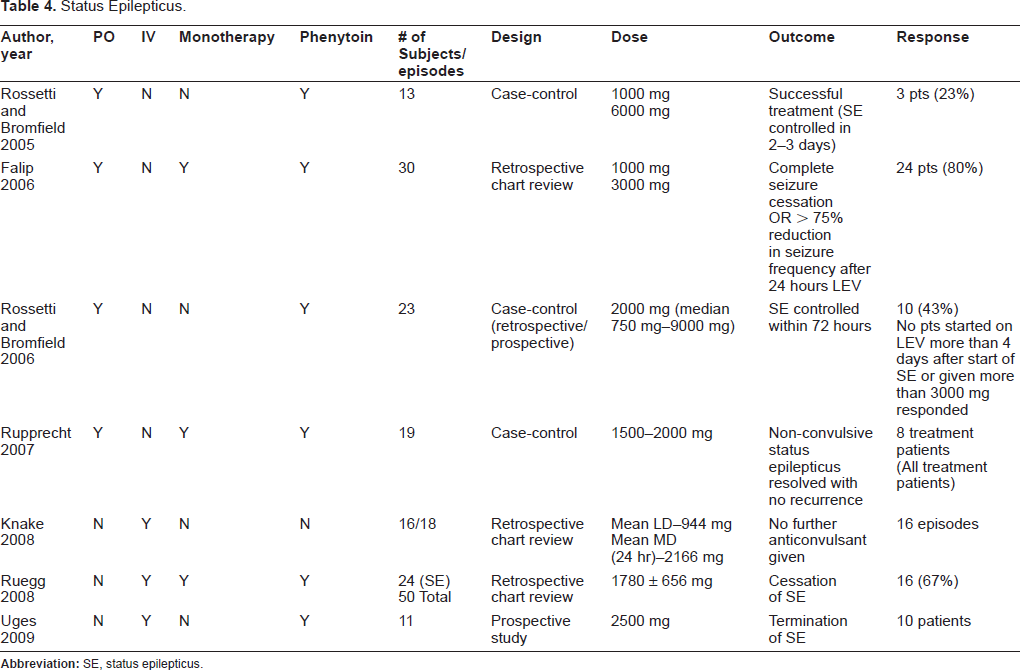
Rossetti and Bromfield retrospectively looked at 13 patients (treatment group) matched to 26 other control cases, matched for age and gender, (control group) in which levetiracetam was administered during treatment of status epilepticus. 2 Oral doses of levetiracetam ranged from 1000 mg to 6000 mg. Concomitant phenytoin was seen more often in the control group. Three patients (23%) were classified as having successful treatment, which was defined as termination of status epilepticus within 2 to 3 days of receiving the study drug. All 3 patients had received levetiracetam for epilepsy treatment prior to status epilepticus onset and dosage was increased 50%-100% during status episodes. 2
Falip and colleagues conducted a retrospective chart review of 30 patients with repetitive seizures. Levetiracetam was administered after failure of phenytoin and valproic acid in 26 patients; 4 patients received levetiracetam monotherapy. 48 Results revealed 24 patients (80%) had either complete cessation or a > 75% reduction in seizures within 24 hours of receiving oral levetiracetam. All four patients who received levetiracetam as monotherapy were effectively treated. Somnolence and dizziness were reported by 3 patients after receiving levetiracetam. 48
Rossetti and Bromfield conducted a second observational, case-control study. 46 Twenty-three patients (8 retrospective, 15 prospective) were evaluated on a median dose of levetiracetam oral of 750 to 9000 mg. Seven patients were receiving levetiracetam prior to status epilepticus. Ten patients (43%) had their status epilepticus controlled within 72 hours. Concomitant antiepileptics were administered in all cases during the 72 hours prior to control. Of note, is that none of the patients started on levetiracetam more than 4 days after the start of status epilepticus or those given more than 3000 mg responded to therapy. 46
Levetiracetam was evaluated as a treatment option for 8 patients presenting with NCSE in a study conducted by Rupprecht and colleagues. 44 In all but one case levetiracetam was added to first line medication (valproate, phenytoin, or benzodiazipines). One patient, who was alert but had continuous epileptic discharges on EEG, received levetiracetam monotherapy. No additional antiepileptic medications were added after starting levetiracetam for all patients. Levetiracetam was administered via nasogastric tube and titrated up to 2000 mg per day. Non-convulsive status epileticus was terminated within 1 to 3 days after start of levetiracetam in all patients. In 5 of the 8 patients (62.5%) coadministered anticonvulsants were tapered off and levetiracetam was continued as monotherapy; levetiracetam was added to phenytoin or oxcarbazepine in the remaining 3 patients (37.5%). One patient experienced dizziness after starting levetiracetam, but no other adverse effects were reported. All patients on levetiracetam monotherapy remained seizure free with no adverse effects at the 2-36 month follow-up. The authors concluded that levetiracetam as add-on therapy for patients with NCSE may be safe and effective. 44
Knake and colleagues evaluated 18 episodes (16 patients) of status epilepticus treated with IV levetiracetam through retrospective chart review. 47 Three patients were treated with oral levetiracetam prior to admission. All status epilepticus episodes were controlled by administration of a combination of antiepileptic drugs; all patients received a benzodiazepine prior to levetiracetam administration. Levetiracetam was the last drug administered in 16 episodes. The mean loading dose of levetiracetam was 944 mg with a maintenance dose of 2166 mg per 24 hours. Sixteen of 18 episodes resolved with no further antiepileptic given to the patients. Sedation was a side effect reported by 2 patients after receiving the study medication. All patients were sent home on oral levetiracetam (mean dose 2058 ± 532 mg). 47
Ruegg and colleagues retrospectively evaluated 50 critically patients who received IV levetiracetam for treatment or prophylaxis of seizures; 24 patients (48%) received IV levetiracetam for status epilepticus. 52 Within the status epilepticus and seizure subset of patients (n = 43), 8 patients received IV levetiracetam as monotherapy treatment. The average dose of levetiracetam for all patients was 1780 mg (±656 mg). Status epilepticus resolved in 16 patients (67%) after levetiracetam administration. In 4 of those patients levetiracetam was first line treatment. There were no serious side effects reported related to administration of IV levetiracetam. Two of 50 patients (4%) developed transient thrombocytopenia as a result of levetiracetam administration. The authors concluded that IV levetiracetam has promising efficacy and safety for us in this population. 52 It is important to note that NCSE was excluded from this evaluation.
A study conducted by Uges and colleagues evaluated the use of IV levetiracetam 2500 mg as add-on in adult patients with status epilepticus (n = 11). 49 Administration was done over 5 minutes. Status epilepticus was established clinically, except for non-convulsive cases which were confirmed by EEG. All patients received benozdiazipines prior to levetiracetam IV administration. One patient received phenytoin and one patient valproate prior to levetiracetam administration. After 24 hours of observation 10 of 11 patients had termination of status epilepticus. Of note, 5 patients still showed signs of confusional behavior 24 hours after administration, but no other adverse events were seen. Patients who were already receiving 2000 mg or more of levetiracetam were excluded from this trial. The authors concluded that add-on of levetiracetam IV to standard status epilepticus treatment is feasible. 49
Although levetiracetam does not have an FDA indication for treatment of status epilepticus, it appears that levetiracetam is a potential option for treatment in refractory patients (Table 4). Since the approval of the IV formulation of levetiracetam, more studies are being conducted evaluating the use of levetiracetam in status epilepticus.
Status Epilepticus.
Use in Pediatrics
In pediatric patients, levetiracetam is FDA approved for use in children ≥4 years old with partial onset seizures and for children >12 years old with myoclonic seizures. The efficacy of levetiracetam in pediatric patients has been established largely through pharmaceutical company initiated trials (UCB) for labeling and marketing. Similar to many adult trials, these trials were placebo controlled for adjunct treatment. Some of these trials have not been published in peer-reviewed journals and the only details available are from the package inserts. The reported pediatric response rates for refractory partial seizures was 44.6% for levetiracetam versus 19.6% for placebo (p = 0.0002) and for myoclonic seizures, 60.4% versus 23.4% (p = 0.0001), respectively. 19
There are several nonrandomized, open-label and retrospective studies which have looked at the use of levetiracetam in pediatric patients as adjunct therapy and as monotherapy. A majority of studies have shown that levetiracetam has the most benefit for partial seizures but it does have benefit in generalized tonic-clonic seizures, infantile spasms, neonatal seizures, absence seizures, localization related seizures, nonconvulsive status epilepticus and various other epilepsy syndromes.3,53–59 Patients as young as 2 days old have had positive experiences with levetiracetam with few to no adverse effects. Dosages have ranged from 6 mg/kg/day to 314 mg/kg/day and duration was anywhere from 1 week to 3 years. 60 The longest published experience with levetiracetam in children was observed by Peake and colleagues who reviewed the use of levetiracetam in 200 children (0.3-19 years) who were maintained on levetiracetam therapy for 3 weeks to 46 months. 61 A majority of these children were on several concomitant antiepileptic drugs (56% on 3-5 AEDs) with only 7 children achieving monotherapy. The retention rate was 49% at more than 12 months. Of these children who were on levetiracetam for more than 12 months, 32% had a positive response rate and 5% obtained seizure freedom.
A compilation of several pediatric studies have shown levetiracetam response rates ranged from 20%-64% and seizure free rates ranged from 2.6%-27% as both adjunct therapy and monotherapy for various chronic seizures.60,62 The large response rate range is likely due to confounding factors in most of the pediatric studies such as use of open-label trials, various concomitant antiepileptics and the many different seizure types. All of the available literature in pediatric patients seems to describe the efficacy and benefit of utilizing levetiracetam in pediatric patients for many seizure types including status epilepticus and NCSE.53,58,59 All of these SE incidences were case reports which described successful seizure control with levetiracetam.
Levetiracetam is not currently FDA approved for status epilepticus or NCSE in critically ill children. However, there are 2 successful pediatric cases of levetiracetam use in NCSE and a retrospective analysis of 10 pediatric patients who received IV levetiracetam; 4 for status epilepticus, 1 for neuroprophylaxis and 1for maintenance of acute seizures.58,59,63 Levetiracetam is frequently utilized off-label in our pediatric intensive care unit for status epilepticus, neurotrauma prophylaxis, seizures due to traumatic brain injury, and refractory seizure of various etiologies. Levetiracetam is especially ideal in this population because of its reliable pharmacokinetics and good tolerability. A lot of successful outcomes have been observed and one of the authors is currently conducting a retrospective review of the IV experiences.
Safety, Tolerability and Adverse Effects
Levetiracetam has a well accepted safety and tolerability profile; especially when compared to first generation antiepileptics. Although there are very few direct tolerability comparisons, levetiracetam tolerability has been reviewed in several instances. The overall incidence of adverse effects in adults appears to range from 42%-84%.21–23,38 The adverse effects that have been observed in clinical trials (>5%) have included asthenia, dizziness, headache, nausea, somnolence, hostility, nervousness, irritability, infection, rhinitis, flu syndrome, pharyngitis, pain, accidental injury, urinary tract infections, psychosis, and pruritus. 64 When levetiracetam has been utilized as a monotherapy agent, it has been well tolerated with the most frequent and severe complaints being somnolence, nervousness, and irritability. The most frequently listed reasons for withdrawal of levetiracetam in studies include somnolence, nausea, vomiting, asthenia, headaches, dizziness, convulsions/tremors, nervousness, irritability, depression, and increased seizures.27,37,40,64 When overall tolerability was directly compared to controlled-release carbamazepine, a larger amount of carbamazepine subjects withdrew due to adverse effects compared to levetiracetam; 19.2% versus 14.4%, respectively. 42 Depression and insomnia was reported more often with levetiracetam whereas back pain was reported more often with carbamazepine. 42
A systematic safety review of all the adverse effects that have occurred during levetiracetam development trials was conducted and published by UCB. 65 In this database 3347 patients were exposed to levetiracetam for various reasons besides epilepsy including Alzheimer's disease, head injury, stroke and anxiety disorders. The daily doses utilized in the various studies ranged from 125 to 6000 mg per day. Upon analysis of lab changes (specifically complete blood count with differential, serum chemistry and liver enzymes) and hypersensitivity reactions among all the patients exposed to levetiracetam, the incidence of events were found to be extremely small; less than 5%. Adverse effects which were seen in the cohort of patients enrolled only in the placebo-controlled epilepsy trials (levetiracetam = 769 versus placebo = 439, respectively) were all infections (30.2% vs. 26.9%), behavioral problems (13.5% vs. 6%), cognition (1%-1.6% vs. 0.3%-1.4%), convulsions (6.9% vs. 8%), worsening of seizures (14% vs. 26%), accidental injuries (10.7% vs. 15.5%), and sleep disturbance (3.6% vs. 2.5%).
Somnolence and behavioral symptoms appear to be the most commonly reported and troubling adverse effects often associated with levetiracetam therapy. Somnolence has occurred in epilepsy clinical trials at rates of 14.8% (n = 769) compared to 8.4% (n = 439) in placebo groups. 65 The overall incidence when compiling epilepsy clinical trials before 2001, appears to be 6.1%-44.7% compared to 3.8%-25.5% for adjunct levetiracetam therapy versus placebo respectively.21–23 A meta-analysis by Zaccara and colleagues compared the central nervous system effects of various new antiepileptic drugs from randomized double-blind trials and found that levetiracetam was significantly associated with somnolence. 66 Although this meta-analysis did not find a relationship between somnolence and drug dose, other clinical trials have seen a dose response relationship. 21 Bootsma and colleagues have reported that somnolence or tiredness appeared to be more common in patients without mental handicap compared to those with mental handicaps. 67
Behavioral effects appear to be a frequent and distinct effect of levetiracetam. The behavioral side effects are more commonly observed in long-term analysis (>1 year),38,67 in pediatric patients and in patients with underlying psychiatric disturbances or learning disabilities.62,68,69 A retrospective analysis to compare psychiatric and behavioral side effects of various antiepileptic drugs was conducted by Weintraub and colleagues. 69 Psychiatric and behavioral side effects were categorized as either anxiety, behavioral changes not otherwise specified, depression, irritability/moodiness, or psychosis. Of the 1029 newly started AED patients reviewed, 521 (50%) were started on levetiracetam and of these there was a 15.7% incidence of psychiatric/behavioral side effects. Comparatively, gabapentin had a 0.6% incidence, lamotrigine 4.8%, zonisamide 9.9%, topiramate 6.3%, oxcarbazepine 5.6%, felbamate 3.6% and vigabatrin 0%. 69 Tiagabine had a similar incidence of psychiatric/behavioral side effects to levetiracetam at 15.8%. However, the number of patients on tiagabine was much lower (n = 19) and not statistically significant in comparison. They also found that a patient with a past psychiatric history was more than twice as likely to experience psychiatric/behavioral side effects. 69 Despite this, one long-term study has described positive effects (increased energy, activity and cheerfulness) attributed to levetiracetam use in 7% of patients at 3, 6, 9 and 15 months. 67 One randomized double-blind study directly compared the difference in anger, hostility, and mood between levetiracetam (500 mg-2000 mg/day) and lamotrigine (50 mg-200 mg/day) over a 20 week period. 70 Both anticonvulsants were given as adjunctive therapy and titrated up over 8 weeks. The primary outcome assessed was the change in score from baseline of the Anger-Hostility subscale score of the Profile of Mood States (POMS). Results showed that mean lamotrigine (n = 132) scores showed improvement compared to levetiracetam (n = 136) for the Anger-Hostility subscale (p = 0.024) and Total Mood Disturbance (p < 0.05). Morevoer, lamotrigine showed improvement relative to levetiracetam for other POMS subscales including Depression-Dejection, Vigor-Activity, Fatigue-Inertia, and Confusion-Bewilderement. 70 One other study that compared the clinical cognitive, neurophysiologic effects of carbamazepine, oxcarbazepine and levetiracetam in healthy volunteers found that levetiracetam was the most favorable of the 3 for causing the least amount of deficiencies. 71
Paradoxical increases in seizure activity have been reported.12,72–74 These are believed to occur at higher doses of levetiracetam and in patients with preexisting mental disabilities.12,74 Nakken and colleagues observed 18% of their adult population had an increase in seizure frequency with 3 patients developing status epilepticus and in children, 43% had increases in seizure frequency with 3 children developing new seizure types and 4 developing different types of status epilepticus. 12 A systematic retrospective evaluation of this adverse event has been conducted by Scuzs and colleagues. 74 Paradoxical effects were defined as increases in seizure frequency or the occurrence of more severe seizures such as generalized tonicclonic within 1 month of levetiracetam initiation. They observed an incidence of paradoxical effects of 14% (207 patients) and a majority of these incidences occurred in mentally challenged patients (p < 0.001). 74
There have been isolated case reports of levetiracetam causing hyponatremia, liver failure, enterocolitis, thrombocytopenia, depression, and myoclonic encephalopathy due to renal failure.75–80 Most of these case reports did not give enough details to completely prove causality nor did rechallenge occur. Rechallenge did occur with the cases of hyponatremia and the liver failure requiring a transplant. The liver failure patient's liver enzymes increased post liver transplant which correlated with reintroduction of levetiracetam. 77 Another case report has reported liver failure in conjunction with both carbamazepine and levetiracetam therapy. 77 Although none of these effects have been observed in any of the large prospective or retrospective reviews, it is important to be aware of the reported cases.
Conclusion
Levetiracetam is an antiepileptic agent which exhibits efficacy for several seizure types, reliable pharmacokinetics, an excellent drug interaction profile, and few adverse events. It is an ideal agent for patients who have new onset partial seizures and could possibly replace phenytoin for use in acute status epilepticus. In comparison to many of the first generation antiepileptics (carbamazepine, phenytoin, valproic acid and phenobarbital) levetiracetam is preferred in terms of ease of administration, dosing, drug interactions and tolerability. In comparison to the new generation antiepileptics, levetiracetam is as efficacious but has fewer drug interactions and better tolerability. Behavior problems tend to be the most problematic adverse effect but it appears to occur more often in people who have underlying behavior or cognitive problems. Levetiracetam would be recommended as the drug of choice for partial epilepsy and for most patients with refractory epilepsy of different seizure types in both adults and pediatric patients. However, it should probably not be a first line option in anyone with a significant behavioral or psychiatric history. It is definitely a viable option for status epilepticus and could be tried in favor of phenytoin.
Disclosure
The authors report no conflicts of interest.
