Abstract
Objectives
To provide background information on diabetic peripheral neuropathic pain (DPNP), current treatment options for DPNP, and information about lacosamide. To determine the safety and efficacy of lacosamide as a treatment option for DPNP.
Methods
A literature search was performed in order to describe DPNP, locate pertinent information about lacosamide, and determine the safety and efficacy of lacosamide for the treatment of DPNP in various trials. A search was also performed to review the effects of other medications on DPNP for the education of the reader as well as to make a comparison to lacosamide.
Results
DPNP is a common occurrence in patients with diabetes. Options for the treatment of DPNP include anti-depressants, anti-convulsants, opiates, and other alternative medications. One of the most recently studied drugs for DPNP is lacosamide, an anti-convulsant usually prescribed for the adjunct treatment of partial onset seizures. Three RCTs using lacosamide and an extension to one of the trials was found and reviewed. Rauck et al found that lacosamide titrated up to 400 mg divided twice daily to treat DPNP caused 60% of patients to have a significant decrease of 3.11 on the Likert Pain Scale as compared to 50.8% of patients in the placebo group having a decrease of 2.21 on the Likert Pain Scale from the beginning of treatment until conclusion of the trial. Shaibani et al performed an extension to the trial performed by Rauck et al and determined that the effects of lacosamide used for DPNP were maintained over a 2.5 year period. Wymer et al reproduced the results of the study done by Rauck et al by using 200 mg, 400 mg, and 600 mg of lacosamide divided twice daily and comparing them to placebo to see the difference in the Likert Pain Scale scores from the beginning of the trial to conclusion of the trial. The Likert Pain Scale differences were 1.8, 1.9, 2.5, and 2.0 for the placebo, 200 mg, 400 mg, and 600 mg dosage groups, respectively. In the 400 mg lacosamide daily group, 58% of subjects experienced a significant decrease in the Likert Pain Scale score. The 200 mg and 600 mg dosage groups showed no difference in the Likert Pain Scale scores. Ziegler et al studied the changes in pain scores from baseline to the last four weeks of maintenance therapy using lacosamide 400 mg and 600 mg versus placebo. No significant changes were seen in either group. The 400 mg and 600 mg groups lowered the pain scores -0.40 and -0.36 points below placebo, respectively. However, when broken into sections, the trial did show significant changes in pain scores. There were no significant differences in adverse effects across all trials using the 400 mg dose of lacosamide. No trials were found that compared lacosamide to any other drug for the treatment of DPNP.
Conclusion
Mixed results do show that lacosamide has the potential to effectively and safely lower pain scale scores in individuals with DPNP. However, due to a lack of consistent results and comparison studies with more established and accepted treatments, lacosamide cannot be recommended first line.
Introduction
The incidence of diabetic peripheral neuropathic pain (DPNP) is increasing as the number of patients with diabetes increases. 1 Factors such as abdominal girth and lack of diabetes control also increase the risk of developing DPNP. When a patient develops DPNP, there are many therapeutic options that they may choose from. One of the newest agents used for the treatment of DPNP is lacosamide, which is used for both partial seizures and the treatment of DPNP.
Pathophysiology of Diabetic Peripheral Neuropathic Pain
The exact cause of DPNP is uncertain and could be variable between different patients. However, in all patients, pain is felt in certain regions of the body. 2 This pain is generally described as a shooting sensation, much like a bolt of energy moving from an extremity up the nerve cell and toward the torso. However, most patients only feel a tingling, like small pins touching the skin, but not truly causing pain, as with the first symptom. The tingling is followed by progressive numbness throughout the appendage, which places the patient at an increased risk of severe infection, as they are unable to sense a wound is present on the appendage. Additionally, some patients may note a burning sensation in the affected appendage that will cause them to seek medical attention.
Patients that experience any of the symptoms associated with DPNP have already had permanent neurologic damage to certain key nerves. Pain sensations, such as those related to DPNP, seem to develop from hyperexcitability of afferent nerves in the periphery that send signals to the central nervous system indicating that the tissue surrounding the nerve is undergoing a pain stimulus. However, no actual pain stimulus is present. It appears that over time the number of sodium and potassium channels increases along this pathway, which then allows for progression of the disease, but also allows for certain medications to be used to modify and lessen the effects of the disease. Another mechanism for disease progression is the loss of inhibitory monoamines, such as serotonin, which may magnify the effects of nerve damage. This occurs because reductions in the levels of serotonin in certain nerves cause an increase in the number of pain transmissions that go through the cell, therefore intensifying the feelings of pain in the patient.
In addition to the above possibilities, there are other reasons patients may experience neuropathic pain and other complications of DPNP, including gastroparesis. There are a series of different medications that work to reduce the impairments caused by DPNP by targeting the potential pathways of nerve function loss.
Current Therapies
All patients with DPNP should have their glucose control medications maximized to obtain goal glucose levels. Increased blood glucose can cause significant damage to nerves, which therefore leads to DPNP. Significant damage continues to occur as long as the patient is not at the therapeutic glucose control goal.
Anti-depressants
As mentioned above, there is some role for serotonin in DPNP. Thus, medications that impact serotonin, such as selective serotonin reuptake inhibitors (SSRIs), should have an impact on neuropathic pain. Studies have found that there is a mild improvement of pain symptoms with SSRIs, 3 however newer data demonstrates that therapies targeting more than one neurotransmitter, such as tricyclic antidepressants (TCAs) and serotonin and norepinephrine reuptake inhibitors (SNRIs), may have a larger impact on the control of pain symptoms.
Tricyclic antidepressants (TCAs) are a popular therapeutic option for patients with neuropathic pain. In addition to having an impact on serotonin and norepinephrine, these agents alter sodium channel functions. This additional mechanism may explain why they are viewed as being superior to SSRIs. However, TCAs have several significant side effects, which can limit tolerability. TCAs are proarrhythmic, which will make some providers less likely to use them. The effectiveness of TCAs for the treatment of neuropathic pain has been documented in the literature. One study found that about 70% of patients using amitriptyline had at least a 50% reduction in neuropathic pain within six weeks of starting the medication as compared to 41% of patients using placebo. 4
The SNRIs impact serotonin and norepinephrine like TCAs, but do not have an impact on sodium channels. The use of the SNRIs is reduced due to the cost of the available medications, though their tolerability is probably higher due to less significant side effects as compared to TCAs. SNRIs have been shown to be effective in the treatment of neuropathic pain. One trial found that 48% of patients using duloxetine had a greater than 50% reduction in their reported pain intensity compared to only 27% in the placebo group. 5 As expected, side effects were low with the most frequent effects being nausea (and other GI effects) and somnolence. Each of these side effects was reduced if titration occurred and decreased in intensity and frequency over time.
Anticonvulsants
Anticonvulsants that target either sodium or calcium channels in the management of epilepsy also seem to have a positive impact on the treatment of neuropathic pain. The most frequently used agent, although controversial, is gabapentin. Other agents that are used include topiramate, pregabalin, and carbamazepine/oxcarbamazepine. However, the strongest data showing effectiveness is with gabapentin and pregabalin.
Gabapentin is believed to have an impact on calcium channels inside the central nervous center and will therefore help to stimulate the inhibitory functions of the central nervous system. Though gabapentin has been shown to be effective in reducing the self-reported pain scores from patients with neuropathic pain, it requires a significant titration plan (from 100 mg daily to 3600 mg daily) to allow for the full effect of the medication. 6 If the medication is titrated too quickly, it will produce significant side effects, such as drowsiness and dizziness, which will reduce long term compliance. One trial comparing moderate dosing of gabapentin (1200–1800 mg daily) to amitriptyline (75 mg to 100 mg) found that the two agents at the doses studied had similar rates of effectiveness. 7
Another medication that is similar to gabapentin is pregabalin. It is more selective for specific binding sites that influence calcium channel activation, thus it is reported to have a reduced frequency of side effects. However, it is considered a controlled substance in some countries and will be more highly regulated than gabapentin. At dosages of 600 mg per day, about 46% of patients reported a 50% reduction in neuropathic pain compared to only 22% of patients receiving placebo. 8 Side effects, though reduced in frequency, are similar to those of gabapentin.
Opiates
Opiate and opioid medications are effective in the management of DPNP. Even mixed agonists such as tramadol have been shown to significantly impact mild to moderate neuropathic pain.2,9 However, if the patient is experiencing more significant pain, other agents such as hydrocodone or oxycodone should be recommended for the short and long term management of DPNP. As would be expected, significant side effects will occur along with the potential for abuse with these agents.
Alternative Treatments
Acetyl-L-Carnitine (ALC) has been shown to be effective in reducing not only the pain associated with DPNP, but also some of the nerve conduction issues which impact other aspects of the patient's life. The supplement must be given at higher doses (greater than 2 g/day) and be administered for at least one month before any clinically significant improvement is seen. 10 The supplement is tolerated well and can be obtained from eating a diet higher in red meat.
Though several different classes of agents have been shown to be effective in the management of DPNP, there is still not an agent that has been found to work more than about two-thirds of the time, and there is room for newer, more effective agents with fewer side effects to enter the market. One agent attempting to fill this role is lacosamide.
Lacosamide
Mechanism of Action
Lacosamide [(R)-2-acetoamido-N-benzyl-3-methoxy-propionamide], which is sold under the trade name, Vimpat™, was approved on October 28, 2008 for use in the adjunct treatment of partial-onset seizures for individuals over 17 years of age in the United States. 11 Lacosamide works by stabilizing hyperexcitable neuronal membranes and inhibiting repetitive neuronal firing. 12 It does this by enhancing the slow inactivation of sodium channels, therefore leading to a decrease in the number of action potentials produced by the overactive neurons. 13 The novelty behind lacosamide's mechanism of action is its specificity to enhance the slow inactivation of voltage gated sodium channels (VGSCs) and not the fast inactivation of VGSCs, 14 in which the majority of anti-epileptic medications, such as phenytoin and carbamazepine, act. This specificity is important because the majority of epileptic neurons are slightly depolarized as compared to a normal membrane resting potential. The slow inactivated VGSCs take seconds to minutes to work, while the fast inactivated VGSCs take milliseconds to work. By enhancing the slow inactivation of the VGSCs, lacosamide decreases the ability of neurons to undergo depolarization and sustained firing. This mechanism allows lacosamide to work on specific neurons that are typically depolarized (epileptic foci) as opposed to the normally functioning physiological neurons. 13 It is also believed that lacosamide interacts with collapsin response mediator protein-2 (CRMP-2), a phosphoprotein involved in the downregulation of N-methyl-D-aspartate receptor subunit NR2B.
The exact effect of this action on epilepsy is currently not known, but it is believed to decrease pain transmission in patients with DPNP.
Schedule Information
Lacosamide was placed in Schedule V of the Controlled Substances Act (the lowest level of abuse potential in the United States of America) on June 22, 2009 due to evidence that when self-administered, lacosamide was taken more than saline. 11 Lacosamide also showed behavioral effects in animal and human models similar to that of alprazolam and phenobarbital, even though the behavioral effects were transient compared to these drugs.
Pharmacokinetics
Lacosamide is available as an oral tablet in strengths of 50 mg, 100 mg, 150 mg, and 200 mg, as well as an intravenous (IV) formulation consisting of 10 mg/mL in a 20 mL vial. 12 The oral dosage form is rapidly and completely absorbed (100% bioavailability) from the gastrointestinal tract and has negligible first pass effect. 15 Lacosamide's absorption is also unaffected by food, age, or sex. Lacosamide exhibits linear kinetics leading to peak concentrations being reached one to five hours after oral administration and immediately after the end of intravenous administration, while steady state concentrations are reached after three days of twice daily dosing. The volume of distribution is 0.6 L/kg, and the drug is less than 15% protein bound. Lacosamide is primarily eliminated through renal excretion and hepatic CYP2C19 metabolism, with an elimination half-life of 13 hours. The oral and IV forms are interchangeable on a 1:1 dosing basis. However, it is recommended that patients should not stay on the IV formulation for more than 5 days due to lack of data beyond that period.
Indications and Dosing
Lacosamide is dosed as an adjunctive medication for partial-onset seizures (FDA indication) beginning at 50 mg twice daily and is increased on a weekly basis in increments of 100 mg per day. The typical dosage range for the approved indication is 200–400 mg divided twice daily. The IV formulation can be given as either 30 or 60 minute infusions twice daily. The IV formulation can either be given undiluted or can be given diluted with normal saline, lactated ringers, or dextrose 5% in water. If the formulation is diluted, it is only good for 24 hours and the remainder should be discarded. Prescribers and pharmacists should be aware of renal and hepatic function adjustments. See Table 1 for pharmacokinetic information regarding AUC (area under the curve) and dosing adjustments. No dose adjustment is considered necessary in mild and moderately renally impaired subjects. However, a maximum dose of 300 mg/day is recommended for patients with severe renal impairment (CrCL ≤ 30 mL/min) and in patients with endstage renal disease. For patients with hepatic impairment, the dose should be titrated with caution. A maximum dose of 300 mg/day is recommended for patients with mild or moderate hepatic impairment. The pharmacokinetics of lacosamide have not been evaluated in severe hepatic impairment, therefore lacosamide use is not recommended in this group of patients.
Renal and Hepatic Function Effects on Lacosamide. 15
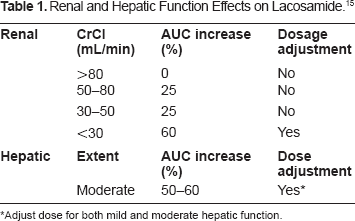
Adjust dose for both mild and moderate hepatic function.
Adverse Effects, warnings, and Monitoring
The most common adverse effects associated with lacosamide include dizziness, headache, nausea, double vision, syncope, ataxia, somnolence, diarrhea, and vomiting. Lacosamide has been shown to increase the PR interval, thus it should be used with caution in patients with conduction problems. There is currently not enough information to give an adequate pregnancy rating, but there are two registries for patients who have been exposed to lacosamide while pregnant, to call in order to better provide information for other females who are taking this medication or physicians who are thinking of prescribing this medication to a pregnant or sexually active female. Careful monitoring while on lacosamide should be practiced in order to avoid possible complications. A baseline ECG should be performed before beginning this medication and after the patient has reached steady state. Patients with decreasing renal and hepatic function should be closely monitored for changes in function due to the need for dosing adjustments. Also, patients should be monitored for suicidal thoughts or behavior as well as depression while taking lacosamide.
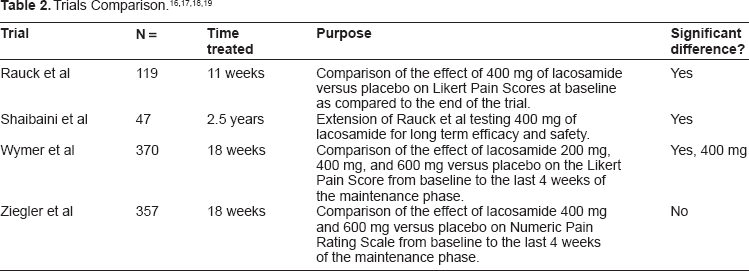
Trials
Rauck et al
There are multiple trials that have studied the effects of lacosamide in the treatment of DPNP. The first Phase 2, randomized, placebo-controlled trial using lacosamide in the treatment of DPNP was performed by Rauck et al. 16 In order to be included in this trial, patients had to be >18 years of age, have a diagnosis of diabetes mellitus (Type 1 or 2), a Likert Pain Score of at least 4, HbA1c < 10% for 3 months, and painful DPNP for at least 1–5 years. Patients that were pregnant, on certain listed medications (which could be used for DPNP), had elevated LFT's, and decreased creatinine clearance were excluded from the trial. A total of 119 patients were randomized to receive placebo or a maximum of 400 mg per day of lacosamide, which was started at 100 mg per day for three weeks then titrated up at 100 mg per day on a weekly basis for the next three weeks. The highest dose reached and/or tolerated was maintained for a four-week period thereafter.
At the end of the maintenance period, patients were titrated backwards in order to discontinue the medication. Safety data was monitored and included lab values, ECG's, and reported adverse events. The primary outcome was the patient recorded pain score using the 11-point Likert Pain Scale ranging from 0 (no pain) to 10 (greatest possible pain), which was compared at baseline and at the conclusion of the trial. The measure of a clinical response to lacosamide was a reduction of at least 2 points in the Likert Pain Score. Secondary endpoints, which included other types of recognized scales and tests were used in order to further confirm the primary outcome. These tests included Short Form-McGill Pain Questionnaire (SF-MPQ), Profile of Mood States (POMS), Patient Global Impression of Change (PGIC), Clinical Global Impression of Change (CGIC), and a count of pain free days.
Rauck et al demonstrated that 400 mg divided twice daily of lacosamide significantly decreased Likert Pain Scores in patients with DPNP. This trial showed that 60% of patients on 400 mg daily of lacosamide had at least a 2-point decrease in their pain score compared to 50.8% of those patients on placebo. Significant differences in pain scores were actually noticed after the first week of treatment with lacosamide and continued throughout the treatment period. Adverse effects were similar in the placebo and lacosamide groups and included mostly headache (18% lacosamide; 22% placebo), dizziness (15% lacosamide; 8% placebo), nausea (12% lacosamide; 7% placebo), and diarrhea (5% lacosamide; 12% placebo). These adverse effects were mostly mild to moderate in intensity and occurred mainly during the titration phase. There were slight findings of an increased QTc interval but these findings were not shown to be clinically significant by the authors. In terms of hepatic impairment, there was a case where a patient had an elevated alkaline phosphate reading and another case that included a patient having increased AST and ALT levels, both of which decreased after discontinuation of lacosamide. However, due to the isolated cases, the authors did not find a causal relationship.
Shaibani et al
Upon the conclusion of the above-mentioned trial, Shaibani et al offered the option of a two-year open label extension trial to assess the safety and efficacy of long-term treatment with lacosamide for painful DPNP. Of the patients who completed the previous double-blind trial (Rauck et al), 47 patients elected to continue lacosamide in the extension phase. 17 The entire trial spanned more than 2.5 years with a total of 34 patients completing the entire trial period. The pain reduction seen in Rauck et al was maintained throughout the extension period with a mean reduction of 3.10 on the Likert Pain Scale, which is significantly different from placebo. The modal dose used in these patients was 400 mg daily. This trial also showed that patients on lacosamide for this extended period of time did not have any significant drug related or clinical concerns associated with the drug. There were no significant differences in the amount of adverse effects that occurred in the maintenance phase (Rauck et al) versus the extension phase.
Wymer et al
A separate trial to study lacosamide in DPNP by Wymer et al randomized 370 patients into a 1:1:1:1 ratio in order to receive placebo, 200 mg, 400 mg, or 600 mg of lacosamide during an 18-week trial period in order to test the safety and efficacy of the 400 mg dose that was found to be efficacious in the trial by Rauck et al. 18 This trial used similar inclusion criteria as Rauck et al with the exception of these trial subjects having HbA1c < 12% for 3 months and pain from DPNP for 6 months to 5 years prior to entry into the trial. Exclusion criteria were similar between the two trials as well. This trial included a 6-week forced titration of lacosamide at 50 mg twice daily and then increasing by increments of 100 mg per day once weekly until the assigned dosage of the subject was met. The forced titration period was followed by a 12-week maintenance phase. After completion of the maintenance phase, patients were weaned over a one-week period and underwent a two-week safety follow-up. The primary efficacy variable was the within-patient change in average daily pain score from baseline to the average pain score over the last 4 weeks of the maintenance phase using the 11-point Likert Pain Scale mentioned earlier. Safety evaluations were performed during the trial as well. As mentioned for the previous trials, other secondary tests were performed as well in order to confirm the clinical significance of lacosamide in treating DPNP.
For patients randomized to the lacosamide 400 mg daily group, the mean daily Likert Pain Score decreased by 2.5 points (38.5%), from 6.5 to 4. For placebo-treated patients, it decreased by 1.8 points (27.3%), from 6.6 to 4.8, which showed a significant decrease in the pain score for the lacosamide 400 mg daily group versus placebo. This decrease in the Likert Pain Score was actually noticed at week 4 of titration and remained significant throughout the rest of the titration and maintenance period. In the lacosamide 400 mg daily group, 58% of patients achieved at least a 2-point or 30% reduction in Likert Pain Score, compared to 46% of placebo-treated patients. There were pain score decreases in the 200 mg and 600 mg daily lacosamide groups as well, however these differences were not significant. The most common adverse events occurring in at least 5% of patients randomized to any of the lacosamide groups included dizziness, nausea, fatigue, headache, and tremor. These effects were seen after dosages of 500 mg or greater were reached. In fact, these adverse effects caused a larger percentage of withdrawal between the different treatment groups. The withdrawal rates for placebo, 200 mg, 400 mg, and 600 mg of lacosamide were 9%, 9%, 23%, and 40%, respectively.
During the safety analysis of this trial, no significant safety issues were discovered. It was found that lacosamide did not affect HbA1c levels, which showed the improvement in pain score was not due to tight glycemic control. Liver tests showed no effects of lacosamide on liver function values. ECG data showed no change in heart rate or QTc interval, but did, however, show a small prolongation of the PR interval in those patients taking lacosamide. Even though there were ECG changes, none of them showed a significant clinical effect.
Ziegler et al
The most recently published study, which examines the safety and efficacy of lacosamide in comparison to placebo, was performed at 52 sites across Europe. 19 This randomized, placebo-controlled trial included individuals with Type 1 or Type 2 diabetes who were ≥18 years of age, and had a pain rating of ≥4 on an 11-point Numeric Pain Rating Scale (NPRS) due to DPNP for the last six months to five years. Patients were excluded from participation in this study if they had other conditions contributing to chronic pain, myocardial infarction or clinically relevant cardiac dysfunction in the last year, elevated LFT's, CrCl < 50 mL/min, decreased or elevated diastolic blood pressure, chronic alcohol or drug abuse within the past year, or any drug use that might interfere with trial results. Participants were not allowed to take pain medications during the trial or within the seven days prior to randomization. A total of 357 patients were randomized in a 1:2:2 ratio to receive either placebo, lacosamide 400 mg (titrated either slow or standard), or 600 mg of lacosamide daily. This was an 18-week trial, which included a one week washout period, a one week baseline pain assessment period, a six week titration phase, and a twelve week maintenance phase. The primary endpoint of the study was to detect a significant intra-individual change on the NPRS from baseline to the average of the last four weeks of the trial.
Baseline characteristics were similar, except for the fact that only two participants in this trial were not of Caucasian ethnicity. Secondary measures as mentioned in the previous trials were also performed as well as the within subject change in pain score in each phase of the trial (titration, maintenance, and entire trial). Standard titration of the lacosamide 400 mg dose included 100 mg per day, which was increased by 100 mg per day weekly until reaching the target dose by weeks 4–6. The slow titration included 100 mg daily for three weeks, then increased by 100 mg daily every week until the target dosed was reached by the end of the titration phase. A safety analysis of adverse effects was also performed.
Ziegler et al found that there was no statistical difference in the studied doses of lacosamide on the lowering of DPNP. The primary endpoint showed a difference of -0.40 and -0.36 NPRS points between placebo and the lacosamide 400 mg and 600 mg doses, respectively at the end of the trial. Both of these changes were not significantly different from placebo. However, if each individual section of the trial is broken apart (titration, maintenance, and entire period), they all show significant differences in the NPRS between placebo and the lacosamide 400 mg and 600 mg doses used in the study. During the titration phase, there was a -0.34 and -0.46 point difference in NPRS between placebo and the 400 mg and 600 mg lacosamide doses, respectively. The maintenance phase showed a -0.66 and -0.79 point difference between placebo and the 400 mg and 600 mg lacosamide doses, respectively. The entire treatment period showed a -0.40 and -0.36 difference between the placebo and the 400 mg and 600 mg lacosamide doses, respectively. The reason that the primary endpoint measure isn't significantly different from placebo could be explained by a possible increased placebo effect during the last four weeks of the trial period. Almost 25% of the placebo group experienced a 50% pain reduction during the last four weeks of the trial. Such high placebo effect responses are not unusual in neuropathic pain studies.
Other secondary studies such as patient's perception of pain on activity and sleep worked in lacosamide's favor by showing a significant decrease in scores from baseline as compared to placebo. Serious adverse events were slightly higher in the 600 mg lacosamide group than the 400 mg and placebo groups (8.3%, 7.3%, and 4.1%, respectively). The most common adverse effects experienced were dizziness, fatigue, nausea, vertigo, headache, and vomiting, which all seemed to be dose related. The authors never mentioned whether significant differences existed between the groups. It is of importance to mention that nearly 23% of patients in the lacosamide 600 mg group that dropped out of the trial, did so because of adverse effects. That compares to 11.3% of dropouts in the 400 mg lacosamide group and 5.4% of the dropouts in the placebo group. Lacosamide did seem to have consistency in its NPRS lowering effects, and it also showed that there were statistically significant changes in the secondary endpoints studies.
Summary of Trials
Three trials mentioned showed significant decreases in the Likert Pain Scale Score in individuals taking 400 mg of lacosamide divided twice daily. Refer to Table 2. Rauck et al first demonstrated this by titrating lacosamide up to 400 mg daily. Shaibani et al then offered an extension to the trial and showed that the decrease in the Likert Pain Score was maintained and there were no significant increases in the amount of adverse effects experienced over this extended time period. Wymer et al then reproduced the effects of the 400 mg lacosamide dosage along with trying a 200 mg and 600 mg lacosamide dosage as well. They found that the 200 mg and 600 mg dosages of lacosamide didn't show a significant decrease in the LIkert Pain Score. In fact, the higher dose caused an increase in the amount of adverse effects and led to more withdrawal from the trial. In contrast, Ziegler et al conducted a trial that showed neither lacosamide 400 mg or 600 mg had a significant effect in lowering NPRS scores from the beginning of the trial until the last four weeks of the trial versus placebo. This trial, however, when broken down into three sections did show that both dosages had significant NPRS changes in comparison to placebo. In regards to adverse effects, the changes seemed to be dose related and it can be assumed that the increase had an effect on the dropout rate of the larger dosage group.
Place in Therapy
These trials do not show a consensus that lacosamide has a statistically significant effect in lowering pain in patients suffering from DPNP. The trials by Rauck et al, Shaibaini et al and Wymer et al demonstrated that using 400 mg divided twice daily of lacosamide was clinically safe and effective in reducing the Likert Pain Score of individuals suffering from DPNP. Higher doses of lacosamide (>400 mg per day) were shown to decrease the pain score, but were not found to be significant. The higher doses also showed an increase in adverse effects and withdrawal from the studies. The 400 mg dose of lacosamide showed an optimal balance between efficacy and safety as compared to the other two dosage options that were studied. However, the study by Ziegler et al shows no difference in using lacosamide 400 mg or 600 mg versus placebo in the treatment of DPNP. If not for the placebo effect, one or both of the dosages could have shown a significant NPRS lowering in order for the primary endpoint to correlate with the significant increase in quality of life that subjects experienced. The majority of data shows that lacosamide is a potential monotherapy for patients suffering from DPNP, but there is also data that shows there is no statistical difference. Due to a lack of head to head analysis with other drugs used for DPNP, it is difficult to assume that lacosamide has better efficacy than other medications. Several considerations need to take place before choosing a medication for DPNP. Lacosamide is a controlled substance, which will lead to tighter regulations over the medication. Also, lacosamide has several unwanted adverse effects. It also has the potential to affect cardiac and hepatic function. Even though these effects are not shown to have a significant clinical difference, there have been reported cases in clinical trials. Lacosamide does not carry an indication from the FDA warranting use for DPNP, while some other medications do. Price may also become an issue for patients that are taking medications due to DPNP because the majority of these patients are taking many other medications for their diabetes as well. As with the initiation of any medication, patient specific factors need to come into consideration. Patients should be screened for any factors that would predispose them to potential adverse effects of each of the mentioned medications. Depending on the type of patient seen determines the type of medication prescribed.
Conclusion
Lacosamide has potential as an agent for DPNP with the majority of data available supporting safety and effectiveness. However, one must keep in mind that there is also data showing no statistical difference. When compared to other agents, which have been used by patients for years to decades, along with inconsistencies in current trial results, it is difficult to recommend the use of lacosamide except as second or third line. As more long-term trials are completed, as well as head to head trials versus the standards of therapy, the place in therapy for lacosamide will be determined.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.